Abstract
Background
Arising from the neuroendocrine cells disseminated all over the body, over 90% neuroendocrine neoplasms (NENs) occur in the gastrointestinal tract or the lung, and the rest arise in the other endocrine tissues.1,2 Although the exceedingly rare occurrence of laryngeal neuroendocrine carcinomas (NECs) severely impeded the investigation, in recent years there have been some improvements in understanding the pathology. In the 4th edition of World Health organization (WHO) classification of Head and Neck Tumors presented in 2017, the laryngeal NECs are divided into 3 groups: the well-differentiated NEC, the moderately differentiated NEC, and the poorly differentiated NEC. The poorly differentiated NEC is further divided into 2 subgroups: the small cell carcinoma (SCNEC) and the large cell carcinoma (LCNEC). 3 LCNEC is recognized as a distinct entity, as opposed to being a part of moderately differentiated NEC. In the 5th edition presented in 2022, the stratification is overarching because neuroendocrine tumor (NET) broad category (divided into G1, G2, and G3 subtypes, based on mitoses, Ki67, and necrosis) is separated from NEC (divided into small and/or large cell subtypes), just like other organ systems. 4 The treatment of primary laryngeal NENs (especially NECs) is substantially extrapolated from that of extra-laryngeal ones, with chemoradiotherapy as a cornerstone. 5 Emanated from dismal survival data, one may speculate surgical resection as an alternative to provide reasonable disease control. In the context of our case series in practice, upfront surgery was the first-line of choice, as opposed to chemoradiotherapy. Our practice may attribute to the world literature, providing additional data for discerning optimal management of this disease.
Case Presentation
Status and Management of Laryngeal NENs
A total of 6 patients were admitted from 2008 to 2019. The following inclusion criteria were used: NENs with clinical, radiological, and histopathological confirmation. The diagnosis was confirmed by examination of primary surgically resected specimens or biopsy. Symptoms, anatomic sites, radiological features, pathology, tumor node metastasis (TNM) classification, treatment modalities, recurrences, metastasis, and the survival data were evaluated.
The demographic data were listed in Table 1. The patients included were adults from 59 to 78 years of age. All male patients had a history of tobacco use. The most common presenting symptom was hoarseness, followed by sore throat and dyspnea. A representative endostroboscopic observation is shown in Figure 1. None of the patients presented with clinical manifestations of ectopic hormone production. Three patients had a stage Iva disease, 2 had stage III, and 1 had stage II. SCNEC was the most encountered subtype in this cohort, followed by LCNEC/NET G1/NET G2. Two of 3 SCNEC patients had 1 site of metastasis, while the LCNEC patient had metastasis to multiple sites.
Clinical Characteristics of 6 Cases.
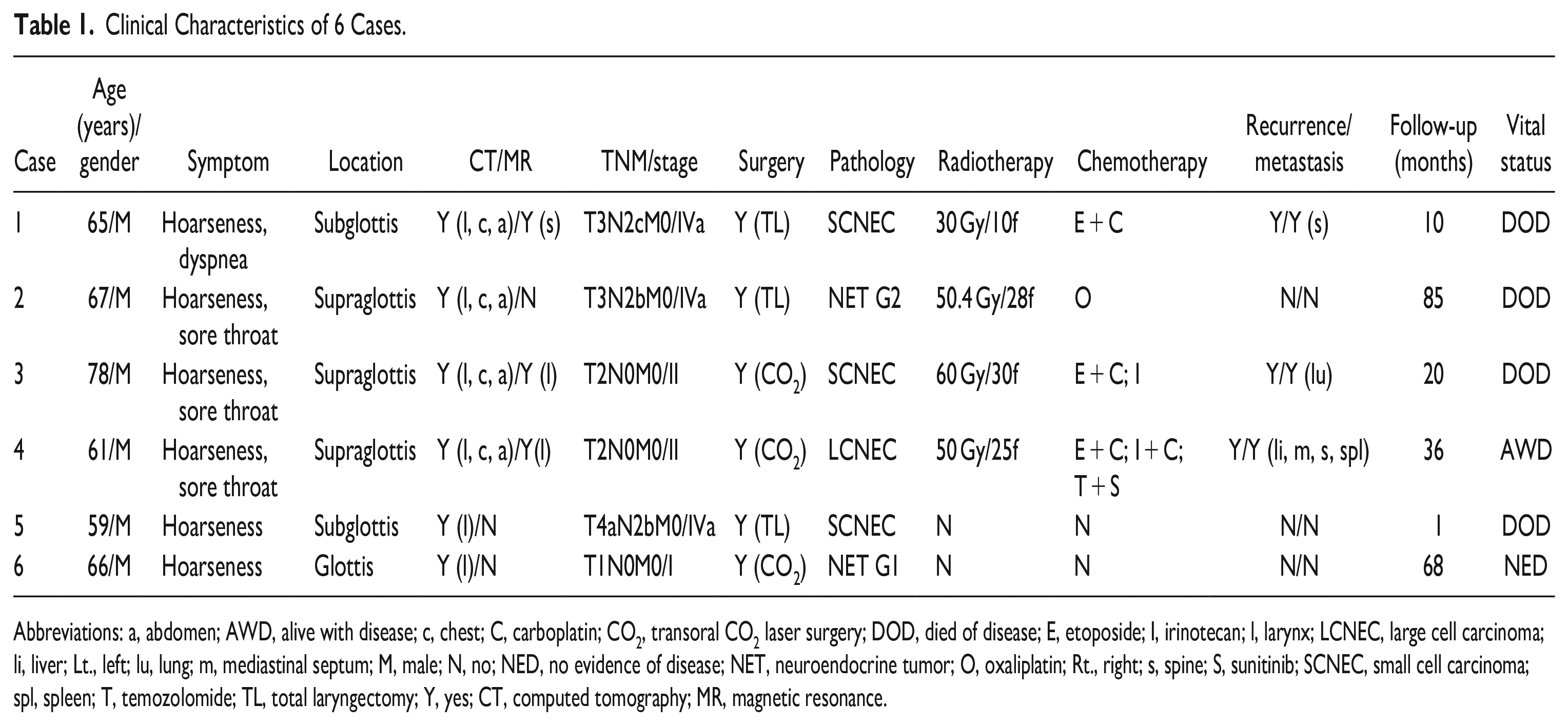
Abbreviations: a, abdomen; AWD, alive with disease; c, chest; C, carboplatin; CO2, transoral CO2 laser surgery; DOD, died of disease; E, etoposide; I, irinotecan; l, larynx; LCNEC, large cell carcinoma; li, liver; Lt., left; lu, lung; m, mediastinal septum; M, male; N, no; NED, no evidence of disease; NET, neuroendocrine tumor; O, oxaliplatin; Rt., right; s, spine; S, sunitinib; SCNEC, small cell carcinoma; spl, spleen; T, temozolomide; TL, total laryngectomy; Y, yes; CT, computed tomography; MR, magnetic resonance.
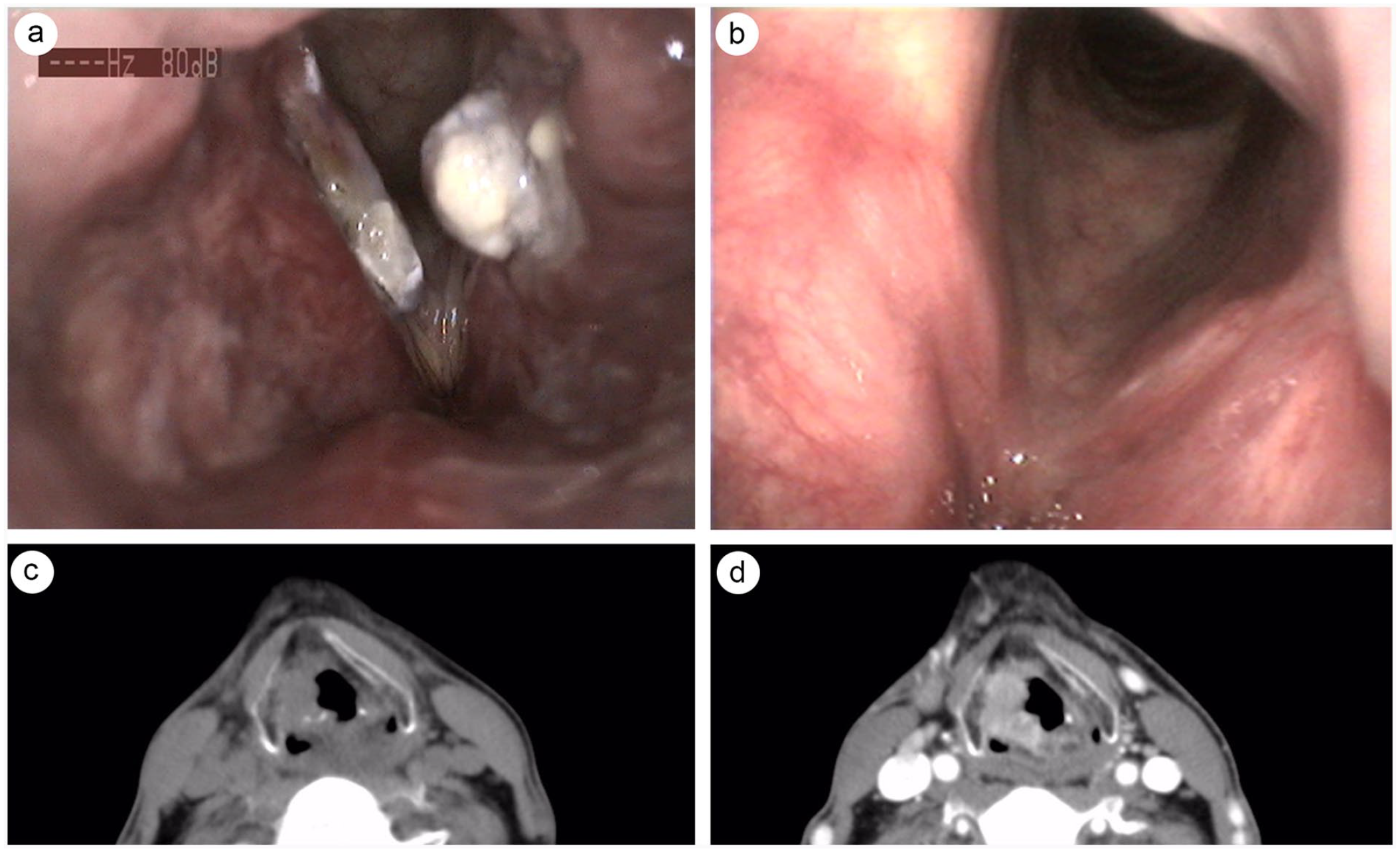
Endostroboscopic and CT examination. Case 4. (a) Laryngeal NEC on presentation. (b) Four months later after CO2 laser surgery and chemoradiotherapy. Case 2. Coronal CT scan, unenhanced (a) and enhanced (b). NEC, neuroendocrine carcinomas.
Radiological Survey
Radiological survey of the masses was conducted. A representative CT is shown in Figure 1. The NENs were isodense or mild hyperdense on CT. The aggressive nature of the NECs was demonstrated by invasion of adjacent structures. In case 1, the arytenoid was partially destroyed. In case 5, the thyroid cartilage was invaded. Contrast-enhanced CT imaging was obtained in 2 cases, which showed homogeneous enhancement (case 2) and heterogeneous enhancement (case 3). On magnetic resonance imaging (MRI), the signal intensity of the lesions was mild hypointense/isointense on T1-weighted MRI; however, it was variable on T2-weighted imaging, with the majority showing mild hyperintense/mixed-isointense signal. Enhanced MR was obtained in 1 case (case 3) showing heterogeneous enhancement.
Upfront Surgical Treatment
All patients underwent surgery and 4 of them received postoperative chemoradiotherapy. The patients were all treated with curative and function-preserving intent whenever possible. For stage I/II laryngeal NENs (cases 3, 4, and 6), the transoral CO2 laser surgeries without neck dissection were carried out. In case 2, the arytenoid was partially resected. The resection of the right aryepiglottic fold from petioles to the arytenoid was performed by CO2 laser. Biopsies were taken from the resection margins to assure adequate removal. All surgical margins were free of tumor. For stage III/IVa laryngeal NENs (cases 1, 2, and 5), total laryngectomies with bilateral (case 1) or ipsilateral neck dissection (cases 2 and 5) were performed. The frequently involved jugular nodes were dissected. The posterior triangle nodes and the paratracheal nodes were also dissected in case 1 and case 5, as the lesions were transglottic. 5 out of 56, 2 out of 21, and 4 out of 22 metastatic lymph nodes were identified in the neck dissection specimen of cases 1, 2, and 5, respectively. Among these 3 cases, the lesion of case 5 not only invaded upwards to the bilateral true vocal cords, anterior commissure, right false vocal cord, and ventricle, but also through the thyroid cartilage. Of all the surgeries, there were no surgical mortalities, and no major complication was noted after operations.
Histological Analysis
The histological findings were shown in Figure 2. Microscopically, cells shared a similar morphology and growth pattern. The different subtypes were distinguished from each other by the cellular architecture, size, pleomorphism, mitotic rate, and evidence of necrosis. The widely used and reliable immunohistochemical markers included chromogranin A and synaptophysin. In this case series, these 2 markers were consistently positive. The epithelial marker cytokeratin was also positive in all cases. S-100, as the most commonly identified negative marker, was not detectable in 2 tested cases (cases 2 and 6). TTF-1 was positive in case 4 and case 5.
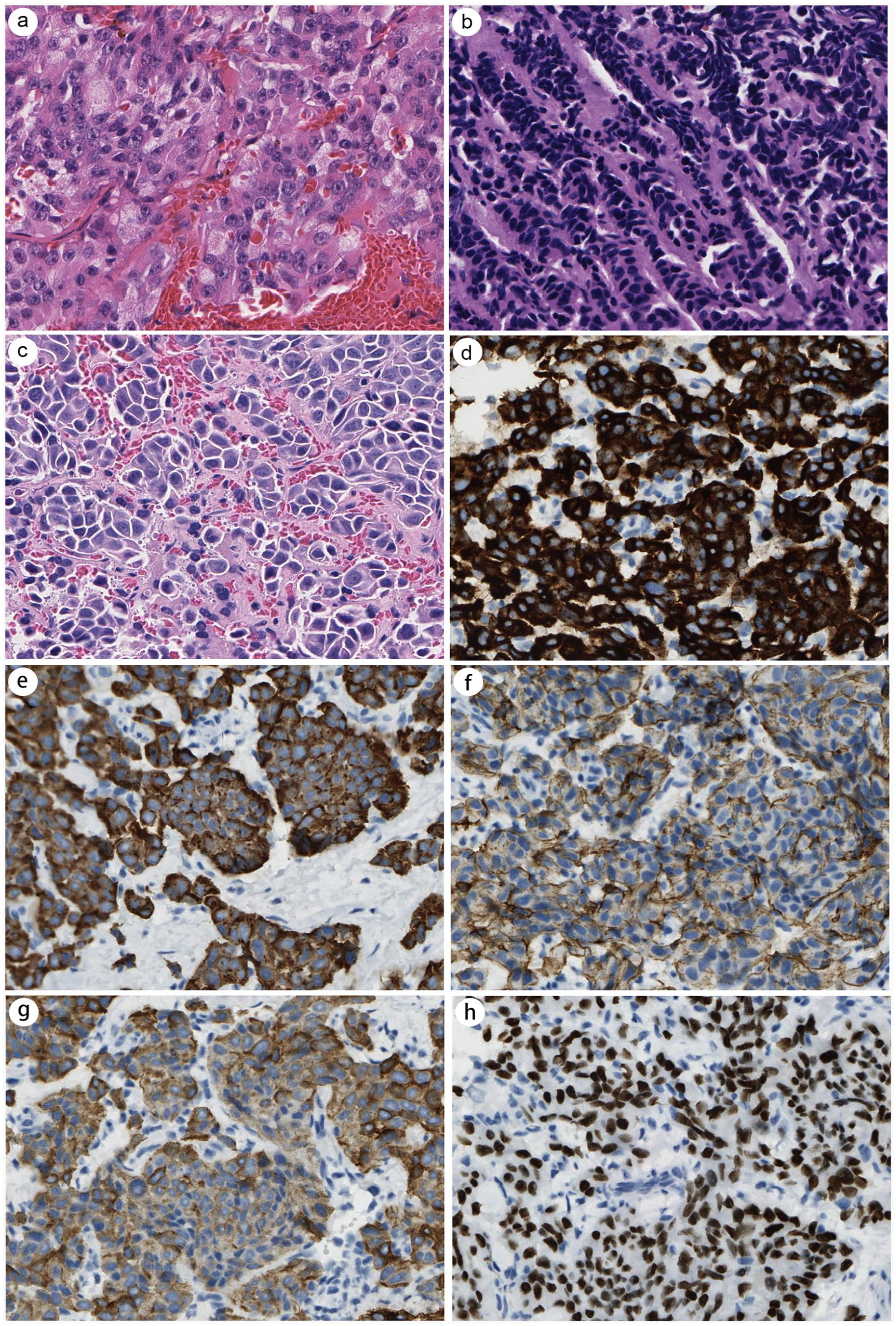
Histologic findings. (a) Case 2. Laryngeal NET G2 (H&E, ×40). (b) Case 3. Laryngeal SCNEC (H&E, ×40). (c) Case 4. Laryngeal LCNEC (H&E, ×40). Case 4. Tumor cells were positive for (d) chromogranin A, (e) synaptophysin, (f) CD56, (g) cytokeratin, and (h) TTF1 (IHC, ×40). H&E, hematoxylin and eosin; LCNEC, large cell carcinoma; NET, neuroendocrine tumor; SCNEC, small cell carcinoma; IHC, immunohistochemical; TTF1, thyroid transcription factor-1; CD56, neural cell adhesion molecule.
Postoperative Chemoradiotherapy
Postoperative adjuvant 3-dimensional conformal radiotherapy by Pinnacle (Philips Radiation Oncology Systems) was employed in 4 of 6 patients, including 2 SCNEC cases (cases 1 and 3), 1 LCNEC case (case 4), and 1 NET G2 case (case 2). The median total dose was 30–60 Gy, administered daily in identical fractions. Postoperative concurrent chemotherapy was also employed in these 4 cases. The carboplatin (100 mg, intravenous drip (VD), on days 1 through 3) and etoposide (200 mg, VD, on days 1 through 2) were concomitantly applied by cycles in 3 cases (cases 1, 3, and 4). Oxaliplatin (100 mg, VD, once a week) were applied in case 2. For case 4, etoposide and carboplatin caused frequent diarrhea, then he started the second-line option of irinotecan (200 mg, VD, day 1) and cisplatin (40 mg, VD, on days 1 through 3) instead. However, due to a worsening of performance status, the third-line option of temozolomide (300 mg, qd) and sunitinib (37.5 mg, qd) was carried out.
Patient Follow-Up
The mean follow-up time was 36.7 months (range: 1–85). Three patients had recurrent locoregional cervical nodal disease (cases 1, 3, and 4) shortly after the surgeries. The time to recurrence was 5, 6, and 11 months (cases 1, 3, and 4). Subsequent revisional bilateral salvage neck dissection was performed. Two out of 7, 1 out of 5, and 8 out of 38 lymph nodes from level II–V of cases 1, 3, and 4 were identified to be positive, separately. However, unfortunately these 3 patients further developed distant metastasis to the spine (n = 2), lung (n = 1), liver (n = 1), spleen (n = 1), or mediastinal septum (n = 1), which led to the eventual demise of patients 1 and 3. Tumor stage on presentation was also a strong predictor of prognosis. Those who presented with a stage I or II NEC had significantly better prognosis than those with stage IVa disease. The 3 patients associated with lymphadenopathy all died; in contrast, 2 out of 3 patients without lymph node disease were still alive at the last follow-up. The prognosis was also strongly dependent on the histological subtype, as all 3 SCNEC patients expired.
Discussion
A Revised Stratification for the Laryngeal NENs Is Crucial
The latest 5th edition of the head and neck WHO tumor bluebook is undoubtably a big step toward a uniform stratification framework for the NENs across various organ systems based on the heterogenic high throughput genetic data.6,7 The International Agency for Research on Cancer and WHO suggest a consensus proposal named “NEN-WHO 2018,” in which one of the salient key points is the distinction between well-differentiated NETs and aggressive poorly differentiated NECs, despite the anatomic location. 8 A most recent microRNA study focused on laryngeal NECs evidenced a peculiar signature likely related to laryngeal poorly differentiated NEC. 9 In line with the proposed universal nomenclature, former well-/moderately differentiated NECs termed in the 4th edition are now recognized as NETs (graded as G1, G2, and G3 by proliferation) in the 5th edition, while laryngeal LCNEC and SCNEC are still termed as NECs. 10 NETs have site-specific incidence and prevalence. Compared to the alimentary tract, the laryngeal NET G3 has never been reported, and pulmonary NET G3 is under study with mounting evidence. 11
LCNEC Is a Distinct Poorly Differentiated NEC Subset and Has Unfavorable Clinical Course
Accurate categorization and grading are of paramount importance for the laryngeal NENs. 12 LCNEC, previously assigned to moderately differentiated NEC but behave differently, has been reclassified in the 4th edition of WHO spectrum as a NEC subtype. LCNEC can be differentiated from NET and SCNEC for its high mitotic rate and its large-sized polygonal cells with coarse salt-and-pepper chromatin and prominent nucleoli, separately. The nucleoli of SCNEC are inconspicuous or absent, and the chromatin is finely granular.13,14 A considerable portion of LCNEC patients had a predilection for advanced stage. Ninety percent of laryngeal LCNEC whilst 66.7% of laryngeal SCNEC patients presented with stage IV disease.13,15 For laryngeal LCNEC, the 3-year overall survival was 11%. 15 In contrast, laryngeal SCNEC has a five-year OS rate of 7.7%. 16 The locoregional lymph node metastasis rate of laryngeal NECs varies. In a recently reported Indian cohort, the lymph-node metastasis was detected in 85.7% of NEC patients. 17 In another earlier report, 87.5% laryngeal LCNEC and 58.9% laryngeal SCNEC patients present with metastasis nodal disease.18,19 About 70% LCNEC and 80% SCNEC of the larynx developed distant metastasis. Approximately 81% of LCNEC and 58% of SCNEC patients develop a recurrence.13,20
LCNEC Patients Appeared to Benefit the Most From Upfront Surgery
With regard to treatment modalities, the available evidence points to chemoradiotherapy as the preferred therapeutic option for laryngeal NECs. It offers universally limited benefit in terms of the locoregional relapse, distant metastasis, and long-term overall survival, highlighting the insufficiency of nonsurgical management.21,22 However, there is limited evidence about the clinical rationale or efficacy of upfront surgery as a preponderant first-line therapy for resectable laryngeal NECs.23,24 In the present cohort of patients, upfront surgery rather than chemoradiotherapy was the first-step care for LCNEC, and it achieved the likelihood of improved survival in patients presented with resectable late-stage laryngeal LCNEC in the best-case scenario. Despite the small size of our cohort making it prone to biases and impossible to give meaningful conclusions, this study, corroborated by other previous studies, underscores that surgical management with subsequent chemoradiotherapy could be beneficial for locoregionally advanced LCNEC, which have a high propensity to metastasize. In some highly selected cases, minimally invasive approaches, such as transoral CO2 laser surgery may be performed.
Conclusion
In summary, laryngeal NENs are a diverse group of oncological entities whose prognosis was dismal. Given the challenges of contradicting sparse reports by different institutions over a protracted timeline and various diagnostic/therapeutic modalities, a more concrete approach is the next best step. The infrequency of laryngeal NEN cases makes case series valuable. For future appraisal of homogenous data, consistent adoption of this 2022 WHO consensus for cogently stratifying patients to delineate the ideal clinical management is strongly recommended. In this scenario, further pooled real-world data will enable us to determine optimal treatment strategies and to develop appropriate, analogous, and coherent treatment principles, as no single institution will have a large enough group for a randomized controlled trial to alleviate the concerns.
Footnotes
Acknowledgements
We thank Ling Nie (a pathologist) and Haijian Fan (a radiologist) for assistance and helpful discussions.
Author Contributions
Study concept and initial design: X.Q.: study design and statistical analysis: X.Q., Y.G., and D.L.: acquisition of data and data analysis and interpretation: D.L., Y.G., and H.Z.: all authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
This study was approved by the Institutional Review Board of Affiliated Drum Tower Hospital of Nanjing University Medical School and conducted in accordance with the Declaration of Helsinki.
Informed Consent
Informed consent was waived given the retrospective nature of this study.
