Abstract
Objective
Adenoid hypertrophy (AH) is one of the common childhood diseases. Surgical and non-surgical treatment of AH in children is planned according to the severity of symptoms and associated complications. In recent years, treatment methods with intranasal sprays have been reported quite frequently in uncomplicated cases. We aim to evaluate the effectiveness of a new combination of azelastine – fluticasone (AZE-FLU) (137mcg azelastine and 50mcg fluticasone) nasal spray in children with uncomplicated AH.
Methods
Sixty-five children diagnosed with AH were included in the study. The mean age of the children was 7.42 ± 2.26 (4–13 years). The cohort consisted of 29 males and 36 females. All children were evaluated clinically and endoscopically. AZE-FLU nasal spray was applied to both nostrils twice a day for three months. Adenoid/choana ratio and symptom scores were evaluated before treatment and at the end of the 12th week.
Results
At the end of 24 weeks of AZE-FLU application, there was a statistically significant decrease in both adenoid/choana ratio and symptom scores. While the initial adenoid/choana (A/C) score was 3.57 ± 0.58, it decreased to 1.74 ± 0.61 following treatment. A dramatic decrease in total symptom scores was observed. The total symptom score average was 15.63 ± 1.28 before treatment, while it was 2.31 ± 1.4 after the treatment with the difference being statistically significant (P < .01).
Conclusion
In this study, the effectiveness of AZE-FLU nasal spray on AH was investigated for the first time. This treatment provides an effective alternative to the surgical approach in children with uncomplicated adenoid hypertrophy. Using this protocol, 96% of patients were removed from the surgery list.
Level of Evidence
is IV
Introduction
The adenoid is a pyramidal cluster of lymphoid tissues located on the roof and posterior wall of the nasopharynx. Normally it is a center of resistance to upper respiratory tract infections but can cause recurrent and chronic sources of infection. Adenoid hypertrophy (AH) is a common childhood disease. 1 AH causes breathing through the mouth, nasal congestion, hyponasal speech, snoring, obstructive sleep apnea, chronic sinusitis, and recurrent otitis media. In the long term, it can lead to complications such as obstructive sleep apnea syndrome (OSAS), growth retardation, cardiovascular morbidity, neurocognitive abnormalities, learning and behavioral problems, hyperactivity, and inadequate attention. 2 Treatment planning of AH in children is done according to the degree of airway obstruction and associated morbidity. If surgical treatment is indicated, the individual risk/benefit analysis of the patients should be evaluated in terms of anesthesia and postoperative complications. Studies on intranasal steroid administration as a non-surgical approach in uncomplicated cases have been presented in the literature.3-8
Azelastine hydrochloride (AZE) has histamine receptor antagonists, anti-inflammatory and mast cell stabilizing properties. Fluticasone furoate (FF) is an anti-inflammatory agent effective on mast cells, eosinophils, neutrophils, macrophages, and lymphocytes. Recent developments include Meda Pharma’s AzeFlu (MP-AzeFlu, Dymista; Mylan, Inc. Canonsburg, PA) that contains azelastine hydrochloride (AZE) and fluticasone propionate (FP) in a single spray. 9 The formulation has been reported to be well-tolerated and safe after three months of continuous use in children with allergic rhinitis. MP-AzeFlu was approved by the FDA in February 2015 for use at a spray dose per nostril twice a day in children aged 6–11 years.10,11 In this study, we aimed to evaluate the effectiveness of AZE-FLU on adenoid tissue size and symptoms of adenoid hypertrophy, as well as to investigate the need for surgical treatment after AZE-FLU administration in children with uncomplicated AH.
Material and Methods
Sixty-five children who were examined with a pre-diagnosis of AH in otolaryngology clinic between July 2019 and April 2020 were included in the study. The procedure was approved by Ethics Committee for a prospective, randomized longitudinal study (protocol number:-100-7989). The mean age of the children was 7.42 ± 2.26 (4–13 years). It consisted of 29 males and 36 females. All children were evaluated clinically and endoscopically. The control group was not created because it would not be ethical either not to treat children or treat with placebo.
Those with a previous history of adenoidectomy and a history of positive allergies or atopy, those with craniofacial malformations including labiopalatal clefts and genetic diseases (e.g., Down’s syndrome), neurological disorders, cardiovascular diseases, acute infections of the nose, palate or nasopharynx, and immunodeficiency disorders or hypersensitivity to azelastine 12 were excluded from the study. Allergic rhinitis diagnosis was made when a child had typical allergic symptoms and showed a positive result on the Prick test. Children were administered AZE-FLU (137 mmcg AZE and 50 mcg FP per spray) as one spray per nostril twice a day, in the morning and evening. The daily total dose was 548 mcg for AZE-Flu and 200 mcg for FP.
The parents of the patients were informed about the aim of the study and the procedure for treatment, and consent was obtained from the parents. Patients who had symptoms related to adenoid hypertrophy for at least six months, whose tonsil dimensions were normal, and who did not have tonsillectomy indications due to chronic tonsillitis were included in the study.
Initially, routine ear nose throat examination and nasal endoscopy were performed on each child. Clinical history was taken from the parents using a questionnaire. The patient's history included age, gender, personal and family history of atopy, and drug use. AH diagnosis was confirmed by nasal endoscopy. Treatment efficacy was evaluated pre-treatment and after a 24-week treatment period with nasal endoscopic examination of symptoms and changes in adenoid tissue. Among the symptoms evaluated were nasal obstruction, rhinorrhea, cough, snoring, nocturnal cough, and obstructive sleep apnea.
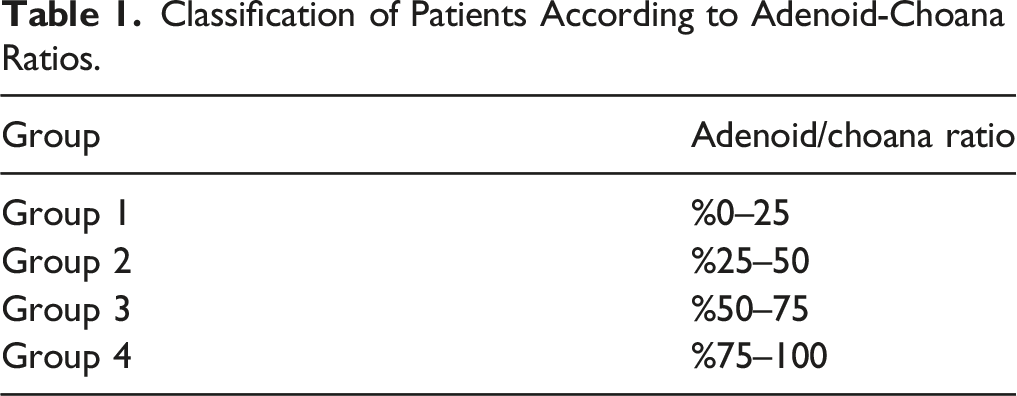
Classification of Patients According to Adenoid-Choana Ratios.
Statistical Analysis
Number Cruncher Statistical System (NCSS) program was used for statistical analysis. Descriptive statistical methods (mean, standard deviation, median, frequency, percentage, minimum, maximum, Q1, and Q3) were used while evaluating the study data. Wilcoxon signed-rank test was used for intragroup comparisons of quantitative variables that did not show normal distribution. Statistical significance was accepted as P < .05.
Results
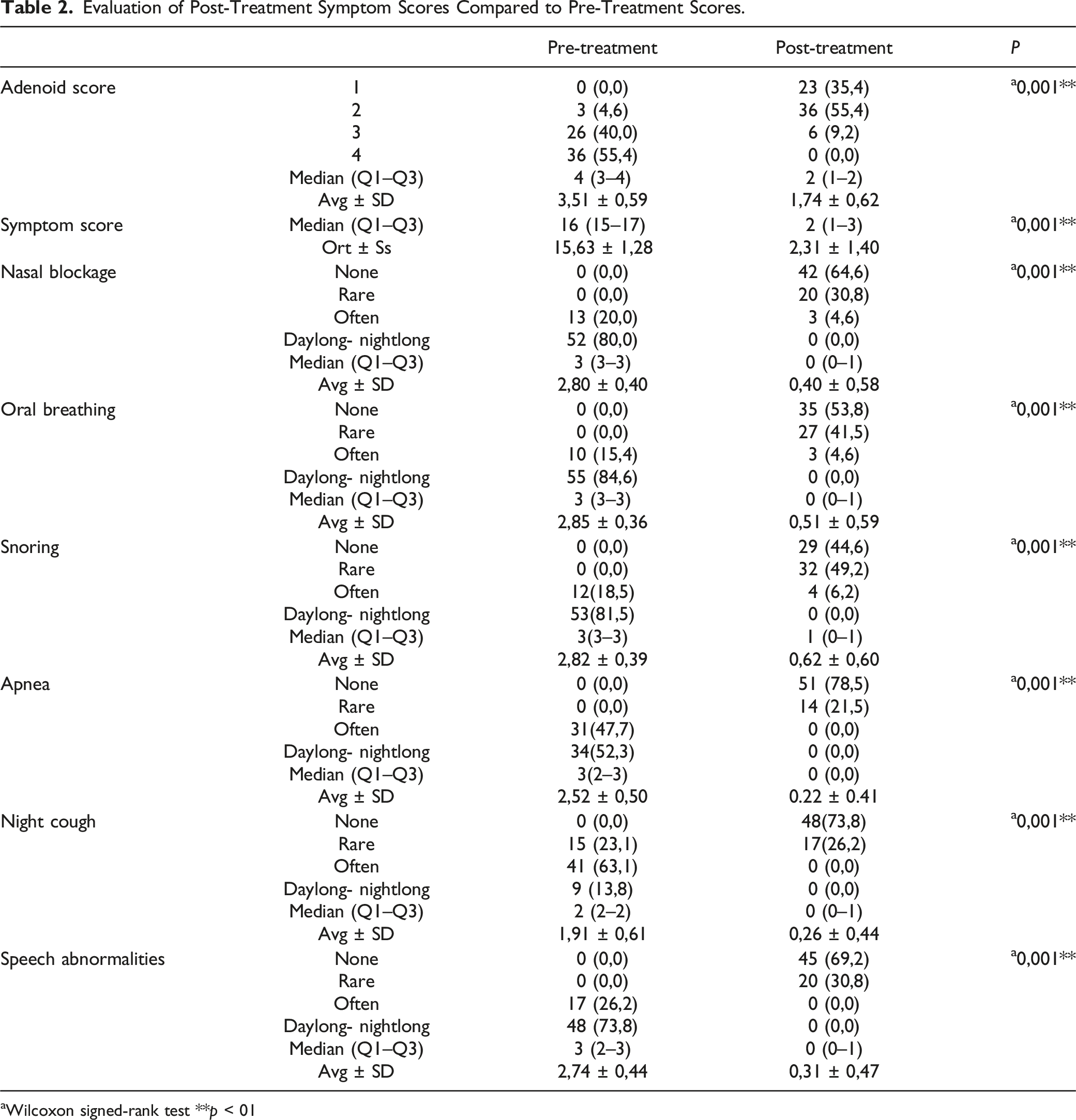
Evaluation of Post-Treatment Symptom Scores Compared to Pre-Treatment Scores.
aWilcoxon signed-rank test **p < 01
Considering all symptoms, the mean total symptom score before and after treatment was calculated and compared. After 24 weeks of treatment, the mean total symptom score decreased from 15.66 ± 27 to 2.29 ± 1.4. This difference was shown to be statistically significant (P < .001). (Figure 1) Symptom scores before and after the treatment.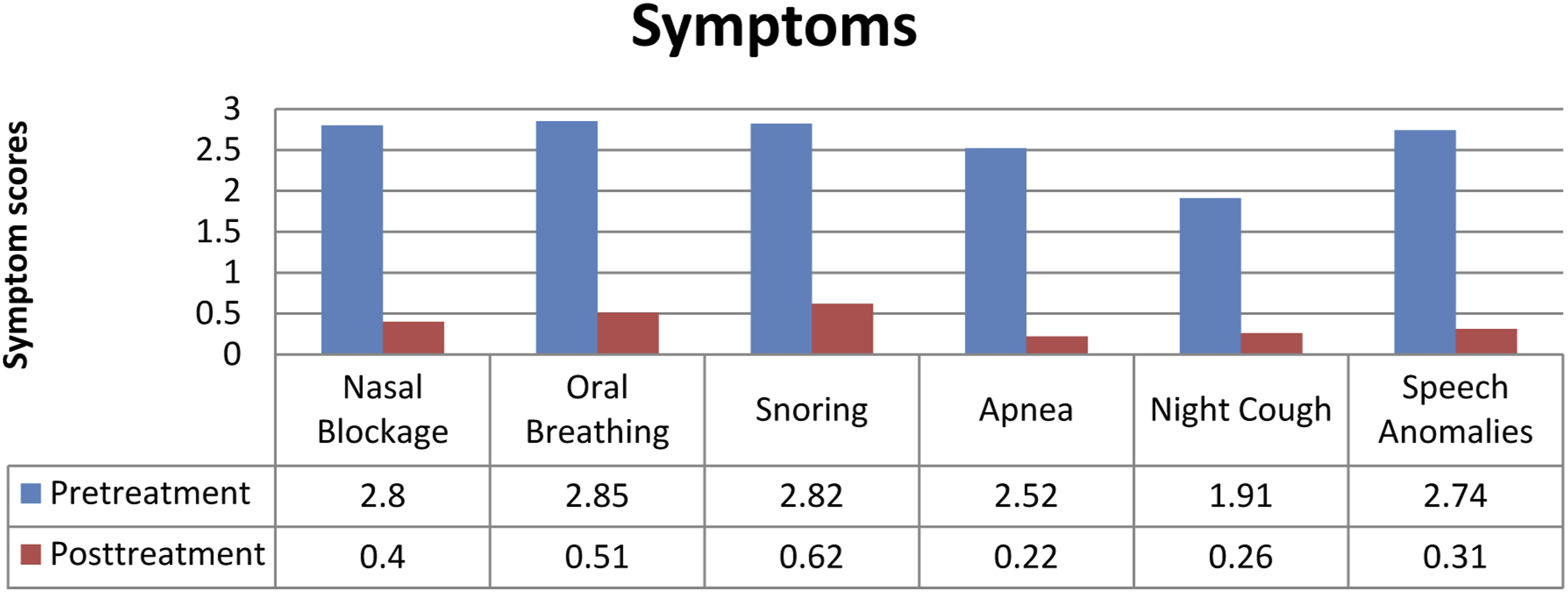
After the 24-week treatment regimen, the adenoid/choana (A/C) ratio decreased from 3.51 ± 0.59 to 1.74 ± 0 (Figure 2). The difference was statistically significant (P < .01). The decrease in the adenoid score after the treatment compared to the pre-treatment was found to be statistically significant (P = .001; P < .01). Distribution of adenoid scores before and after treatment.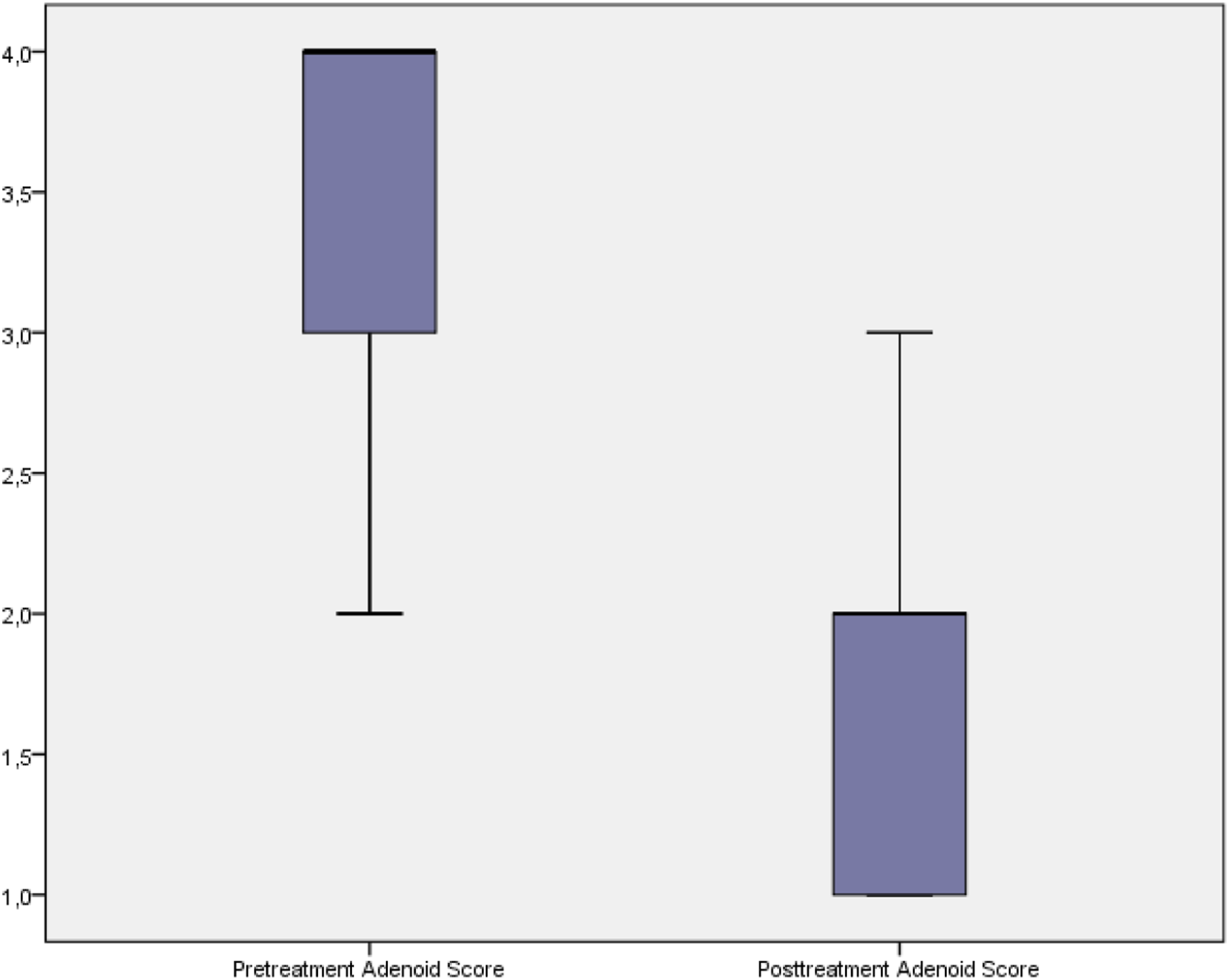
The mean nasal obstruction symptom score was 2.8 ± 0.4 prior to treatment. Following the treatment, the mean score decreased to 0.4 ± 0.58 which was statistically significant (P < .01). Similarly, a statistically significant decrease was observed in mouth breathing symptom scores which were 2.85 ± 0.36 before treatment and 0.51 ± 0.59 after the treatment (P < .01). The snoring symptom scores significantly decreased from 2.82 ± 0.36 before the treatment to 0.62 ± 0.60 after the treatment (P < .01). Concordantly, apnea symptom scores decreased significantly from 2.52 ± 0.50 to 0.22 ± 0.41 before and after the treatment, respectively (P < .01). Night cough symptom score was 1.91 ± 0.61 before the treatment, which decreased significantly after treatment to 0.26 ± 0.196 (P < .01). Moreover, the speech abnormality symptom scores were 2.74 ± 0.44 before the treatment and 0.31 ± 0.47 after the treatment with the difference being statistically significant (P < .01).
There was no statistically significant difference in tympanometry test results between the two groups before and after the treatment. None of the patients had a personal or family history of allergies or atopy, previous surgery, any medication in the last four weeks, or immunodeficiency.
In this study, 62 out of 67 patients did not need surgery after the treatment. The remaining 5 children were informed that they did not fully respond to the treatment. However, parents of three children did not want their children to be operated on. Adenoidectomy was performed on the remaining two children.
Discussion
Nasal obstruction is a common symptom in children and one of the main indications of AH. 14 The etiology of AH is unknown, but inflammatory mechanisms may play an important role. Local and systemic inflammatory markers and proinflammatory cytokines are increased in these children which stimulate lymphoid tissue proliferation. Appropriate treatment of this condition is necessary to control nasal obstructive symptoms. It has been suggested that systemic or topical anti-inflammatory agents play a role in the treatment of adenoid hypertrophy.14,15 AH is also one of the most common surgeries in childhood, and adenoidectomy is generally considered the definitive treatment for nasopharyngeal obstruction. As adjunctive treatments, several non-surgical alternatives can reduce the size of the nasal passages. Some authors have suggested the use of topical nasal steroids to protect immunologically active tissue and to reduce AH to avoid the anesthesia and surgery risks inherent in adenoidectomy.7,16
AH obstructs the nasal passages and eustachian tubes and blocks the clearance of nasal mucus. Adenoid hypertrophy and obstructive sleep apnea are associated with increased expression of various mediators of inflammatory responses. It responds to anti-inflammatory agents, such as corticosteroids. Corticosteroids can reduce adenoid hypertrophy with lympholytic or anti-inflammatory effects. Intranasal corticosteroids reduce cellular proliferation and production of proinflammatory cytokines in the adenoid mixed cell culture system. Atopy in atopic juveniles is associated with an increased number of immunoglobulin-E positive cells and FcyRI+cells in adenoidal tissue. 17
Glucocorticoid receptor expression is evident in the adenoid tissue of children with obstructive sleep apnea syndrome (OSAS). The glucocorticoid receptor-α/-β ratio is higher with high glucocorticoid receptor-α expression in OSAS patients than in those with recurrent upper airway infections. 18
The successful use of intranasal steroid therapy in children with AH was first introduced by Demain and Goetz. 3 Although the mechanisms by which steroids reduce nasal airway obstruction are not yet clear, there are some logical theories. Some of these include reduction of adenoid size by direct lympholytic effect. The anti-inflammatory effect of steroids helps to reduce adenoidal and nasopharyngeal inflammation or reduces the likelihood of the adenoid acting as an infection reservoir. 3
Corticosteroids can reduce adenoid hypertrophy with lympholytic or anti-inflammatory effects. 19 The mechanism by which topical steroids improve nasal airway obstruction symptoms has yet to be determined. In some studies, a significant correlation has been observed between the A/C ratio and the nasal symptom score. 3 These results show that the improvement in nasal airway obstruction symptoms is related to the effect of intranasal corticosteroids on adenoid size. On the other hand, it should be noted that the use of fluticasone propionate increases the nasal airway volume and, in addition, reduces the soft tissues of the inferior turbinate, resulting in improvement of symptoms. 20 It is more common in children with allergic diseases such as adenoidal hypertrophy, allergic rhinitis, bronchial asthma, or atopic dermatitis (40.4%) than in healthy children (22.3%). Allergic inflammation in the nasal mucosa may contribute to adenoidal hypertrophy. 21 Several mechanisms are believed to be involved, such as direct lymphocytic action, inhibition of inflammation, and changes in the adenoid bacterial flora. 3
Brouillette et al. reported that six weeks of fluticasone nasal spray administration reduced the severity of pediatric OSAS symptoms. 19 Demain and Goetz showed that 24 weeks of topical nasal corticosteroid therapy reduced adenoid size and improved symptoms of nasal airway obstruction. They also reported a 29% decrease in the average A/C ratio (3). Cengel and Akyol used intranasal mometasone furoate monohydrate in their study and reported a 50% reduction in initial adenoid size. 7 They observed a 20% (12.5–32.5) reduction in mean choanal obstruction in the mometasone group and a 0.0% (0.0–0.0) reduction in the placebo group. Ciprandi et al. showed a significant reduction (P < .05) in mean adenoid size after 8 weeks of flunisolide treatment compared to normal saline treatment in this study, and we aimed to evaluate the effects of AZE-FLU combination therapy in children with adenoid hypertrophy.
A study has been reported on the use of topical azelastine for the treatment of AH. In this study, the reduction in the A/C ratio was reported as 35.6% after 12 weeks of topical azelastine use (22).
It was concluded that nasal corticosteroids affect the anatomical component of OSA by reducing inspiratory upper airway resistance at adenoidal level.8,22
In controlled studies, azelastine nasal spray was well tolerated in adults and children (12 years of age) for treatment times of up to four weeks. 23 In our study, AZE-FLU combination treatment was well tolerated by all patients for 12 weeks.
In the two studies including results regarding the need for surgery, Criscuoli et al. reported that in their first long-term study of 100 weeks, 13 out of 24 patients (54%) who responded well to initial therapy still needed surgery, 24 while Usta et al. reported that only 2 out of 39 patients indicated for adenoidectomy after mometasone furoate therapy needed surgery. 25 In our study, 62 out of 67 (92%) patients who received treatment did not require a surgical approach. Adenoidectomy was performed in two patients (1.5%). The remaining three patients (6%) were informed about their ongoing surgery needs, but their parents refused surgery, suggesting that this treatment had improved their condition sufficiently.
Azelastine nasal spray (Allergodil, Lastin, Afluon; MedaAB, Stockholm, Sweden) is a fast-acting, effective, and well-tolerated H1 receptor antagonist for the treatment of rhinitis (26). Mast cell has stabilizing and anti-inflammatory properties, reducing its concentration. Leukotrienes, kinins, and platelet-activating factor reduce inflammatory cell migration in vitro and in vivo, as well as in rhinitis patients. 26 Well-controlled studies in patients with seasonal allergic rhinitis, perennial rhinitis, or vasomotor rhinitis suggest that azelastine nasal spray has a rapid onset of action and it improves nasal symptoms associated with rhinitis such as nasal congestion and postnasal drip.27,28
Intranasal corticosteroids also reduce vascular permeability and edema. Therefore, these profound anti-inflammatory effects may reduce the immunological activation shown in hypertrophic adenoid tissue and reduce the adenoid size. 29
In the efficacy data obtained in children with allergic rhinitis taking MP-AzeFlu for three months, a significant (P = .0410) reduction in nasal symptoms was reported compared to those who received FP alone. 30 Additionally, intranasal azelastine therapy has been reported to be beneficial in the treatment of AH in the general pediatric population with AH. 31
Our patients were followed up for three months after completing the treatment. None of them complained of severe symptoms as at the beginning of the study.
Our study showed that AZE-FLU treatment applied in children with AH is very effective in reducing symptoms and improving children. Thus, there was no need for a possible adenoidectomy operation to be performed on children. In this way, possible complications of the operation performed with general anesthesia are prevented. The additional cost of surgical intervention to the treatment has also been eliminated. The problem of being away from school that such an operation could cause has also been resolved. Since AH is a pathology that affects children's school success, it should be expected that children's academic performance will improve with this treatment. Since this treatment is also a nasal spray, it is easy to apply and does not interfere with the daily life of children. Our patients who participated in the study reported that they had no problems with the application in the controls.
Although the aim of the study is not a comparison of the efficacy of topical steroid alone with a combination of topical steroid and azelastine, our personal observation during the study is that combination therapy is more effective than topical steroid alone. However, whether these observations are due mainly to one of the components, or a synergistic effect of both, a question which should be the subject of future studies.
Conclusion
The safety of AZE-FLU is widely accepted for the pediatric population. Although the mechanism itself has not yet been clearly and completely explained, it is important to determine the role of intranasal azelastine-corticosteroids in the treatment of children with adenoid hypertrophy. This method provides an effective alternative to surgical treatment in children with adenoid hypertrophy. With the protocol applied in this study, 92% of the patients were excluded from surgery. Intranasal corticosteroids are well tolerated by children; however, the most appropriate drug, the most effective dose, and the optimal duration of treatment should continue to be investigated and determined by prospective and randomized studies.
Footnotes
Author Contributions
All authors have substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data. All authors have taken part by drafting the article or revising it critically for important intellectual content. All authors have final approval of the version to be published. All authors agreed to be accountable for all aspects of the work.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: This study was conducted in accordance with the tenets of the Declaration of Helsinki.
Funding
The authors received no financial support for the research and/or authorship of this article.
Ethics Approval
was obtained from the local Research Ethics Committee prior to the study.
Informed Consent
Every participant patient was informed comprehensively about the study and informed consent was obtained from each of them.
Data Availability
All data generated or analyzed during this study are included in this article [and/or] its supplementary material files.
