Abstract
Cribriform-morular thyroid carcinoma (CM-TC) is a rare entity that usually occurs in association with familial adenomatous polyposis (FAP) but may be sporadic. Herein, we present a new case of cribrifo
Introduction
Cribriform-morular thyroid carcinoma (CM-TC) is an uncomm
Herein, we report a new case of CM-TC occurring sporadically in a 28-year-old-woman with no history of FAP.
Case report
A 28-year-old-woman presented to the outpatient department with neck swelling. She complained of dysphagia, dyspnea
On gross examination, the right lobe revealed an encapsulated firm tan-white tumor measuring 55 × 50 mm whereas the left lobe showed a fleshy well-circumscribed nodule of 33 × 30 mm.
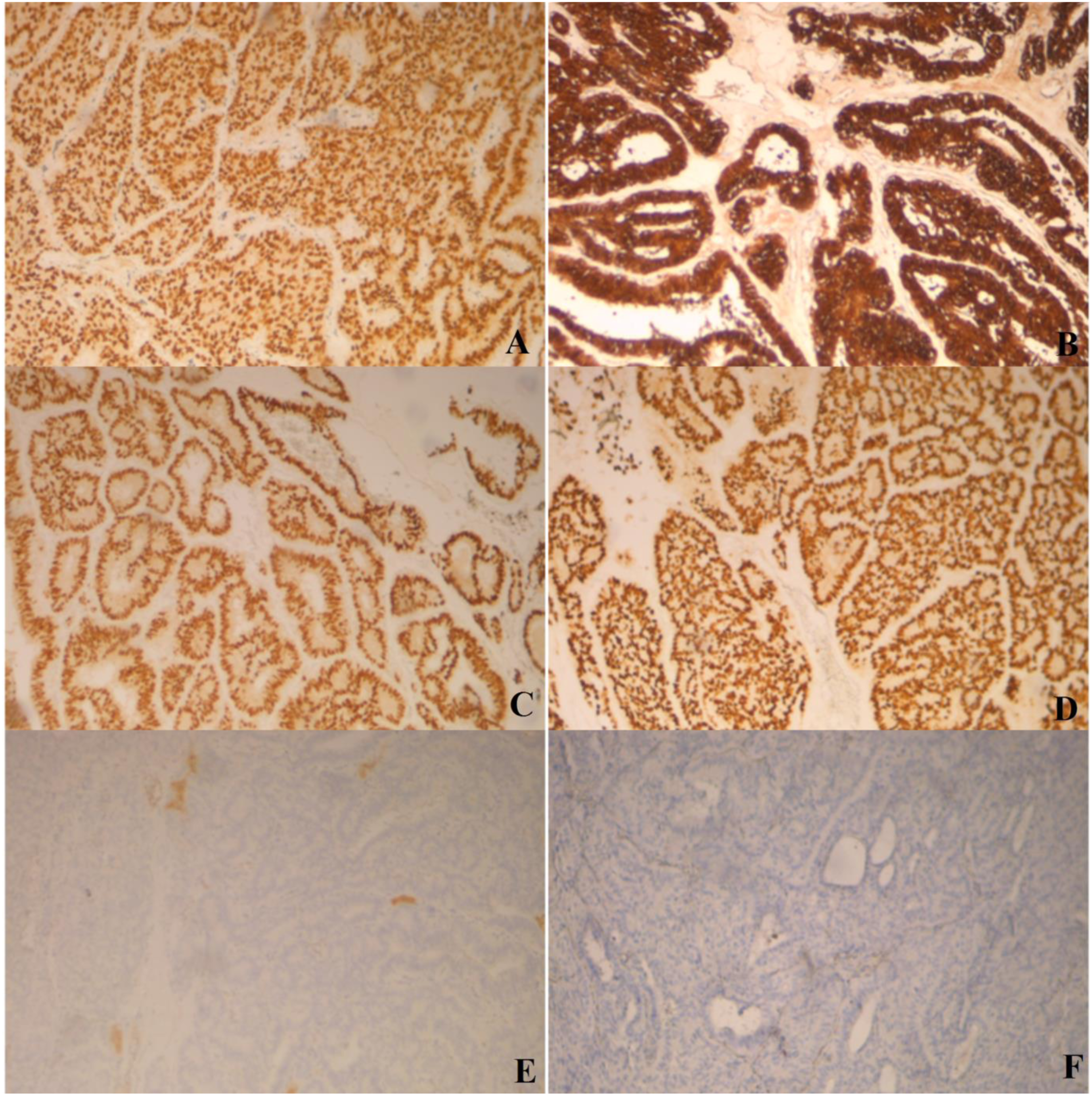
Histology of the right thyroid nodule showed an encapsulated tumor with glandular, cribriform, solid (A) Tumor histology with cribriform pattern of growth (H&E stain, X200). (B) Glandular structures (H&E stain, X100). (C) Tumor histology with papillary architecture (H&E stain, X100). (D) Solid architecture of the tumor (H&E stain, X200). (E) Solid architecture of the tumor (H&E stain, X400). (F) Tumor cells were columnar with nuclear grooves (H&E stain, X400). The results of immunohistochemistry analysis. (A) Diffuse and strong staining with TTF1 (X100). (B) Tumor cells showed nuclear and cytoplasmic expression of β-catenin (X100). (C) Diffuse and strong staining with estrogen receptors (X100). (D) Diffuse and strong immunoreactivity with progesterone receptors (X100). (E) Staining with thyroglobulin was negative (X100). (F) CDX2 negativity is illustrated (X100).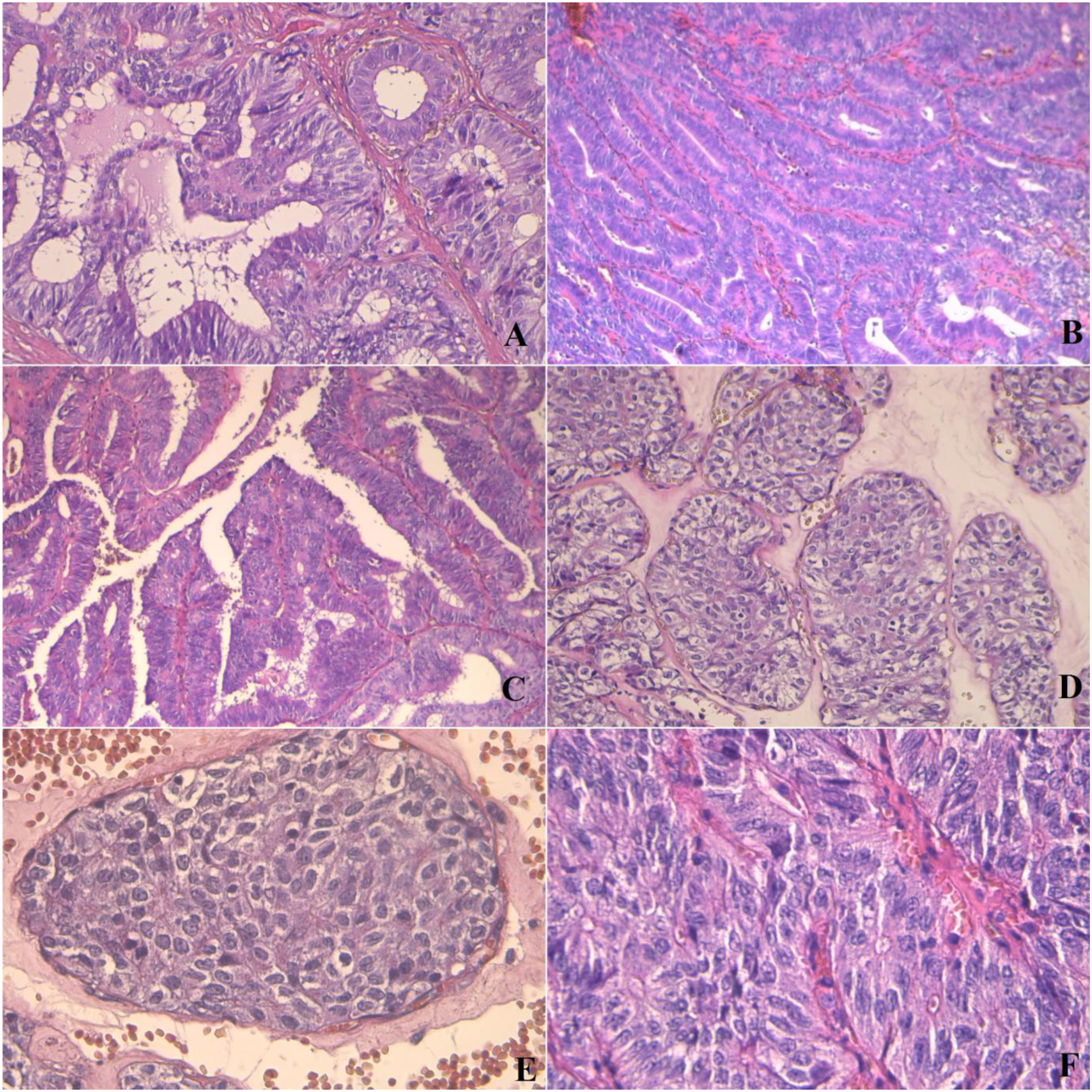
The left thyroid nodule corresponded to an encapsulated follicular variant of PTC. In addition, there were two foci of papillary microcarcinoma of 2 and 1.5 mm involving the left thyroid lobe.
The remaining thyroid parenchyma displayed lymphocytic thyroiditis. Moreover, five perithyroid lymph nodes were negative for malignancy but revealed granulomatous inflammation with slight fibrinoid necrosis (Figure 3). The admission course was uneventful. The patient was discharged on the third postoperative day. She was prescribed with thyroxine supplement and subsequently underwent adjuvant radioactive iodine therapy. Colonoscopy was performed 10 days after surgery and showed no polyposis. However, screening for Adenomatous Polyposis Coli (APC) gene mutation Non-caseating granuloma in perithyroid lymph node (H&E stain, X100).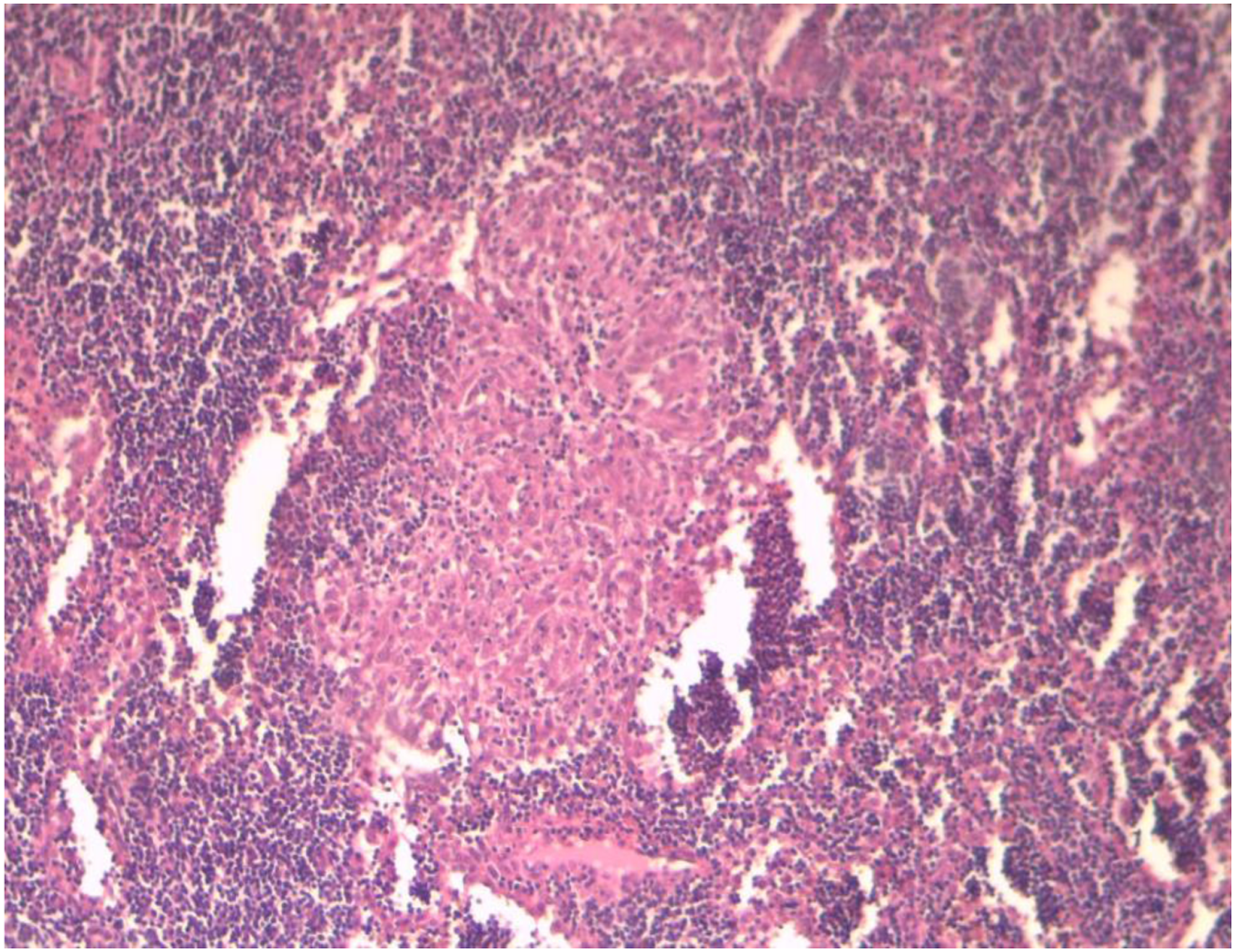
At present, after 11 months of follow-up
Discussion
Cribriform-morular thyroid carcinoma (CM-TC) is an extremely rare form of malignancy. 2 In 1994, Harach and coworkers first recognized the neoplasm as a unique and distinct variant of papillary thyroid carcinoma occurring in the setting of familial adenomatous polyposis (FAP). 3 Later in 1999, Cameselle-Teijerio and Chan reviewed four cases with similar morphology occurring sporadically in patients without evidence of FAP. 4
FAP is a rare genetic disorder characterized by the development of hundreds to thousands of adenomas in the colon and rectum.
5
The disease is inherited in an autosomal dominant manner.
6
It is the result of germline mutation
Giving the fact that up to 90% of FAP-associated thyroid carcinoma have cribriform-morular histology, total colonoscopy and APC gene analysis are recommended once CM-TC is diagnosed.9,10 Similarly, patients with FAP should be regularly screened for thyroid nodules. 10 Interestingly, it is reported that CM-TC could be diagnosed 5 years before FAP manifestations. 11 As a result, a normal colonoscopy cannot rule out a familial form with certainty.
Additionally, sporadic CM-TC can harbor somatic mutation
In contrast to follicular cell-derived thyroid carcinoma, CM-TC does not harbor BRAF V600E mutations and only rarely displays RAS or PIK3CA mutations 1 .
CM-TC affects almost exclusively young females. 2 Clinically, it is usually presented as a neck mass discovered incidentally on palpation or on imaging. 11
Cytologic features of CM-TC on fine needle aspiration include cribriform architecture, morules, nuclear clearing, papillary architecture, spindle cells, hyaline materials, foamy or hemosiderin-laden histiocytes, and the absence of colloid in the background. 13
On gross examination, most tumors are well-circumscribed or encapsulated, tan to white, solid and fleshy with no necrosis or hemorrhage. There is no lobe predilection.14,15 In FAP, tumors are frequently multifocal, whereas sporadic cases appear usually as a solitary nodule. 16
Histologically, CM-TC is usually an encapsulated tumor displaying a variety of architectural patterns
Morules are frequently observed and are considerably suggestive of CM-TC. They consist of small cell nests displaying peculiar biotin-rich clear nuclei and resembling mulberries.6,11 These epithelial nests can resemble squamous metaplasia in follicular cell-derived thyroid neoplasms. 19 However, morules in CM-TC do not represent squamous metaplastic epithelium since they do not exhibit keratinization or intercellular bridges and they stain negatively for p63 and p40.6,19 Moreover, the morular cells are strongly positive for CD10, bcl-2, and E-cadherin. 11 These cells may express both CK5 and CD5, raising the hypothesis of thymic/ultimobranchial pouch-related differentiation. 19
Non-morular cancer cells exhibit immunoreactivity for CK7, CK19, vimentin, bcl2, thyroid transcription factor-1(TTF1), P53, galectin-3, estrogen, and progesterone receptors.6,20 Immunoreactivity for thyroglobulin and HBME-1 is variable and can be either focal or negative. 11
β-catenin is strongly expressed in both cytoplasm and nucleus of morular and non-morular tumor cells. By contrast, the normal thyroid parenchyma shows membranous staining pattern. The diffuse expression of β-catenin represents the hallmark of this malignancy in both FAP-associated and sporadic cases.7,11
Other thyroid carcinomas should be considered in the differential diagnosis of CM-TC, namely: (1) Diffuse sclerosing variant of PTC shows squamous metaplasia, which can mimic morules in CM-TC. However, diffuse sclerosing variant exhibits dense sclerosis with patchy to dense lymphocytic inflammation, abundant psammoma bodies, and extensive lymphovascular invasion.
21
(2) Columnar cell variant of PTC is associated with a fatal course. Histologically, it is composed of thin papillae lined by pseudostratified epithelium. By contrast to CM-TC, columnar cell variant lacks the conventional nuclear features of PTC and is devoid of squamous morules.21,22 Besides, the neoplastic cells may show supranuclear and subnuclear vacuolization and they are positive for thyroglobulin. Nuclear expression for β-catenin is absent.18,21
(3) Tall cell variant of PTC is composed of cells that are two to three times as tall as they are wide with distinct cell borders, abundant oncocytic-like cytoplasm, and exaggerated papillary nuclear features. Follicles are stretched and elongated with «tram-track» appearance.
21
Surgical treatment depends on whether it is FAP-associated or sporadic CM-TC. In this regard, lobectomy is recommended for sporadic cases, while total thyroidectomy is recommended for familial cases. However, extensive lymph nodes dissection is not necessary. 16
In general, CM-TC is considered as an indolent neoplasm with a favorable outcome regardless of whether it is FAP-associated or sporadic.2,17
Recurrence rate is overall low and disease-related mortality is uncommon. 11 However, a few reports in the literature described an aggressive biologic behavior. A case of CM-TC that metastasized to the lung and brain had been reported. 18 Another case of sporadic CM-TC with bone metastasis had been described in association with a telomerase reverse transcriptase (TERT) promoter mutation. 23
Conclusion
CM-TC is a rare entity having unique clinical, pathological
Footnotes
Author contributions
All authors made a substantial contribution to the acquisition, analysis and interpretation of data in addition to writing-review &editing. All authors approved the final version.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Our institution does not require ethical approval for reporting individual cases since we anonymously reported clinical and imaging information concerning our patient’s case.
Informed consent
A written informed consent was obtained from the patient to publish this paper.
Data availability
The data that support the findings of this work are available from the corresponding author upon reasonable request.
