Abstract
Introduction
Determining the appropriate clinical and pathological stage is important for cancer diagnosis, treatment, and research. Cancer staging aims to establish treatment plans and predict prognosis by grouping cancers based on their anatomical structure. The American Joint Committee on Cancer (AJCC) introduced the tumor–node–metastasis (TNM) staging system in 1977. The AJCC TNM staging system is commonly used for head and neck cancers.
Oropharyngeal squamous cell carcinoma (OPSCC) incidence has dramatically increased over the past few decades. The dramatic increase is primarily due to human papillomavirus (HPV) infection among young individuals aged <60 years, men, and Caucasians.1-3 HPV-positive OPSCC was epidemiological and clinically distinctive from HPV-negative OPSCC. 4 HPV-positive OPSCC shows a much higher survival rate than HPV-negative OPSCC. 5 Therefore, the most recent version of the 8th AJCC TNM staging system distinguished HPV-positive OPSCC from HPV-negative cancers. 6 However, changes in the classification of HPV-negative OPSCC are limited. In the 7th and 8th editions, locally advanced diseases invading the surrounding structures were classified as T4 tumors, while T1-T3 tumors were classified according to the primary tumor size. Therefore, the tumor size range included in the T4 stage may vary. Despite the heterogeneity in the size of T4 OPSCC, to the best of our knowledge, no single study of prognosis according to primary tumor size has been reported in T4 OPSCC. Recent guidelines recommend radiotherapy (RT) alone or surgery followed by RT or CCRT if indicated. for early-stage OPSCC. For advanced OPSCC, CCRT is recommended as standard of care and surgery followed by RT or CCRT if indicated after surgery as an option. 7 HPV-related OPSCC can be considered a distinct disease primarily as a consequence of its anatomical location and its viral etiology. Its optimal treatment approach is still not well-defined. For sure, HPV-related OPSCC is extremely sensitive to radiation exposure and patients generally are complete responders and long-term survivors. Therefore over the years scientific interest has shifted to new stratagems to potentially improve functional outcomes. 8
Recent studies have shown that the acquisition of invasiveness and metastatic potential may occur early in the onset of cancer. 9 Recent data from several cancers support these results by showing a paradoxical relationship between survival rate and tumor diameter. For instance, in prostate, colon, and breast cancer, patients with very small cancers show a worse survival rate than patients with large cancers.9-11 These results suggest that small tumors spreading rapidly may be related to extreme aggressiveness, possibly due to genetic and epigenetic features that allow local invasion and distant metastasis. However, whether these relationships can be applied to patients with OPSCC is not yet known.
Therefore, we sought to determine whether very small, locally advanced (T4) OPSCC was associated with worse disease-specific survival (DSS). To test this hypothesis, we analyzed patients with T4 OPSCC according to tumor size using the United States Surveillance, Epidemiology, and End Results (SEER) data from 2010 to 2016 before using HPV status for cancer staging.
Materials and Methods
This study was conducted according to the ethical standards of the institutional and/or national research committee and in accordance with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. This study was reviewed and approved by the Institutional Review Board (IRB) of the Kyung Hee University Hospital at Gangdong. The requirement for informed consent was waived because all data used in this study were deidentified.
Data Sources
The SEER database was built using patient data collected from several geographically different populations representing the United States population from urban to rural areas. Oropharyngeal cancer cases in the National Cancer Institute’s SEER database from 2010 to 2016 and their detailed information were queried using SEER*Stat version 8.3.6. This database includes incidence and population data stratified by race, age, sex, and year of diagnosis.
Study Population
This retrospective cohort study was approved by the IRB of the Kyung Hee University Hospital at Gangdong. The requirement for informed consent was waived because all the data used in this study were de-identified. We collected and reviewed the records of patients with oropharyngeal cancer in the SEER database from 2010 to 2016. The diagnosis of OPSCC in the SEER database was confirmed pathologically. Patients with non-T4 OPSCC and those with incomplete clinicopathological information were excluded.
Clinicopathological Variables
Clinicopathological data were analyzed, including age, sex, race, tumor site, cancer stage, TNM stage, pathological grade, type of treatment, and survival outcome. The tumor sites were classified as tonsils (C090-C099), the base of the tongue (C019), and others (C024, C051, and C100-C109) using the World Health Organization International Classification of Diseases for Oncology. The staging of all cases was based on the seventh edition TNM classification of the AJCC staging manual. 12 Immunohistochemical markers were used to define HPV status as HPV-positive or HPV-negative. Tumor size was categorized as ≤1 cm, 1 to 2 cm, 2 to 3 cm, 3 to 4 cm, and >4 cm. For statistical analysis, we additionally compared the 2 size groups (≤1 cm vs > 1 cm and >4 cm vs 1 < T ≤ 4 cm). The histological features of the tumor were classified as undifferentiated, poorly, moderately, or well-differentiated according to cytological findings and architecture. 13 Treatment modalities (surgery + radiotherapy, others) were analyzed.
Statistical Analysis
DSS was defined as the time from diagnosis to death specifically due to the disease. The Kaplan–Meier (KM) survival curve was used to analyze DSS, and statistical differences in DSS were calculated using the log-rank test. A survival time of 60 months (5 years) was defined as the endpoint for the KM estimate. Statistical analysis of clinicopathological variables was performed using Fisher’s exact or Pearson’s chi-square test. Propensity score matching (PSM) using the cardinality method was used to match 2 groups of people on a one-to-two basis according to their propensity score to calibrate the effects of baseline clinicopathological differences (i.e., age, sex, race, primary site, TNM stage, histological grade, and treatment). Univariate Cox proportional regression analysis was used to calculate hazard ratios (HRs) for the poor DSS. All statistical analyses were performed using R version 4.2.0 (R Foundation for Statistical Computing, Vienna, Austria). Statistical significance was defined as P < .05.
Results
A total of 13,081 patients diagnosed with OPSCC between 2010 and 2016 in the SEER database were identified (Figure 1). A total of 1153 patients with T4 OPSCC (735 HPV-positive and 418 HPV-negative) were enrolled in this study after excluding those with incomplete clinical information.
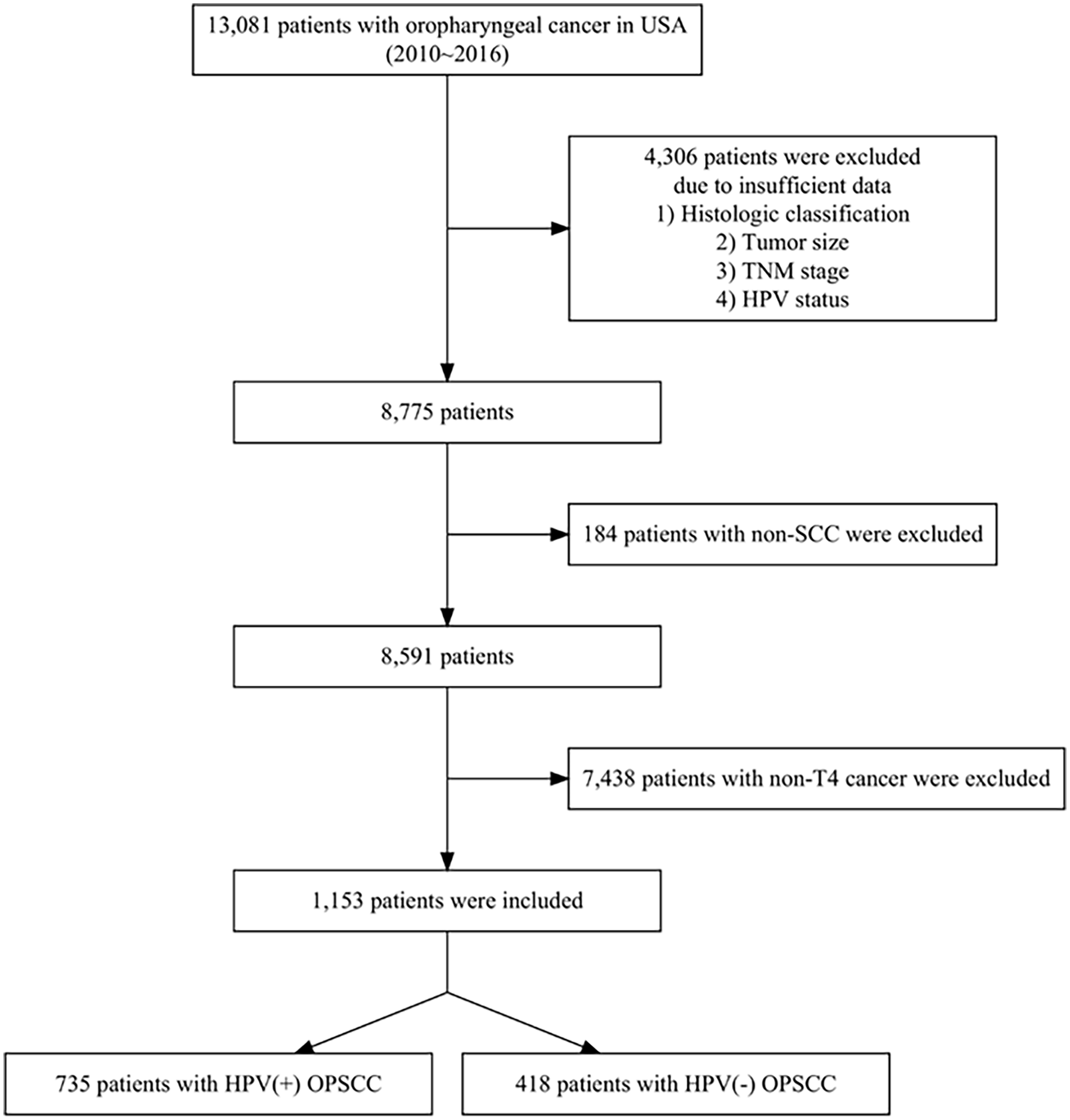
Flowchart of the study population selection. SCC, squamous cell carcinoma; OPSCC, oropharyngeal squamous cell carcinoma; HPV, human papillomavirus.
The detailed characteristics of the enrolled patients are presented in Table 1 (N = 1153). The number of HPV-positive cancers was nearly twice that of HPV-negative cancers (N = 735 vs N = 418). Overall, 65.9% of patients were <65 years and 34.1% were >65 years. The predominant sex was male (84.0% vs 16.0%). Patients were more likely to be white (78.14%), followed by black (12.23%) and others (9.6%). The primary sites of OPSCC were the tonsils and the base of the tongue (43.37% vs 42.32%). Most patients (70.9%) had N2 or N3 neck metastases, and 29.1% had N0 or N1 neck metastases. Distant metastasis was identified in 65 (5.7%) patients. Most cancers showed moderate-to-poor histologic differentiation (well, 4.9%; moderate, 44.7%; poor, 49.1%; undifferentiated, 1.3%). Of the patients, 29.0% were treated with surgery and radiotherapy, and 71.0% were treated with other modalities, such as definitive radiation therapy, chemoradiation therapy, and surgery only. The number of patients classified according to tumor size was 17 (≤1 cm), 55 (1-2 cm), 195 (2-3 cm), 274 (3-4 cm), and 612 (> 4 cm). Patient variables showing statistical differences between HPV-positive and HPV-negative cancers were sex, race, primary site, histologic grade, and treatment modality (P < .001, P < .001, P < .001, P < .001, and P < .012, respectively). However, no statistical differences were observed in neck metastasis, distant metastasis, or tumor size.
Demographic and Clinicopathologic Characteristics in T4 OPSCC.
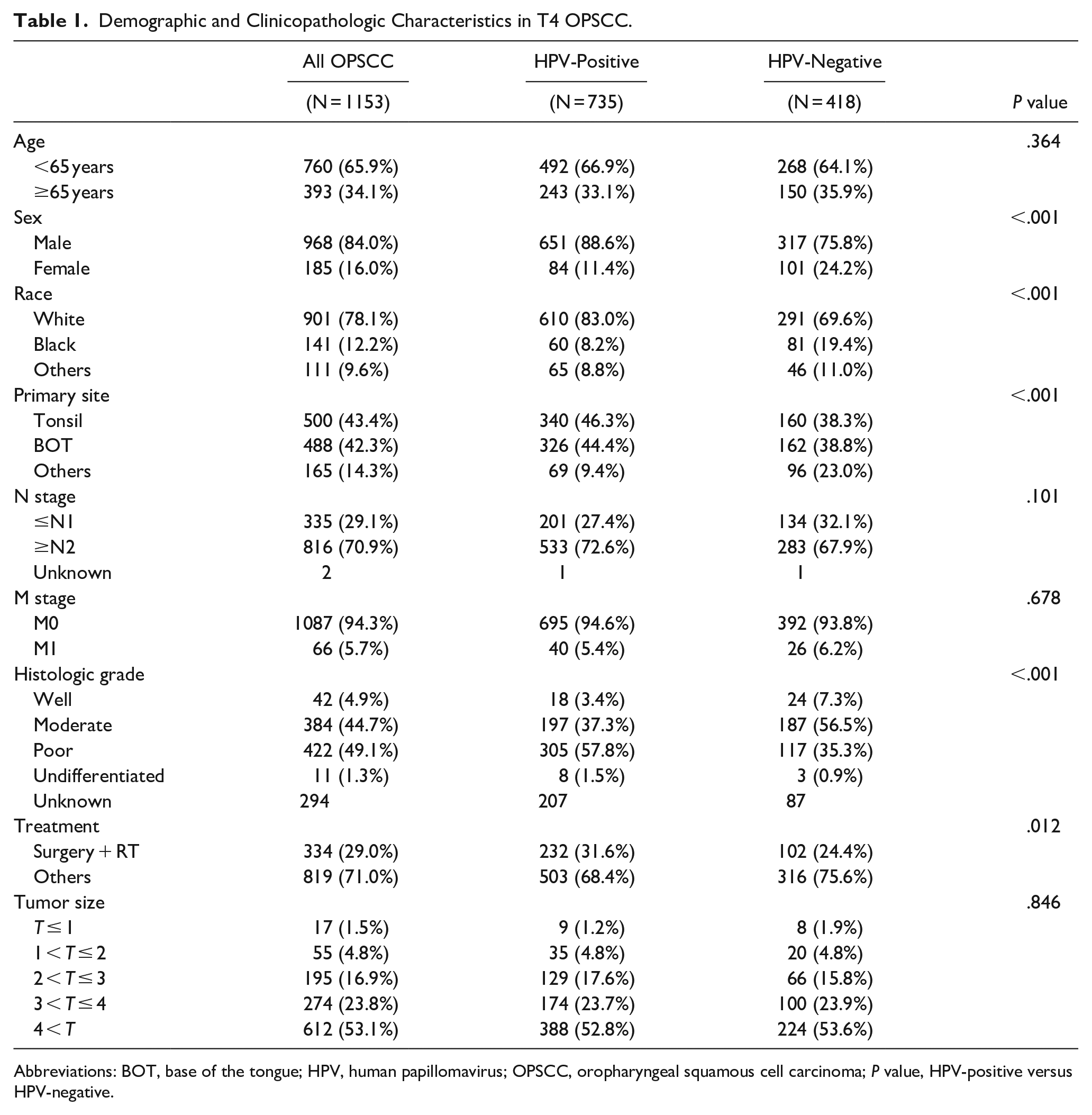
Abbreviations: BOT, base of the tongue; HPV, human papillomavirus; OPSCC, oropharyngeal squamous cell carcinoma; P value, HPV-positive versus HPV-negative.
KM plots according to HPV status and tumor size are shown in Supplemental Figure 1. The 5-year DSS rate of all patients was 55.7%. Tumors 1 to 2 cm (58.1%) had the best 5-year survival rate, followed by 2 to 3 cm (50.0%), 3 to 4 cm (46.9%), >4 cm (40.2%), and ≤ 1 cm (32.3%) (P < .0001*). To determine whether the results differed according to HPV status, we conducted a subgroup analysis by HPV status (Supplementary Figure 1B and C). Tumors ≤1 cm had the worst prognosis in HPV-negative T4 OPSCC; however, this was not the case in HPV-positive cancers.
Tumors >4 cm showed a poor survival rate compared to tumors 1 to 4 cm in size, regardless of HPV infection status (Figures 3A–C). This result was consistent with oncological knowledge that the larger the tumor size, the worse the prognosis. In contrast, tumors ≤1 cm showed a significantly worse survival rate than tumors >1 cm in size only in HPV-negative cancers (P < .001, Figure 2A–C). This result is consistent with our hypothesis that very small T4 cancers may have aggressive potency. Despite the clear statistical significance, only 17 very small T4 cancers were included in this study, of which approximately half were HPV-negative (N = 8, Table 2). Therefore, we selected a trait-matched control group using PSM to reduce the bias caused by the small sample size. A double number (N = 16) control group was selected using PSM to increase the statistical power. After PSM, the statistical differences in patient characteristics between very small tumors and other tumor sizes were reduced in HPV-negative cancers (Table 2). Even after selection bias correction through PSM, the relatively poor survival rate of tumors ≤1 cm in HPV-negative cancers was statistically significant (Figure 2D–F). Therefore, these results indicate that tumors ≤1 cm in size have a poor prognosis in patients with HPV-negative T4 OPSCC.
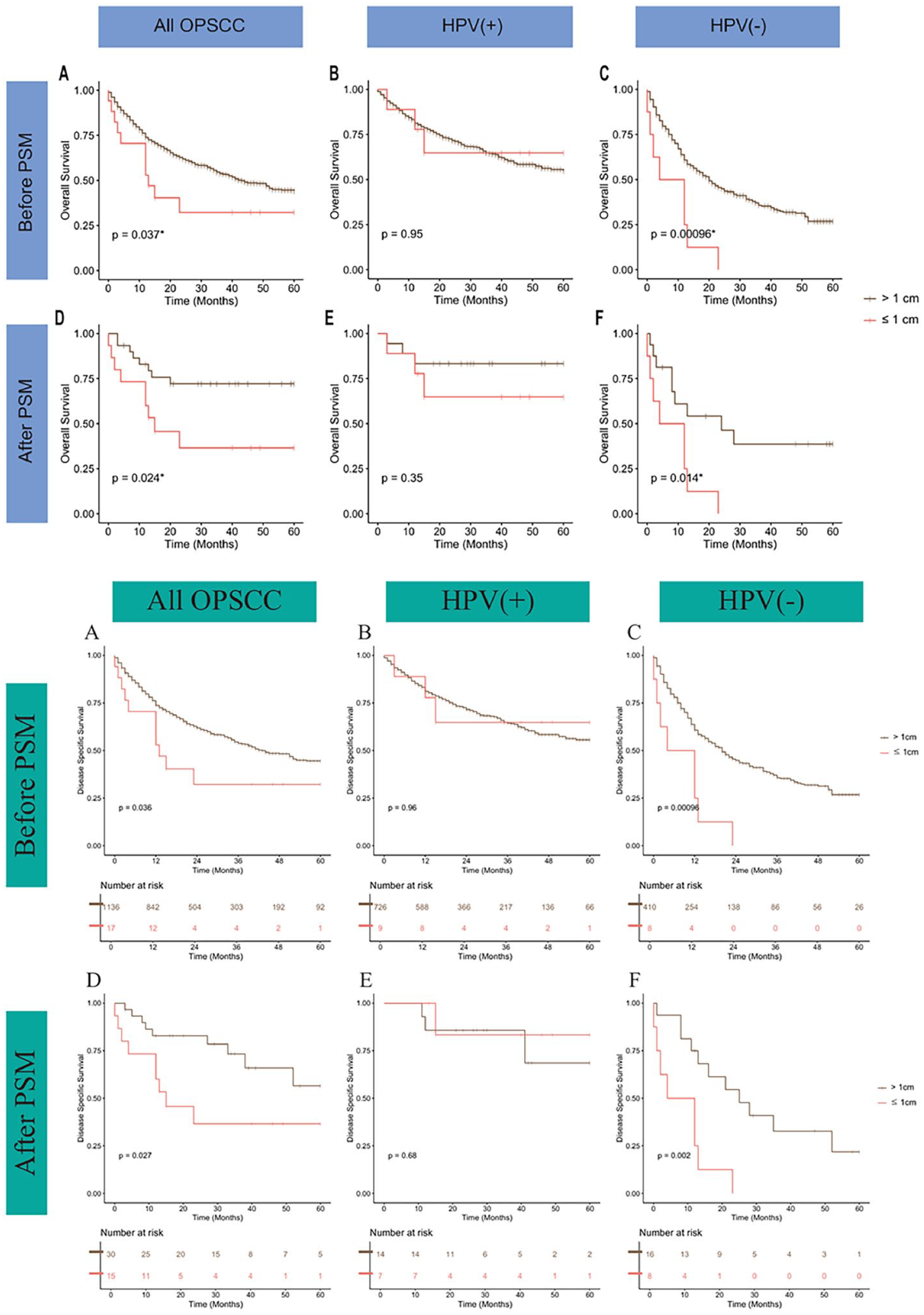
KM survival plots of patients with T4 OPSCC divided by 1 cm. (A and D) all patients; (B and E) HPV-positive patients; (C and F) HPV-negative patients; (A, B, and C) before PSM; (D, E, and F) after PSM. HPV, human papillomavirus; PSM, propensity score matching; KM, Kaplan–Meier.
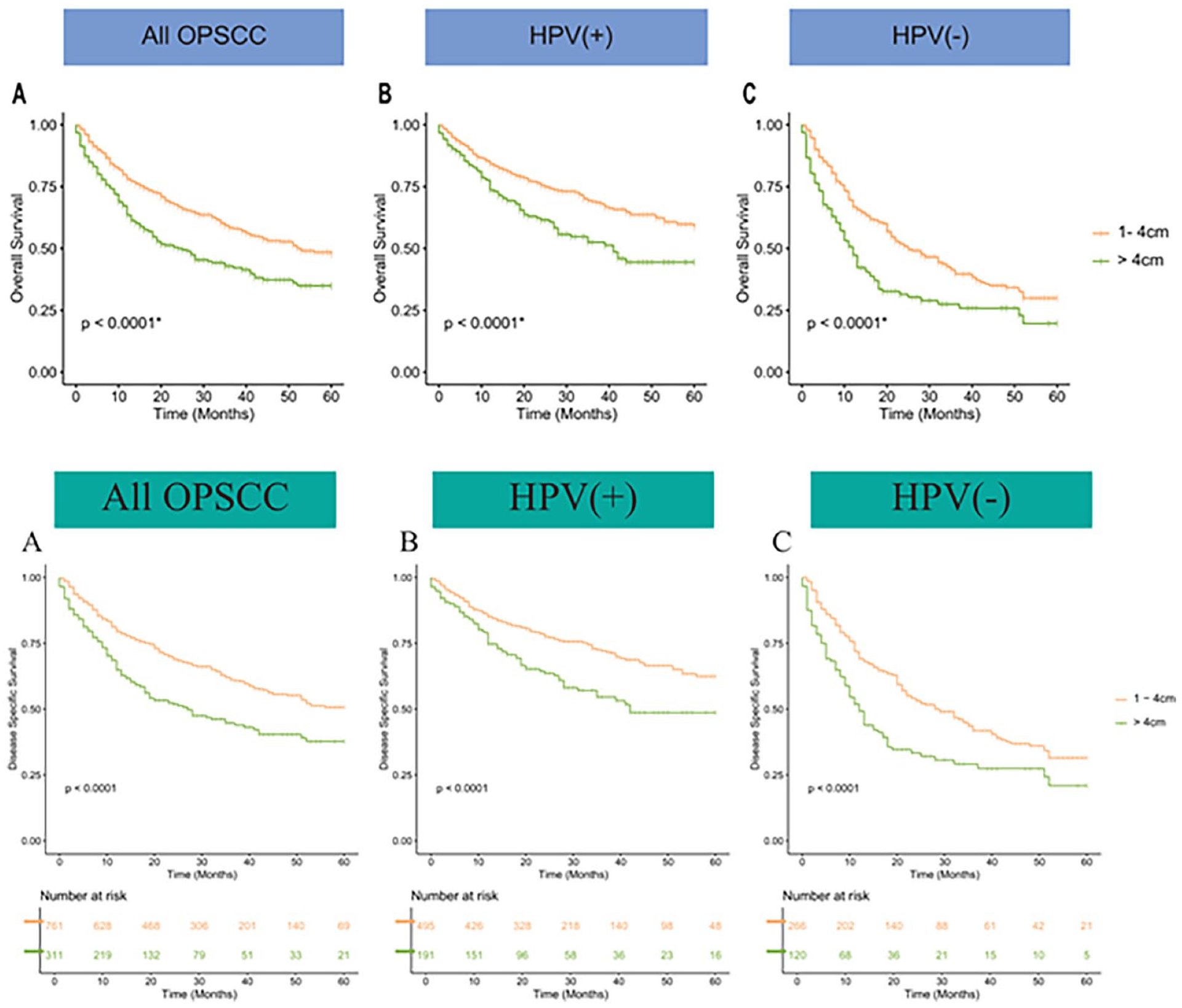
KM survival plots of patients with T4 OPSCC divided by 4 cm. (A) all patients, (B) HPV-positive patients, and (C) HPV-negative patients. HPV, human papillomavirus; KM, Kaplan–Meier.
Characteristics of HPV-Negative T4 OPSCC Divided by 1 cm Before and After PSM.
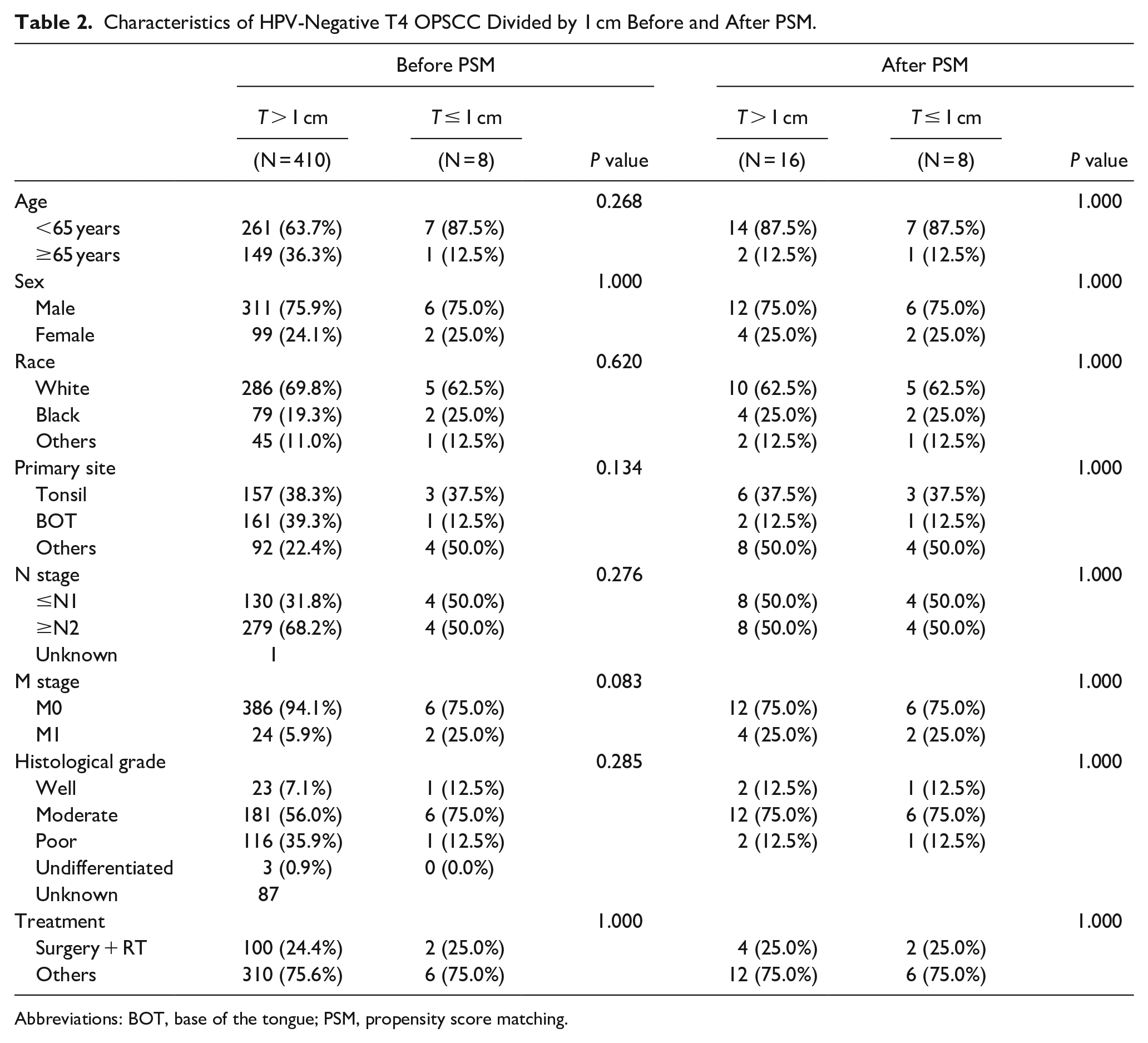
Abbreviations: BOT, base of the tongue; PSM, propensity score matching.
Univariate Cox analysis of patients with HPV-negative T4 OPSCC is shown in Figure 4. Tumors ≤1 cm were the worst prognostic factors among the multiple characteristics of T4 OPSCC (HR = 2.8, P < .006). The HR for tumors ≤1 cm was higher than that for tumors > 4 cm (HR = 1.6, P < .001) and distant metastases (HR = 2.6, P < .001).
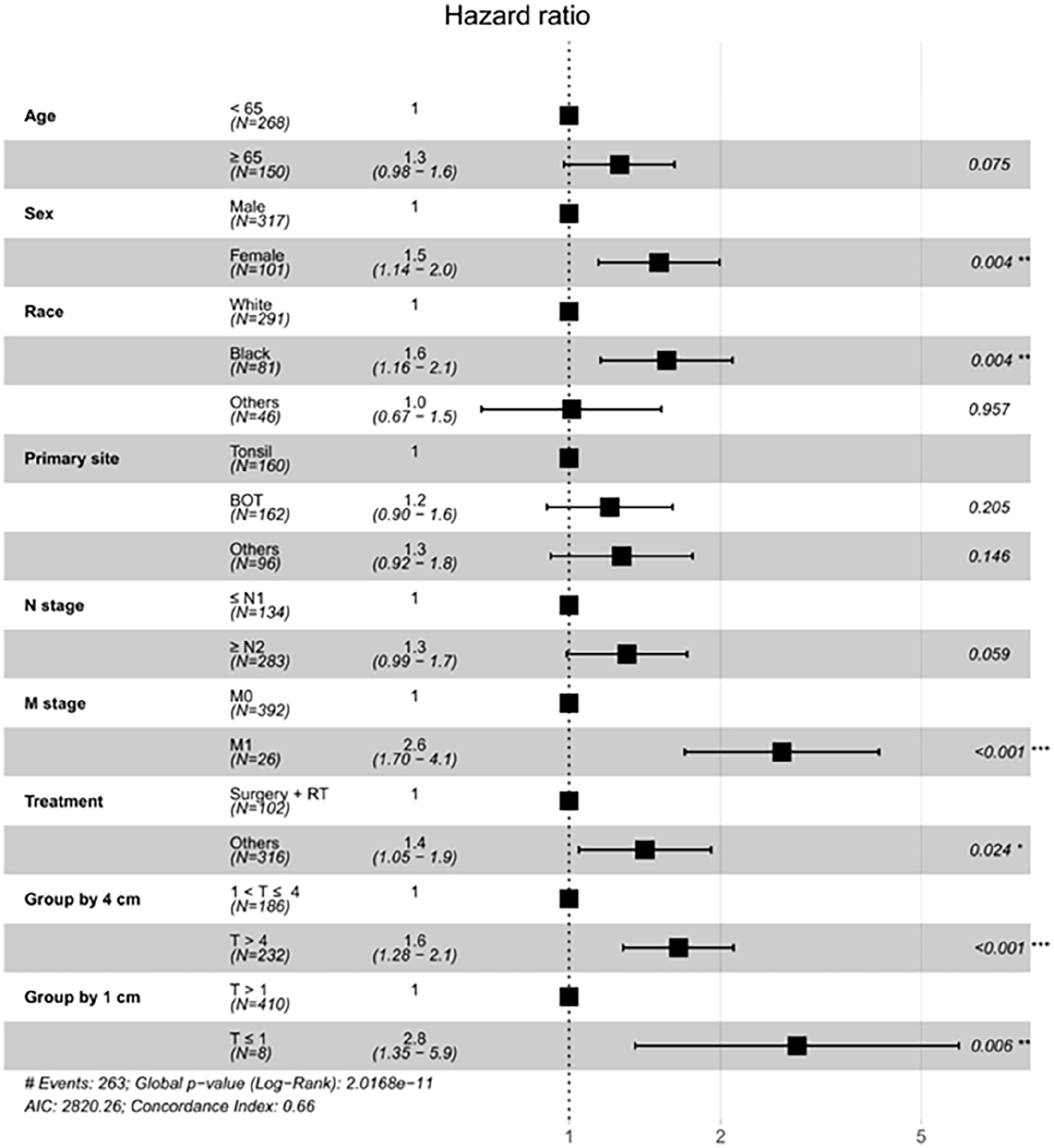
Univariable Cox proportional hazards regression model analysis in HPV-negative T4 OPSCC.
Discussion
We confirmed that tumors ≤1 cm in size were associated with a poor prognosis in patients with HPV-negative T4 OPSCC. In particular, the DSS of tumors ≤1 cm was significantly worse than that of tumors >4 cm in HPV-negative T4 OPSCC. The HR for tumors ≤1 cm was almost equivalent to that for distant metastases. This extraordinary finding conflicts with previous findings suggesting that a smaller tumor size would be associated with better survival.
It is unclear whether these results are limited to our study owing to the large difference between our results and those from previous studies. However, our literature search revealed that other research groups had reported similar results regarding very small tumor sizes in various cancers. A study reported that very small breast cancers with metastatic lymph nodes represent higher cancer-related mortality than large breast cancers. 9 They suggested that a very small tumor size could be a predictive factor for biological aggressiveness. Another study revealed poor outcomes of small pancreatic adenocarcinoma with lymph node metastasis. 11 These findings support that small tumors with the capability to make early lymph node metastasis may have more aggressive biological characteristics.10,13 Based on these similar results, we suggest that early regional invasiveness (T4) of ≤1 cm tumors in patients with OPSCC may be related to poor survival.
This study did not observe a poor prognosis for tumors ≤1 cm in HPV-positive cancers. However, the reason for this result remains unclear. However, we can presume that these results are related to the histopathological and physiological characteristics of HPV-positive OPSCC cases. A characteristic of HPV-positive OPSCC is that the primary tumor size is small (low T) at diagnosis, and lymph node metastasis is common (high N). 14 Nevertheless, in most cases, HPV-positive OPSCC tends to show better DSS than HPV-negative OPSCC. 14 The remarkably good prognosis of HPV-positive OPSCC, regardless of the advanced stage, may be the major reason for our study’s failure to find statistical differences. Recently, the favorable biological behavior of HPV-positive OPSCC is leading to an increasing interest in de-escalation trials. Two trials in this area are the appropriateness of de-escalated adjuvant therapies in HPV-positive OPSCC. The trials, postoperative adjuvant treatment for HPV-positive tumors and evaluation of de-escalated adjuvant radiation therapy for HPV-Associated Oropharynx Cancer, reduced intensity adjuvant treatment in patients who have undergone surgery.15,16 Another Phase II study, NCT03416153, is the appropriateness of initial de-escalated radiotherapy for HPV-positive OPSCC.
The ability of cancer cells to infiltrate adjacent tissues distinguishes them from healthy cells. T4 stage of small tumors indicates early infiltration into the surrounding tissue. Several molecular biomarkers involved in this process have been reported previously. For example, cadherins are cell adhesion molecules that maintain epithelial integrity. A low expression of E-cadherin is related to increased motility of tumor cells allowing surrounding tissue invasion and metastasis. 17 Matrix metalloproteinase (MMP) is an extracellular protease that destroys surrounding tissues and is needed in the invasion and metastasis of tumor cells. 17 High expression of MMP-1 is related to advanced cancer stage and metastatic lymph nodes in OPSCC. 18 There is a potential that these biomarkers might be related to the early infiltration event in ≤1 cm T4 OPSCC. Identifying biomarkers associated with early infiltration is necessary for developing targeted therapies.
This study had some limitations. First, the SEER database may include miscoded or missing data. However, the reliability of the SEER database was randomly applied to individual data. Thus, the risk of misclassification bias is not significantly high. The SEER database does not include detailed treatment information. However, we assumed that adequate treatment would have been administered in most cases. Second, HPV-positive OPSCC shows increased cellular chemo- and radio-sensitivity, particularly in the case of the p16/HPV-DNA combined positivity and in the absence of smoking history, since smoking habits may have impacted the HPV-positive cluster prognosis. 19 As the SEER database did not provide the smoking habits, we could not adjust for this in our study. Despite these limitations of this study, we could analyze rare T4 OPSCC ≤1 cm using SEER big data. Our study provides a reliable risk analysis of tumor size using the SEER database. Nevertheless, our analyses must be repeatedly validated in other databases and interpreted with caution.
This study revealed that very small tumors (≤1 cm) had significantly worse DSS than larger tumors in patients with T4 OPSCC. A deeper understanding of the early infiltration process in very small T4 OPSCC is crucial for the development of more effective and personalized treatments.
Supplemental Material
sj-jpg-1-ear-10.1177_01455613241271686 – Supplemental material for Poor Survival of Patients With Very Small But Locally Advanced (T4) Oropharyngeal Cancer
Supplemental material, sj-jpg-1-ear-10.1177_01455613241271686 for Poor Survival of Patients With Very Small But Locally Advanced (T4) Oropharyngeal Cancer by Jeong Wook Kang, Joo Kyung Noh, Min Kyeong Lee, Yeon Seo Lee, Young Chan Lee, Jung-Woo Lee, Moonkyoo Kong, Seong-Gyu Ko and Young-Gyu Eun in Ear, Nose & Throat Journal
Footnotes
Author Contributions
Jeong Wook Kang has involved in investigation and writing—original draft; Joo Kyung Noh has been involved in visualization; Young Chan Lee has been involved in data curation and methodology; Jung-Woo Lee has been involved in writing—review and editing; Moonkyoo Kong has been involved in writing—review and editing; Seong-Gyu Ko has involved in statistical method design; Young-Gyu Eun has involved in conceptualization, supervision, and writing—review and editing.
Data Availability
All data used in this study were available in the US SEER database. Access must be granted by the National Cancer Database program. Data are also available from corresponding author upon reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Research Foundation of Korea grant funded by the Korean government (MSIT) (no. 2020R1A5A2019413) and by a grant of the Korea Health Technology R&D Project through the Korea Health Industry Development Institute, funded by the Ministry of Health and Welfare, Republic of Korea (grant number: HI20C1205).
Ethics Statement
The study was approved by the IRB of the Kyung Hee University Hospital at Gangdong. The IRB waived informed consent requirements because this study did not use any identifiable individual information.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
