Abstract
Objective
The purpose of this study was to analyze the difference of survival rates in paitents with oropharyngeal keratinizing squamous cell carcinoma (KSCC), nonkeratinizing squamous cell carcinoma (NKSCC), basaloid squamous cell carcinoma (BSCC), and papillary squamous cell carcinoma (PSCC).
Materials and methods
Patients diagnosed with oropharyngeal squamous cell carcinoma between 2004 and 2015 were collected from the SEER database. Cox proportional hazards models and Kaplan–Meier curves were used for survival analysis. Propensity score matching (PSM) was performed to adjust for the effect of confounding variables. Due to the small sample size of PSCC, this study did not perform PSM between it and other subtypes.
Results
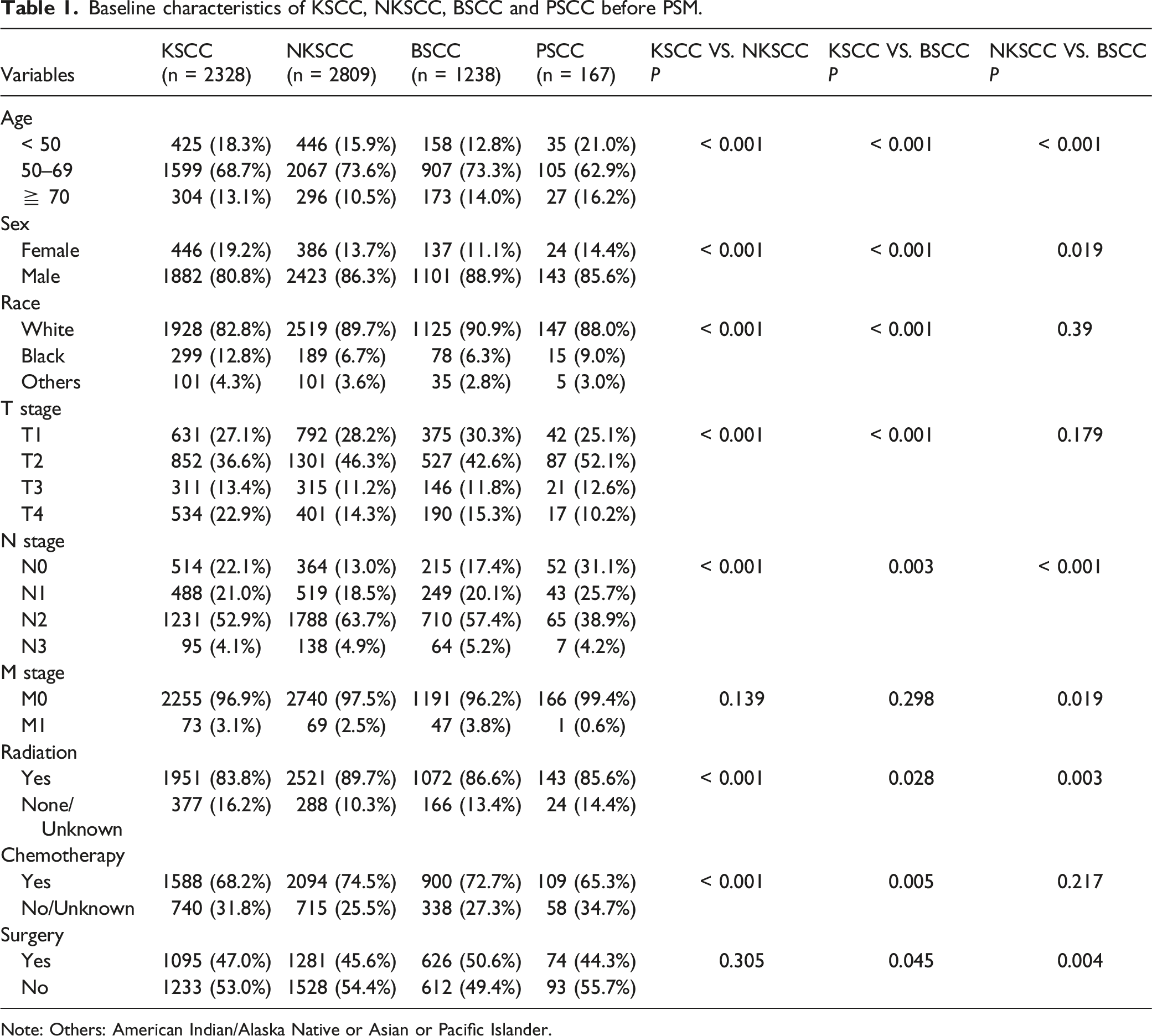
The 5-year cancer-specific survival (CSS) rate of PSCC was higher than that of KSCC, NKSCC, and BSCC (0.627 vs. 0.812 vs. 0.789 vs. 0.875, P < 0.05); And the CSS rate of KSCC was lower than that of other subtypes both before and after PSM. In addition, the 5-year and 10-year CSS rates of BSCC were not different from NKSCC (P > 0.05), but not as good as NKSCC in the long term (P = 0.028). After PSM, the 5-year, 10-year, and long-term prognosis of BSCC were significantly worse than those of NKSCC (P < 0.001).
Conclusion
The 5-year CSS of PSCC was better than the other three subtypes. The short-term prognosis of BSCC was not significantly different from NKSCC, but the long-term survival was lower than that of NKSCC, and the difference was more obvious after PSM. Meanwhile, the prognosis of KSCC was worst.
Introduction
Oropharyngeal squamous cell carcinoma (OPSCC) is one of the common malignant tumors located on the mucosal of the upper aerodigestive tract, accounting for 10–12% of all upper aerodigestive tract tumors, 1 composed of tonsil cancer, tongue base cancer, soft palate cancer carcinoma, and uvula carcinoma. 2 As with oral cancer, 90% of oropharyngeal cancers are squamous cell carcinomas (SCC). 3 In recent years, the incidence of OPSCC has been increasing, and it has gradually become the most common malignant tumor of head and neck cancer in the United States. 4 This is mainly due to the increase in oropharyngeal cancer caused by HPV infection. It has been widely confirmed that the prognosis of HPV-positive OPSCC patients was better than HPV-negative.5,6 In the latest American Joint Committee on Cancer (AJCC-8) staging system HPV-positive and HPV-negative OPSCC were classified as separate entities for the first time, while p16+ used as a surrogate marker for HPV.6-8 However, several studies have reported 5–20% of p16-positive OPSCC but HPV-negative.6,7,9,10 In addition, although OPSCC at the tonsils and base of the tongue tend to be more HPV detectable than other sites, with approximately 56-70% being HPV-positive compared to 19–22% of tumors at other sites and the prognostic value of HPV at other sites seems less reliable. 2
Studies have found that the histological subtype of SCC can be used as an independent factor to evaluate the prognosis of patients in other tumors, such as lung SCC, 11 nasopharyngeal SCC, 12 and so on. The main subtypes of OPSCC are keratinizing squamous cell carcinoma (KSCC), nonkeratinizing squamous cell carcinoma (NKSCC), basaloid squamous cell carcinoma (BSCC), and papillary squamous cell carcinoma (PSCC). 13 Most of NKSCC, BSCC, and PSCC and 15–25% of KSCC are associated with transcriptionally active HPV infection.2,14,15 Although parts of these types are related to HPV, and the prognosis of HPV-positives was better than that of non-positives, the prognosis of each of these subtypes and the differences between them cannot be fully evaluated only through HPV status. There were few and controversial studies on histological subtypes of OPSCC, such as BSCC, which was previously thought to be associated with aggressiveness. However, recent studies have shown that its prognosis was better than that of conventional SCC.16,17 Therefore, we compared these subtypes by the data from the Surveillance, Epidemiology, and End Results (SEER) database and used propensity score matching (PSM) to adjust for confounding factors to explore the prognostic significance of histological subtypes in OPSCC.
Materials and methods
Data acquisition
Data on patients diagnosed with OPSCC from January 1, 2004 to December 31, 2015 were collected from the SEER 18 plus database using SEER*Stat software (version 8.4.0; www.seer.cancer.gov). Because the SEER database is a public database, there is no need for institutional review of patient informed consent. The inclusion criteria: (1) Patients over 18 years old; (2) The International Classification of Tumor Diseases 3rd Edition (ICD-O-3) site code were “C01.9-Base of tongue, NOS,” “C02.4-Lingual tonsil,” “C05.1-Soft palate, NOS,” “C05.2-Uvula,” “C09.0-Tonsillar fossa,” “C09.1-Tonsillar pillar,” “C09.8-Overlapping lesion of tonsil,” “C09.9-Tonsil, NOS,” “C10.0-Vallecula,” “C10.2-Lateral wall of oropharynx,” “C10.3-Posterior wall of oropharynx”, “C10.4-Branchial cleft”, “C10.8-Overlapping lesion of oropharynx,” and “C10.9-Oropharynx, NOS.” The morphological codes were as follows: “8052/3: Papillary squamous cell carcinoma (PSCC)”, “8071/3: Squamous cell carcinoma, keratinizing, NOS (KSCC),” “8072/3: Squamous cell carcinoma, large cell, nonkeratinizing, NOS (NKSCC),” “8073/3: Squamous cell carcinoma, small cell, nonkeratinizing (NKSCC),” and “8083/3: Basaloid squamous cell carcinoma (BSCC)”. (3) OPSCC was the only cancer or the first diagnosed primary tumor; (4) Pathological diagnosis. Exclusion criteria were: (1) The age of the patient was less than 18 years old; (2) The survival time was less than or equal to 1 month; (3) Patients with unknown age, sex, T stage, N stage, M stage, treatment information, and cause of death. The study was conducted in accordance with the Declaration of Helsinki.
Variables
This study collected the data of age, sex, race, T stage, N stage, M stage, surgery, radiotherapy, and chemotherapy of four different subtypes of OPSCC. In our study, the outcome variable was cancer-specific survival (CSS). CSS was defined as the death of a patient due to OPSCC.
Statistical analysis
Categorical variables were expressed as percentages, and the Pearson chi-square test was used to compare the differences of variables between different OPSCC subtypes. To minimize the influence of confounding factors, PSM was used to match KSCC with NKSCC, KSCC with BSCC, NKSCC with BSCC by the radio of 1:1, and the logistic regression model was used to calculate the propensity score of each patient, of which the following variables were included: age, sex, T stage, N stage, M stage, surgery, radiotherapy, and chemotherapy. When matching, the method of matching without replacing the caliper was adopted. The caliper value was set to 0.001. Due to the small number of patients with PSCC, we did not perform PSM between it with other subtypes in this study. After PSM, we again compared the baseline characteristics of the different between-group variables. And the standardised mean difference (SMD) was used to evaluate whether the matched cohorts were balanced in terms of variables before and after matching, with the SMD value < 0.10 being the criterion for adequate balance. 18 Kaplan–Meier method was used to draw survival curves, and the differences in survival before and after histological subtype matching were compared by log-rank test. The prognostic factors of OPSCC were evaluated through univariate and multivariate COX regression analysis. All statistical analyses were performed by R statistical software (version 4.0.3). P-value < 0.05 was considered statistically significant.
Results
Baseline characteristics
As shown in Figure 1, we collected data on 9420 OPSCC patients from the SEER database, of which a total of 6542 patients met the study criteria. There were KSCC: 2328 cases; NKSCC: 2809 cases; BSCC: 1238 cases; and PSCC: 167 cases. Table 1 summarized the baseline characteristics of patients in each subtype, and the results of the chi-square test between variables for KSCC, NKSCC, and BSCC before matching. It can be seen that the subtypes of OPSCC patients were mainly concentrated in 50–69 years old, the proportion of males was much higher than that of females, the rate of lymph node metastasis was high, and radiotherapy and chemotherapy were more commonly used in treatment. Additionally, there were significant differences in some variables among KSCC, NKSCC, and BSCC. The flowchart of this study (KSCC: Keratinizing squamous cell carcinoma; NKSCC: Nonkeratinizing squamous cell carcinoma; BSCC: Basaloid squamous cell carcinoma; PSCC: Papillary squamous cell carcinoma). Baseline characteristics of KSCC, NKSCC, BSCC and PSCC before PSM. Note: Others: American Indian/Alaska Native or Asian or Pacific Islander.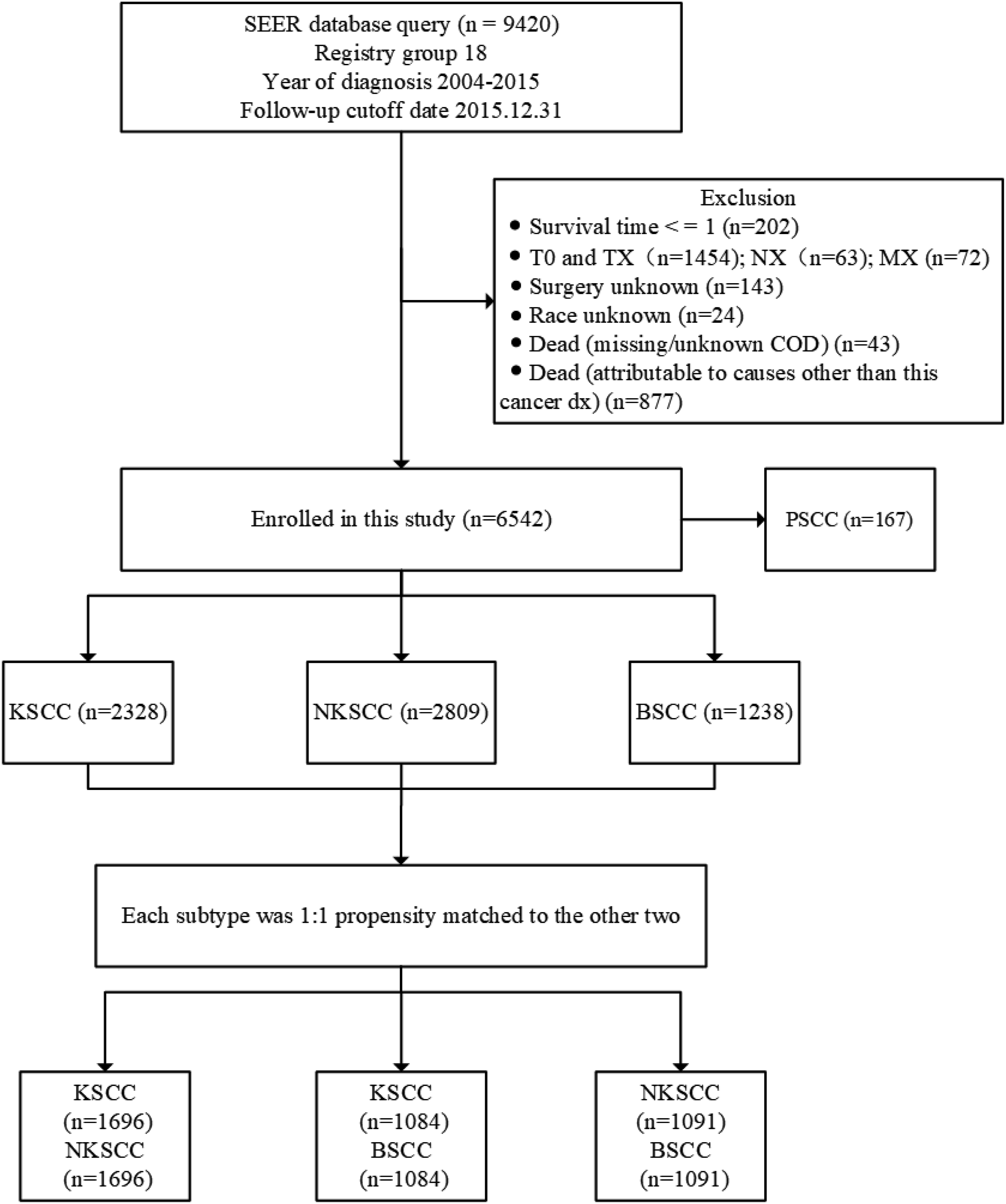
Survival comparison before PSM
The long-term prognosis of KSCC was the worst, followed by BSCC, and there was no significant difference between NKSCC and PSCC (Figure 2A). Furthermore, we calculated 5- and 10-year CSS rates for different subtypes to more accurately compare their differences (Supplementary Table 1). The 5-year CSS rates of KSCC, NKSCC, BSCC, and PSCC were: 0.627, 0.812, 0.789, and 0.875. The 5-year CSS rate of PSCC was better than KSCC, NKSCC, and BSCC (P < 0.001, P = 0.042, and P = 0.013). There was no statistical difference in 5-year CSS between BSCC and NKSCC (P = 0.190). In contrast, the 5-year CSS rates of NKSCC and BSCC were higher than that of KSCC (P < 0.001 and P < 0.001). The 10-year CSS rates of KSCC, NKSCC, BSCC, and PSCC were 0.557, 0.761, 0.711, and 0.744 respectively. The CSS rate of PSCC was still better than that of KSCC (P < 0.001) but not statistically different from that of BSCC and NKSCC (P = 0.058 and P = 0.290); the 10-year CSS rates of NKSCC and BSCC were higher than that of KSCC (P < 0.001 and P = 0.290), while there was no statistical difference found between BSCC and NKSCC (P = 0.062). Specific survival curves of the four subtypes of OPSCC before PSM (A), the cohort of KSCC and NKSCC after PSM (B).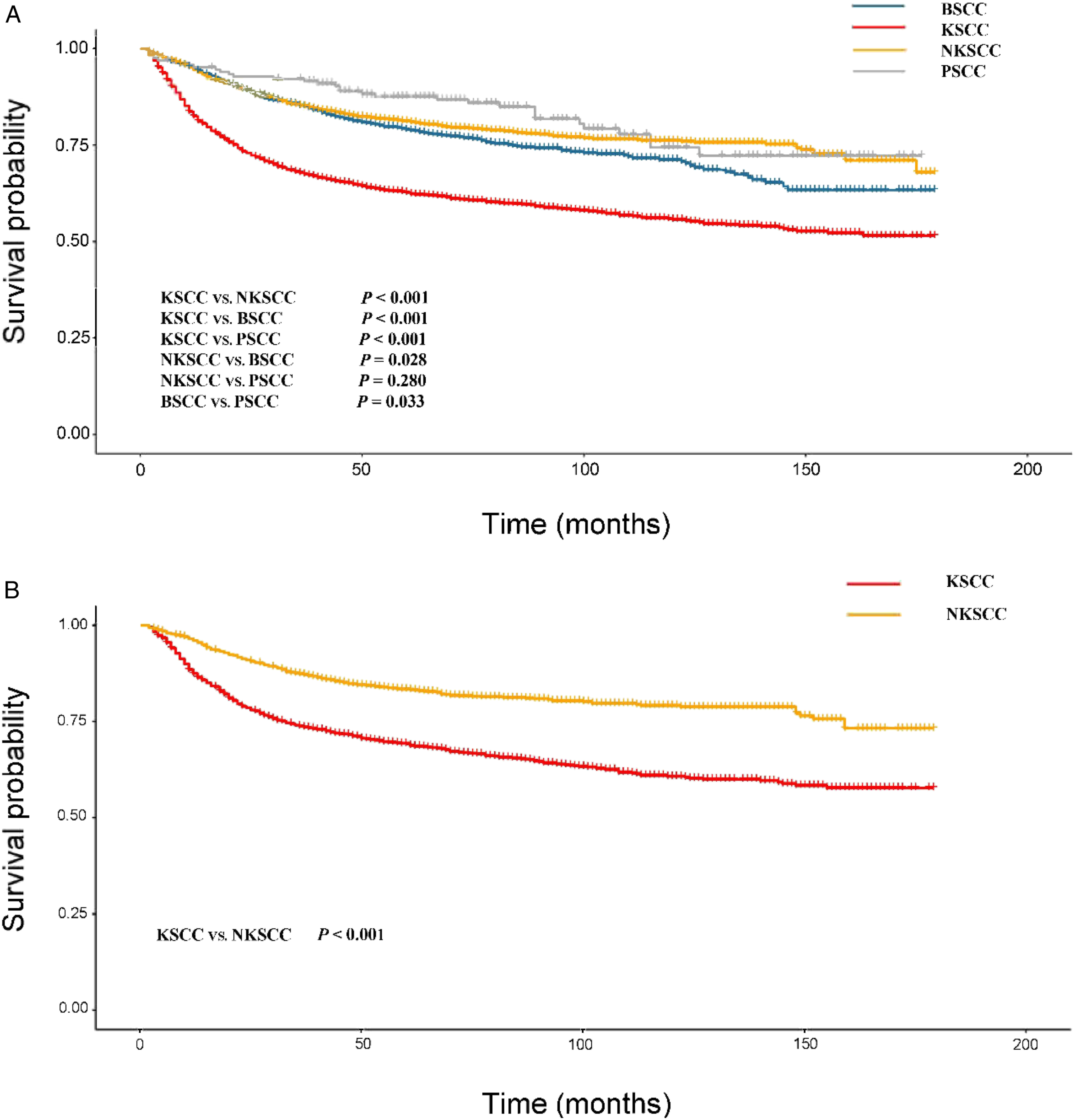
Survival comparison after PSM
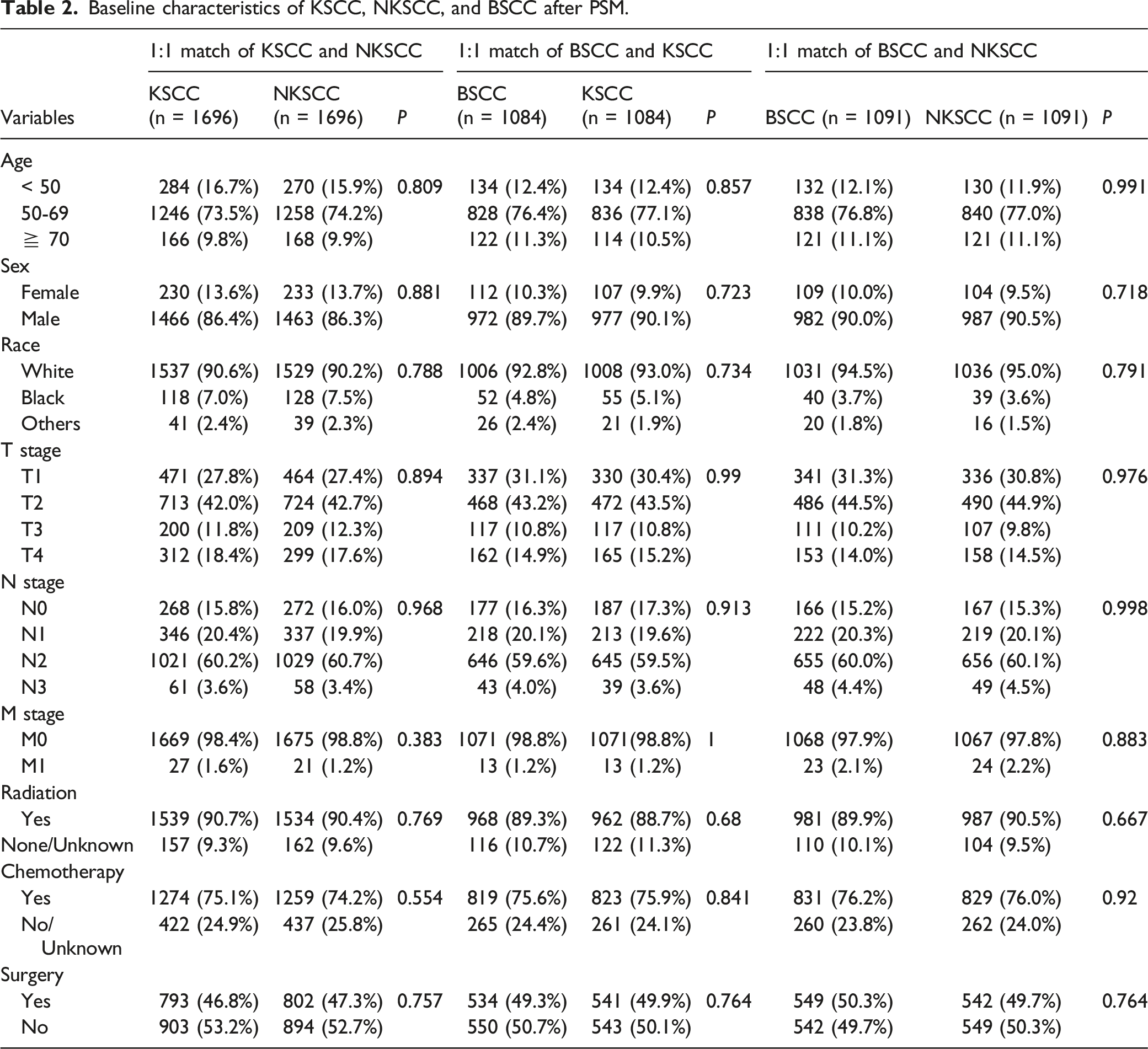
Baseline characteristics of KSCC, NKSCC, and BSCC after PSM.
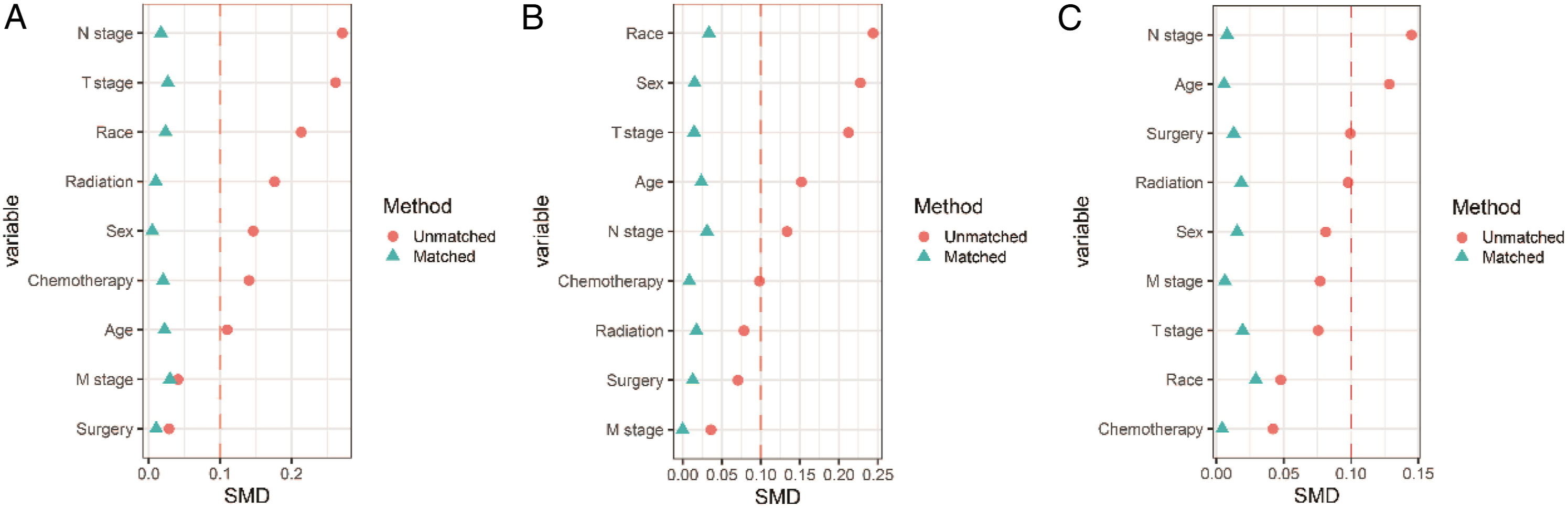
Plot of differences between variables before and after matching of the cohort of KSCC and NKSCC (A), the cohort of KSCC and BSCC (B), and the cohort of NKSCC and BSCC (C). SMD less than 0.1 is the criterion for balance.
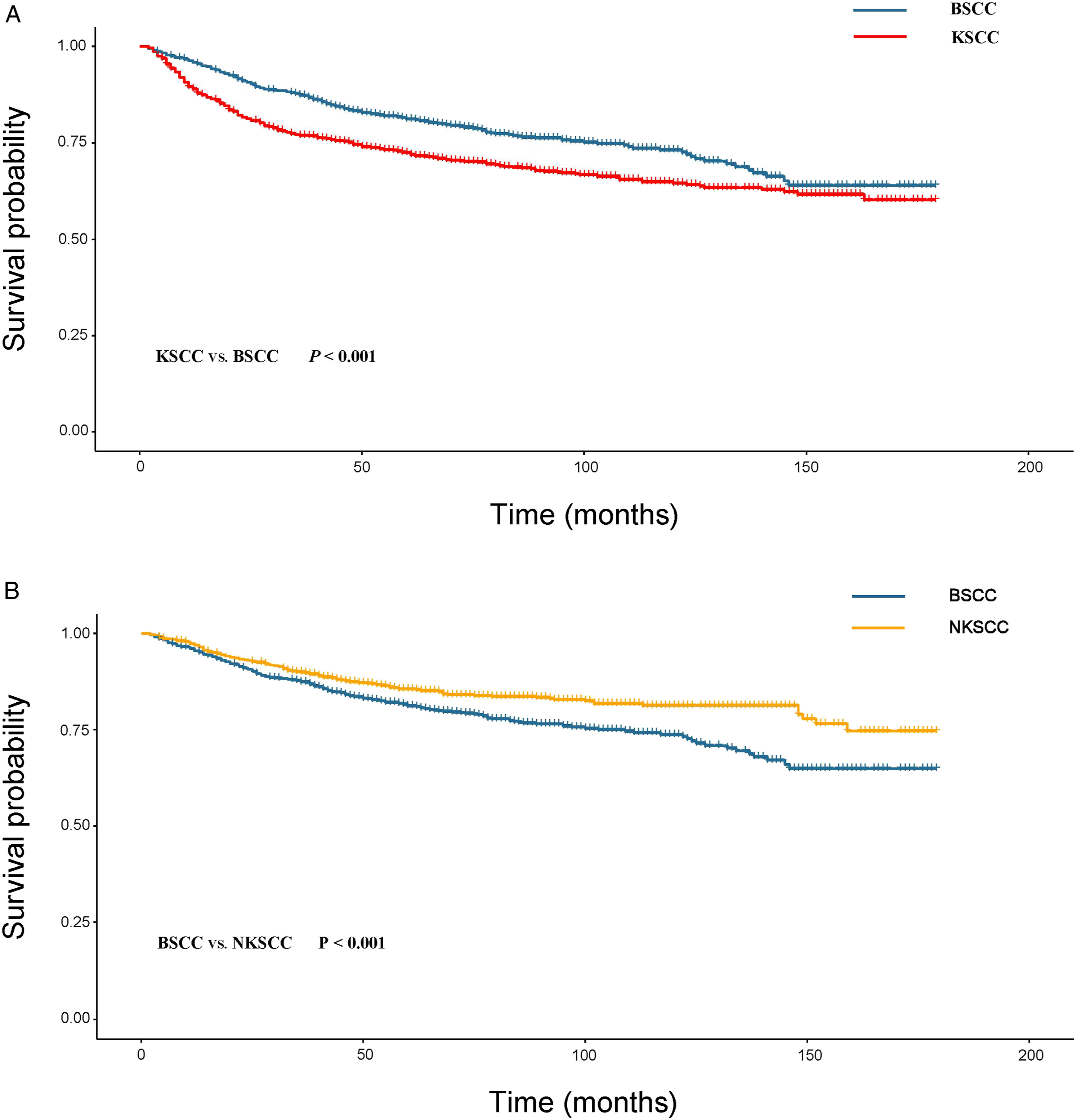
Specific survival curves of the cohort of KSCC and BSCC after PSM (A), the cohort of NKSCC and BSCC after PSM (B).
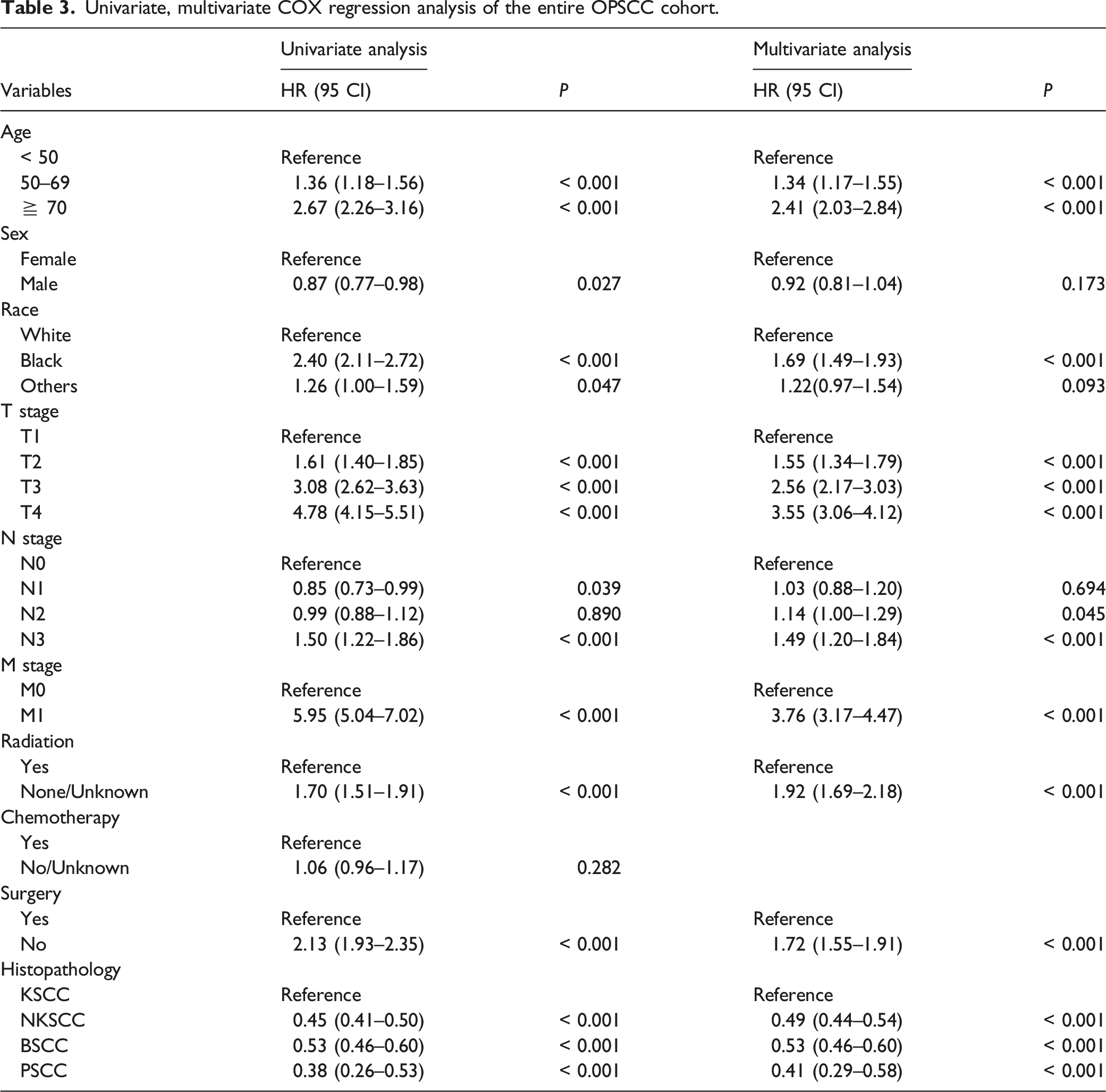
Univariate, multivariate COX regression analysis
Univariate, multivariate COX regression analysis of the entire OPSCC cohort.
Subgroup analysis between matched groups
We performed subgroup analyses of matched cohorts to assess differences in survival among the subvariables of patients in each matched cohort. As displayed in the Figure 5, the prognosis of NKSCC was better in the cohort of KSCC with matched NKSCC, but there was no statistical difference in others in race, N3 stage, or M1 stage (P > 0.05). BSCC has a better prognosis than KSCC in the matched KSCC and BSCC cohort, but there was no statistical difference in age < 50 years, age > 70 years, female, others in race, T1 stage, N1 stage, M1 stage, or radiotherapy None/Unknown (Supplementary Figure 1); NKSCC had better prognosis than BSCC in the cohort of NKSCC and BSCC after matching, but there was no statistical difference in age < 50 years, age >70 years, female, black in race, others in race, T3 stage, T4 stage, N1 stage, M1 stage, or radiotherapy None/Unknown (Supplementary Figure 2). The forest plot of subgroup analysis between KSCC and NKSCC after PSM.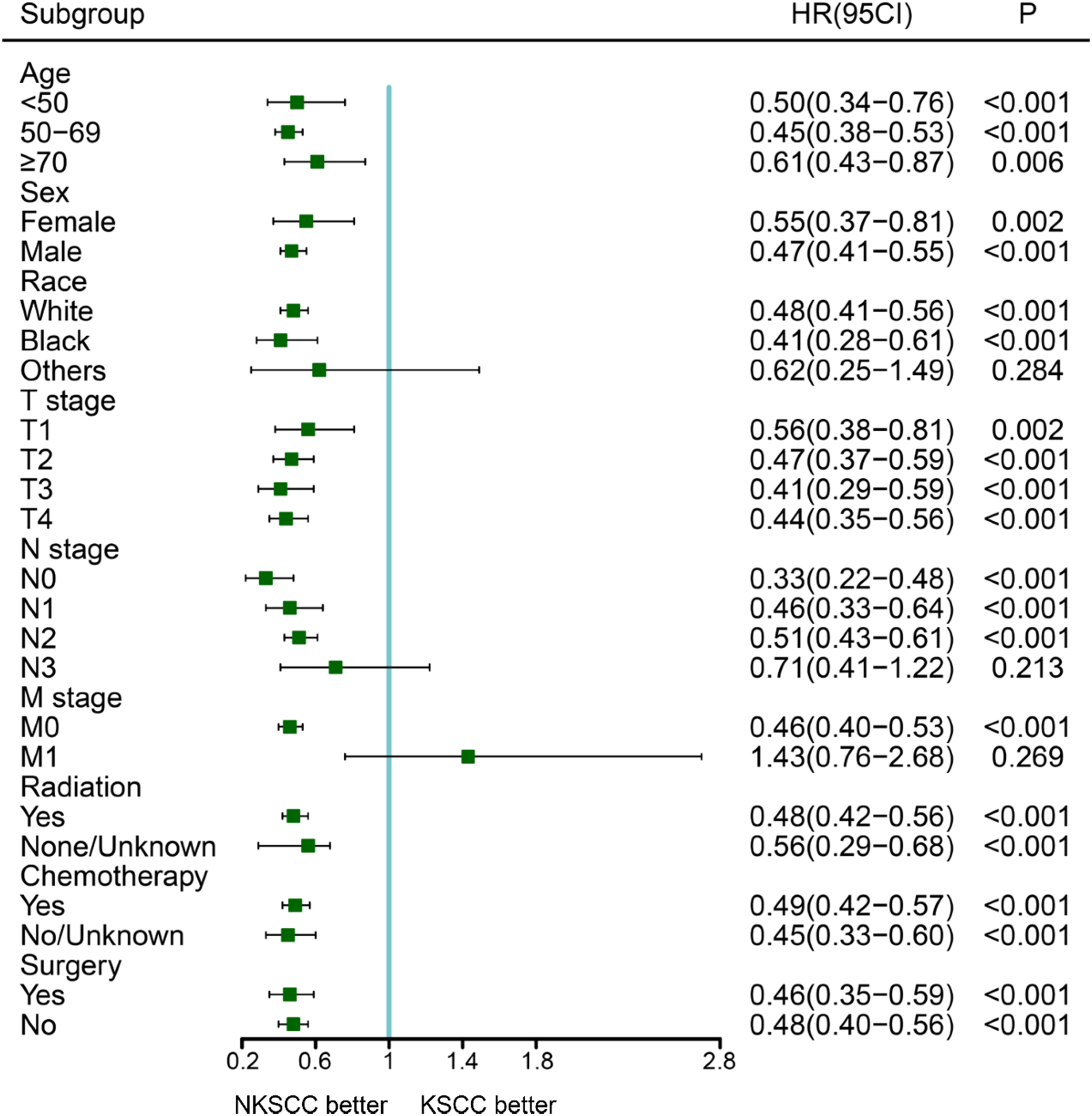
Discussion
The more common subtypes in OPSCC are KSCC and NKSCC and BSCC and PSCC are rare, especially PSCC, so there are few studies on BSCC and PSCC. Here, we compared the prognosis of these four subtypes of OPSCC using data from SEER for the first time. KSCC was the subtype with the worst prognosis before and after PSM, which is in line with our understanding of it. In addition, the prognosis of BSCC was controversial, and since it was first described by Wain et al. in 1986, BSCC was generally considered to be more aggressive than SCC. 19 However, conflicting data exist regarding the prognostic significance of BSCC. Two small case-control studies of BSCC versus head and neck SCC have come to different conclusions, one showing that BSCC was not beneficial for survival and the other reporting that BSCC was not statistically different from moderately or poorly differentiated SCC.17,20,21 Jacobi et al. suggested that Oropharyngeal BSCC has a certain correlation with HPV infection and has a good prognosis. 22 The different conclusions of different studies may be related to their different sample size and understanding of BSCC. There were also studies using the SEER database finding that the Hazard Ratio (HR) in oropharyngeal BSCC was less than 1 compared with SCC.16,17 However, it only compared the difference between BSCC and conventional SCC, the differences between these subtypes were not analyzed in detail. It can be seen that BSCC has a better prognosis than KSCC in our study. Moreover, Unsal et al. reported that 1-year and 5-year survival rates of BSCC were similar to NKSCC, but the 10-year long-term survival rate was significantly decreased. 23 It is worth noting that similar findings were also found in our study. The 5-year and 10-year CSS rates of BSCC were not statistically different from that of NKSCC, but it can be seen that the survival curve of BSCC decreased significantly after 10 years, and the long-term prognosis was worse than that of NKSCC (Figure 2A). Furthermore, after PSM adjusted the influence of confounding factors, it was obvious that the prognosis of BSCC was inferior to that of NKSCC.
PSCC, a variant of SCC, which was individually identified and classified in the World Health Organization (WHO) classification. 24 It was characterized by exogenous papillary growth, with a satisfactory 5-year OS and complete disease resolution in more than 80% of cases. 25 Oropharyngeal PSCCs differ markedly from their oral and throat counterparts, they tend to have NK morphology and are more often associated with transcriptionally active HPV. 26 Due to the small number of cases, there are fewer studies comparing the survival difference of PSCC with other subtypes of OPSCC. We found that the 5-year CSS rate of PSCC was significantly better than that of the other three subtypes, but with the prolongation of survival time, there was no significant difference between it and NKSCC. In addition, interestingly, there was no statistical difference in 10-year CSS between PSCC and BSCC in our study. These may echo the fact that PSCC mostly have NK morphology. In addition, Wang et al. found that radiotherapy and surgery were independent prognostic factors for oropharyngeal cancer, 27 which was confirmed in all our cohorts. However, chemotherapy was not a prognostic factor, which may be related to its incomplete information.
There are some limitations of our study. First of all, in the SEER database, a large number of patients with oropharyngeal SCC had to be excluded because their exact histological subtypes were not available, which could lead to missing patient numbers and selection bias. Second, the database lacks detailed information on smoking history and radiotherapy and chemotherapy. 28 Finally, in order to minimize the influence of interfering factors, we performed PSM. Although PSM removed the effect of confounding factors, the PSM analysis itself may also introduce potential selection bias and the level of evidence was still lower than that of randomized controlled clinical trials. 29 Taking these limitations into account, multicenter, prospective, randomized controlled trials are needed to minimize confounding and confirm these findings.
Conclusion
The 5-year CSS rate of oropharyngeal PSCC was superior to the other three subtypes, and the short-term and long-term survival rates of KSCC were significantly lower than those of the other subtypes. Although the short-term prognosis of BSCC was not significantly different from that of NKSCC, the long-term survival was not as good as that of NKSCC. And the difference between them became more obvious after PSM. Given some limitations of this study, large cohorts, multicenter and prospective studies are required to confirm our findings.
Supplemental Material
Supplemental Material - Survival comparison of different histological subtypes of oropharyngeal squamous cell carcinoma: A propensity-matched score analysis based on SEER database
Supplemental Material for Survival comparison of different histological subtypes of oropharyngeal squamous cell carcinoma: A propensity-matched score analysis based on SEER database by Tao Li, Yi Wang, Xianwang Xiang and Chuanjun Chen in Ear, Nose & Throat Journal
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Fundamental Research Funds for the Central Universities (No. WK9110000146).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
