Abstract
Importance
Most French Olfactory Questionnaires are time consuming, which may affect the patient participation.
Objective
To validate a short French version of the Olfactory Disorders Questionnaire (Fr-ODQ).
Design
Prospective controlled study.
Setting
Multicenter study.
Participants
Patients with long-lasting olfactory dysfunction (OD) treated with platelet-rich plasma into the olfactory clefts and asymptomatic subjects.
Intervention
Development and validation of a short ODQ.
Main Outcomes
Individuals completed the full Fr-ODQ. The Threshold, Discrimination, and Identification (TDI) test was performed in OD patients. A combined statistical analysis was performed to determine the most informative items of the Fr-ODQ to develop a shorter version. The internal consistency was determined with Cronbach’s alpha. The reliability and external validity were evaluated through a test-retest approach and by correlating with the Fr-ODQ. Both the minimal clinically important difference (MCID) and the threshold of the short ODQ version were determined.
Results
A total of 263 patients (173 [65.8%] females) and 129 controls (92 [71.3%] females) completed the evaluations. The mean age of patients was 51.2 ± 15.3 years. The mean duration of OD was 42.4 ± 54.3 months. The biostatistical models selected 10 essential items composing the Fr-ODQ-10. The baseline Fr-ODQ-10 was significantly correlated with the TDI (rs = 0.228; P = .001) and the Fr-ODQ (rs = 0.875; P = .001), demonstrating high external validity. Fr-ODQ-10 was significantly lower in controls compared to OD patients (P = .001), highlighting high internal validity. The internal consistency was good (α = .796). The external consistency was adequate, with significant correlations between the test-retest Fr-ODQ-10. The Fr-ODQ-10 significantly decreased from baseline to 4 months post-treatment. A Fr-ODQ-10 score >7.5 was considered abnormal (sensitivity: 84.8%, specificity: 84.2%). The MCID of Fr-ODQ-10 was established at >3.
Conclusion and Relevance
The Fr-ODQ-10 is a valid and reliable clinical instrument, demonstrating correlation with the psychophysical olfactory assessment.
Introduction
Olfactory Dysfunction (OD) may affect 2.7 to 22.2% of the general population. 1 OD substantially affects the patient’s quality of life, with an increase in mental health disorders, depression, and anxiety in patients suffering from OD.2,3 The primary causes of OD include sinonasal disorders, post-viral OD, neurological diseases and post-traumatic lesions of the olfactory nerve. 4 The prevalence of OD significantly increased throughout the coronavirus disease 2019 (COVID-19) pandemic, with approximately one-third of patients reporting long-lasting OD.5 -7 The development of a valid and reliable patient-reported outcome questionnaire (PROM) assessing OD and the related impact on quality of life is therefore warranted to improve both the baseline and follow-up evaluations of OD patients. In 2005, Frasnelli and Hummel developed the Olfactory Disorder Questionnaire (ODQ), which is a 34-item PROM reporting features of OD and the related impact on QoL. 8 In 2022, a French version of ODQ (Fr-ODQ) was validated and is used in our clinical practice. 9 Based on our experience of more than 2000 patients, the Fr-ODQ has proven to be a valid and reliable PROM; however, its length—comprising 34 questions—can be time-consuming and may negatively impact patient compliance and willingness to complete the ODQ at each visit.
The objective of this study was to validate a short version of the Fr-ODQ, including the most predictive items for olfactory function change according to psychophysical olfactory testing.
Methods
Ethical Statement
The study protocol was approved by the Institutional Ethics Committee (n° CHUSP20032020). Patient and healthy individual informed consent was obtained.
Patients and Settings
The study protocol was approved by the Institutional Ethics Committee (n° CHUSP20032020).
Patients were recruited from 2 medical centers between January 2021 and February 2025 and informed consent was obtained to participate. The OD consisted of long-lasting (>6 months) quantitative or qualitative impairments related to viral infection or head trauma. Patients with severe neurological diseases limiting the understanding of the study protocol or those who were not native French-speakers were excluded. A control group of healthy subjects was composed, matching the age and gender of the study group. Controls had no neurological, otolaryngological (sinonasal) history, current post-viral OD, or general disorders that could impact olfaction.
Olfactory and Nasal Evaluations
Participants completed the Fr-ODQ. Psychophysical olfactory evaluations were performed with the Threshold, Discrimination and Identification test (TDI; Medisense, Groningen, Netherlands), a standardized and validated evaluation of olfaction. Normative studies reported that anosmia was defined as a TDI score ≤16 points, and hyposmia was defined as a TDI score ≤30.75. TDI > 30.75 was considered normosmia. 10 The clinical response was based on the minimal clinically important difference (MCID) of the TDI (5.5 points). 11
Questionnaire Development
The short version of the French ODQ was developed after the collection of the Fr-ODQ (full version) by patients and controls. This approach was chosen to ensure a comprehensive database including all Fr-ODQ items, which are necessary to determine the most appropriate items to keep in the short version.
The validation processes were conducted by a multidisciplinary team including otolaryngologists of the olfactory research group of young otolaryngologists of the international federation of otorhinolaryngological societies (YO-IFOS), a psychologist, a linguist and 2 statisticians. The team used a database of patients who prospectively completed the Fr-ODQ from pre- to 4-month post-injection of platelet-rich plasma (PRP) into the olfactory cleft 12 for long-lasting OD. All patients were prospectively followed with both Fr-ODQ and the TDI. 10 From this database, biostatisticians used Random Forest analysis to assess the relative importance of each ODQ item in predicting olfactory PRP treatment success, defined as clinically significant improvement (Z ≥ 6, where Z = TDI1-TDI0). Three separate models were trained to examine the temporal and structural dimensions of prediction, including pre-treatment data (identifying the predictive factors present before the intervention that can anticipate significant improvement after treatment), post-treatment data (identifying items which, after treatment, distinguish patients who responded well from those who showed little or no improvement), and post-pre difference data (based on the difference between post-treatment and pre-treatment responses, making it possible to identify the items whose evolution is most strongly associated with clinical improvement).
Additionally, multiple complementary statistical approaches were used, including the Shapiro-Wilk tests assessing normality of distributions and guiding the choice of non-parametric tests; the Wilcoxon paired tests identifying items demonstrating significant pre-post treatment changes; Exploratory Graph Analysis (EGA) applied community detection algorithms to identify underlying dimensional clusters of related items; Extended Bayesian Information Criterion Graphical LASSO (EBICglasso) estimated sparse partial correlation networks with regularization to reveal robust connections between items; Unique Variable Analysis (UVA) detected items that were either redundant or failed to coherently integrate into identified dimensions; and Centrality measures quantified each item’s importance within the network structure. This multi-method approach enabled the selection of items with optimal psychometric properties, sensitivity to change, and clinical relevance. Based on these successive analyses, the Fr-ODQ items were categorized as “essential,” “optional,” and “unimpactful.” Only the essential items were retained for composing the short version of the Fr-ODQ. The final version of the Fr-ODQ-10 is provided in Figure 1. The 10 selected items are rated through a 4-point Likert-scale ranging from “Strongly disagree” to “Strongly agree.” Figure 2 proposed an English version, which needs to be validated.

The Fr-ODQ-10. Fr-ODQ-10, French version of the Olfactory Disorders Questionnaire; OD, Olfactory dysfunction.
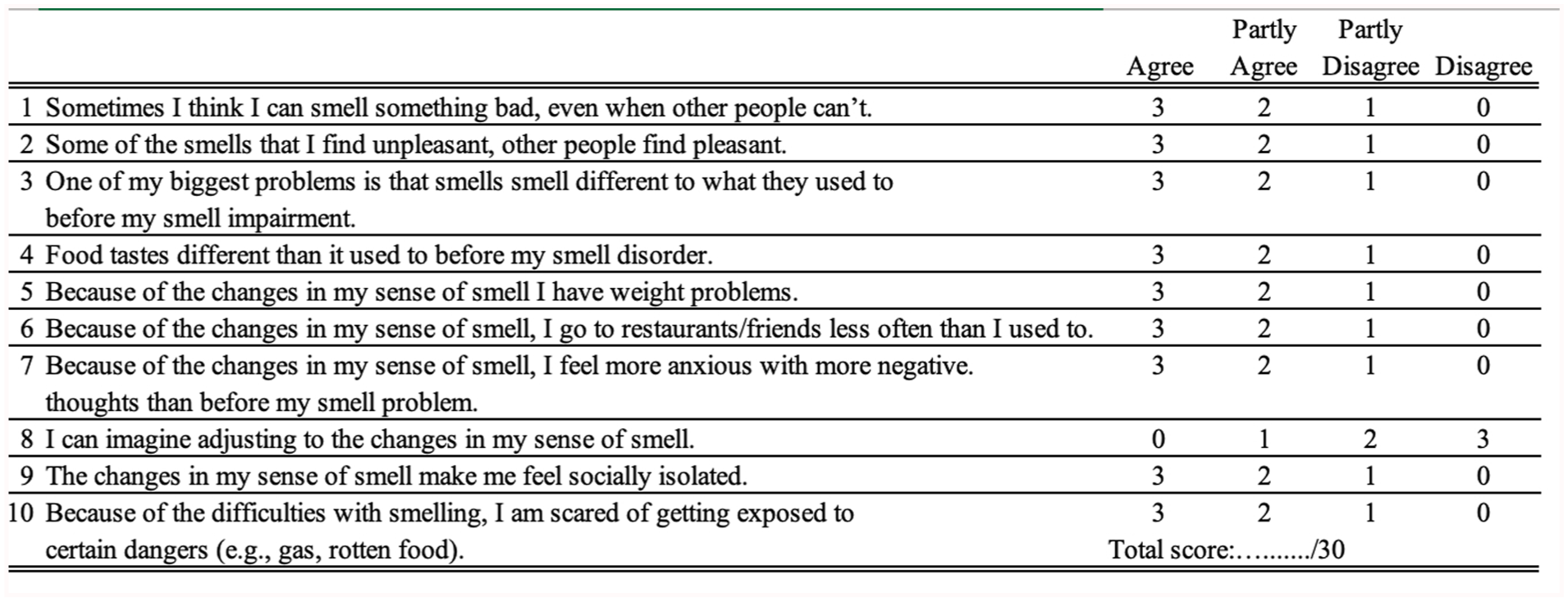
An English version of the ODQ-10. Fr-ODQ, French version of the Olfactory Disorders Questionnaire; OD, Olfactory dysfunction; ODQ, Olfactory Disorders Questionnaire.
Validity, Reliability and Responsiveness to Change
The statistical analyses were performed with Statistical Package for the Social Sciences for Windows (SPSS version 29.0; IBM Corp, Armonk, NY, USA). The Shapiro-Wilk test was applied to all items, revealing a non-normal distribution that indicated the use of non-parametric tests. Only the essential items of Fr-ODQ, composing the short Fr-ODQ-10, were kept for the validity and reliability analyses. The test-retest reliability was evaluated through a correlation analysis between the baseline (test) and 7 day (re-test) completed short Fr-ODQ-10 with the Spearman correlation coefficient. The internal consistency was assessed with Cronbach’s alpha. External validity was assessed by a correlation analysis between the Fr-ODQ-10 and the full version of Fr-ODQ (Spearman correlation coefficient). The internal validity was assessed with a comparison of the Fr-ODQ-10 scores between patients and healthy individuals (Mann-Whitney U test). The pre- to post-treatment (PRP injections into the olfactory clefts) changes of the Fr-ODQ-10 were evaluated with the Wilcoxon Rank test to assess the responsiveness to change. Only patients with an improvement of TDI scores (MCID = 5.5 points) 4 months after the PRP injections were selected for the responsiveness to change analysis. 11 From this information, the MCID of the Fr-ODQ-10 was determined using the standardized effect size approach, calculated as half the standard deviation of baseline scores (0.5 × Standard deviation), a widely accepted threshold representing a moderately perceptible clinical change. The threshold suggesting a pathological score was calculated with the receiver operating characteristic (ROC) curve, considering data of OD patients and controls. A level of significance of P < .05 was used.
Results
Patients and Settings
A total of 263 patients (173 female, 65.8%) and 129 controls (92 female, 71.3%) completed the evaluations. The mean age of patients was 51.2 ± 15.3 years. Demographic, comorbidity, clinical and etiological data for the patient cohort are summarized in Table 1. The most common comorbidities among patients were hypertension (19.4%), hypercholesterolemia (19.0%), and hypothyroidism (14.8%). The predominant cause of OD was post-viral (77.9%), with smaller proportions attributed to idiopathic (4.6%) and post-traumatic (4.2%) etiologies. The mean duration of OD was 42.4 ± 54.3 months. The majority of patients had adhered to an unsuccessful olfactory training protocol before being injected with PRP (77.9%). The intake of zinc (34.6%), B12 vitamins (23.2%), and A vitamins (10.3%) was the additional protocols considered by patients before PRP therapy (Table 1). The baseline Fr-ODQ of patients was 36.8 ± 14.1. Patients and controls did not differ in gender and age range characteristics.
Epidemiological and Clinical Outcomes of Patients.
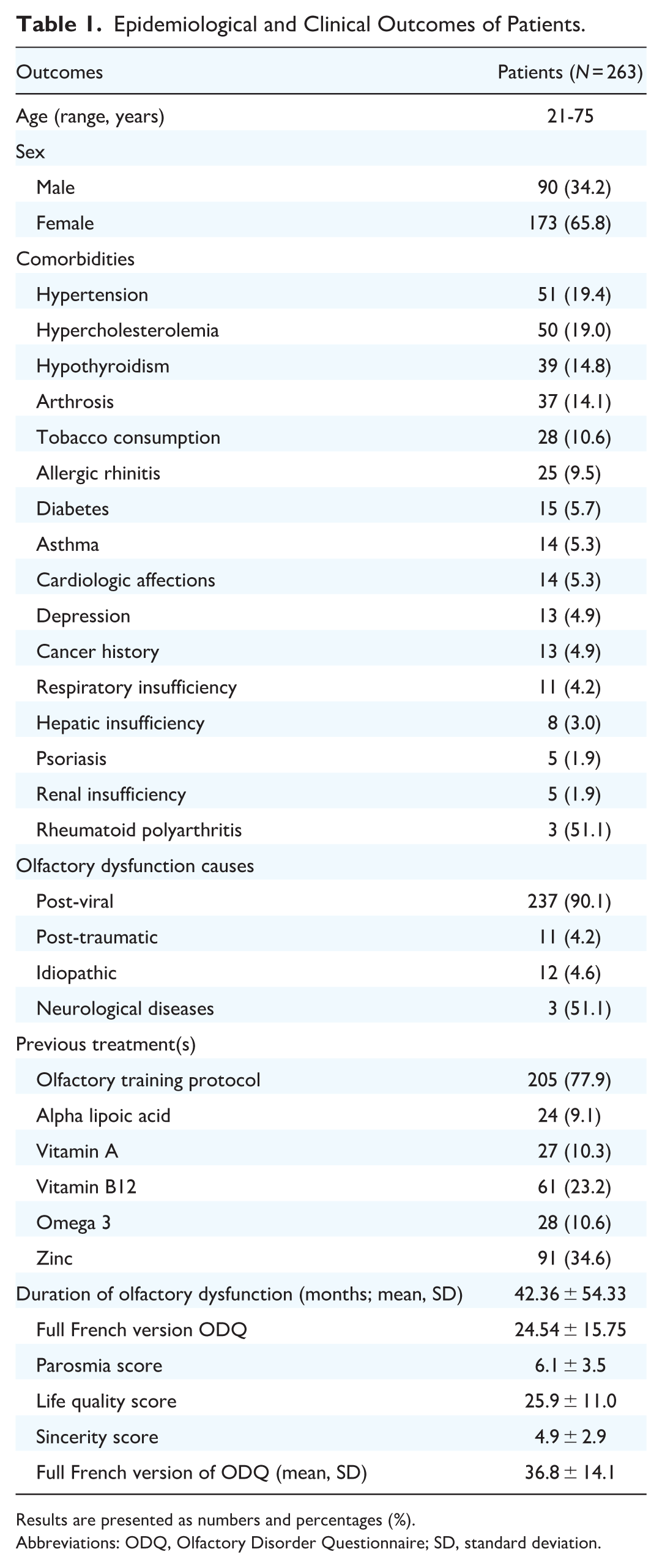
Results are presented as numbers and percentages (%).
Abbreviations: ODQ, Olfactory Disorder Questionnaire; SD, standard deviation.
Development of Short Fr-ODQ-10
The Random Forest models, EBICglasso, EGA and UVA combined analyses led to the identification of 10 essential and 10 optional items, which are specific to 1 to several models, highlighting the value of combining several analysis approaches (initial state, final state, evolution) to capture the different dimensions of patient-related olfactory experience and response to treatment (Table 2). The Bayesian and random forest analysis-based relevance for including the 10 essential items in the short version of ODQ is described in Table 2. The Fr-ODQ-10 and a potential English version (ODQ-10) are described in Figures 1 and 2, respectively. The total score of Fr-ODQ-10 ranges from 0 (no OD/impact on quality-of-life) to 30 (severe OD with significant impact on quality-of-life).
Biostatistical Analysis Results.

Abbreviations: EBICglasso, Extended Bayesian Information Criterion Graphical LASSO; EEU, Combined EGA/UVA/EBICglasso; EGA, Network analysis; OD, Olfactory dysfunction; RFM, Random forest method; UVA, Unique variable analysis; +, Present.
Validity, Reliability, Responsiveness to Change, and Associations
The Fr-ODQ-10 was significantly correlated with the Fr-ODQ (rs = 0.875; P = .001), demonstrating high external validity. No significant association was found between full Fr-ODQ and TDI. The internal validity analysis is reported in Table 3. The significantly higher Fr-ODQ-10 score compared to healthy controls demonstrated adequate validity. Among item analyses, 1 item (“Some of the smells that I find unpleasant, other people find pleasant”) did not report a significant difference with controls, but random effect and Bayesian analyses suggested a significantly predictive value on the post-treatment outcomes (Table 2). The Cronbach’s alpha was .796 (95% Confidence interval [CI]: 0.764-0.826), which consisted of good internal consistency (>.700). Test-retest reliability findings are reported in Table 4, which support adequate external consistency. The mean TDI score significantly increased after 4 months post-PRP injections. Similarly, the mean Fr-ODQ-10 significantly decreased after treatment, which supported adequate responsiveness to change property (Table 3).
Comparison of Olfactory Questionnaire Between Patients and Healthy Individuals.

Abbreviations: ODQ-10, olfactory Disorder Questionnaire-10; PRP, Platelet-rich plasma.
Test-Retest Reliability.

Abbreviations: ODQ-10, olfactory Disorder Questionnaire-10.
The Spearman correlation coefficient analysis reported that there was a mild but significant association between the baseline Fr-ODQ-10 and the baseline (rs = 0.228; P = .001) and post-treatment (rs = 0.262; P = .001) TDIs. Moreover, there was a significantly positive association between the baseline Fr-ODQ-10 and the post-treatment Fr-ODQ-10 (rs = 0.663; P = .001).
MCID and Fr-ODQ-10 Threshold
The ROC curve analysis is available in Figure 3. The area under the curve was 0.896 (95% CI: 0.864-0.928). A Fr-ODQ-10 >7.5 was considered abnormal (Youden’s Index), which was associated with a sensitivity of 84.8% and a specificity of 84.2%. The MCID of Fr-ODQ-10 was established at >3. After 1 PRP injection, 123 (46.8%) patients achieved the TDI MCID (5.5 points), and 84 (31.9%) patients achieved the Fr-ODQ-10 MCID.

ROC curve analysis. Fr-ODQ, French version of the Olfactory Disorders Questionnaire, ROC, receiver operating characteristic.
Discussion
The development of short and time-saving PROMs is important for patient participation in personalized medicine and clinical studies. 13 In this study, we introduce the Fr-ODQ-10—a short form of the French ODQ—created through a rigorous, multidimensional statistical approach. The Fr-ODQ-10 demonstrates strong validity and reliability for French-speaking populations. The development of such practical PROMs is important regarding the increase of OD in the general population, which may be attributed to several factors, including the pandemic sequelae, the aging of Western countries’ populations, and the increase of some disorders associated with OD.4,14
The internal validity of Fr-ODQ-10 is high based on significantly higher Fr-ODQ-10 scores in OD patients compared to controls, corroborating the findings of the German and Portuguese versions of ODQ, where authors demonstrated significantly higher scores of ODQ in OD patients compared to control groups.8,15 In the present study, the external validity of Fr-ODQ-10 was reported as high through the strong positive association with the validated Fr-ODQ (rs = 0.875). In the German ODQ, Frasnelli and Hummel evaluated the external validity of ODQ through a correlation analysis with the Beck Depression Inventory and Mood Inventory. 8 They reported mild-to-moderate but significant associations between all questionnaires, ranging from 0.18 to 0.47, which supported an adequate external validity. Langstaff et al. did not find significant external validity by comparing TDI and ODQ scores. 16 The comparison/association between ODQ and psychophysical olfactory assessment was similarly performed by Frasnelli and Hummel, who showed a weak but significant correlation between the QOD-NS and the TDI score (r = −0.15, P = .034). 8 In the present study, the baseline Fr-ODQ-10 was significantly correlated with the TDI (rs = 0.228; P = .001), which may be attributed to the robust statistical approach used that considered the association between ODQ items and TDI scores to determine the essential items of Fr-ODQ-10.
Internal consistency refers to the extent to which items within a PROM correlate with each other, demonstrating they collectively measure the same underlying construct. The Cronbach alpha value of Fr-ODQ-10 was .796, which is comparable to the French and German ODQ versions exhibiting α = .827 and α between .54 and .93 for negative and positive statements, respectively,8,9 while the internal consistencies of the English and Italian versions of ODQ were significantly higher (α = .90).16,17 From psychometric and biostatistical standpoints, the Fr-ODQ-10 Cronbach’s alpha of .796 was not surprising, because it reflects a deliberate balance between brevity and psychometric integrity. Our rigorous statistical approach prioritized discriminative items with high diagnostic and prognostic values (predictive value of Fr-ODQ-10 on pre- and post-treatment TDI) rather than maximizing internal consistency alone, which often occurs through item redundancy in longer questionnaires, such as the German, English, and French full versions of ODQ. Although this explanation can be extended to test-retest reliability analysis, the Fr-ODQ-10 reported adequate item and total score test-retest reliability, with correlation coefficients being >.6 for most items and total scores. These values are comparable with those found in the German (0.71 and 0.78 for negative and positive statements) and English (0.56 to 0.77) ODQs.8,16 The test-retest reliability of both Italian and French ODQ full versions was higher, ranging from 0.944 to 0.969.9,17
Responsiveness to change is a key psychometric parameter in the validation of a PROM, describing the capability of the PROM to highlight therapeutic changes. 18 Because there was no effective treatment for many ODs, including post-viral, post-traumatic, and idiopathic OD, for a long time, the responsiveness to change of most olfactory PROMs was not assessed.
The development of PRP injections into the olfactory clefts and the related promising results for COVID-19 and non-COVID-19 post-viral and post-traumatic OD,19 -23 made possible the assessment of responsiveness to change and the MCID of Fr-ODQ-10. Although the Fr-ODQ-10 was evaluated after a single PRP injection, statistical analysis revealed a significant reduction in Fr-ODQ-10 scores 4 months after the injections. The threshold of Fr-ODQ-10 for identifying potential OD associated with quality-of-life impairments was determined to be 7.5, and the MCID was evaluated at 3.04, which renders the Fr-ODQ-10 directly usable in clinical practice. Similarly to Fr-ODQ-10, the full version of Fr-ODQ reported adequate responsiveness to change. 9
The primary limitations of this study are the low number of asymptomatic individuals and the lack of TDI in this population. The lack of TDI in control group may lead to the inclusion of patients with subtle OD and related symptoms included in the Fr-ODQ-10. The establishment of a control group of individuals with normal TDI (>30.75) is mandatory in future studies assessing the psychometric properties of Fr-ODQ-10 or other versions in order to improve the internal validity analyses. Moreover, the high proportion of post-viral OD patients may have influenced the validity and reliability outcomes, with different validity and reliability properties for patients with chronic rhinosinusitis with nasal polyps, rhinitis, and other etiologies. However, the primary inclusion of post-viral OD patients reflects the current anosmic and hyposmic populations of Western countries. The German and English versions of ODQ were both validated before the pandemic and, therefore, included more patients with non-COVID-19 OD. Contrarily, the Portuguese, Italian, and French ODQ were validated after the pandemic, including a greater proportion of post-viral patients. Note that in the present study, an English translation was proposed by our group (Figure 2). However, further validation is still required before potential use in clinical practice.
Future studies are needed to evaluate the reliability of Fr-ODQ-10 in patients with chronic rhinitis, rhinosinusitis, posttraumatic, or idiopathic ODs. Indeed, while these conditions may be associated with OD and related impaired quality-of-life, they may share different pathophysiological mechanisms underlying potential differences in OD clinical presentation. Moreover, the olfactory recovery process in rhinitis and rhinosinusitis may lead to different ‘responsiveness to change’ findings of Fr-ODQ-10. Large cohort study with several disease subgroups may refine the use of the Fr-ODQ-10, while determining the importance of some specific outcomes.
The main strengths of this study are the evaluation of the responsiveness to change and a robust statistical approach to select the essential items of the Fr-ODQ full version, leading to the construction of a time-saving, reliable, and valid PROM for clinical otolaryngology practice. Concerning the responsiveness to change features, future studies are needed to investigate this parameter after several PRP injections, which appear to be associated with better recovery than 1. 24
Conclusion
The Fr-ODQ-10 is a short, valid and reliable PROM for assessing the olfactory dysfunction and the related impact on quality-of-life of French speaking patients.
Footnotes
Acknowledgements
Roxane Geerts and the Audiology team for the Sniffin-stick tests. S. Soylu for the database of controls.
Author Contributions
Jérôme R. Lechien, Giovanni Briganti, Antonino Maniaci: design, acquisition of data, data analysis & interpretation, drafting, final approval, and accountability for the work; final approval of the version to be published; agreement to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. Leigh Sowerby, Justin Michel, Thomas Radulesco, Luigi Vaira, Lea Lamoulen, Floren Carsuzaa, Sven Saussez, Eishaan Kamta Bhargava, Giovanni Cammaroto, Giannicola Iannella, Maria R. Barillari, Paolo Boscolo-Rizzo, Giovanni Salzano, Nicolien Vander Poel, Miguel Mayo-Yanez, Alberto M. Saibene, Kalamkas Sagandykova (Yo-IFOS Group): data analysis & interpretation, drafting, final approval, and accountability for the work; final approval of the version to be published; agreement to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Availability of Data and Materials
Available on request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethics Committee
The study protocol was approved by the Institutional Ethics Committee (n° CHUSP20032020). Patient and healthy individual informed consent was obtained.
Consent for Publication
Patient and healthy individual informed consent was obtained.
