Abstract
Schwannomas account for a large proportion of tumors in the head and neck regions; however, schwannomas of the larynx are rare. An 11-year-old boy had a sore throat for one month, and his symptoms gradually worsened such that he needed to visit our otolaryngology clinic. The preoperative investigation revealed a smooth mass in the left arytenoid cartilage. Endoscopy-assisted transoral resection of laryngeal masses was performed under general anesthesia, and the resected tissue was histopathologically diagnosed as a laryngeal schwannoma. The postoperative recovery was very good. During the 1-year follow-up, there was no recurrence of the schwannoma or related symptoms. Although laryngeal schwannomas are rare, they should be considered in the differential diagnosis of these tumors. Sufficient preoperative imaging should be performed before surgical resection, and surgery is the preferred treatment option.
Introduction
Schwannomas, also called neurilemmomas, are neurogenic tumors. Schwann cells that originate from the peripheral nervous system can form benign encapsulated tumors.1,2 Approximately 25%–45% of schwannomas are located in the head and neck, mostly in the parapharyngeal spaces. Laryngeal schwannomas are very rare, accounting for 0.1%–1.5% of all benign laryngeal tumors.3,4 Here, we describe a case of laryngeal schwannoma in an 11-year-old boy and provide a review of relevant literature reports.
Case Report
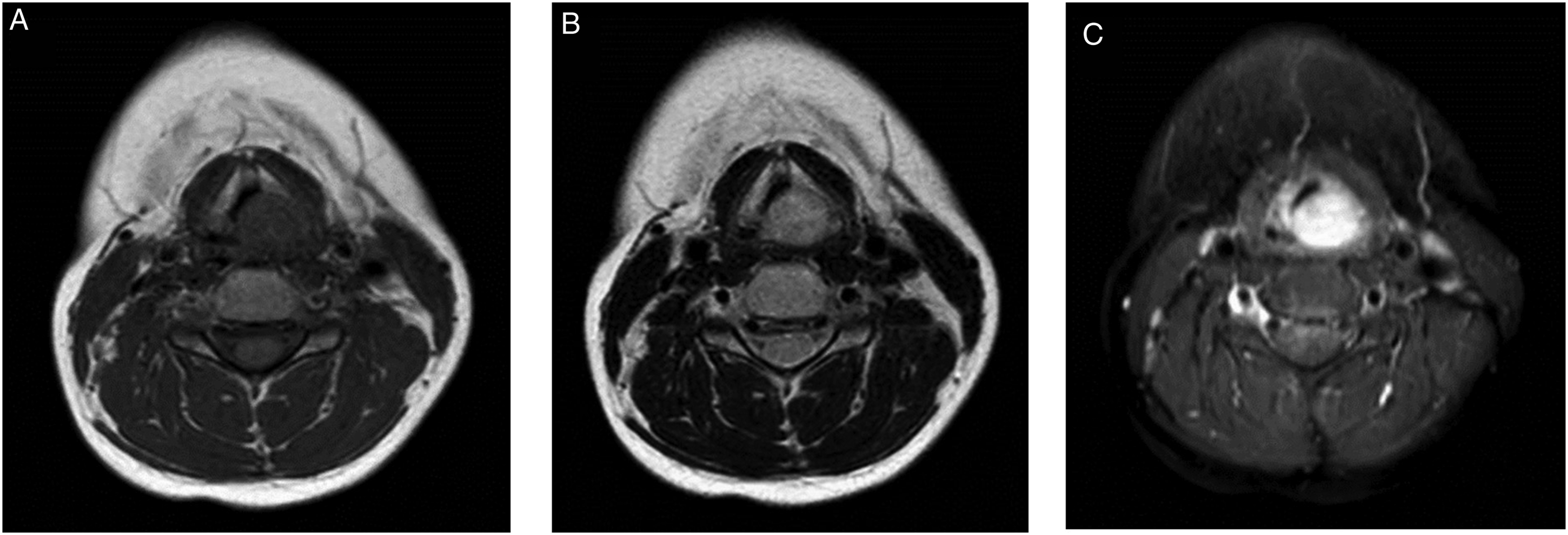
An 11-year-old boy presented to the otolaryngology department with a sore throat that had persisted for one month and gradually aggravated symptoms. Laryngoscopy showed a left arytenoid submucosal mass with a pseudomembrane on the surface. His vocal cord movement was normal (Figure 1A), and the other head and neck examinations revealed no abnormalities. Magnetic resonance imaging (MRI) revealed a soft tissue mass measuring 20 mm × 16 mm × 15 mm in the neck, with a nodular mass in the left aryepiglottic fold that compressed the airway. MRI provides superior soft tissue images, and neurilemmomas show different intensities on T1-weighted images (Figure 2A) and high homogeneous or heterogeneous intensities on T2-weighted images (Figure 2B), which usually enhance after gadolinium injection (Figure 2C). The patient underwent laryngeal microsurgery with a plasma knife to remove the lesion. Laryngoscope adopted 0-degree Hopkins, and an intraoperative photograph is shown in Figure 1B. The histopathology confirmed that the schwannoma was present in 2 different areas: Antoni A and Antoni B. The Antoni A area contained spindle-shaped cells (Figure 1C), and immunohistochemical detection of the S100 protein was positive (Figure 1D). The MRI showed no obstruction or abnormal density in the laryngeal cavity 3 months postoperatively. No recurrence of laryngeal schwannoma was found during the 1-year follow-up. (A) Laryngoscopy showed a submucosal mass in the left arytenoid cartilage, with partial occlusion of the glottic opening. (B) Intraoperative photograph. (C) Hematoxylin-eosin stain (×200 magnification). (D) Positive stain for S-100 (×50 magnification). Cervical MRI. The lesion is well delineated and its topography submucosal, with T1-weighted (A) and T2-weighted (B) isointensity. After contrast injection there is homogeneous enhancement, with no signs of infiltration (C).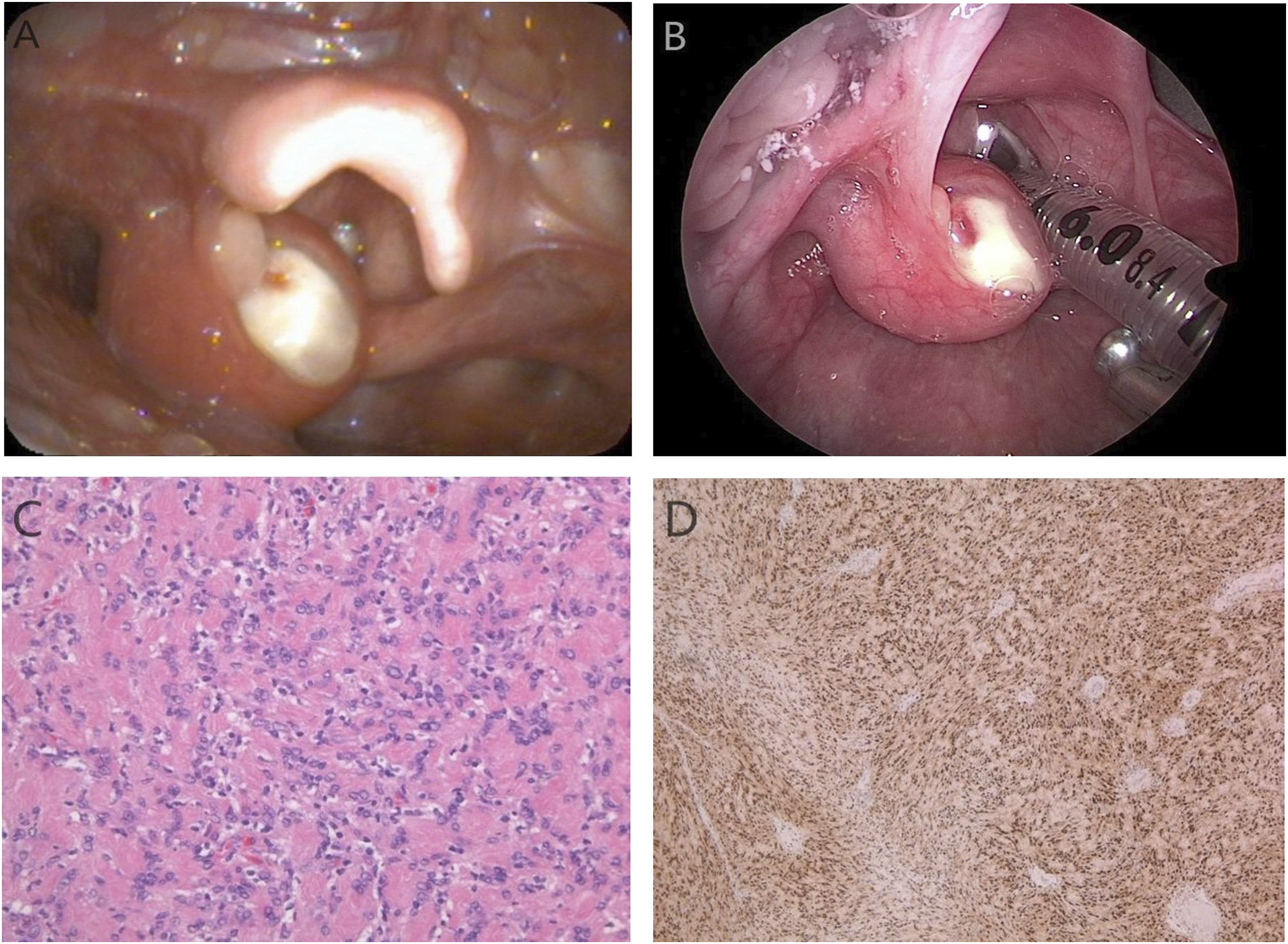
Discussion
Laryngeal neurogenic tumors include neurofibromas and schwannomas, and laryngeal schwannomas are very rare and were first reported in 1925. 5 Laryngeal schwannomas accounted for 0.1%–1.5% of all benign laryngeal tumors. The internal branch of the superior laryngeal nerve is considered to be the origin of supraglottic Schwannoma. 4 They are usually solitary, slow-growing, 6 encapsulated tumors that grow eccentrically away from the nerve trunk. The arytenoid epiglottic fold and arytenoid cartilage are the most common anatomical sites. 7 The peak age of onset is between 25 and 50 years, and the tumors are more common in women. 6 The common symptoms include hoarseness, dysphonia, sore throat, sore swallowing, dysphagia, and abnormal sensations in the throat. 1 Dyspnea may occur when the tumor blocks the glottis, and in the initial stage, most patients are asymptomatic.8,9
Clinical manifestations of laryngeal schwannomas closely relate to the size and location of the mass. If the mass is small and its location is not critical, patients are usually asymptomatic. Patients may present non-specific symptoms such as abnormal sensation, dysphagia, and hoarseness. Hoarseness may be caused by a decline in the motor ability of vocal cords. If the schwannoma originates from the vocal cords, the vocal cords function is often impaired owing to the influence of the tumor mass. Patients with severe conditions may have dyspnea or even symptoms of acute airway obstruction, which can be life-threatening. 10
The imaging findings of schwannomas and neurofibromas are similar, and the 2 tumors are usually indistinguishable. On Computed Tomography (CT), schwannomas present similar findings to muscles with low density and contrast enhancement. 11 On MRI T1-weighted images, schwannomas have moderate signal intensity similar to muscle, while on T2-weighted images, they showed high signal intensity.11,12 They are enhanced strongly, and most show low-strength edges consistent with the capsule. Fusiform and target signs can be seen on T2-weighted images and on enhanced images of schwannomas and neurofibromas. Masses relative to the nerves on ultrasound, CT, or MRI suggest the possibility of schwannomas, and heterogeneity with cystic degeneration is common.11,13
Histopathology is the gold standard for the diagnosis of schwannomas. In 1988, Enzinger and Weiss proposed 3 histological criteria for the diagnosis of schwannoma: (1) Antoni A and B regions, (2) encapsulation, and (3) S-100 protein positivity. 14 The Antoni A region is characterized by the tight arrangement of spindle-shaped Schwann cells and an arrangement of nuclei in the palisade to form Verocay bodies. There were few cells in the B region of Antoni and fusiform Schwann cells in the mucoid matrix.
Complete surgical resection is the first choice of treatment for laryngeal schwannoma. Schwannomas are radiation-resistant and therefore do not respond to radiotherapy. 15
Various surgical methods have been reported in the literature. Excision depends on the location and size of the tumor. 16 Regardless of the surgical method used, surgeons must completely remove the tumor to prevent recurrence. In addition, efforts should be made to maintain the integrity of the mucosa around the tumor and to retain normal laryngeal function. In the present patient, the tumor originated from the left arytenoid cartilage. We prefer to use endoscopy-assisted plasma knife resection, which provides satisfactory hemostasis, compared with cold excision.
Conclusion
Laryngeal schwannomas are extremely rare, slow-growing, and benign tumors that present with multiple symptoms. The overall prognosis for laryngeal schwannomas is good, and surgical resection remains the preferred treatment. We believe that the best surgical approach can be determined based on the size, location, and extent of the lesion. According to this case report, endoscopic plasma knife resection of laryngeal schwannoma provides a safe and minimally invasive method to completely remove the lesion. A long-term follow-up is required to ensure that there are no signs of recurrence.
Footnotes
Declaration of Conflicting Interests
The authors(s) declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclose receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Beijing Hospitals Authority’s Ascent Plan (grant number: DFL20191201) and the Respiratory Research Project of National Clinical Research Center for Respiratory Diseases (grant number: HXZX-20210201).
Ethical Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Verbal informed consent was obtained from the patient for anonymized information to be published in this article.
Data Availability
Data and materials can be obtained from the corresponding authors upon reasonable request.
