Abstract
Cochlear implants (CIs) are effective interventions for treating severe-to-profound sensorineural hearing loss. In patients with complex inner ear anatomy due to conditions such as incomplete partition type II (IP-II) and enlarged vestibular aqueduct, they present unique challenges, increasing the risk of surgical and postoperative complications, including facial nerve stimulation (FNS), cerebrospinal fluid leaks, and vestibular dysfunction. The 32 year-old subject of this case study presented with IP-II and experienced persistent FNS, dizziness, and declining CI performance despite an initial revision surgery and appropriate programming strategies of her processor. Interdisciplinary collaboration between audiology, neurotology, and speech-language pathology helped to identify and address contributing factors to the patient’s symptoms. After strategic programming adjustments, including pulse width modification and electrode management in conjunction with aural rehabilitation sessions, the patient’s speech perception scores and CI quality of life metrics improved markedly over time, as did her confidence and engagement in social and professional settings. This case highlights a challenging example of CI performance issues in the setting of IP-II and aims to examine how CI programming, in response to anatomical and surgical difficulties, affects patient outcomes in cases involving inner ear malformations.
Introduction
Cochlear implants (CIs) are effective interventions for treating severe-to-profound sensorineural hearing loss (SNHL). While most implanted patients have progressive SNHL and normal temporal bone anatomy, CI can be considered for other patients with uncommon causes of hearing loss and anatomical abnormalities. Before pursuing implantation for these patients, several factors should be considered to ensure a safe and effective surgery. Treatment strategies tailored to the individual patient can help improve patient outcomes and overall quality of life throughout their CI journey.
Inner ear malformations such as enlarged vestibular aqueduct (EVA) and incomplete partition type II (IP-II) can result in symptoms including severe hearing loss, dizziness, and vertigo. IP-II is characterized by a deficient modiolus, absent osseous spiral lamina, and absent interscalar septum. These findings, in combination with an EVA, have classically been known as the Mondini malformation. Compared to those with normal anatomy, those with inner ear malformations, including IP-II, may face an increased risk of surgical complications. These can include cerebrospinal fluid (CSF) leakage, perilymphatic gushers, meningitis, and facial nerve injury or stimulation. 1 Facial nerve stimulation (FNS) occurs when the current stimulating the internal electrodes spreads to the facial nerve, causing slight-to-severe facial spasms and discomfort. Factors that increase the likelihood of FNS include malformation, bony dyscrasia (such as otosclerosis), or trauma to the inner ear structures. 2 Although these malformations can complicate CI surgery and programming, with proper care team planning, many patients can obtain successful outcomes.3,4
When CI performance declines in cases with complex inner ear malformations, it can be challenging to distinguish potential causes. Declines could result from anatomical issues, programming errors, or even device dysfunction, which, in rare instances, may include a soft failure. This occurs when there are signs of device failure that are not indicated on manufacturer integrity tests, but may include a decline in performance or other adverse symptoms. These symptoms may improve or resolve after surgical re-implantation. 5 Currently, this is the only way to confirm a soft failure diagnosis. An additional surgery is a challenge for any patient, but for those with inner ear malformations, it can lead to further complications due to the complex anatomy of their inner ear. Due to this, all possible causes of device performance decline should be considered, as symptoms of inner ear malformations can present as a soft failure. Patients with typical anatomy who experience soft failure and undergo re-implantation often regain the same or improved function of their device. 6 Patients with inner ear malformations can be prone to postoperative complications, and secondary surgery should be avoided unless necessary.
In patients with complex inner ear anatomy who experience issues with CI performance, several variables beyond implant function should be considered. A physical shift of the implant, changes in CSF such as leakage and gushers, and the possibility of device failure must all be considered. An interdisciplinary team-based approach, consisting of otologists, radiologists, speech-language pathologists (SLPs), and audiologists, is essential for identifying challenges and achieving optimal patient outcomes. This case highlights a challenging example of CI performance issues in the setting of IP-II and aims to examine how CI programming, in response to anatomical and surgical difficulties, affects patient outcomes in cases involving inner ear malformations.
Case Study
The subject of this case study is a 32 year-old female with a history of asymmetric SNHL and IP-II. She was initially diagnosed with congenital hearing loss as a child and had been a long-time hearing aid user. Following a progressive decline in audiometric thresholds and aided word recognition, she received a left CI (CI522), Slim Straight (lateral wall electrode), from Cochlear Ltd (Sydney, NSW, Australia) at the age of 23.
The device provided satisfactory sound awareness, but she struggled with speech perception outcomes. The utilization of aural rehabilitation and home speech exercises yielded no additional benefit. Performance declined for two years, and ultimately, an integrity test indicated that the device was functioning correctly according to manufacturer specifications. A subsequent X-ray was ordered, revealing a partial insertion of the electrode array.
At this time, revision surgery was performed with implantation of a CI512 Contour Advance, perimodiolar electrode. Performance following the second activation proved significantly improved compared to the first. However, less than a year following the procedure, the patient began experiencing significant involuntary FNS. The audiologist at the time investigated the electrodes and found 5 of the 22 were causing FNS. Prolonged episodes of vertigo also accompanied this, lasting several hours daily. The offending electrodes were subsequently deactivated, which reallocated the frequencies associated with each band. Following the change, FNS decreased significantly, as did the vertigo symptoms.
Balance symptoms dissipated and remained stable until months later, when an abrupt recurrence of vertiginous symptoms occurred. A CT scan indicated she had air within the cochlea stemming from an apparent perilymphatic fistula. The patient agreed with the recommendation for a surgical repair involving packing of the left round and oval window. Following the procedure, she recovered well and saw a reduction in her symptoms. She had no further symptoms for ~6 months.
The patient presented to the clinic for a second opinion with audiology and neurotology. She expressed concern with CI function and decreased speech perception performance over the previous 6 months. Along with reduced performance, episodic FNS, and occasional imbalance were present. Again, a manufacturer integrity test was completed and returned normal device function. She also expressed concerns with not being able to hear and communicate effectively at work and in social situations. The inability to listen properly with her CI and hearing had led her to be less engaged with workplace communication. A reported gradual decrease in CI performance began following a bout of COVID-19, which led to the recurrence of FNS and dizziness. The patient perceived the decrease as a significant hindrance to interpersonal communication, thinking she exhibited symptoms of a soft failure following her research. 5
Audiometric testing was performed using the patient’s Kanso 2 sound processor at a 0° azimuth and 1 m distance from the speaker. The contralateral ear with hearing loss in the severe-to-profound range was plugged. Soundfield audiometry revealed a pure tone average of 30 dB HL. Initial speech perception results revealed AzBio Sentence scores of 81% in the quiet condition, 39% at a +10 signal-to-noise ratio (SNR), 44% with consonant-nucleus-consonant (CNC) words, and 62% on CNC phonemes. This indicates a mild decline as compared to previous records that revealed performance of 90% in the quiet condition. With the reported decline being <10% on speech testing, the recommendation of soft failure could not be made.
Initial inspection of the patient’s daily program revealed a wide dynamic range across the array. A group of 6 deactivated electrodes was located towards the basal end. The most basal, 22, remained activated despite the inactive electrodes, 16 to 21. The large gap could create pitch mismatches, as the programmed bands are no longer tonotopically aligned throughout the cochlea. 7 The patient reported a previous audiologist had decided to disable the electrodes as each of them caused severe facial stimulation, which could not be rectified during that programming session.
Electrically evoked compound action potentials (eCAPs) were measured and found to be slightly above upper comfort levels. With eCAPs typically falling between behavioral threshold and upper comfort levels, this finding indicates a mild understimulation in her programming. 8 The pulse width was increased from 25 to 50 µs. No other programming changes were made during this first session, and the resulting MAP is depicted in Figure 1. This approach enabled an overall reduction in the electric current required while maintaining equal audibility and potentially reducing the occurrence of FNS triggered by higher levels. For simplicity, it was therefore decided to let the patient leave with a MAP to investigate 1 significant variable at a time.
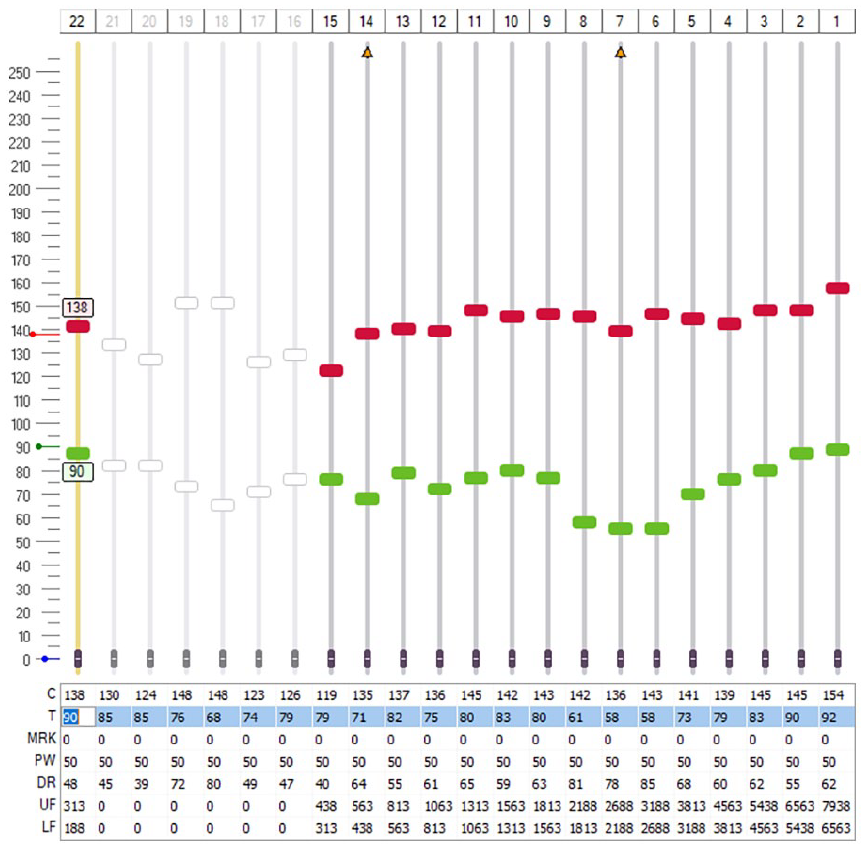
The adjusted programming MAP after the initial visit, following the extension of the pulse width and lowering of stimulation levels. Electrodes 16 to 21 can be seen to be deactivated.
Care was then taken to individually reactivate and investigate the disabled electrodes to determine if any one of them was the cause of the reported FNS. Each of the tested electrodes elicited FNS, and no manipulation of parameters or frequency allocation could eliminate the resulting symptoms. Therefore, the electrodes remained disabled. Electrode 22 remained active to prevent additional mismatch and compression on the remaining electrodes. 9 She was encouraged to use the programming for 2 weeks, keep a journal detailing the presence of any FNS and or balance issues, and return for an evaluation.
The patient’s neurologist speculated that the CI itself might be contributing to her vestibular symptoms. To determine if the symptoms originated from a peripheral or central cause, she did not wear the sound processor for 2 weeks, and an magnetic resonance imaging (MRI) of the brain was ordered. A 3 T strength MRI was requested, and due to the incompatibility of the internal implant, the internal magnet was surgically removed under anesthesia and then replaced after the scan was completed. The MRI ultimately was nonrevealing. After the procedure, 6 weeks passed before she could resume regular processor wear due to discomfort and swelling, and then attend her next audiology appointment.
At this appointment, the patient reported no change in the severity of vestibular symptoms. However, she did note a significant reduction in FNS. Since resuming regular device usage following the MRI, the patient indicated a perceived decrease in the volume of her processor. This resulted in difficulty with everyday communication. Impedances were unchanged from the previous visit Figure 2. Soundfield audiometry revealed worsened thresholds up to an additional 15 dB HL. Speech perception revealed a significant decrease with AzBio Sentence scores of 66% in the quiet condition, 26% with CNC words, and 40.66% on CNC phonemes. Threshold programming levels were significantly increased by varying amounts to improve poor soundfield thresholds, perception of soft sounds, and increase overall intelligibility. The patient was to report back for further testing the following month.
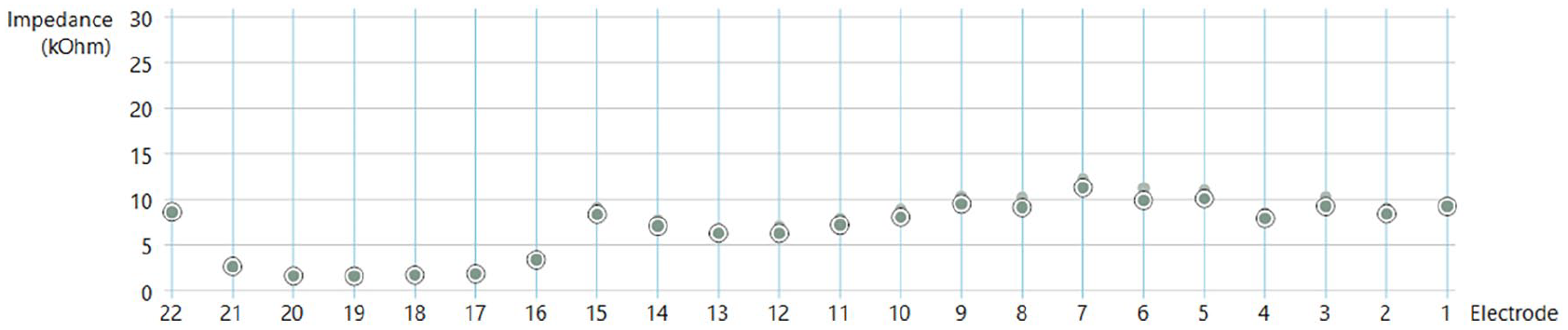
Impedance levels at the start of the second visit. The smaller, filled-in circles from the previous visit can be seen above many of the current electrode levels.
The patient returned for her most recent appointment following resuming consistent, everyday use and reported increased speech understanding, the complete absence of all FNS, and the resolution of vestibular symptoms. Speech perception showed a significant increase, with AzBio Sentence scores of 82% using CI alone and 99% bimodally in the quiet condition. She was now satisfied with her CI outcome, and additional programming changes were unnecessary. Audiometric testing of the contralateral ear completed at this appointment revealed it to be a candidate for a CI. However, the patient remains hesitant due to her experience of complications with the first ear.
Throughout the case, she simultaneously attended coordinated visits with speech-language pathology on the same day as her audiology appointments. The patient completed a thorough aural rehabilitation process, including listening therapy. The process allowed her to reassociate sounds with cognitive meaning, as well as guide CI programming efforts to ensure access to the spectrum of speech frequencies. Additionally, the SLP worked with the patient on advocacy and communication skills to help her better navigate both social and professional interactions. These visits provided her with the confidence to engage more at work and speak up for herself in times of miscommunication.
From the initial presentation to the completion of her latest visit, the scores on her CI Quality of Life-35 assessment questionnaires at her 1 year follow-up appointment indicated a significant improvement. She continues to thrive in the bimodal condition, citing both work and social activities have been drastically improved. The scores from the beginning, middle, and end of the case are shown in Figure 3.
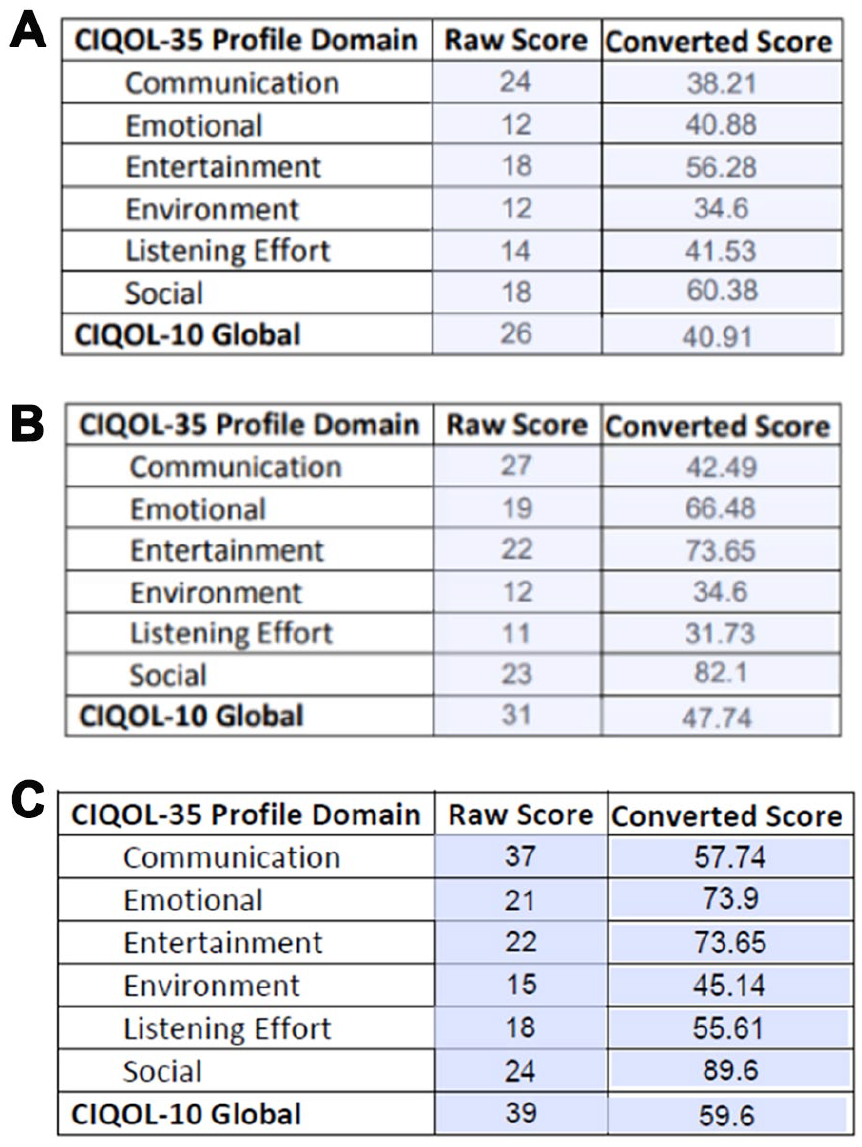
The raw and converted scores of the QOL-35 questionnaire at (A) initial presentation, (B) after intervention, and (C) at the most recent visit. QOL-35, CI Quality of Life-35.
Discussion
The overall goal of this case study is to highlight the consideration of a patient’s anatomy when making changes in CI programming. Utilizing an interdisciplinary team-based approach can significantly enhance patient outcomes. Compared to the patient’s status when they first presented, significant improvements have been achieved in their current performance.
Inner ear malformations such as IP-II can impact hearing outcomes post-CI surgery. Structural and functional changes occur in both the cochlea and proximal vestibular structures as a result of implantation surgery due to their proximity. 10 Patients with inner ear malformations can perform worse than their peers with normal anatomy.11,12 An investigation on the audiological performance of patients with inner ear malformations was conducted by Ozkan et al, 13 which revealed that favorable outcomes increased when the cochlear nerve was visible on MRI. This emphasizes the need for imaging and a care team that has an in-depth understanding of the anatomical complexities to create realistic expectations regarding the patient’s capacity for improvement after surgery.
For this patient, contributing factors such as vestibular symptoms, FNS, and overall hearing ability significantly affected her quality of life. The nature of CI surgery can lead to vestibular disorders post-implantation. 10 However, there is great variability in likelihood that patients will be affected due to variables such as their etiology of hearing loss, surgical technique, intraoperative trauma, and premorbid vestibular function, to name a few. 14 Programming measures, such as strategically disabling electrodes and altering pulse width, can be taken to mitigate FNS to improve function.12,15 Perimodiolar electrodes typically have a lower occurrence of FNS compared to lateral wall electrodes.16,17 This is usually a result of requiring less electrical stimulation due to their proximity to the basilar membrane. However, programming the CI with excessively high current levels or leaving electrodes out of compliance can lead to FNS, regardless of the type of electrode used. Both increasing pulse width and simultaneously lowering her current stimulation levels likely resolved her symptoms. 2 Surgical interventions should only be considered if other adjustments do not elicit benefit. 12 Utilizing a specialized team to determine the cause of any symptoms persisting postsurgery is vital to avoid unnecessary procedures. Extending the pulse width in cases such as this can be an option to reduce the necessary electrical current, thereby providing patients with audibility and reducing the prevalence of FNS.
Investigative measures should be taken before deactivating electrodes. Each electrode deactivated changes the frequency allocation of the remaining electrodes. Removing too many can result in pitch mismatches and increased signal compression, both of which can result in poorer speech perception outcomes.7,9 However, the removal of negatively affected electrodes can aid in reducing FNS or inaudible electrical current if electrodes have migrated out of the cochlea. 12
This case study reviews a patient who required a comprehensive team-based approach to achieve her best performance with a CI. The importance of auditory rehabilitation throughout this process cannot be overlooked in rebuilding this patient’s confidence in device use. Speech-Language Pathologists are integral to the CI process. Following visits with the SLP team, the patient was able to advocate for herself in the workplace and improve her quality of life. This experience led to the current consideration of implanting her right ear. Since at one point, she was unsure of any possible improvement with her CI this is quite meaningful. With continued progress, we have tremendous confidence in continued beneficial outcomes for both the patient and others with similar stories.
Footnotes
Acknowledgements
The content of this case study is solely the authors’ responsibility and does not necessarily reflect the official view of the University of Chicago Medicine or Rush University Medical Center.
Ethical Considerations
As this is a case study, it was deemed exempt from the Institutional Review Board process at the Rush University Medical Center.
Consent to Participate
Written consent was obtained from the subject of this case study, which allowed for its submission.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Information supporting this case study is available from the corresponding author, JS, on request.
