Abstract
In this paper, we described technique of platelet rich plasma injection into the olfactory cleft in a 22-year-old female with 24-month post–COVID-19 anosmia. The technique starts with the blood extraction and the isolation of PRP through a 10-min centrifugation. The supernatant was injected in nasal regions after a local anesthesia through a 0° rigid optic. Several points of .2–.5 mL were performed in the nasal septum in regard of the head of the middle turbine and in the head of the middle turbine in both sides. The baseline threshold, discrimination, and identification scores were 1, 8, and 0, and the Olfactory Disorder Questionnaire score was 51, respectively. The injection of PRP in olfactory cleft was done without complication and mild pain. The patient perception of recovery of smell sense occurred at 3-week post-injection. From this time, the smell sense progressively improved to the 2-month consultation. At 2-month post-injection, the TDI scores reached 16, 16, and 16 (48), while the Olfactory Disorder Questionnaire was 73. The injection of PRP into the olfactory cleft appears to be a safe and easiness new approach that may improve the recovery of smell sense. Future controlled studies are needed.
Keywords
Introduction
Olfactory Dysfunction (OD) affects 1 to 20% of the general population of Western country. 1 The most prevalent causes of OD are sinonasal disorders, post-viral olfactory dysfunction, neurological diseases, and post-traumatic lesions of the olfactory nerve. 1 Since the onset of the coronavirus disease 2019 (COVID-19) pandemic, the prevalence of OD has substantially increased, affecting 30 to 86% of patients.2-4 Depending on the time point of the disease, the OD may include anosmia, hyposmia, phantosmia, or parosmia. 5 To date, there is no treatment of persistent OD. The patients with loss of smell sense or parosmia are advised to adhere to standardized olfactory protocols to increase the recovery probability.6,7
In this paper, we described the injection of platelet rich plasma into the olfactory cleft as an interesting new approach to improve the recovery of smell sense.
Materials and Method
Indications
The following conditions are considered as potential indications: persistent (> 6 months) post-viral anosmia or hyposmia; severe parosmia; recent (<2 years) post-traumatic or idiopathic hyposmia or anosmia or neurological disease related anosmia or hyposmia.
Exclusion Criteria
Patients with acute or chronic sinonasal infection, long-time anosmia or hyposmia (> 2 years), history of nasal cancer or aggressive tumor (e.g., inverted papilloma), or OD related to nasal polyposis. The realization of a nasofibroscopy prior the indication of injection is important to exclude above-mentioned conditions and to explore the olfactory cleft anatomy. The olfactory cleft endoscopy scale has to be scored at 0 8. This scale is a validated scale reporting the findings of discharge, polyps, edema, and scarring or crusting on a scale of 0, 1, or 2 on each side, giving a total score ranging from 0 to 20.
Clinical and Olfactory Outcomes
The French version of the olfactory disorder questionnaire (Fr-ODQ) was completed by patients before and 2-month post-procedure, while otolaryngologist assessed the patient olfaction with threshold, discrimination and identification test. 8 Patient was invited to rate local anesthesia and procedure annoyance and pain with a 10-point visual analog scale ranging from 0 (no pain/annoyance) to 10 (important pain/annoyance). The physician assessed the potential difficulties during the procedure, the occurrence of post-procedure bleeding or post-nasal drip sensation.
Procedure
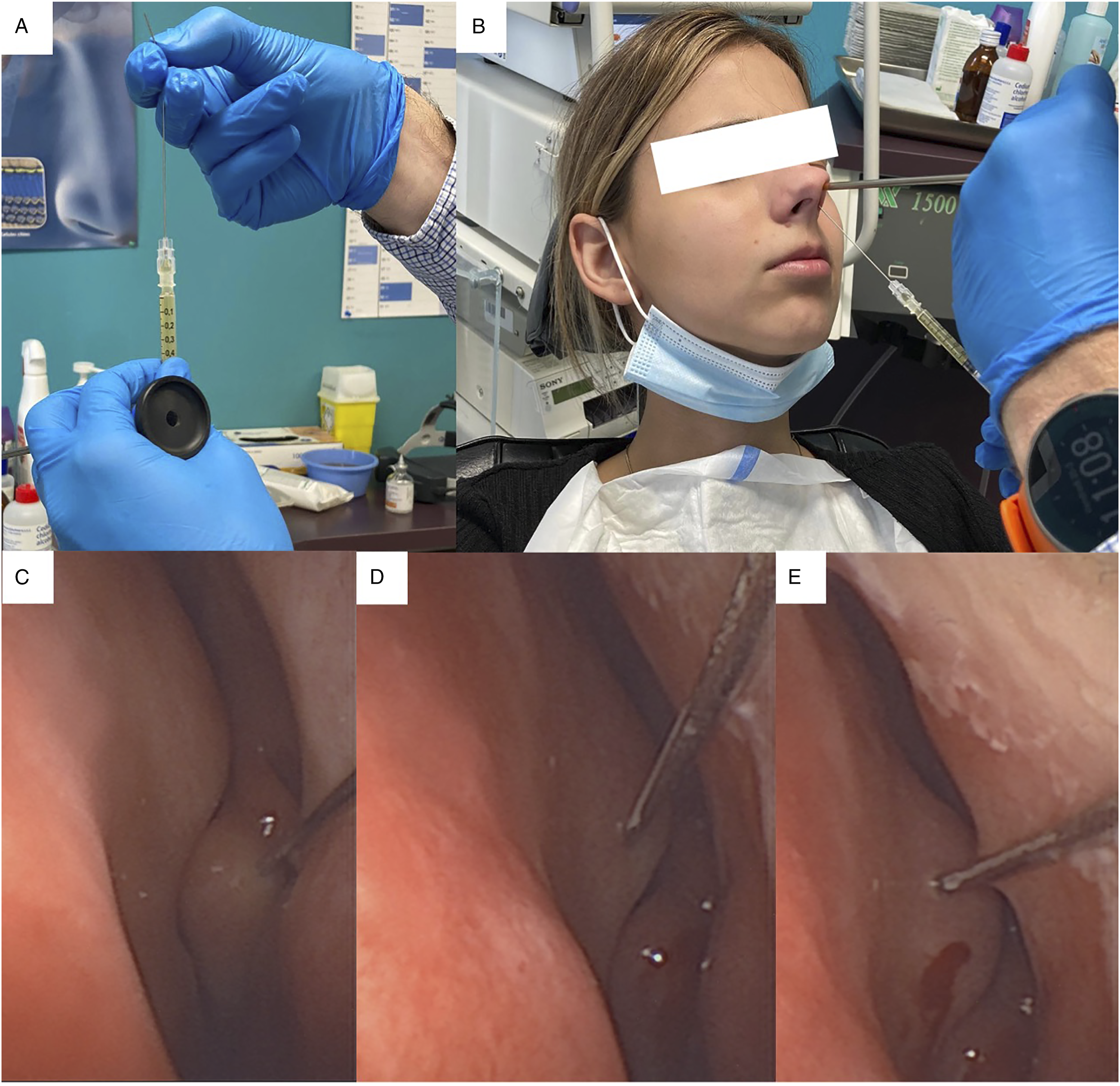
The procedure is described in Figures 1 (A–F) and 2 (A–E). The first step consists of the blood extraction (20 mL; Figure 1A) into a tube with sodium citrate anticoagulant (B), and the isolation of PRP through a 10-min centrifugation at 4200 rpm (C). The supernatant was drawn up into a 10 mL syringes and the otolaryngologist transferred the PRP into a 1 mL syringe (D). A 27-G needle was used for the injection (10 cm; E). The local anesthesia was performed with Xylocain 10% spray 2 minutes after the injection of xylometazoline chlorhydrate drops into the nasal fossae (F). The injection was performed through a 0° rigid optic to guide the needle direction (Figure 2A and B). Note that to have a better access of the anatomical region, we may bend the needle (30°; A). Several points of .2–.5 mL are performed in the middle turbinate (C) and in the nasal septum in regard of the head of the middle turbine, and in the anterior part of the region (D, E). In case of anatomical deviation, the injection is performed closest to the olfactory cleft (D, E). The procedure is similarly performed in the contralateral side. Patient was observed for 15 minutes post-procedure for potential adverse effects and was discharged. Preparation of the platelet-rich plasma and patient. Footnotes: The following steps of the procedure are described: A–B: blood extraction into a tube with sodium citrate anticoagulant; C: centrifugation at 4200 rpm; D: transfer of the PRP into a 1 mL syringe; E: preparation of the 27-G needle for the injection; F: local anesthesia with Xylocain 10% spray 2 minutes after the injection of xylometazoline chlorhydrate drops into the nasal fossae. Procedure. Footnotes: The following steps of the procedure are described: A, B: the injection of the PRP through a bended needle with a 0° rigid optic; C–E: injection of PRP into several points of .2–.5 mL in the middle turbinate (C) and in the nasal septum in regard of the head of the middle turbine and in the anterior part of the region (D, E).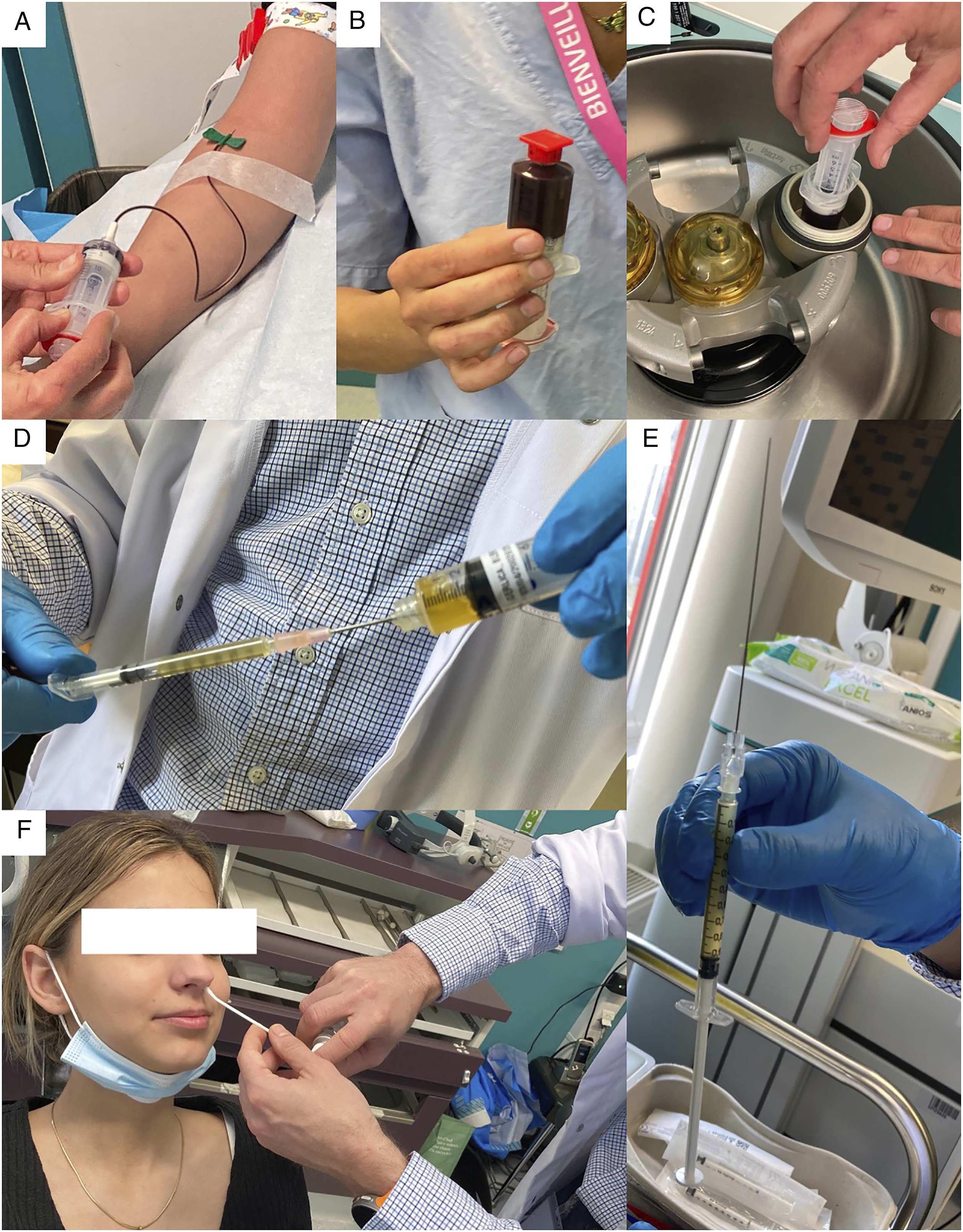
Results
A 22-year-old female with 24-month post–COVID-19 anosmia was recruited for the injection of PRP. The patient medical history was unremarkable. She benefited from 2 doses of vaccine. There was no abnormality in the nasofibroscopic examination and the olfactory cleft endoscopic scale score was 0 9 . The threshold, discrimination, and identification scores were 1, 8, and 0, respectively. The Olfactory Disorder Questionnaire score was 51, with parosmia, life quality statement, and sincerity statement scores of 10, 32, and 9, respectively. The patient benefited from the procedure, which was done without complication. A total of 2.2 mL of PRP was injected in both nasal regions (1.2 mL right, 1.0 mL left). According to the visual analog scale, patient reported pain levels of 2/10 and 1/10 for the blood extraction and the injection, respectively. The local anesthesia with xylocaine 10% was judged as the most annoyance step with a score of 3/10.
The patient perception of recovery of smell sense occurred at 3-week post-injection. From this time, the smell sense progressively improved to the 2-month consultation. At 2-month post-injection, the TDI scores reached 16, 16, and 16 (48), while the Olfactory Disorder Questionnaire was 73 (parosmia: 11; life quality statement: 47; and sincerity statement: 15). Throughout the 2-month post-injection, patient adhered to an olfactory training protocol. 6 The informal consent was signed by patient for this publication.
Discussion
The platelet rich plasma is an old technique used in orthopedic, dermatology, plastic surgery, or rehabilitation to regenerate the injured tissues. In otolaryngology, PRP was used in the management of vocal fold scars, 10 tympanic membrane perforation, 11 or neck fistula, 12 with encouraging results. The first use in olfaction was made in a preliminary case-series by Carol Yan in 2019, 11 while another study is in process in Brussels (not yet published). 14 In this paper, we provide our step-by-step approach to inject PRP into the nasal septum and the middle turbinate; that is, close of the olfactory clefts. The main advantage of this approach is the safety and the easiness of the technique. Because PRP is an autologous biological product derived from the patient blood, there is no reject, risk of disease transmission, or adverse effect. However, the injection has to be performed in the minutes following the end of the centrifugation because there is a risk to coagulate the supernatant. From an anesthesiologic standpoint, the use of xylocaine 10% spray in place of nasal imbibed packing is sufficient and provides adequate pain outcome during the procedure.
Our patient recovered subjectively smell sense 3 weeks after the injection, which matches with the current knowledge about the physiological effect of PRP. Indeed, from a physiological standpoint, the PRP pockets in the mucosa will progressively release anti-inflammatory and pro-regenerative factors of the platelets, leading to the upregulation of some factors by the cells of nasal and olfactory tissues, including growth and transforming factors, vascular endothelial growth molecules, epidermal growth factor, and insulin-like growth factor. 15 Interestingly, some studies supported that PRP may promote axon regeneration and neuroregeneration. 16 The use of PRP in COVID-19 OD is particularly interesting because it has been suggested that the persistence of the virus in the olfactory region and the associated inflammation of the neuroepithelium may account for prolonged or relapsing symptoms of COVID-19, including loss of smell. 17 In that way, the potential anti-inflammatory effect of PRP may reduce the chronic inflammation and cell-related injuries, promoting the regeneration of the olfactory tissues. Because the OD related to COVID-19 is more severe than many other viral OD, 18 future studies are needed to explore the effectiveness of PRP injection in olfactory cleft of large cohort of patients with OD and to better understand the mechanisms of the PRP in this population.
Conclusions
The injection of platelet rich plasma into the olfactory cleft and middle turbinate of patients with COVID-19 related OD may be a promising safe and easiness approach to improve the olfactory recovery process. Future large cohort randomized controlled studies are needed to confirm the effectiveness of this approach.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
