Abstract
Granular cell tumor (GCT) is a rare soft-tissue neoplasm that originates from Schwann cells. Most cases occur in the subcutaneous or submucosal regions, and intramuscular GCT is even more uncommon. Herein, we describe an atypical GCT growing in the sternocleidomastoid muscle. A 41-year-old Chinese man was admitted to our hospital for a right neck mass. In magnetic resonance images, the mass showed higher signal intensity than the adjacent normal muscle on T2-weighted images and intermediate signal intensity on T1-weighted images. Slightly enhanced and higher signal intensity was observed in the peripheral rim on contrast-enhanced fat-sat T1-weighted images, and the “stripe sign” was evident. The tumor was completely resected, and pathological examination indicated an atypical GCT. The patient tolerated the procedure well. This is a rare case report of an intramuscular GCT from diagnosis to treatment. Magnetic resonance imaging might help diagnose such tumors, and we review different magnetic resonance imaging characteristics of intramuscular GCTs at other sites.
Introduction
Granular cell tumor (GCT), otherwise known as granular cell myoblastoma, is an uncommon nerve-derived soft tissue tumor which was first found in the tongue by Abrikossoff in 1926.1-3 GCT is subdivided into benign, atypical, and malignant subtypes according to morphological and immunohistochemical criteria. 4 Most GCTs appear benign and only a few exhibit atypical or malignant behavior. This neoplasm often involves the head and neck, and GCT of the tongue accounts for 1/3 of all cases. It can also be found in various other anatomic sites such as the ovaries, breast, and digestive tract. GCT usually grows in subcutaneous or submucosal regions, and intramuscular GCT is an extremely rare clinicopathological entity.
In this paper, we report a case of GCT growing in the sternocleidomastoid muscle (SCM) that occurred in a 41-year-old Chinese man. To our knowledge, no GCTs of the SCM have been reported in the English literature. Here, we review the pathology, magnetic resonance imaging (MRI), treatment, and prognosis of GCTs and summarize the MRI characteristics of different positions of the intramuscular GCTs.
Case report
A 41 year-old Chinese man was admitted to our hospital due to an indolent right neck mass found in 2018 without obvious incentive. The mass was solid, slow-growing, painless, and without any inflammatory reaction. He reported no dyspnea, difficulty swallowing, hoarse voice, blood-stained sputum, or other symptoms. On physical examination, a solid mass could be palpated and it developed along the right SCM with unclear boundaries; the mass could be pushed left and right but not up and down.
Cervical color Doppler ultrasonography revealed a hypoechoic mass approximately 31.8 mm × 22.5 mm in size, a well-delimited margin and uneven internal echogenicity in the right subcutaneous soft tissue layer of the neck. Fiberoptic nasopharyngolaryngoscopy showed no abnormalities of the pharynx or larynx. Magnetic resonance imaging (MRI) of the neck (Figure 1A-E) showed an oval-shaped lesion with a well-defined margin, approximately 36 mm × 32 mm × 34 mm in size developed in the right SCM. The mass exhibited higher signal intensity (SI) than adjacent normal muscle on T2-weighted images (T2-WI), intermediate SI on T1-weighted images (T1-WI), and uneven increased SI in fat-sat contrast-enhanced T1-WI with patchy low signal areas within. (A-E) Magnetic resonance imaging. Coronal T2-weighted images (T2-WI) (A and B). Axial spectral attenuated inversion recovery (SPAIR) T2-WI (C). Axial T1-weighted images (T1-WI) (D). Fat-sat contrast-enhanced axial T1-WI (E). The mass showed slightly increased SI in T2-WI (A). The mass did not contain fat on the fat signal image (B). The mass showed obvious hyperintensity compared to normal muscle on SPAIR images, and no fat component was visible in the mass (C). The tumor showed an intermediate SI that resembled the adjacent normal SCM in traditional T1-WI and presented the “stripe sign” (D). The mass had slightly enhanced and higher SI on the peripheral rim in T1-WI with contrast and also showed the “stripe sign” (E).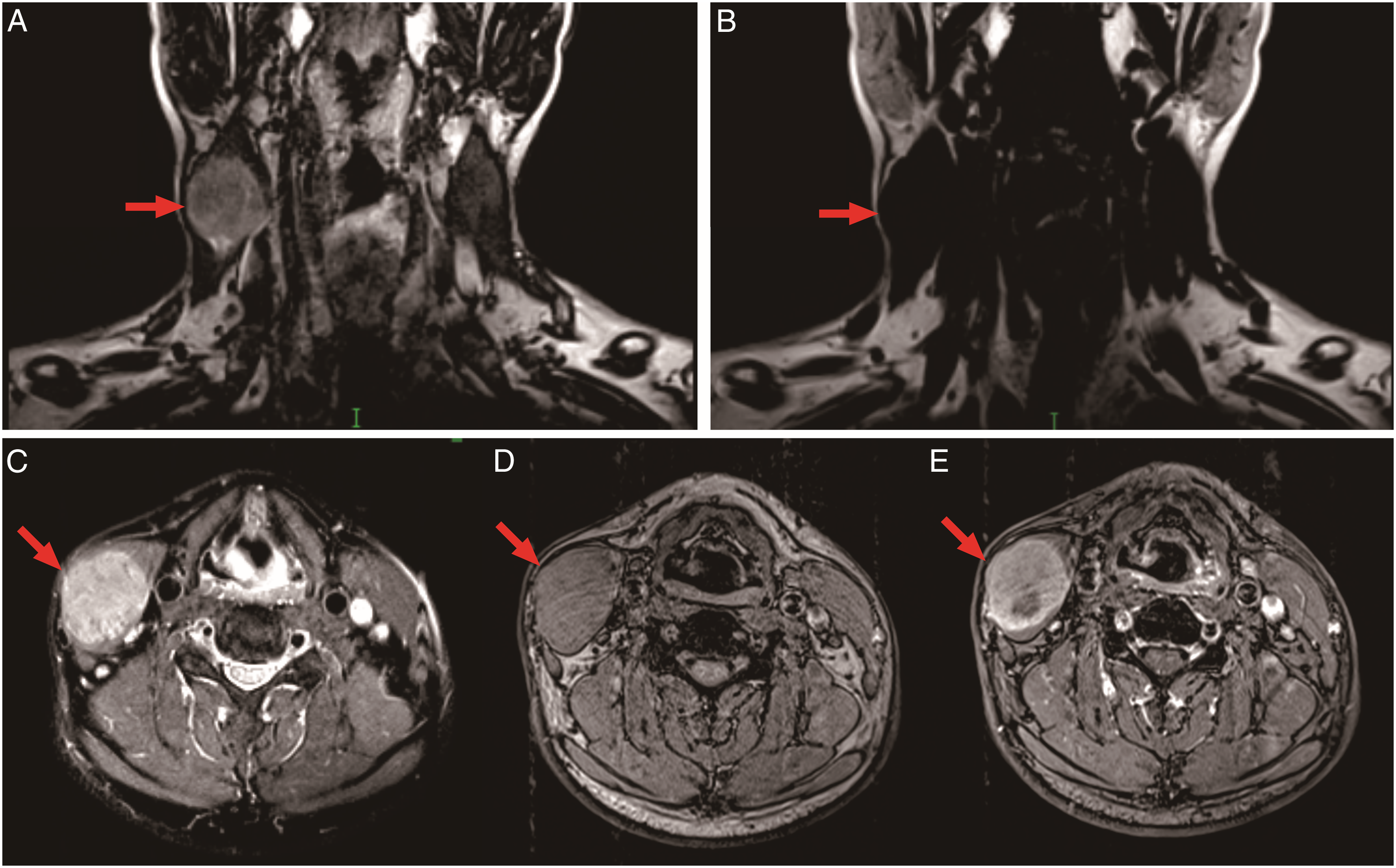
Preoperative examinations were performed, and all laboratory tests showed no obvious abnormality, which excluded contraindications to surgery. Because a malignant tumor could not be completely excluded in our patient, the mass was completely resected together with a margin of the normal surrounding muscle. Selective neck dissection was also performed due to a finding of enlarged lymph nodes at levels II, III, and IV in the right neck observed during the operation. A cycle of radiotherapy (PCTV1 54 Gy/27 F, PCTV 2 50 Gy/27 F) was carried out after surgery in the cancer center of our hospital.
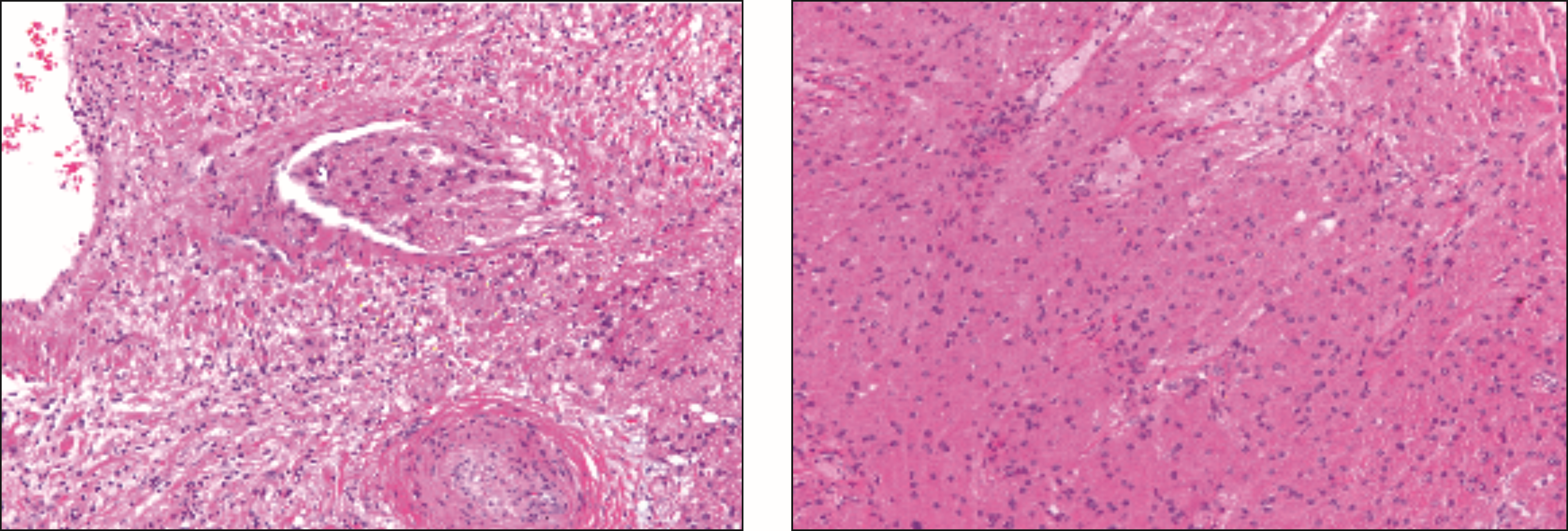
By eye, we observed that the mass was solid and the interior was gray-white in color with a clear periphery (Figure 2). Under the microscope, the tumor cells could be seen growing within the muscle, infiltrating the surrounding tissues, and invading the vasculature. Some of the cells exhibited enlarged nuclei and small nucleoli in some areas. There were only a few mitoses, and there was no evidence of neoplastic necrosis in the cells. It can be seen that the tumor entrapped some fibers of skeletal muscle (Figure 3). Immunohistochemical studies revealed that the tumor was positive for S100 and SOX10, but negative for CD68, PCK, SAM, CD34, Desmin, Syn, and HMB-45. The proliferative index of the neoplastic cells evaluated by Ki-67 labeling was 2%. Histopathological analysis of the biopsy specimen indicated that the right neck mass was an atypical granular cell tumor (AGCT). The neoplasm was adjacent to one side of the cut edge, and there was no metastasis in the dissected lymph nodes at levels II, III, or IV of the right neck. No severe adverse implications occurred in the patient after surgery. The patient is now alive without local recurrence or distant metastasis after a 10-month follow-up. Cross section of a resected lesion. Pathological images of the intramuscular tumor.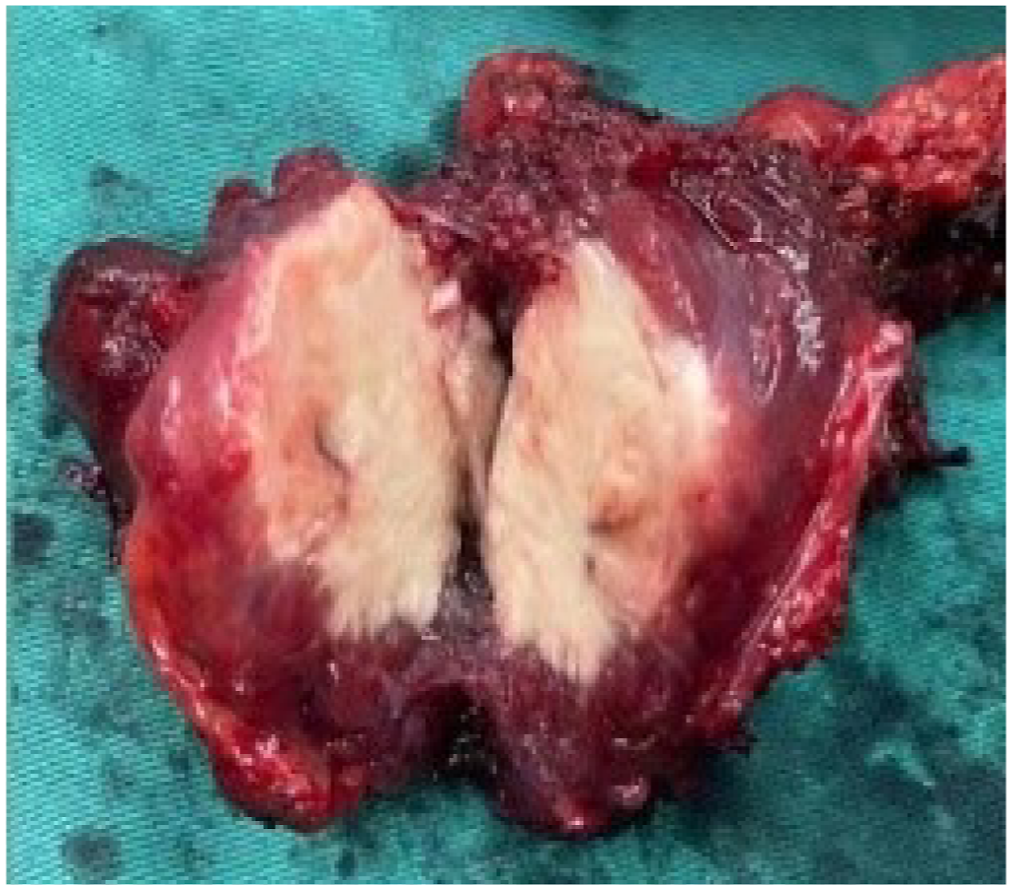
Discussion
GCT is a rare soft-tissue tumor originating from Schwann cells. It occurs in people of all ages causing a painless mass. Most cases are diagnosed in adults between the ages of 30 and 50 years. Some studies have reported that GCTs are more likely to occur in women rather men, with a ratio of approximately 2:1.
GCTs are divided into three different types: benign, atypical, and malignant, and the majority of GCTs are benign.4,5 According to the six diagnostic criteria first described by Fanburg-Smith et al, 4 an AGCT means it meets one or two of the criteria. Only one of the recognized criteria was observed in our patient, so the tumor was classified as atypical. The histology of GCT is usually characterized by small nuclei with dense chromatin in the center, the absence of necrosis and mitoses, positive staining with periodic acid Schiff (PAS) and diastase resistance of the granules, and a nest of granular cells partially separated by bands of connective tissue. Immunohistochemical studies of GCT showed that the majority of GCTs express the S100 protein, but some cases called “non-neural GCT” are negative for S100.6,7 Other proteins such as Vimentin, CD68, SOX10, SAM, and PCK could also be positive. The Ki67 proliferative index should be used as one of the characteristics to differentiate benign or atypical GCT from the malignant variant, rather than for an absolute classification as benign, atypical, or malignant based on Ki67 LI values of less than 5%, 5–10%, and >10%, respectively. 8
Reports of intramuscular granular cell tumor.
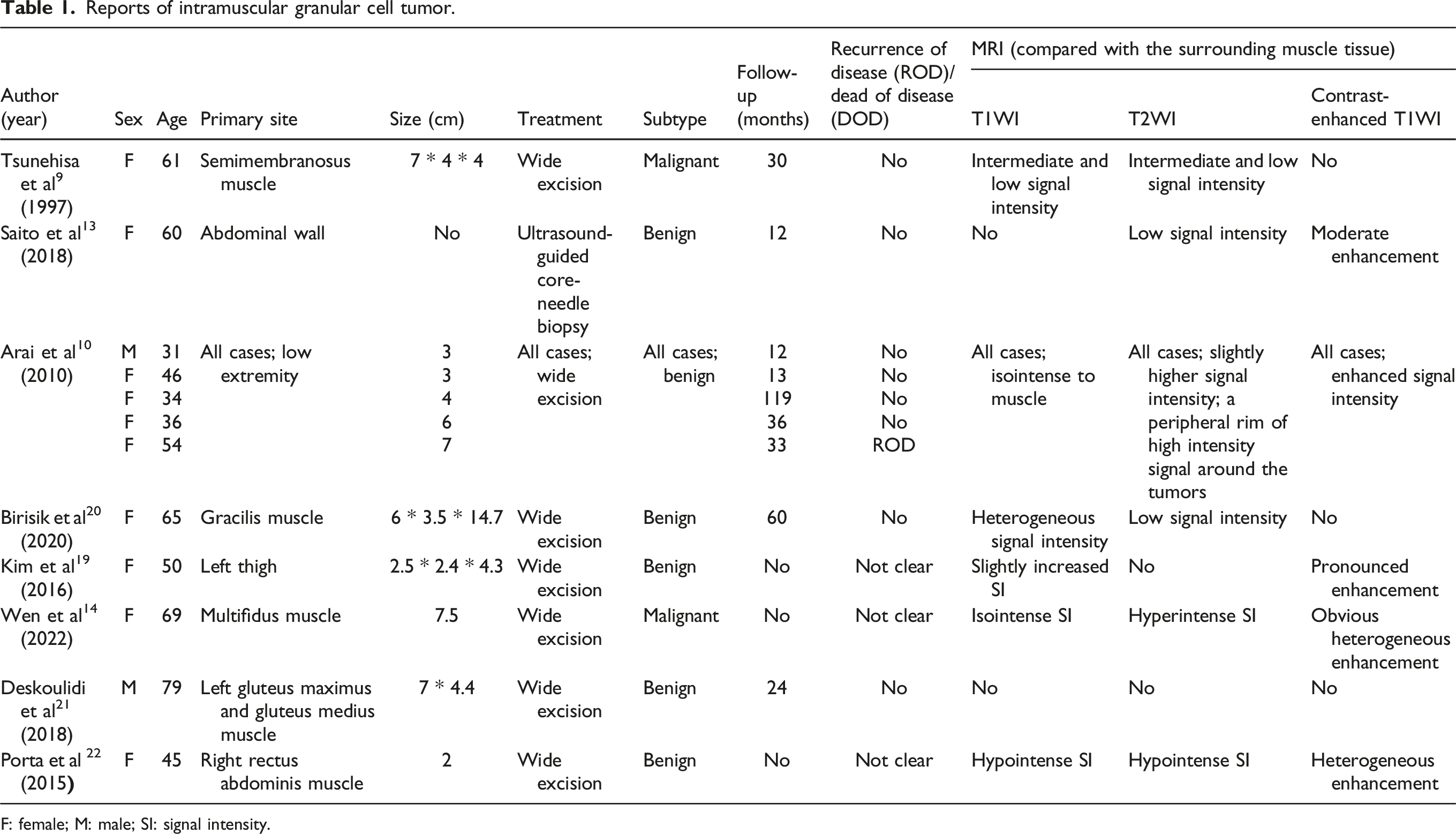
F: female; M: male; SI: signal intensity.
GCT lacks standardized diagnosis and treatment, and at present, complete surgical resection with safe margins is the main treatment. Most benign GCT cases have a favorable prognosis after total excision, while the prognosis of malignant GCT (MGCT) is worse due to recurrence or metastasis after total resection.23,24 The clinical manifestations of benign GCT (BGCT) and most AGCTs are slow growth and a clear margin, but infiltrative growth and possible vasculature invasion are occasionally found.7,25 Machado et al. 7 and Battistella et al 25 concluded that vascular invasion and other invasive features should not be considered to indicate an unfavorable prognosis but an increased risk of metastasis. A study of the risk of recurrence and the status of the margin in breast granular cell tumors conducted by Papalas et al 26 suggested that positive margins do not mean a higher risk of recurrence. The above features made it controversial to conduct postoperative adjuvant therapy on BGCTs and AGCTs with infiltrative growth, vasculature invasion, or positive margins. In a study of AGCTs and MGCTs in Japan, it was found that close attention should be paid and long-term follow-up provided to AGCTs that had histological metastatic potential. 27 Moreover, a wide resection is necessary for both AGCTs and MGCTs, which means better local control. Postoperative adjuvant therapy, such as radiotherapy or chemotherapy, plays a limited role in recurrence or metastasis, does not improve the prognosis much in AGCT, and is therefore usually used for the treatment of recurrent GCTs or the control of GCTs.27,28 Moreover, it is important to obtain a differential diagnosis to rule out other possibilities including metastatic tumors, leiomyosarcoma, and malignant fibrous histiocytoma.
In conclusion, our case report describes the MRI and histological characteristics of AGCT in the SCM in detail and retrospectively analyzes the MRI characteristics in some other cases. Limitations of the study include that it reports only one case, the follow-up period is short, and it lacks sufficient evidence to apply postoperative radiation in our case. The MRI features of GCT described here could be helpful for the diagnosis of GCT. Total resection and long-term follow-up are necessary for the treatment of GCT, and more cases are necessary to improve our knowledge of this rare neoplasm.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
This case does not constitute research at our institution.
Informed consent
Verbal informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
Data availability
The data used to support the findings of this study are available from the corresponding author upon request.
