Abstract
Objective
Patients with advanced nasopharyngeal carcinoma (ANC) often experience chronic pain. Opioids are generally recommended to treat tumor-related pain, but increased opioid use may lead to detrimental aftereffects, particularly with respect to tumor progression, resulting in reduced quality of life and increased risk of death. Our objective was to investigate whether the high size of opioid prescriptions is associated with poor overall survival (OS) in patients with ANC.
Methods
A consecutive cohort of patients with newly diagnosed ANC who underwent high or low opioid prescription size treatment during 2012–2019 was retrospectively identified from our medical institutions. Survival was estimated with the Kaplan–Meier method with a log-rank test. Multivariate binary logistic regression was used to assess the association between opioid use and OS, adjusting for age, sex, body mass index (BMI), Eastern Collaborative Oncology Group performance status (ECOG PS), and ANC histology. The criterion to distinguish between the high opioid prescription size group [HD] and the low opioid prescription size group [LD] was 5 mg of oral morphine equivalents (OME) per 24 hours.
Results
The cohort consisted of 244 consecutive patients (HD: n = 120, median age = 66 years [range, 40–81 years]; LD: n = 124, median age = 65 years [40–82 years]. Patients who underwent treatment with a high opioid prescription size had a worse median OS than those who underwent treatment with a low opioid prescription size (5.1 vs 6.6 months), and the high opioid prescription size was associated with a remarkable 48% higher risk of death than the low opioid prescription size (HR 1.48, 95% CI 1.11–1.98; P = .005). The cumulative dose of opioids greater than or equal to 500 mg of OME was associated with a higher risk of death, adjusted for age, sex, BMI, ECOG PS, and ANC histology.
Conclusions
In patients with newly diagnosed ANC experiencing palliative care, a high opioid prescription size may be associated with shorter OS than a low opioid prescription size.
Introduction
Pain has been associated with early mortality in patients with advanced nasopharyngeal carcinoma (ANC). 1,2 Opioids are safe and effective for tumor-related pain and have been the mainstay of analgesia in these patients. 3,4 However, opioids can be associated with neurotoxicity, nociception, and poor survival benefits. 5,6 The early combination of palliative care and opioid use can play a key role in preventing irregular pain episodes in patients diagnosed with ANC. 6 However, opioid overdose in ANC patients can result in respiratory depression and pain-induced immunosuppression, which can be associated with an increase in ANC-related mortality. 7 Opioid-related deaths reported each year continue to increase, with more than half of deaths linked to high opioid prescriptions. However, it remains uncertain whether high opioid prescription treatment results in poor overall survival (OS) in ANC patients. 8-10 Although opioids do not appear to accelerate death in sanatorium settings, they may have unique antineoplastic activity and have an impact on OS. 11 However, recently a randomized trial 12 of a humanized mouse model demonstrated that morphine, one of the most commonly used opioids, provides analgesia in sickle mice without affecting survival. Inconsistently, other studies 13-15 of a humanized mouse model showed that chronic morphine promotes tumor progression associated with cross-activated signaling pathways and affects survival.
Previous studies 16-18 have shown that the high size of the opioid prescription is independently associated with poor OS in individuals with other malignant solid tumors. However, it is not clear whether the independent association remains in patients with ANC. Although opioids may have a negative effect on survival attributable to promoting tumor cell growth, it is equally possible that increased opioid use associated with disease progression poses a great threat to OS. 11,17 To date, accessible studies on whether there is a survival benefit for ANC patients undergoing opioid treatment remain lacking. Furthermore, data on the association of opioid overdose with OS in patients with ANC remain unclear. Therefore, we launched a retrospective study to assess survival outcomes associated with opioid use in the treatment of chronic pain related to ANC and to investigate whether the high size of the opioid prescription is associated with poor OS in patients with ANC.
Patients and Methods
Clinical Data
Ethical approvals were obtained from Wuhan Fourth Hospital; Tongji Medical College, Huazhong University of Science and Technology (Grant number: 179323). A consecutive cohort of newly diagnosed ANC patients who underwent high or low opioid prescription size treatment from January 2012 to January 2019 was retrospectively identified from our medical institutions. The inclusion criteria used in this study included patients with clinically and histopathologically confirmed ANC; patients who continued high or low opioid prescription size treatment until death from any cause; and Eastern Collaborative Oncology Group performance status (ECOG PS) of 0-2. Key exclusion criteria included unclear opioid intake; unknown tumor stage; incomplete follow-up information; unclear time to diagnose ANC; unclear baseline mu-opioid receptor (MOP-R) score; 19 nonpalliative treatment of previously diagnosed ANC (immunotherapy, targeted therapy, combination therapy, chemoradiotherapy, and even limited surgical interventions); use of coadjuvant drugs similar to opioids; and discontinuation of opioid use.
Study Design and Management
A retrospective multicentre study was implemented in which patients with newly diagnosed ANC had received continuous high or low opioid prescription size treatment. The criterion to distinguish between high and low opioid prescription sizes was 5 mg oral morphine equivalents (OME) per 24 hours. OME ≥ 5 mg OME per 24 hours was considered in the high opioid prescription size group (HD). Less than 5 mg of OME per 24 hours was considered in the low opioid prescription size group (LD). The calculation of opioid usage was performed as previously reported. 16 Prescriptions for opioids were documented using the prescription drug monitoring program in China. Opioid prescriptions varied as a function of pain severity. All prescribed opioids used in patients with ANC were available in this study. The OME was determined on the basis of an equianalgesic conversion table as presented in previous studies. 16 Duplicate prescriptions were excluded. The development of a medication plan and the recording of adverse drug reactions and drug cycles were decided by high-volume physicians at their respective medical institutions. The procedure for opioid treatment was consistent with a previous study. 16
Outcomes and Assessments
The primary outcome measure was OS defined from the introduction of initial opioids to death from any cause. Primary analysis was carried out using multivariate binary logistic regression, adjusted for age, sex, body mass index (BMI), ECOG PS, and ANC histology. Baseline pain was assessed according to a previous study. 19 Follow-up occurred every month until death or the end of the study.
Statistical Analysis
We conducted a descriptive analysis to assess the characteristics of both groups of patients. Continuous demographic variables recorded as means and SD or median were compared with the Mann–Whitney U tests. Categorical demographic variables recorded as frequency tables were compared with a chi-square test. Survival was estimated using the Kaplan–Meier method with a log-rank test. Multivariate binary logistic regression was applied to evaluate the association between opioid use and OS, adjusting for age (40 ≤, <60 and 60 ≤, <90), gender (female and male), BMI, ECOG PS (0, 1, and 2), and ANC histology (undifferentiated, poorly differentiated, and unclear). A 2-tailed P < .05 was considered statistical significance. The analyses were implemented with SAS 9.6 (Inc., Cary, NC) or GraphPad Prism 9.0 (La Jolla, California).
Results
Demographic Characteristics
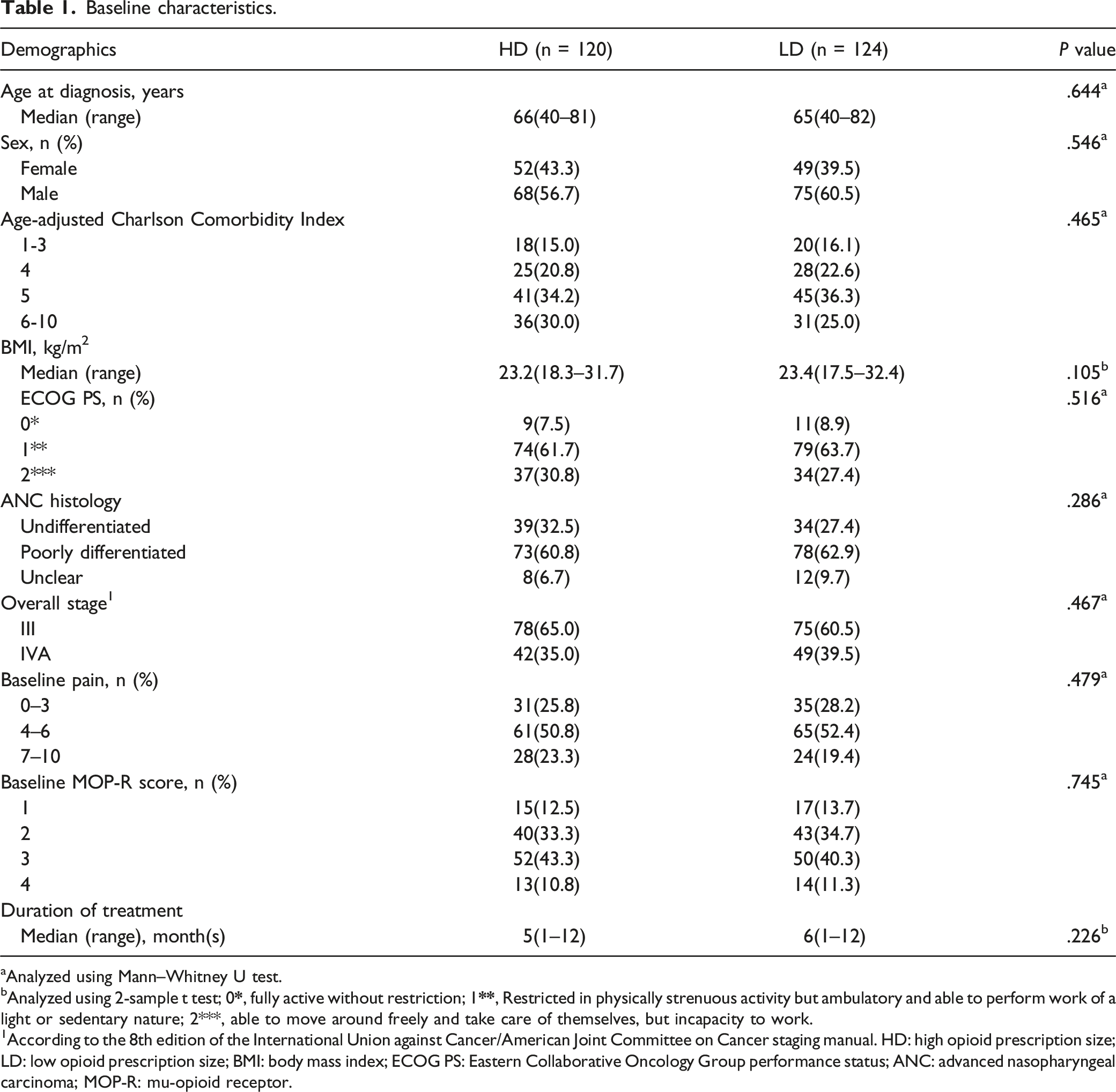
In total, 316 consecutive patients with newly diagnosed ANC who experienced palliative care were evaluated for availability. Seventy-two (22.7%) individuals were excluded according to the present exclusion criteria, leaving 244 individuals for the endpoint analysis, as detailed in Figure 1 (HD: n = 120, median age = 66 years [range, 40–81]; LD: n = 124, median age = 65 years [40–82]). In both groups, the number of males predominates (56.7% with HD vs 60.5% with LD). The ECOG PS was 0 in 7.5%, 1 in 30.8%, and 2 in 61.7% with HD vs 0 in 8.9%, 1 in 27.4%, and 2 in 63.7% with LD (P = .516). ANC histology was undifferentiated in 32.5%, poorly differentiated in 60.8%, and unclear in 6.7% with HD vs undifferentiated in 27.4%, poorly differentiated in 62.9% and unclear in 9.7% with LD (P = .286). Baseline pain was 0-3 in 25.8%, 4–6 in 50.8%, and 7–10 in 23.3% with HD vs 0–3 in 28.2%, 4–6 in 52.4%, and 7–10 in 19.4% with LD (P = .479). The baseline MOP-R score was 1 in 12.5%, 2 in 33.3%, 3 in 43.3%, and 4 in 10.8% with HD vs 1 in 13.7%, 2 in 34.7%, 3 in 40.3%, and 4 in 11.3% with LD (P = .745). The median duration of the drugs was 5 months (range 1–12) with HD and 6 months (1–12) with LD (P = .226). The demographic characteristics of the individuals included in the present study were well balanced between both groups, as detailed in Table 1. Flow diagram showing the methods used to identify patients to assess survival outcomes associated with opioid use in the treatment of chronic pain related to ANC. Baseline characteristics.
aAnalyzed using Mann–Whitney U test.
bAnalyzed using 2-sample t test; 0
1According to the 8th edition of the International Union against Cancer/American Joint Committee on Cancer staging manual. HD: high opioid prescription size; LD: low opioid prescription size; BMI: body mass index; ECOG PS: Eastern Collaborative Oncology Group performance status; ANC: advanced nasopharyngeal carcinoma; MOP-R: mu-opioid receptor.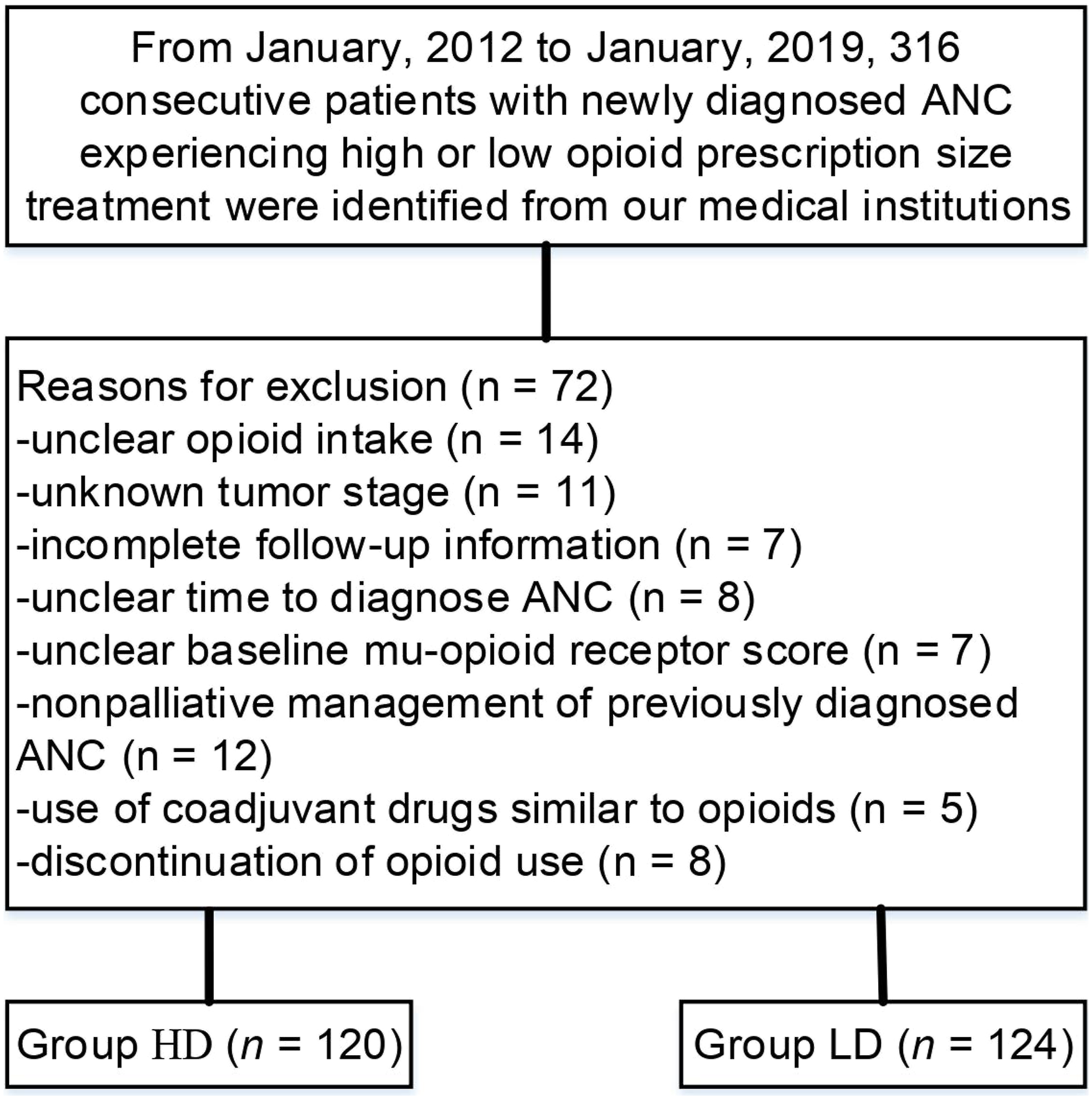
Overall Survival
At a median follow-up of 8 months (range 1–12 months), 189 deaths were detected (77.0% [188/244]; 99 patients [40.5%] with HD vs 89 patients [36.5%] with LD). For patients who received high opioid prescription size treatment, 74 deaths (61.7%) were attributed to disease progression, and 25 (20.8%) were not; for patients who received low opioid prescription size treatment, 65 deaths (52.4%) were attributed to disease progression, and 24 (19.4%) were not. A notable discrepancy was detected in the median OS between groups (5.1 months [95% CI, 4.7–6.4] for HD vs 6.6 months [95% CI, 5.4–7.2] for LD). Patients who underwent high opioid prescriptions had a worse median OS than those who underwent low opioid prescriptions, and patients who underwent high opioid prescriptions were associated with a remarkable 48% higher risk of death than those underwent low opioid prescriptions (HR 1.48, 95% CI 1.11–1.98; P = .005), as presented in Figure 2. A notable distinction of nearly 1.5 months was observed in the median OS, and the differences between groups tended to be significant because both survival curves remained separated until the final follow-up. Kaplan–Meier curves for overall survival. The median overall survival was 5.1 months (95% confidence interval [CI], 4.7–6.4) for HD and 6.6 months (95% CI, 5.4–7.2) for LD (HR 1.48, 95% CI 1.11–1.98; P = .005). *The hazard ratio was calculated using a Cox proportional hazards model, with age, sex, BMI, ECOG PS, and ANC histology used as covariates and intervention provided as time-dependent factor.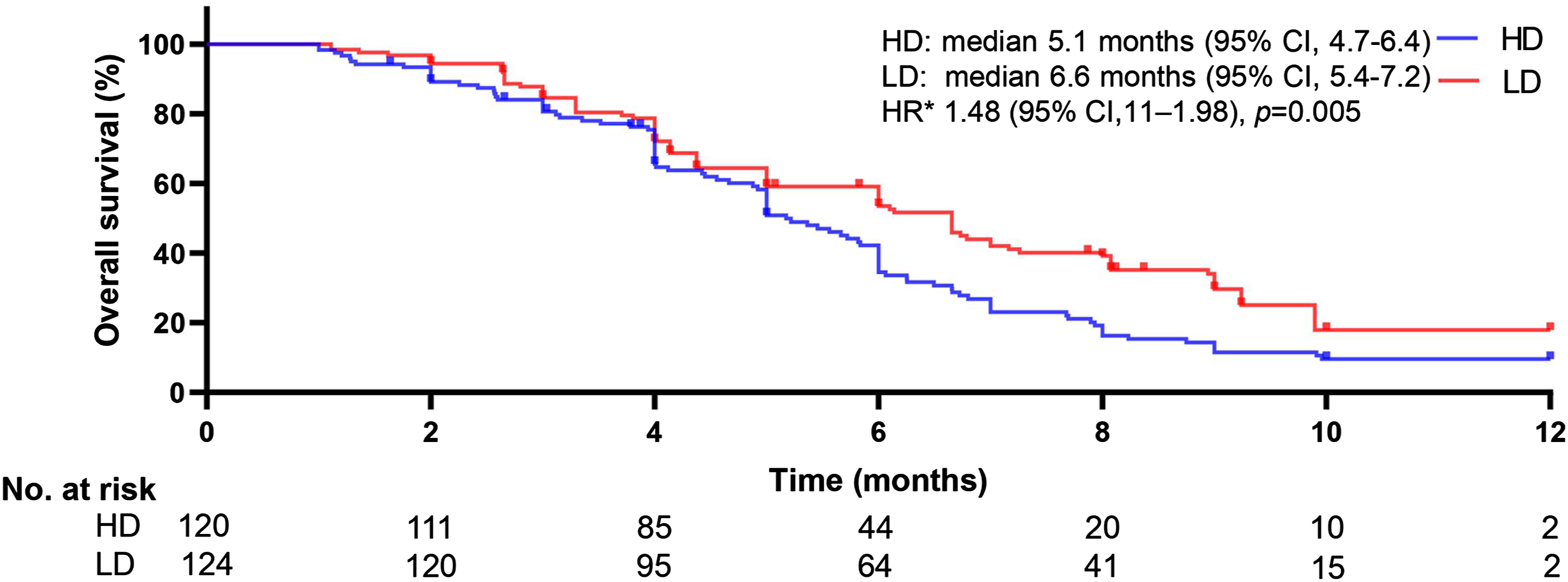
Multivariate Binary Logistic Regression Analyses
Multivariate binary logistic regression analysis of factors associated with OS in patients with newly diagnosed ANC.
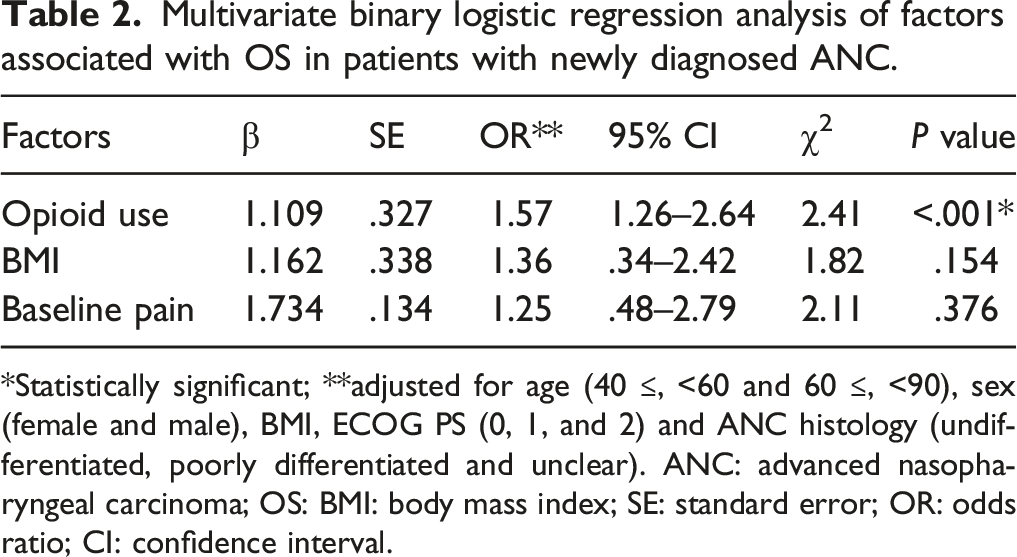
*Statistically significant; **adjusted for age (40 ≤, <60 and 60 ≤, <90), sex (female and male), BMI, ECOG PS (0, 1, and 2) and ANC histology (undifferentiated, poorly differentiated and unclear). ANC: advanced nasopharyngeal carcinoma; OS: BMI: body mass index; SE: standard error; OR: odds ratio; CI: confidence interval.
Multivariate binary logistic regression model for the cumulative dose analysis of opioids in patients with newly diagnosed ANC.
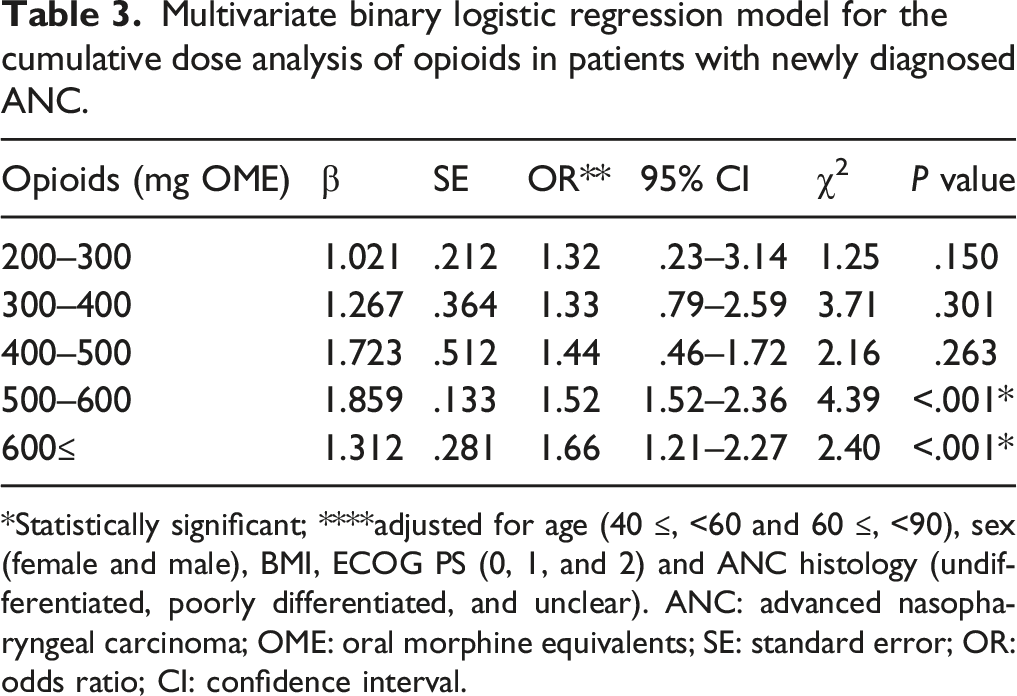
*Statistically significant; ****adjusted for age (40 ≤, <60 and 60 ≤, <90), sex (female and male), BMI, ECOG PS (0, 1, and 2) and ANC histology (undifferentiated, poorly differentiated, and unclear). ANC: advanced nasopharyngeal carcinoma; OME: oral morphine equivalents; SE: standard error; OR: odds ratio; CI: confidence interval.
Discussion
The results of the retrospective multicentre study that evaluated the survival outcomes associated with opioid use in the management of chronic pain related to ANC showed that the high opioid prescription size produced shorter OS than the low opioid prescription size in patients with ANC who received palliative care. The findings of shorter OS in ANC patients with high opioid prescriptions suggest the negative effect of high opioid prescription exposure on OS. The OS of high opioid prescriptions may be conditioned by palliative care. Furthermore, it remains controversial to achieve persistent relief from pain through opioid use.
Our findings were consistent with the results of a previous study 19 of 1386 individuals with newly diagnosed advanced nonhematologic malignancies evaluating adverse outcomes. Their results showed that patients with high opioid prescriptions had shorter median survival than those with low opioid prescriptions (5.5 vs 12.4 months; P < .01). Their conclusion suggested that a high prescription size for opioids is associated with shorter survival. A prospective cohort study 20 of 150 patients with advanced tumors assessing whether opioids are associated with survival showed that high opioid prescription use was a key predictor of shorter survival, independent of the influence of other variables. Their finding may be attributed to respiratory depression, drowsiness, and the onset of extensive pain progression. However, a previous study 21 on the impact of opioid use on survival in individuals with advanced tumors failed to suggest a remarkable association between opioid use and survival. A retrospective cohort study 22 of 223 patients with terminal tumors that explored whether high or low opioid prescription use has an effect on survival demonstrated that patients who underwent high opioid prescriptions had a longer median OS than those who did not (22 vs 9 days; HR .45; 95% CI, .34–.60, P < .01), and these distinctions continued after adjustment by baseline variables. In their study, the Kaplan–Meier curves were similar between the high and low opioid prescription cohorts, and a drug dose–response relationship with survival was not observed. Their conclusion suggested that the high size of opioid prescriptions resulted in increased survival. A retrospective study 23 of 317 tumor patients failed to show an association between different opioid doses and survival. Although well-defined opioid-related mechanisms propel promising pain relief outcomes in patients with advanced tumors, opioid use related to tumor progression and possible adverse events caused by opioids can result in premature death. Furthermore, in patients who receive larger opioid prescriptions and develop tolerance to opioids, continued opioid use can have a negative effect on survival, 24 suggesting that opioids may have immunosuppressive effects related to infectious diseases or tumor progression. 25
Advanced cancer is commonly associated with chronic pain that requires opioid intervention. 9,11,25 However, the use of opioids has been implicated in instigating disease progression, which ultimately reduces survival. 16,26 Evidence-based studies 19,20 have shown the negative impact of opioids on survival. Numerous mechanisms used to elucidate this negative correlation have been reported, but given the heterogeneity of the patient’s response to opioids, these mechanisms may have limitations. 11,13,20,27 In particular, a high size of opioid prescriptions was associated with adverse events that can weaken organ function and shorten survival. 16 Furthermore, immunosuppression related to the high size of opioid prescriptions has been insinuated. 28,29 There is an increasing trend in the application of opioid overdose that can result in a large number of patients developing some tolerance to the pain medications currently available (not just opioids). 3,28 Drug-induced tolerance has not yet been found to be a perfect solution. 3,21 In clinical practice, drug-induced immunosuppression and antitumor-induced immune system enhancement can coexist. 3,4,17
The prognostic variables established to influence survival in individuals with ANC include age, tumor burden, opioid consumption, comorbidities, and level of cardiopulmonary function. 3,26,27 Consistent with published studies, 11,16 the findings of the present study showed that high opioid prescriptions tended to result in reduced survival and we failed to detect any unfavorable effects of other prognostic variables on survival, although several researchers 5,30 claimed that this association exists. Individuals with a larger tumor burden may be associated with more pain and thus more opioid use and naturally be doomed to suffer from shorter survival. 3,4,20 Furthermore, patients with less opioid use attributed to less pain may have improved organ function contributing to longer survival. 11,17,20 In our multivariate analysis, we evaluated the factors that influence survival, showing that the high size of opioid prescriptions is still an independent factor associated with poorer survival.
Several drawbacks must be recognized. First, the retrospective design used in the present study and the small study population limit the reliability of the conclusion. Strict inclusion and exclusion criteria may lead to an overestimation of survival. Second, established medication schedules are lacking in the management of patients with ANC undergoing palliative care, which might inevitably introduce treatment bias. Third, the recording of pain levels can be influenced by patients, their families, and assessors, and the recording of pain data has a certain period of time, although the criteria for assessing pain are the same. Fourth, a quantitative assessment of tumor burden is missing, which can have an impact on the reliability of the outcome, as more bone metastases can produce more pain, which in turn leads to an increase in opioid demand, a reduced quality of life, and ultimately shortened survival.
Conclusions
The reported results contribute to an increasing body of evidence that a high size of opioid prescriptions tends to be associated with poorer survival than a low size of opioid prescriptions in newly diagnosed ANC patients undergoing palliative care. The exact mechanism of action for the negative survival effect of high opioid prescription size treatment is not yet clear. Alternatives to reducing opioid use, such as nerve blocks, should be considered. Given the retrospective nature, multicenter trials with a large sample are required to validate whether the present findings will persist over time.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
All methods were carried out according to the relevant guidelines and regulations of the institutional and/or national research committee and the 1964 Helsinki declaration. This study was approved by both Medical Ethics Committees (Wuhan Fourth Hospital; Tongji Medical College, Huazhong University of Science and Technology [Grant number: 179323]), and informed consent was waived by the Medical Ethics Committees.
Data Availability
The datasets generated during and analyzed during the current study are not publicly available due to the protection of patient privacy but are available from the corresponding author on reasonable request.
