Abstract
Purpose
To improve pain management after tonsillectomy (TE) by comparing individual analgesic management by demand versus a fixed-scheduled analgesic treatment protocol in a prospective trial.
Patients and Methods
Forty consecutive patients received individual pain treatment by demand (control group) followed by 40 patients who were treated by a fixed-scheduled four-staged escalating analgesic protocol (intervention group) after TE. Minimum and maximum pain as well as pain on ambulation (NRS 0-10) on the first postoperative day were defined as primary objectives. Secondary endpoints comprised the analgesic score, treatment-related side effects/pain-associated impairments, wish for more pain medication, and patient satisfaction. Patients were surveyed using the standardized and validated “Quality Improvement in Postoperative Pain Treatment” (QUIPS) questionnaire.
Results
Patients of the control group reported comparable minimum (2.03 ± 1.42 vs 2.38 ± 1.79, P = 0.337, r = 0.110) and maximum pain (6.65 ± 2.10 vs 6.93 ± 1.86, P = 0.536, r = 0.07) and pain on ambulation (4.73 ± 2.26 vs 5.18 ± 2.19, P = 0.370, r = 0.10) compared to the intervention group. Patients in both groups were comparably well satisfied with the pain treatment (7.53 ± 2.40 vs 7.73 ± 2.30, P = 0.704, r = 0.04), experienced similar side effects and functional impairments (P > 0.050, Φ < 0.3), and did not ask for much more analgesic medication (P = 0.152, Φ = 0.160).
Conclusion
Pain control following TE was not distinctly affected by applying a fixed-scheduled analgesic treatment protocol compared to individual analgesic therapy. In conclusion, analgesic treatment after TE remains unsatisfying. Consequently, further efforts are needed to achieve a standardized and effective approach to the underlying pathophysiological causes of pain following TE.
Introduction
Tonsillectomy (TE) is one of the most frequent types of surgery in Germany, with 87,107 procedures performed annually (in 2016). 1 Multiple reports have repeatedly shown that patients after TE suffer to a large extent from severe pain over several days despite individual analgesic medication.2-4 In 2013, Gerbershagen et al. 5 reported that TE ranked 24th out of 179 surgical procedures in terms of its postoperative pain levels despite analgesic medication. In this regard, it ranks ahead of kidney transplantation, open hysterectomy, and gastrectomy.
An evidence-based pain management concept leading to sufficient pain control after TE does not yet exist. 4 Because of the high level of postoperative pain explicitly after TE, several countries, including Sweden and France, have developed specific guidelines for its postoperative pain management. Again, a generally applicable sufficient regimen could not be identified or recommended.6,7
According to the German S3 guideline “Treatment of Acute Perioperative and Posttraumatic Pain,” analgesic step-by-step regimens should be established at every hospital for rapid, effective, and safe postoperative pain management. The analgesic medication should be divided into basic and demand medication. The basis of adequate pain therapy is the administration of non-opioid analgesics, which are supplemented by the administration of weakly and strongly acting opioids if pain control is insufficient. 8
So far, studies are inconclusive as to whether postoperative pain management according to a fixed regimen is superior to the administration of analgesics on demand.9-11 Several studies have demonstrated the superiority of fixed-scheduled administration after TE, but these were either in children or in an outpatient setting.12,13 Up to now, no studies have investigated the superiority of fixed-scheduled escalating postoperative pain management after TE in an inpatient setting in adults.
We developed a four-staged escalating analgesic scheme according to the guidelines of the World Health Organization (WHO). The objective of the study was to evaluate the efficacy of this standardized postoperative pain management regimen compared to individualized pain medication by demand with regard to the postoperative pain reduction, analgesic score, treatment–related side effects, and patient satisfaction.
The relevance of the question is high due to the significant pain-related reduction in quality of life and the multiple negative consequences that untreated postoperative pain entails. 14
Patients and methods
The present prospective study was approved by the Ethics Committee of the Friedrich-Alexander-University of Erlangen-Nürnberg (Germany) (approval number 224_18 B). It was conducted according to the Declaration of Helsinki and followed the STROBES guidelines as well as the guidelines of the “Quality Improvement in Postoperative Pain Treatment” (QUIPS) registry for the assessment and comparison of pain outcome parameters in German hospitals. 15 The study was registered in the German Clinical Trial Register (DRKS00016517).
A total of 80 patients presenting for elective bilateral TE between August 6, 2018 and May 12, 2020 in the Department of Otorhinolaryngology, Head and Neck Surgery, Erlangen University Hospital, were included. The first 40 consecutive patients received as-needed postoperative pain management (August 6, 2018–May 23, 2019) at the discretion of the ward physician (control group). Subsequently, the newly developed escalating pain concept was introduced. After repeated training of the staff and a five-week introductory phase (June 3, 2019–July 8, 2019), it was applied to another 40 consecutive patients after TE (July 9, 2019–May 12, 2020) (intervention group).
All patients gave their written informed consent after receiving thorough information about the study. Inclusion criteria were defined as follows: bilateral total TE due to chronic tonsillitis, 16 minimum age of 18 years, sufficient cognitive and linguistic ability to understand the content of the questionnaire, written informed consent capacity, American Society of Anesthesiologists (ASA) status I-III, and body weight of 50–120 kg. Patients with a single-sided or partial TE, a TE à chaud, a TE due to an oncologic indication, a medical history of pre-existing opioid-dependent pain, aged under 18 years, the prevalence of a significant central nervous system disease and/or obstructive sleep apnea, and/or refusal to participate in the study were excluded from the study.
Tonsillectomy was performed by means of “cold” dissection, where the tonsils were bluntly dissected along the tonsil’s capsule with the anterior and posterior palatal arch left intact. After thorough hemostasis via compression and bipolar coagulation, the wound bed was left open for secondary wound healing. 16 The “cold” technique is assumed to result in less tissue damage and lower post-tonsillectomy morbidity. 17
In all patients, anesthesia was induced according to the current hospital standard (fentanyl 1–2 µg/kg body weight (bw), propofol (TCI (target-controlled-infusion) plasma level 4–8 ng/mL, Orchestra® Base Primea, Fresenius Kabi, Bad Homburg, Germany, plasma mode, Marsh mode), and rocuronium 0.3-0.5 mg/kg bw, intravenously (IV)). To maintain anesthesia, plasma propofol TCI was reduced to levels between 2.5 and 4.0 ng/mL, and the strong intraoperative pain stimulus was treated with remifentanil 0.2–0.5 µg/kg/min run rate combined with Fentanyl (1 µg × kg−1 bolus as required). To attenuate propofol injection pain, all patients received 40 mg lidocaine 2% IV before propofol administration.
Pain in the recovery room was treated with intravenous piritramide (0.05–0.2 mg × kg−1) or pethidine (0.4–0.7 mg × kg−1) up to a pain intensity of <3 (NRS). Antibiotics (sultamicillin 750 mg p.o., twice daily) were administered only in case of infection postoperatively.
Maximum and minimum pain as well as pain on ambulation (Numeric Rating Scale, NRS 0 (“no pain”)–10 (“worst pain imaginable”)) on the first postoperative day were defined as primary endpoints. Secondary endpoints comprised analgesic score, treatment–related side effects/pain-associated impairments (cough, fatigue, sleep disturbance, nausea, dizziness, mood, and/or mobility impairment) wish for more pain medication and patient satisfaction.
The four-stage pain regimen was developed according to the principles of the WHO stage scheme (see Figure 1).
18
Ibuprofen combined with acetaminophen were used as basic medication and were applied to all patients on the first 2 postoperative days 3 times or 4 times per day, respectively, per os (p.o.) unless there were contraindications. If this did not result in adequate analgesia, that is, pain at rest NRS <3 and on ambulation <5, weak (tilidine) or strong (hydromorphone) opioids were administered. In addition, there was a rescue medication at each level that could be administered in case of pain spikes (NRS ≥5 at rest). If adequate analgesia was not achieved at level 3, the acute pain service was to be contacted. In addition, an antiemetic and a laxative were prescribed and administered as needed to all patients receiving hydromorphone. Patients with marked dysphagia and hepatic or renal insufficiency received separate pain regimens. Pain was recorded 30–60 minutes after analgesic application and at every shift change, 3 times a day. Four-staged pain protocol. In patients with a history of ulcers, especially with complications of bleeding or perforation, in elderly patients, and for patients requiring concomitant therapy with low-dose acetylsalicylic acid or other drugs that may increase gastrointestinal risk. Abbreviations: NRS, numeric rating scale; p.o., per os; IV, intravenous; s.c., subcutaneous; gtt, guttae; SD, single dose; PCIA, patient-controlled intravenous analgesia.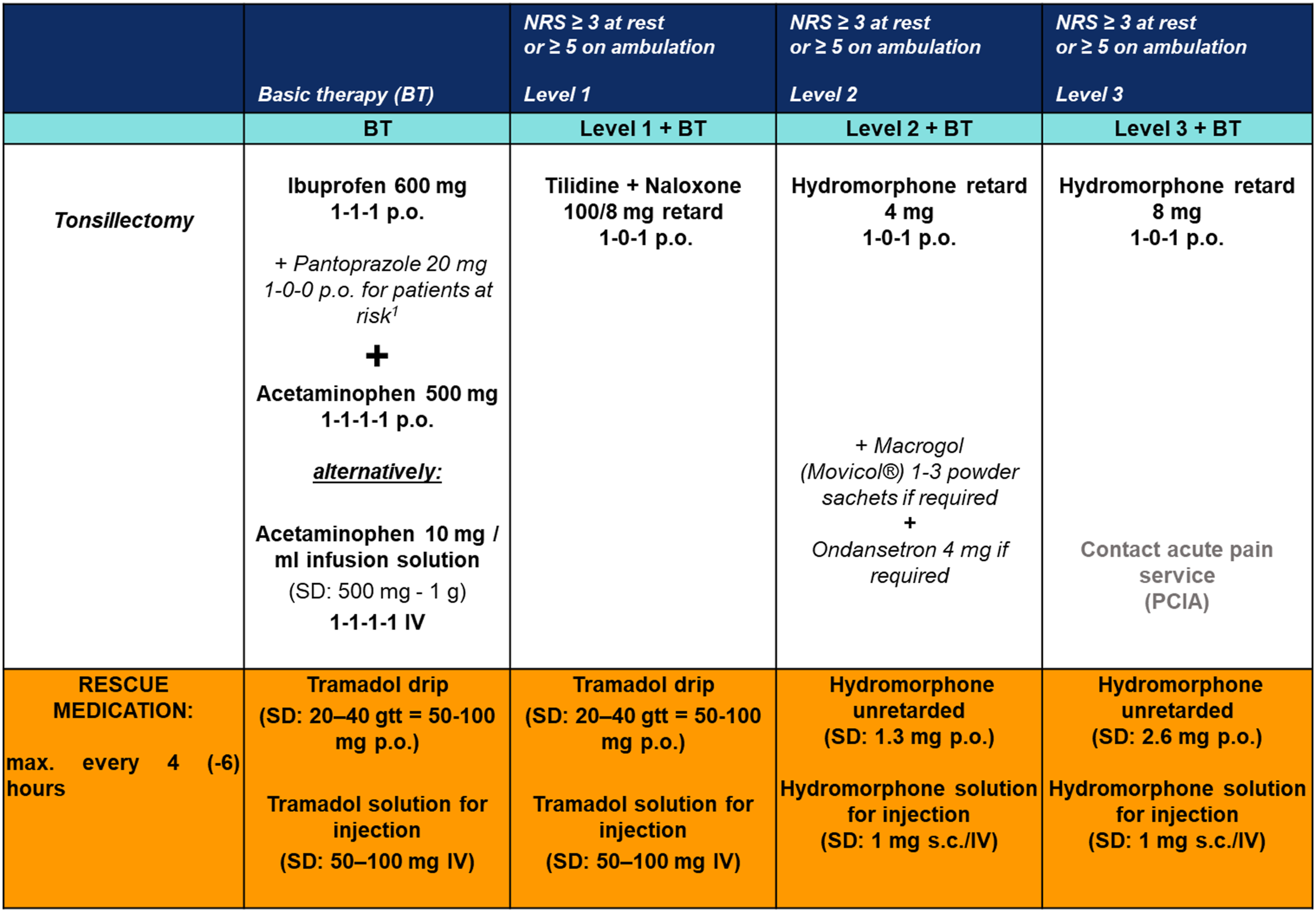
Patients were surveyed on the first postoperative day using the QUIPS questionnaire. QUIPS is a Germany-wide initiative founded in 2003 which is dedicated to evaluating and improving postoperative pain therapy.19,20 The project is open to all hospitals in Germany and has developed into the world’s largest acute pain register. Standardized and validated questionnaires are used to survey patients regarding their pain intensity (minimal, maximal, and during activity), physical impairments, side effects of pain therapy, and their general condition (so-called outcome parameters). 21 This is supplemented by the so-called process parameters, filled out by the investigator (L.S.) of the study, containing patient-relevant information, details on anesthesia and surgery, as well as analgesic consumption.
Sample size estimation was performed for the primary endpoint maximum and average pain perception and was based on Gostian et al. 22 Here, two standardized analgesic treatment protocols were compared regarding postoperative pain perception after tonsillectomy. A medium-sized effect was found for maximum pain (f = 0.251) and pain during activity (f = 0.223). Transferred to the targeted effect postoperative day x group, with a beta of 20% and an alpha of 5%, a sample size of at least 28 patients per group would be required to detect significant differences between the two treatment groups.
Numerical values are presented as mean values ± standard deviation (SD), nominal variables as absolute (n) or relative values (%). Unpaired t-tests with Bonferroni correction were performed for group comparisons of metric variables. For t-tests, effect sizes were given by r (0.1 = small, 0.3 = medium, and 0.5 = strong effects 23 ). Side effects and postoperative bleeding events were analyzed using Chi-square and Fisher´s exact tests, where applicable a P-value of less than 0.05 was considered statistically significant and marked with an asterisk (*). Statistical computations were carried out using SPSS Version 26.0 (IBM SPSS Statistics 26.0; IBM, New York, NY).
Results
A total of 97 consecutive patients were screened. Out of these, 17 patients were excluded due to excessive body weight (>120 kg, n = 2), a missing signature on the informed consent form (n = 1), or due to deviation of the postoperative pain medication from the established pain regimen in the intervention group (n = 14). Thus, a total of 80 patients (40 patients in the control group and 40 patients in the intervention group) were included and contributed to the results of the study. The CONSORT flowchart is depicted in Figure 2. CONSORT Flowchart. Representation of the study process using a CONSORT flowchart 47 to document included and excluded study participants. Abbreviations: n, number of patients.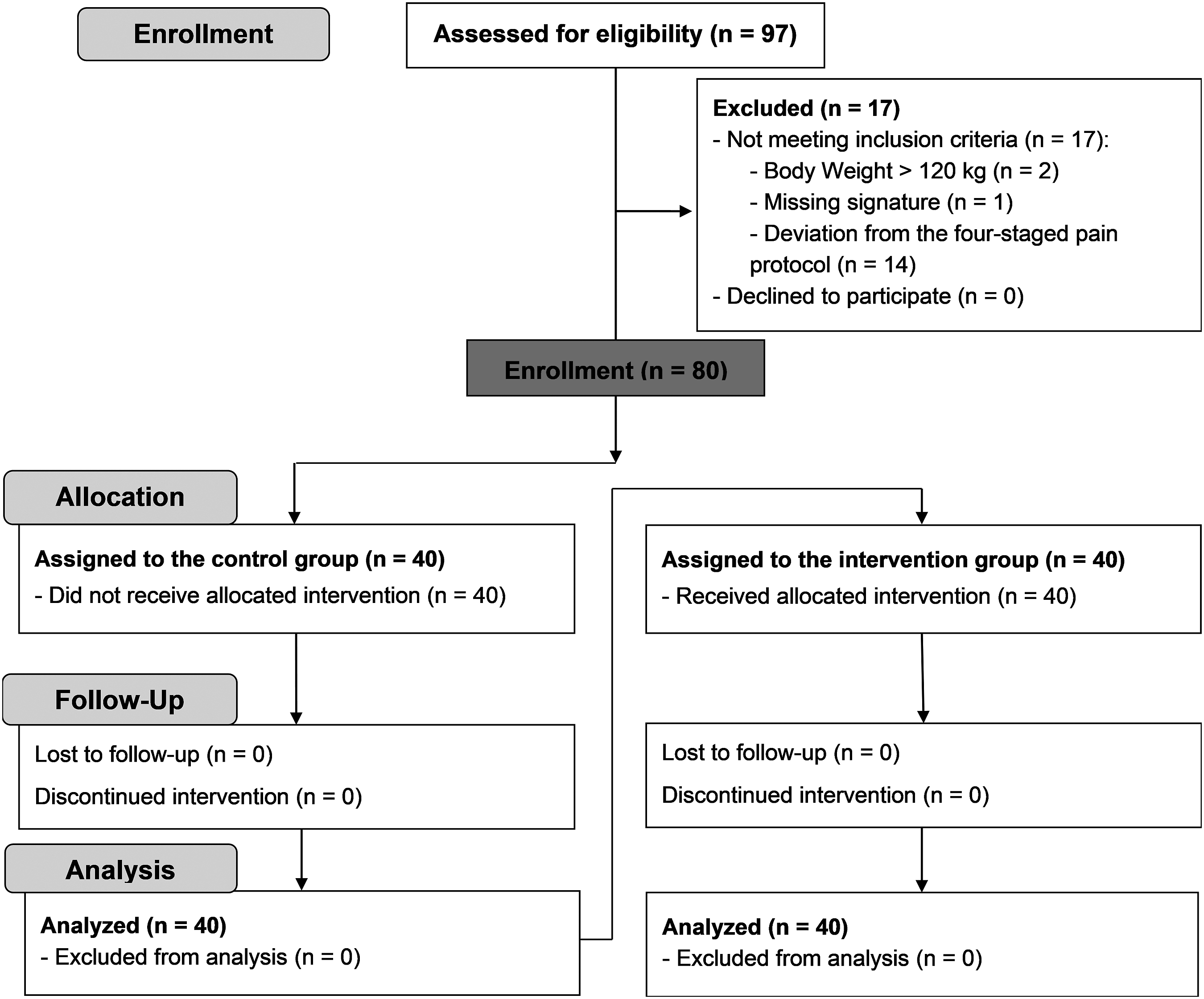
Patient demographic and clinical characteristics.
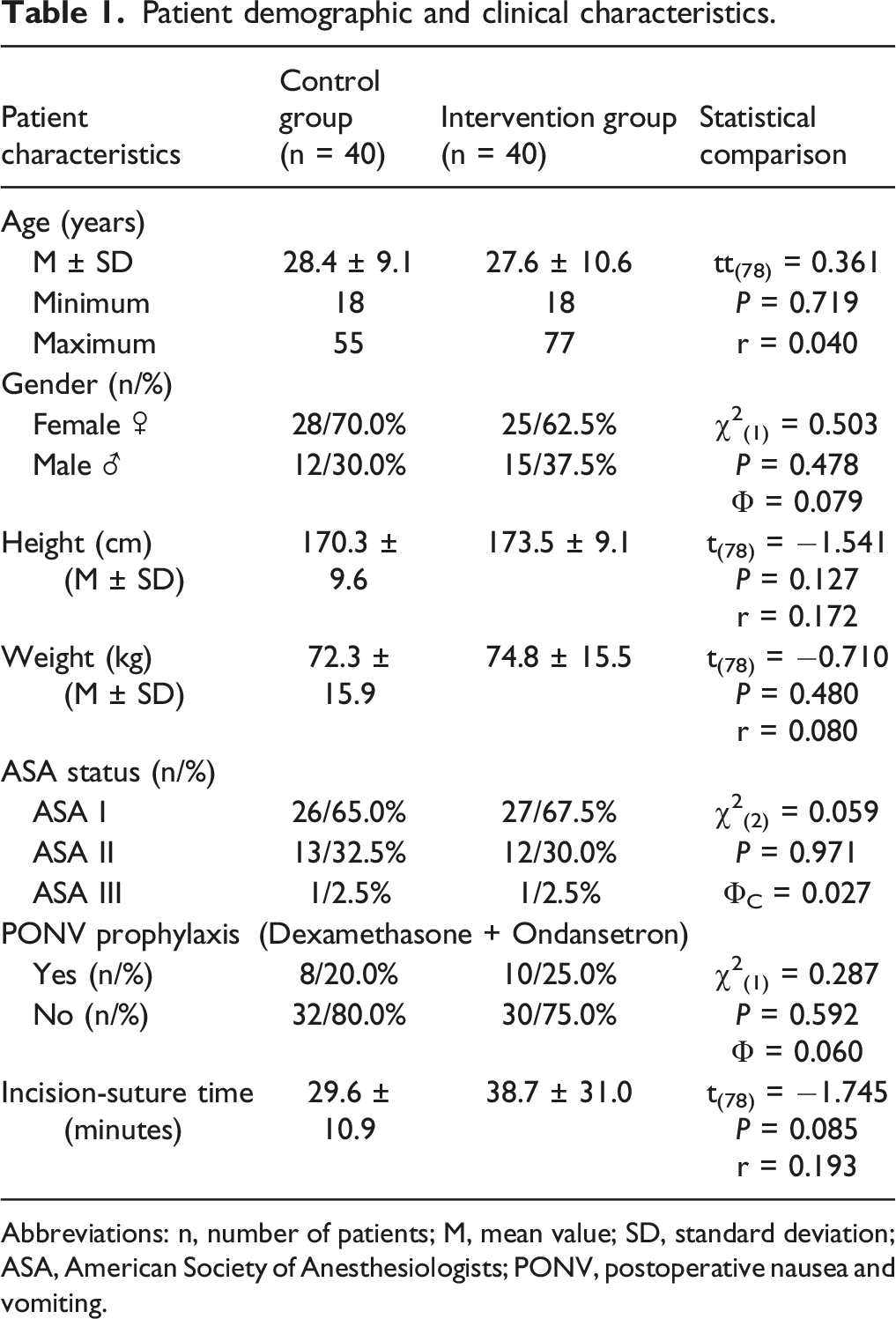
Abbreviations: n, number of patients; M, mean value; SD, standard deviation; ASA, American Society of Anesthesiologists; PONV, postoperative nausea and vomiting.
Intraoperative fentanyl consumption did not differ significantly between the two study groups (control group 0,110 ± 0.03 mg vs intervention group 0.125 ± 0.494 mg, P = 0.106, r =0.297).
In the recovery room, opioids were given to 31 of 40 patients (77.5%) in the control group. Out of these, 29 patients (72.5%) received piritramide IV (5.40 ± 4.08 mg), while 2 patients (5%) received pethidine IV (1.88 ± 8.75 mg). In the intervention group, 33 out of 40 patients (82.5 %, χ2 (1) = 0.000, P = 1.000, Φ = 0.000) received piritramide IV (6.13 ± 5.22 mg, P = 0.491, r = 0.078) and 4 patients (10 %, χ2 (1) = 0.721, P = 0.396, Φ = 0.095) received pethidine IV (3.13 ± 10.11 mg, P = 0.556, r = 0.067).
In the control group, a total of 38 patients received inpatient postoperative pain therapy. Two patients did not ask for any analgesic medication. Metamizole was the most frequently administered analgesic in the control group (23/57.5%). Most patients (n = 32; 80.0%) received combinations of two (n = 16; 40.0%) or more (n = 16; 40.0%), i.e. up to 5, non-opioid analgesics. Two patients (5%) received piritramide (7-25 mg) and 3 patients (7.5%) received unretarded tramadol per os (25-200 mg).
In the intervention group, all patients received the basic medication consisting of ibuprofen and acetaminophen. Three patients (7.5%) received the so-called rescue medication (tramadol) in addition to the basic therapy. One patient (2.5%) received pain medication according to level 1 in addition to the basic therapy. The relative and absolute frequencies of the individual drug administrations are depicted in Figure 3. Analgesic consumption. Absolute and relative numbers (n/%) of patients in the control group and the intervention group receiving the respective analgesics.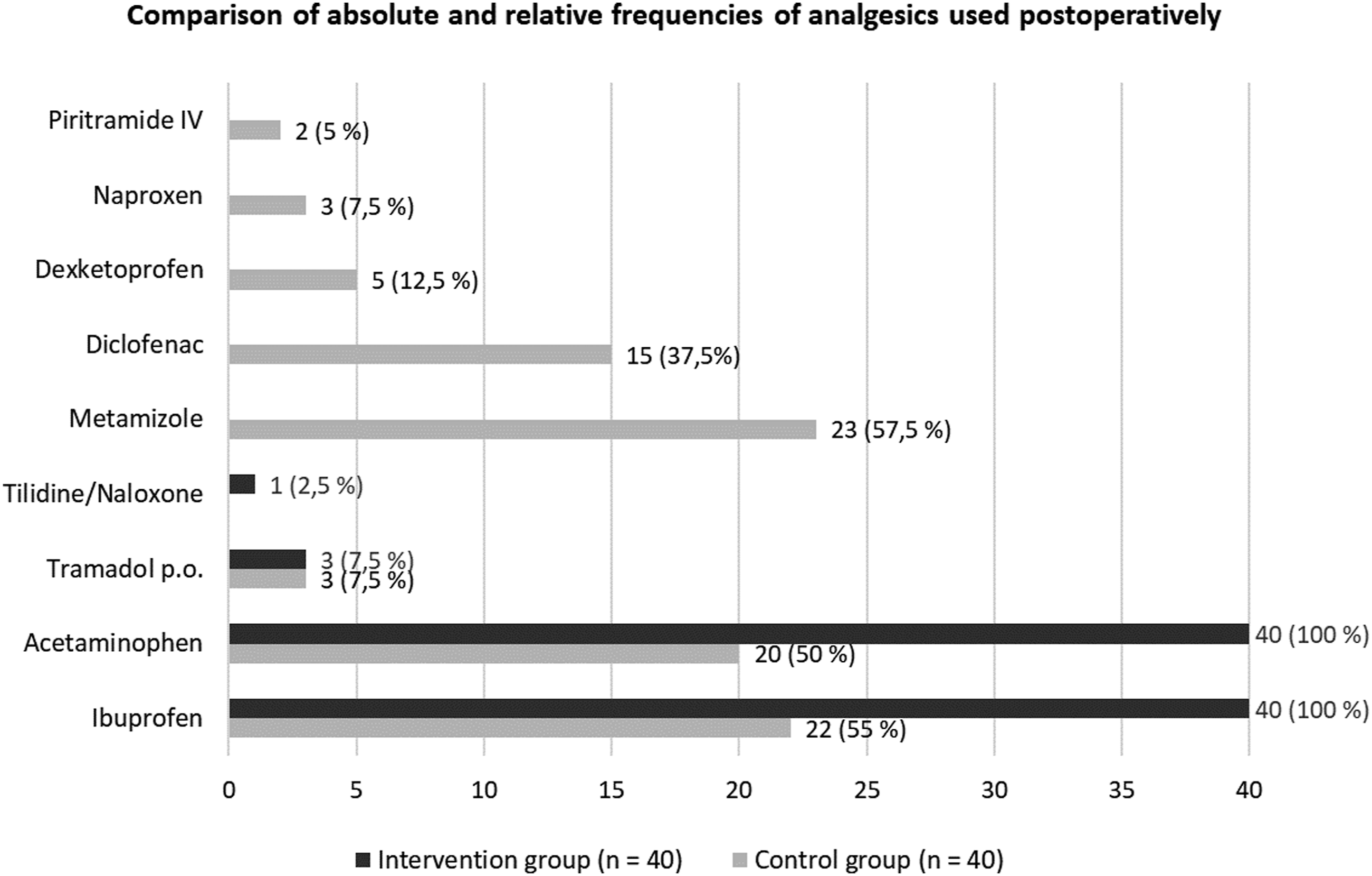
Patients of the control group experienced similar postoperative pain intensities (NRS) compared to the intervention group regarding minimum (2.03 ± 1.42 vs 2.38 ± 1.79, t(78) = 0.967, P = 0.337, r = 0.110) and maximum pain (6.65 ± 2.10 vs 6.93 ± 1.86, t(78) = 0.621, P = 0.536, r = 0.07) as well as pain on ambulation (4.73 ± 2.26 vs 5.18 ± 2.19, t(78) = 0.903, P = 0.370, r = 0.10). The results are presented in Figure 4. Minimum/maximum pain and pain during activity. Data presented as average values (NRS) of all patients included. Abbreviations: Pain min, minimum pain intensity; Pain max, maximum pain intensity.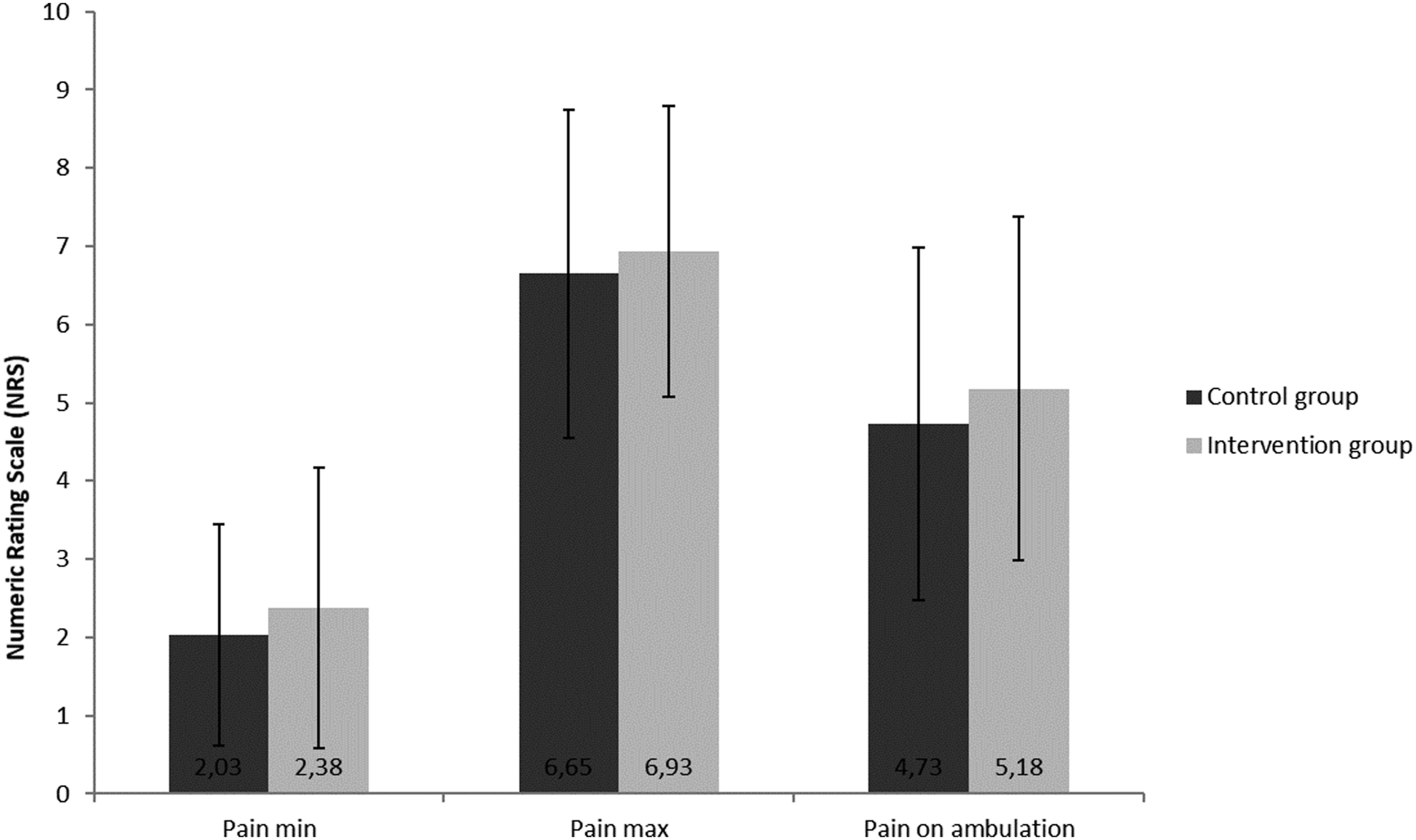
Incidence of functional impairments and treatment-related side effects of all patients.
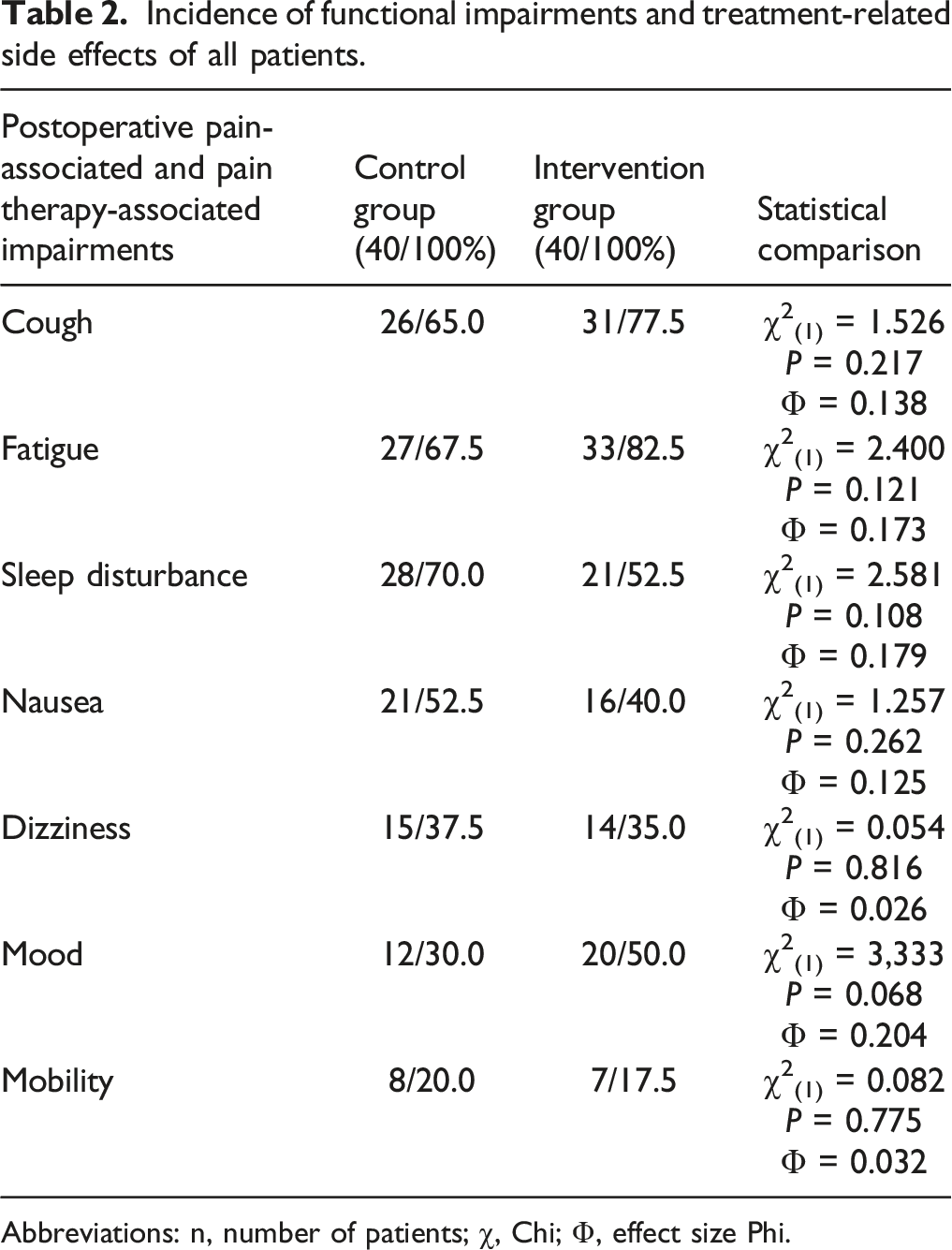
Abbreviations: n, number of patients; χ, Chi; Φ, effect size Phi.
There were no significant differences in the number of postoperative bleeding events. A total of 3 (7.5%) bleeding events requiring intervention occurred in the control group and a total of 5 (12.5%) in the intervention group χ2 (1) = 0.556, P = 0.712, Φ = 0.119).
Overall, patient satisfaction was encouragingly high in both groups with no statistically significant difference (control group: 7.53 ± 2.40 vs intervention group: 7.73 ± 2.30, t(78) = 0.381, P = 0.704, r = 0.04).
Five out of 40 patients in the control group wished for more pain medication (12.5 %) compared to 10 patients in the intervention group (25.0 %) (χ2(1) = 2.051, P = 0.152, Φ = 0.160).
Discussion
This study shows that both postoperative pain management as needed and standard escalating analgesic therapy resulted in unsatisfactory pain control after TE. The four-step escalating analgesic regimen with standardized timely use of the basic medication did not result in better pain management, but comparable side effects. Patients reported high satisfaction with the treatment they received. One advantage of the standardized pain concept was that the absolute number of different analgesic agents used could be reduced (up to five different peripheral analgesics in the control group) without achieving poorer analgesia. Potential pharmacokinetic and pharmacodynamic interactions can thus be avoided and potential side effects better monitored.
Up to now, studies have not clearly shown whether postoperative pain therapy according to a fixed regimen is fundamentally superior to the administration of analgesics on an as-needed basis.9,11 There is only one study in adults by Magdalena et al. 13 that showed better pain reduction and fewer complications on the fourth postoperative day after TE when patients were treated according to a fixed protocol.
Regarding children, Hobson et al. 9 state in their systematic review that “there was limited evidence available to draw any conclusions about the efficacy of PRN (pro re nata) versus ATC (around-the-clock) analgesic administration for the management of postoperative pain in children.” In a study of Sutters et al. (2004), it was found that scheduled dosing of acetaminophen and codeine after TE in children was not superior to on-demand treatment, while children of all study groups did not experience adequate pain relief. 10 In a later study in 2010, the same study group demonstrated the superiority of a fixed-scheduled administration of acetaminophen combined with hydromorphone versus an as-needed administration in children for the first three postoperative days after TE. 12
The German S3-guidelines on the treatment of acute perioperative and posttraumatic pain strongly recommends the use of graduated balanced analgesic concepts for sufficient pain management, where non-opioids should always form the basis. 8 In case of high expected pain, the combination of several non-opioids can be advantageous, as they can additionally reduce opioid-related side effects. 8 In a previous study, we found that the majority of patients needed pain treatment consisting of two non-opioid analgesics after TE. 22 Definitive statements about an ideal combination of non-opioids are currently not evidence-based. 8 We chose the combination of ibuprofen and acetaminophen because its efficacy, good tolerability, and additive/synergistic effect have been repeatedly demonstrated.24,25 In children it has proven to be of similar efficacy as codeine plus acetaminophen or even morphine plus acetaminophen with less effect on nausea and vomiting.3,26 In addition, ibuprofen counteracts the inflammatory component that is also thought to cause this severe pain after TE. 5 A potentially higher postoperative risk of bleeding due to ibuprofen, even in combination with corticosteroids, could be excluded by multiple studies.27,28 Due to the much-discussed risk of agranulocytosis, the indication restrictions by the Federal Institute for Drugs and Medical Devices (BfArM), and the elaborate duty to inform, that is, required risk disclosure and safeguarding information, we deliberately decided against metamizole for the fixed pain regimen according to international guidelines.2,3,6,7
The study situation regarding the use of opioids for postoperative pain treatment is poor. For adults, there is only one prospective controlled study that has been able to show that the combination of tramadol and naproxen significantly improves pain relief compared to monotherapy with diclofenac in adults. 29
In addition, postoperative pain after tonsillectomy was not satisfactorily reduced by the use of patient–controlled intravenous anesthesia with morphine. 30
Severe pain after TE has been demonstrated in numerous studies4,31-34 and was again shown in our study collectives. Moreover, considering the results of many controlled clinical trials analyzing great varieties of pain management regimens, it becomes obvious that there is no standard pain therapy after TE with reliable proof of sufficient pain reduction.2,4
Moreover, various treatment attempts far beyond the usual pain medication have been studied.35-38 Most of them have only shown limited effects. The grade of recommendation for the use of local anesthetics is C7,8 and the use of antibiotics for positively influencing post-TE pain does not seem reasonable. 39 However, patients seem to benefit from the use of physical measures such as local cooling8,40 and the perioperative administration of corticosteroids.2,6,8 Preemptive administration of analgesics also showed only a limited effect. 41
It is to be noted that the exact pathophysiology of the severe pain after TE is still unclear, but seems to be a multifactorial event consisting of direct injury to existing nerve endings, a local inflammatory response, mechanical irritation from contractions of the pharyngeal muscles and soft palate, and leaving the wound bed open.3,42
With regard to the molecular level, numerous membrane proteins and ion channels are under investigation, for example, the TRPV1 (= transient receptor potential vanilloid 1), offering possible new targets for future medication. 42
It must also be said that postoperative pain courses after TE have proven to show great variety.43-45 Most patients experience high postoperative pain intensities, but also a small percentage (about 15%) suffer from very limited pain. 43 This was also seen in our patient collective, where 2 patients of the control group did not ask for any pain medication.
The foremost limitation of our study is due to the missing randomization and blinding. Still, both study groups were highly comparable regarding demographic and clinical characteristics that are known to distinctly influence pain. 44 Because of the study design, the present results refer to the outcome status of the first postoperative day, which, however, has been shown to be one of the most painful time points in the course of postoperative pain after TE.
Of the total of 80 patients included, no patient was excluded, which precluded possible bias due to missing responses (nonresponder bias). All study participants were interviewed uniformly on the first postoperative day and by means of validated and standardized, easy-to-understand QUIPS questionnaires, which enabled comparable, low-bias results to be obtained. The interview was always conducted by the same person (L.S.), which excluded bias resulting from different interviewing styles.
Moreover, it may have been possible to detect any minor differences regarding pain control between the 2 postoperative pain regimens with a larger number of patients and an even closer and more rigorous supplemental pain medication.
The optimal postoperative pain treatment after TE remains to be determined. In this regard, further investigations addressing the detection of the mechanisms at a cellular level for the pronounced pain development and its efficient clinical treatment are mandatory to address this up to now unsolved problem. This may allow more desirable, more tailored pain management for individual patients in the future. However, sufficient analgesic therapy should always consist of a regular survey of postoperative pain and the utilization of all supportive therapies. Patients should be offensively educated about the postoperative pain process and pain reduction that is often not possible to an adequate extent. One should always be aware of the high level of suffering patients experience after TE.
Conclusion
Postoperative pain therapy following tonsillectomy by the clock using a standardized escalating treatment regimen did not allow superior pain reduction compared to pain management on demand. Accordingly, pain after tonsillectomy remains unsatisfactorily high. Further efforts are necessary to improve patient care.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
