Abstract
Conductive hearing loss is a common clinical condition caused by disorders of the middle and external ear. Here, we report a case of conductive hearing loss with complete tympanic membranes. Clinical examinations revealed no external and middle ear lesions, but high-resolution temporal bone computed tomography revealed enlarged vestibular aqueducts. Detailed audiological tests and follow-up results indicate that the exclusive air–bone gap is the outcome of inner ear malformation but not middle ear lesions, preventing the need for tympanic assessment.
Introduction
Conductive hearing loss is frequently associated with middle or outer ear disease, easily diagnosed and distinguished by the severe tympanic membrane or external auditory canal pathology. A limited number of patients with intact tympanic membranes and no abnormal symptoms of conductive deafness were misdiagnosed as having otosclerosis or other middle ear diseases, resulting in stapes surgery or ossiculoplasty with no improvement in hearing.1,2 Schuknecht 3 relates the inexplicable air–bone gap (ABG) to inner ear pathology, hypothesizing that conductive hearing loss in these individuals has a pathological third window in the inner ear. Recent research literature describes inner ear third window lesions anatomically, with superior semicircular canal syndrome (SSCD) being the most frequent, followed by vestibular aqueduct enlargement. This work presents a case of a large vestibular aqueduct that exclusively presented with conductive hearing loss.
Case Report
A 39-year-old female patient presented with a 3-year history of right hearing loss and no history of head trauma or otitis media. Ear endoscopy revealed bilateral normal external auditory canals and intact tympanic membranes (Figure 1A and B). Tuning fork test: Rinne’s exam was negative on the right side and positive on the left side; Weber’s tuning fork examination lateralized to the patient’s right ear. Gelle’s test was positive on the right ear. The audiology test showed an ABG of 30 to 40 dB on the right side, whereas the hearing threshold on the left side was normal (Figure 1C and D). Tympanometry revealed an As tympanogram bilaterally and a partially evoked stapedius reflex in the right ear (Figure 1E to H). Wideband acoustic impedance testing showed a resonance frequency of 815 Hz on the right and 1116 Hz on the left, with normal energy absorption rates bilaterally. The vestibular system of the patient was normal, there was no Tullio phenomenon, and the Valsalva maneuver did not induce vertigo or nystagmus. The temporal bone high-resolution computed tomography (HRCT) revealed an enlarged right vestibular aqueduct (Figure 2).
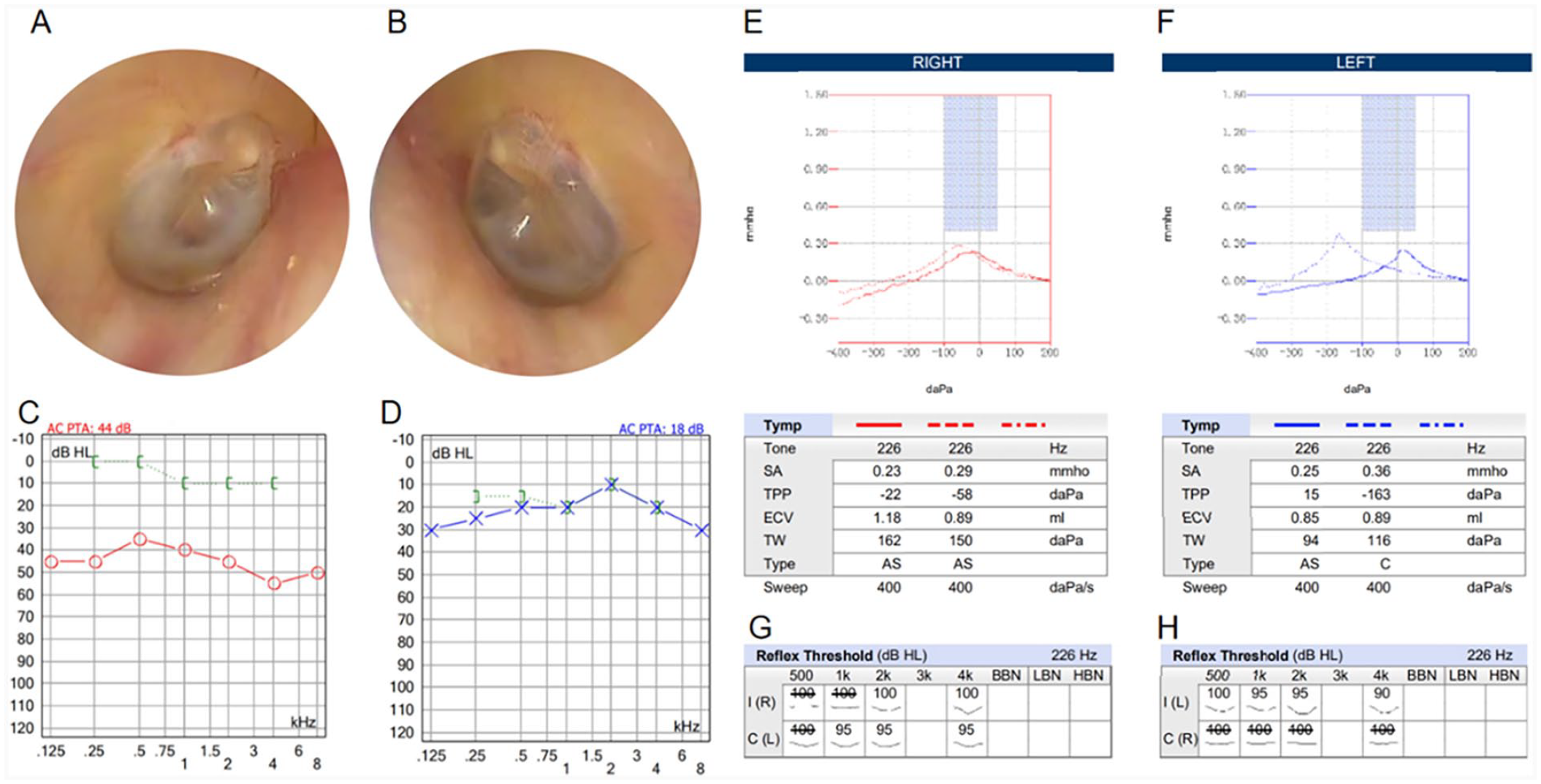
Ear endoscopy images. Bilateral external auditory canals are clear; the tympanic membranes are intact and marked. Right ear (A); left ear (B). (C) and (D): Pure tone audiometry test. The right audiometry shows conductive hearing loss with an air–bone gap of 30 to 40 dB (C); and a normal left audiometric threshold (D). (E) to (H): Acoustic immittance test. The bilateral tympanic curve is of As type (E–F); the ipsilateral stapedial reflex (100 dB at 2 and 4 kHz) and the contralateral stapedius reflex (95 dB at 1, 2, and 4 kHz) are present in the right ear. The ipsilateral stapedial reflex (100 dB at 500 Hz, 95 dB at 1 and 2 kHz, and 90 dB at 4 kHz) is present, while the contralateral stapedius reflex is absent in the left ear (G–H).
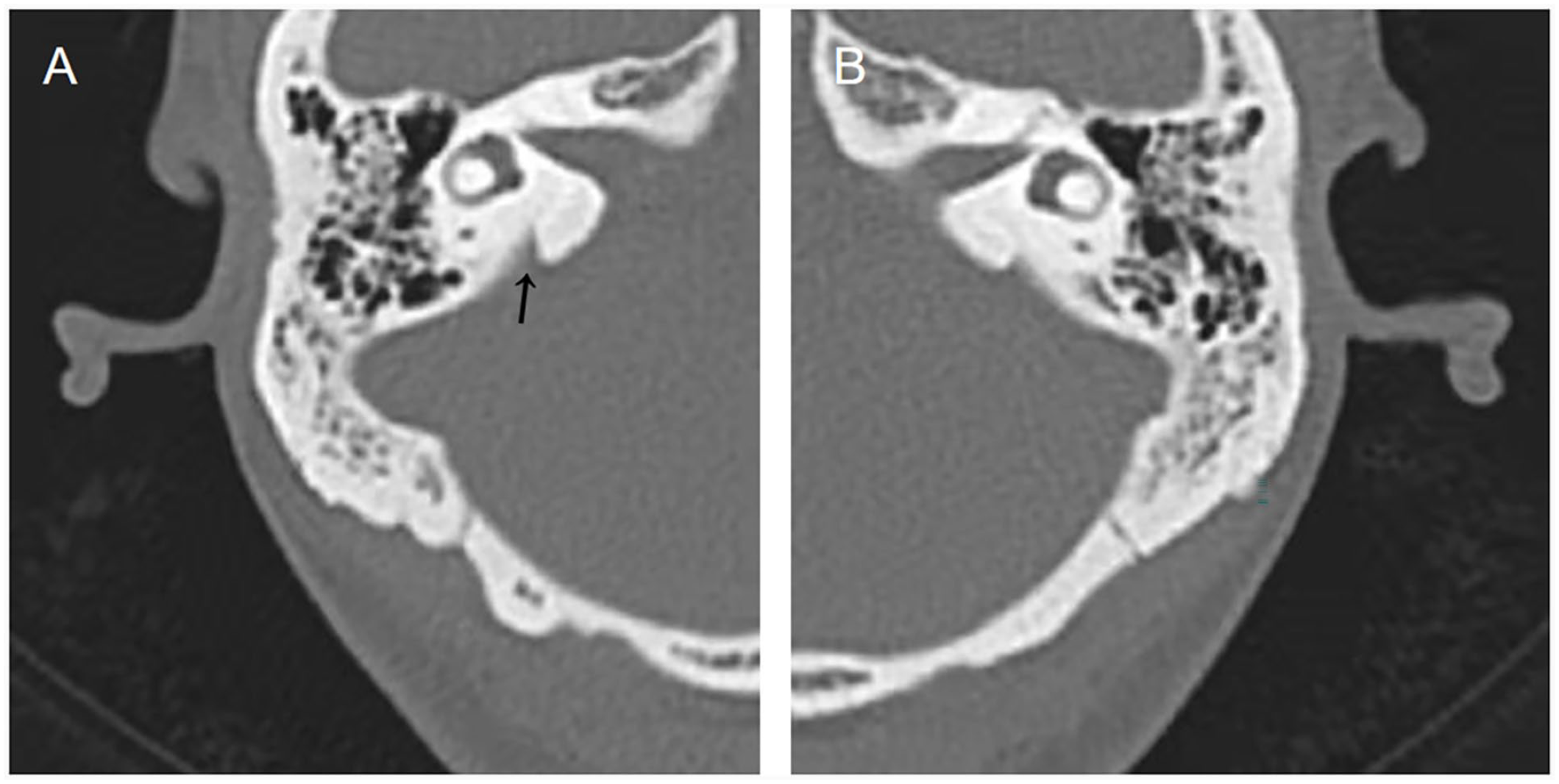
Temporal bone HRCT shows no hyperintensity in the tympanic sinus, tympanic cavity, or mastoid process; the mastoid process is pneumatized bilaterally. Enlarged vestibular aqueduct on the right side (A, arrow). No significant enlargement on the left (B). HRCT, high-resolution computed tomography.
The definite diagnosis was an enlarged vestibular aqueduct (EVA) with right-sided conductive hearing loss. Tympanic exploration or stapedectomy was not recommended. The patient was advised to have regular hearing evaluations to monitor hearing changes, avoid strenuous physical activities and head trauma.
After 3 months, the patient was re-retested for pure tone audiometry, which revealed no significant hearing change in the right ear.
Discussion
Otosclerosis is frequently the primary differential diagnosis in clinical practice for conductive hearing loss with an intact tympanic membrane following the exclusion of middle ear infections. The diagnosis is mainly based on the patient’s medical history and specialized audiological examinations. Otosclerosis patients typically have intact tympanic membranes and well-functioning eustachian tubes but test negative for the Gelle’s test due to stabilization of the footplate of the stapes bone. The stapedius muscle reflex gradually ceases to activate as the condition advances.
In contrast to otosclerosis, patients with inner ear conductive hearing loss frequently elicit acoustic reflexes due to normal middle ear conduction. 4 Wideband acoustic immittance measurement of sound energy absorption rate guides clinical differential diagnosis. Research evidence indicates that otosclerosis patients have a slightly lower sound energy absorption rate between 400 and 1000 Hz, within the normal range. 5 However, patients with larger vestibular aqueducts have sound energy absorption rates near or over the upper limit of the normal range at 400 to 1000 Hz. 6 In addition, monitoring the resonant frequency (RF) with multiple-frequency tympanometry can be beneficial. 5 Patients with otosclerosis have higher RF values because of the stiffness of the middle ear system. 7 On the contrary, patients with EVA have lower RF values than those with otosclerosis. The EVA decreases the mechanical impedance of the inner ear, lowering the resonance frequency of the entire system. 8 The resonance frequency in the present case was lower in the affected right ear compared with the healthy left side (815 vs 1116 Hz), which contradicts the clinical picture of otosclerosis.
Concisely, most frequencies in the right ear elicited acoustic reflexes in this patient, and Gelle’s test was positive. The pure tone threshold curve lacked a Carhart notch, and the middle ear resonance frequency was lower on the afflicted side. The diagnosis of otosclerosis was then ruled out. The patient did not report any symptoms of Tullio’s phenomenon, and the Valsalva maneuver did not cause substantial vertigo or nystagmus. Moreover, the normal vestibular function was not consistent with significant SSCD. A detailed examination of the HRCT scan of the patient’s temporal bone revealed an enlargement of the right vestibular aqueduct, which led to the clinical diagnosis.
Clinical manifestations of large vestibular aqueduct syndrome (LVAS) frequently include mixed hearing loss detected by pure tone audiometry, with an ABG in the low frequencies and severe hearing loss in the high frequencies. 9 In many cases, hearing loss rapidly progresses to severe or profound sensorineural hearing loss, making it impossible to assess bone conduction thresholds and concealing the ABG. Therefore, patients with isolated conductive hearing loss caused by vestibular aqueduct enlargement are extremely uncommon.2,10
The mechanism of the conductive component in LVAS hearing is yet unknown. Scientists have different hypotheses. Merchant and Rosowski 10 suggest that the “third window phenomenon” in the inner ear can explain the low-frequency ABG in LVAS. The EVA, which acts as a third window, changes the compliance of the system, resulting in acoustic energy entering the vestibule via stapes motion being deflected away from the cochlea. Furthermore, the changes allow for an enhancement in bone conduction; several patients show supranormal bone conduction responses. In the present case, pure tone audiometry revealed a better bone conduction hearing threshold on the right side than on the left, reaching 0 to 10 dB. A recent investigation on making predictions based on radiologic findings using an otobiomechanical approach discovered no significant correlation between the radiologic measures and the observed ABG, implying that the pathological third window effect is not the sole factor determining CHL in LVAS. 11 Valvassori and Clemis 12 hypothesized that the conductive component of the LAVS hearing loss could be explained by the increased pressure of the refluxed perilymphatic and endolymphatic fluid, which results in decreased stapes mobility. However, Nakashima et al 13 discovered that acoustic reflexes persist in cases of EVA, which contradicts stapes fixation, and suggests that ABG seen in some cases is caused by ossicular discontinuity as the findings of a temporal bone study demonstrated a 38% incidence of ossicular deformities in EVA cases. 14
Patients with stapedial otosclerosis can benefit from stapedectomy surgery. However, surgical assessment of patients with third window lesions must be done with prudence. Tavora-Vieira and Miller 15 described a case of severe sensorineural hearing loss following stapedectomy due to a misdiagnosed EVA as stapedial otosclerosis. Furthermore, as the cerebrospinal fluid (CSF) is directly connected to the cochlea vestibule, stapedectomy for patients with EVA may result in “perilymphatic gusher,” 16 which increases the risk of CSF otorhinorrhea and intracranial infection if not addressed carefully. Audiological tests and temporal bone HRCT in the present case indicated that the source of straight conductive hearing loss was an EVA rather than otosclerosis; as such, surgical exploration was not recommended.
Conclusion
In addition to otosclerosis, pathological third window alterations in the inner ear should be addressed when evaluating conductive deafness with an intact tympanic membrane. The mechanics of middle and inner ear pathology leading to conductive deafness must be elucidated through accurate audiological and vestibular function testing and a targeted reading of temporal bone HRCT to achieve an appropriate differential diagnosis. Unnecessary tympanic exploration surgery can be avoided with these measures.
Footnotes
Data Availability
The data used to support the results of this study have received hospital ethical approval for use.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Statement
Written informed consent was obtained from the patient (s) for their anonymized information to be published in this article.
