Abstract
Objectives
Despite various solutions to the issue of ipsilateral vessel-depleted neck in microvascular head and neck reconstruction, concrete data on its safety and implementation are scarce. This paper focuses on the feasibility and success rates of contralateral anastomosis in free flap reconstruction in the head and neck region.
Methods
This single-center retrospective study at a tertiary referral center includes all patients who underwent free flap reconstruction of the head and neck with contralateral anastomosis between January 1st, 2007 and February 28th, 2021. Primary objectives were frequency, success, and flap-associated complication rates. Secondary objectives were recipient vessels and flap type.
Results
Of 318 patients who underwent microvascular reconstruction, anastomosis was performed on the contralateral side of the neck in 32 patients (10.0%). Recipient vessels involved mainly the superior thyroid artery (74.2%; n = 23) and the facial vein (51.1%; n = 23). Thirty patients (93.8%) received a radial forearm free flap. Flap-associated complications occurred in 12.5% of included cases (n = 4): one partial flap necrosis (3.1%), one anastomotic insufficiency (3.1%), one venous thrombosis of the microvascular pedicle (3.1%), and one wound dehiscence (3.1%). All of these complications were resolved without complete flap loss.
Conclusion
This study demonstrates that contralateral anastomosis is a successful and safe option in microvascular head and neck reconstruction, especially using a radial forearm free flap. Thus, anastomosing to the contralateral side of the neck can be advocated as a valuable option in the ipsilateral vessel-depleted neck.
Keywords
Introduction
Since 1959, microvascular free flaps have been used in reconstruction in head and neck surgery. 1 The frequently used and well established radial forearm free flap (RFFF) and anterolateral thigh flap (ALT) 2 were first described in 1981 3 and 1984, 4 respectively. The preferred recipient vessels for microvascular free flaps in head and neck reconstruction are the ipsilateral branches of the external coronary artery and their accompanying veins or the internal and external jugular vein because of their constant anatomic location, useful length, sufficient diameter, and their stable perfusion. 5
However, prior surgical and/or radiation therapy to the neck can cause severe tissue damage. This may result in the inability to work with commonly used vessels for the anastomoses, significantly increasing the challenges for the reconstructive surgeon.2,5-7
Various options have been shown to be effective in these challenging cases but usually require the extension of the surgical area beyond the neck.2,5,7-9 In this regard, the use of the contralateral side of the neck has been reported, but data specifically on the outcomes of contralateral anastomoses are scarce. Out of 261 free flaps reported by Hanasono et al., anastomoses to the contralateral side of the neck were used in 8 patients. 10 Corbitt et al. described only four contralateral anastomoses in their series of 40 out of 3090 microvascular reconstructions performed following failure of the initial free flap. 11
The presented work focuses on the results of free flap reconstruction of the head and neck, emphasizing the performance of anastomosis to the contralateral side of the neck. The frequency and success rate of the contralateral anastomosis serves as the primary objective. Secondary objectives include the flap related complications and the choice of the recipient vessels.
Patients and methods
This retrospective study included all patients who underwent ablative surgery for head and neck cancer with concurrent microvascular reconstruction at a tertiary referral center (Head and Neck Cancer Center at the Department for ENT, Head and Neck Surgery, University Clinic Erlangen, Germany) between January 1st, 2007 and February 28th, 2021. The study was approved by the local ethics committee (Application nr 16_21 B). On admission, all patients of the University Clinic Erlangen sign an authorization to use patient data for scientific purposes.
Inclusion criteria were complete medical and surgical charts, treatment due to head and neck cancer, age older than 18 years and the reconstruction with a microvascular free flap with both anastomoses performed on the contralateral side. The presence of a contralateral anastomosis was defined when the vascular pedicle, that is the artery and vein, crossed over the midline from its exit out of the pharyngeal tube to be anastomosed to vessels on the contralateral side of the neck. The following exclusion criteria were applied: total laryngectomy, tumors located in the midline, incomplete medical and surgical charts and age under 18 years. The patients’ medical charts and surgical records were evaluated according to age, sex, tumor entity and tumor stage according to the 7th edition of the TNM classification. Furthermore, previous surgical and radiation therapies applied to the head and neck were evaluated. Regarding the current tumor, characteristics of the performed surgery, the recipient vessels used for the anastomosis, operating time, performing surgeons, the success rate (defined as the flap survival rate), the flap-associated and general postoperative complications (e.g., venous thrombosis of the microvascular pedicle, insufficiency of the anastomosis, flap necrosis, or wound dehiscence), the length of postoperative stay on the intensive care unit and inpatient stay were analyzed.
Metric variables are presented as mean ± standard deviation (SD) and range (minimum (min)–maximum (max)).
Results
Patients’ demographic and clinical characteristics
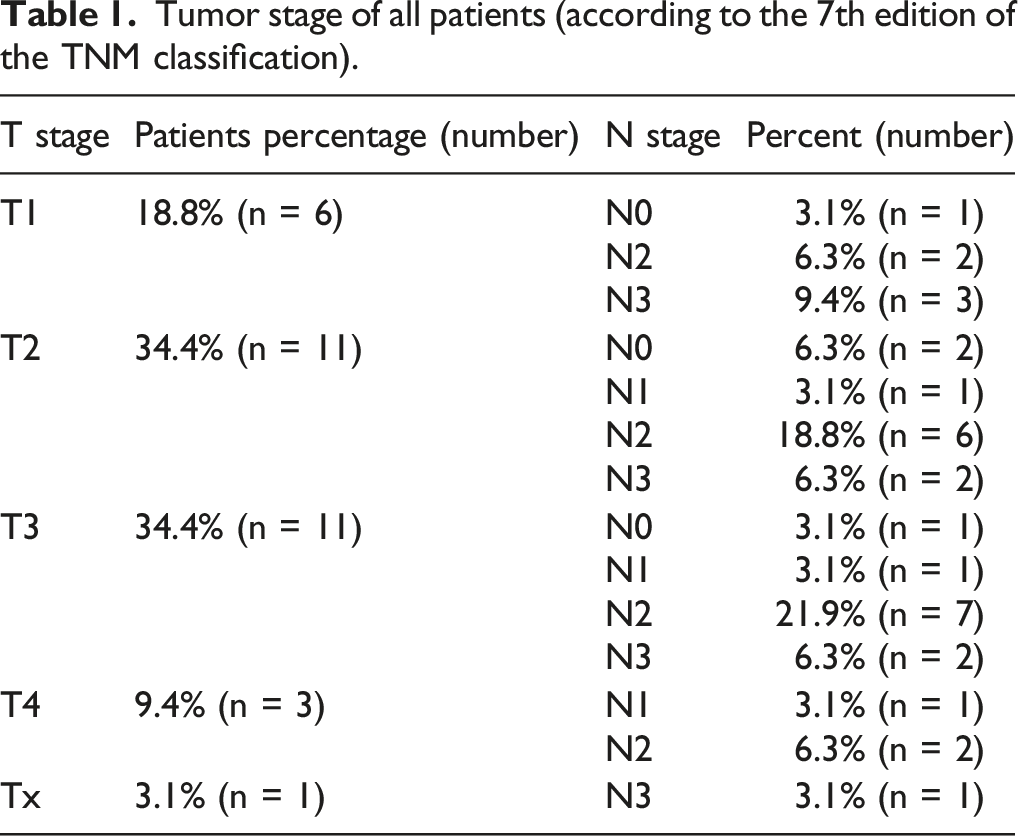
Tumor stage of all patients (according to the 7th edition of the TNM classification).
In total, 5 patients (12.5%) had undergone previous surgical treatments to the neck. One patient (patient no. 2) had a tumor resection and bilateral neck dissection five years earlier due to a right-sided tumor of the oral cavity (T1 N0) and was diagnosed with a left-sided hypopharyngeal tumor (T1 N0 M0) as a second malignancy. Another patient (patient no. 20) developed a second malignancy of the contralateral oropharynx (T3 N0 M0) 24 years after the first oropharyngeal tumor (T3 N3) that had been treated with primary chemoradiation. An elective salvage neck dissection one year later revealed no signs of malignancy within the suspected lymph nodes. One patient (patient no. 6) developed an ulcer and pharyngeal fistula 4 months after tumor resection and bilateral neck dissection (T2 N2a M0 oropharyngeal carcinoma), followed by the ligation of the ipsilateral external carotid artery because of a secondary hemorrhage and adjuvant chemoradiation. Another patient (patient no. 31) suffered from complete flap failure on day 14 after resection of an oropharyngeal tumor (T1 N2 M0) combined with bilateral neck dissection, using a RFFF. On day 12 after primary surgery, after developing a salivary fistula, the patient needed surgical hemostasis due to bleeding from the pedicle requiring ligation of the pedicle.
Two patients had had an incomplete resection shortly before complete tumor resection with simultaneous microvascular reconstruction. One patient underwent a R1-resection of a T2 N0 M0 oropharyngeal carcinoma (patient no. 9), whereas the other (patient no. 27) had a tumor debulking of a T3 N2b M0 oropharyngeal carcinoma.
Surgical characteristics and outcome
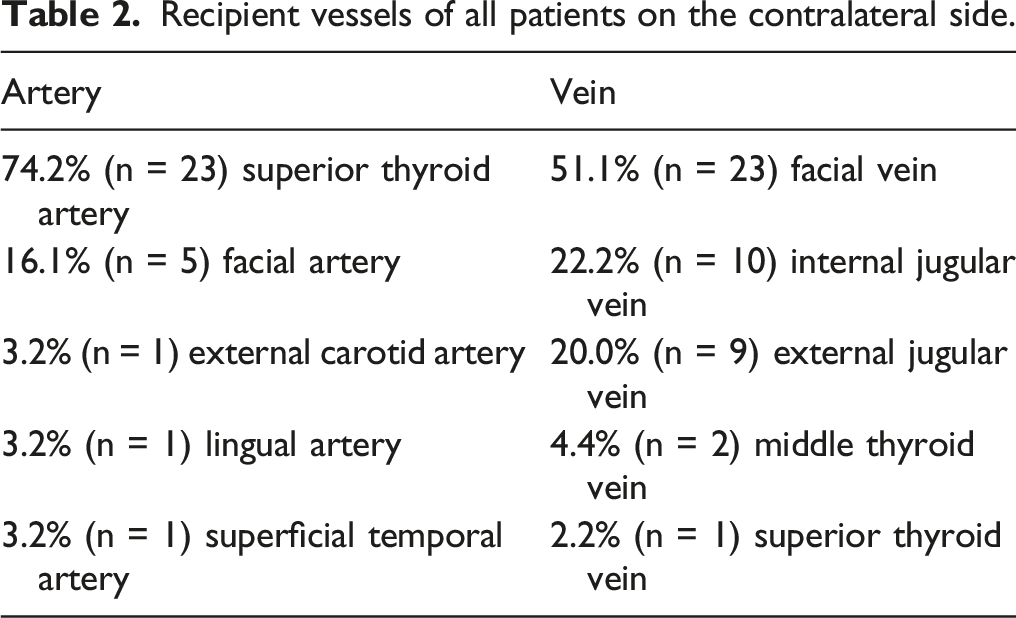
Reconstructions were performed with a RFFF in 30 patients (93.8%; Figure 1) and in two patients (6.7%) with an ALT. In addition to the RFFF or ALT, in 6.3% (n = 2; patient no. 10 and no. 22), a pectoralis major flap was raised simultaneously. The decision to perform a contralateral anastomosis of the microvascular free flap was at the surgeon’s discretion. There were various reasons for the contralateral microvascular anastomosis: 71.9% (n = 23) of the patients had an extended tumor resection surgery or a more radical neck dissection on the ipsilateral than on the contralateral side of the neck, which impaired the vascular status significantly. The recipient vessels were mostly the superior thyroid artery (74.2%; n = 23) and the facial vein (51.1%; n = 23), as shown in Table 2. Contralateral anastomosis of a secondary microvascular reconstruction using a RFF (patient no. 31). (A) RFFF after resection of a left-sided pT1 pN2 cM0 oropharyngeal tumor; (B) exit of the vascular pedicle on the ipsilateral side of the neck; (C) microvascular anastomosis on the contralateral side of the neck (radial artery with superficial temporal artery, cephalic vein with internal jugular vein, and smaller vein with external jugular vein). RFFF = radial forearm free flap. Recipient vessels of all patients on the contralateral side.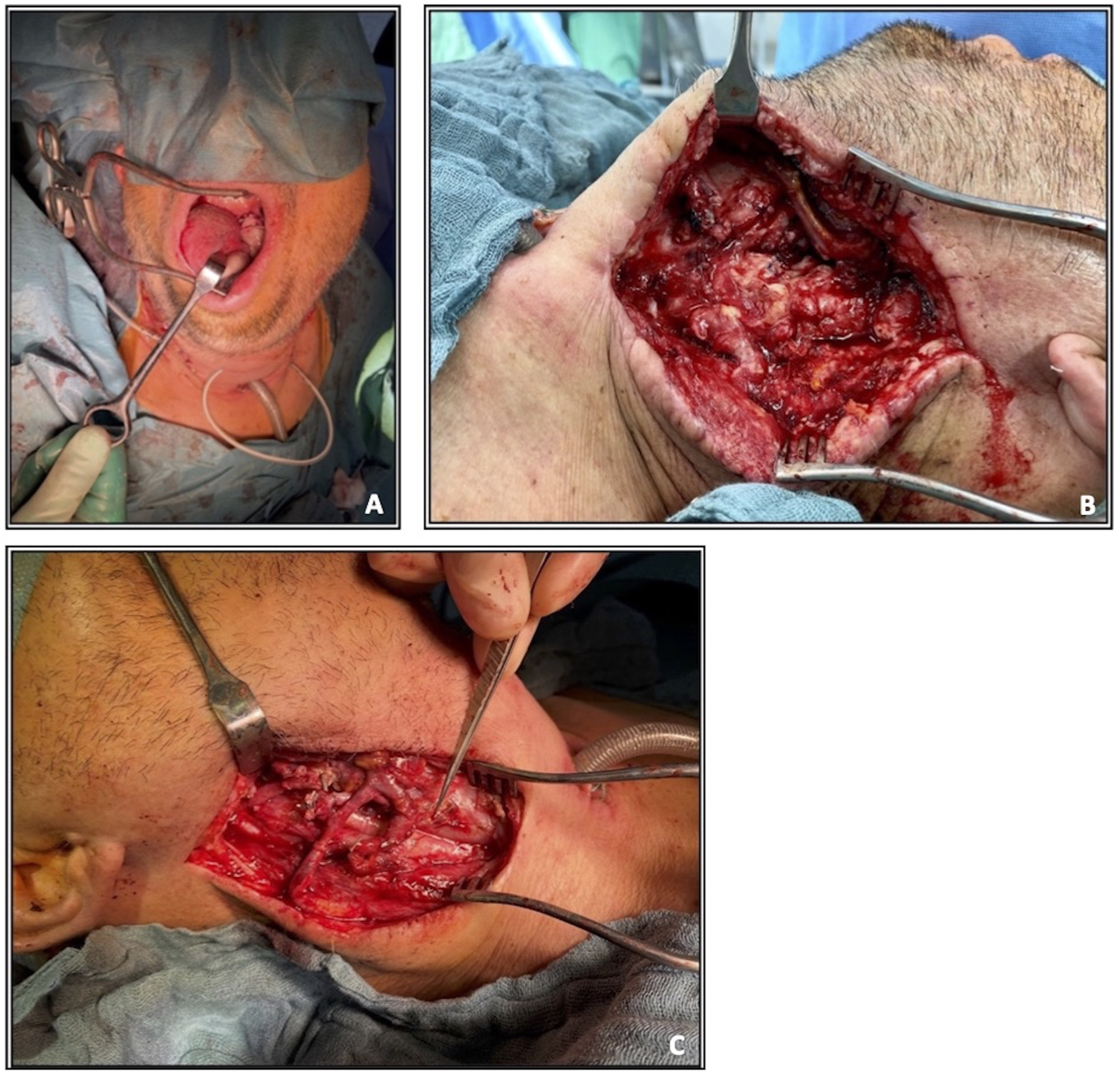
No complete flap failure was observed in any of the patients examined. Flap-associated complications occurred in four patients (12.5%). In one case (3.1%; patient no. 1), partial flap necrosis occurred. After surgical removal of necrotic tissue of the flap on postoperative day 15, primary wound closure was facilitated. One patient developed a venous thrombosis of the microvascular pedicle (patient no. 5), that is the facial vein, and one patient a wound dehiscence (patient no. 30). Both complications were resolved on day 3 and day 28, respectively, by revision surgery. One patient (patient no. 24) started bleeding out of the arterial anastomosis on postoperative day 6. Furthermore, the same patient also started bleeding from his replacement patch surgery of the ipsilateral internal carotid artery which had been performed during the previous surgeries because of tumor infiltration of the ipsilateral internal carotid artery. The patient died because of the massive bilateral bleeding. Another patient (patient no. 27) died in the course of rehabilitation due to a lethal embolism of the pulmonary artery.
The mean length of surgery was 780.8 min ± 117.2 min (562 min to 1124 min). A maximum of 8 surgeons participated in the corresponding operations.
Postoperatively, all patients were transferred to the clinic’s own intensive care unit for a mean length of stay of 6.0 ± 4.5 days (1-27 days). Patients were hospitalized for 23.7 ± 9.0 days (6-46 days) on average.
Surgical characteristics of all patients.
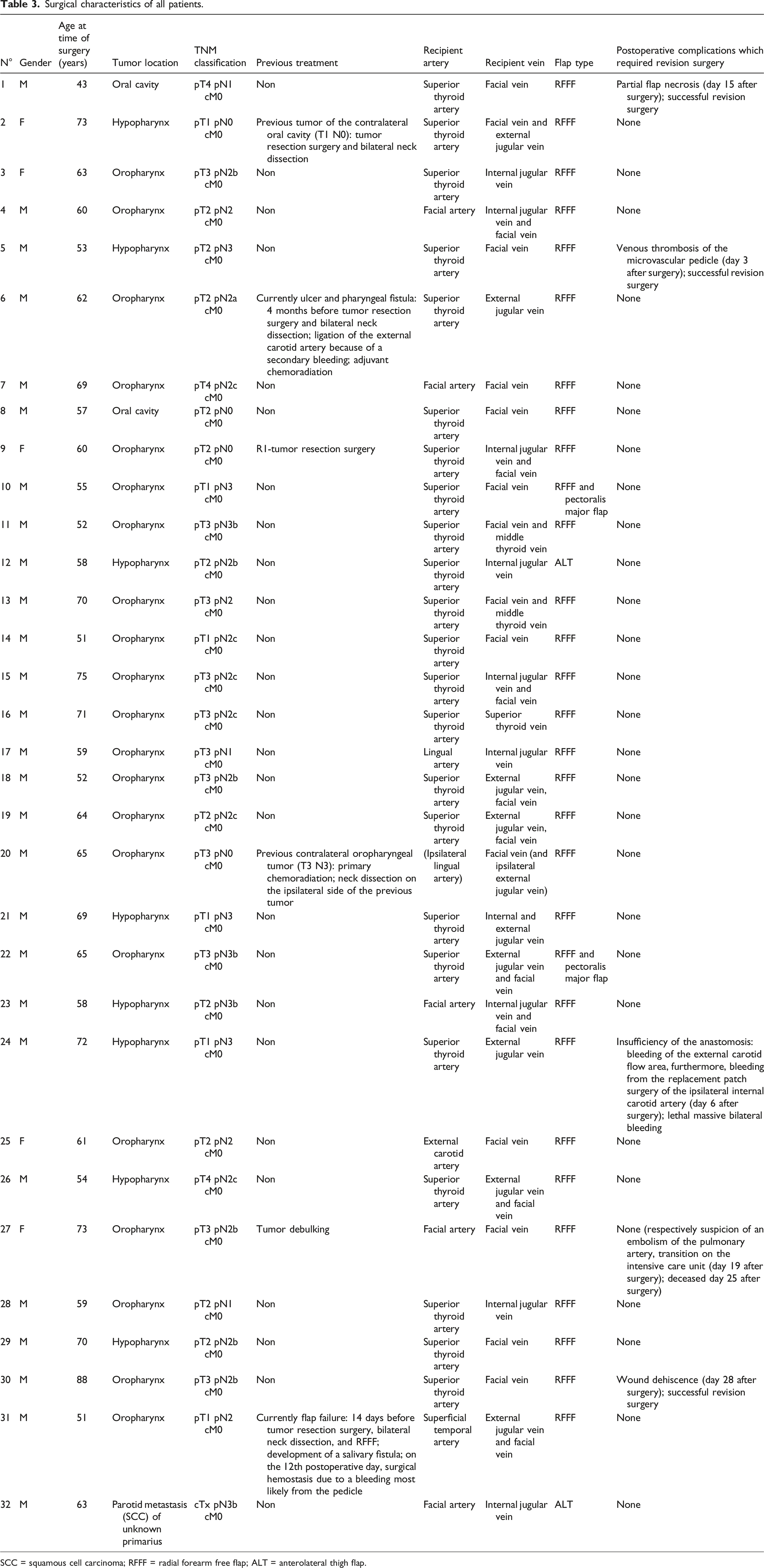
SCC = squamous cell carcinoma; RFFF = radial forearm free flap; ALT = anterolateral thigh flap.
Discussion
This single-center study demonstrates that in vessel-poor necks, contralateral anastomosis is a feasible and effective option in head and neck microvascular reconstruction. In this series, the need to perform contralateral anastomosis was rare (10.0%) but yielded a high success rate (100%) without complete flap loss. The most commonly used RFFF (93.8%) allowed arterial and venous anastomosis predominantly to the contralateral superior thyroid artery (74.2%) and facial vein (51.1%), respectively. Flap-related complications (12.5%) were not significantly increased and were all managed successfully.
Our study is in line with reports on microvascular anastomoses to the contralateral side of the previously untreated neck occurring in 3% (24 out of 675 patients) to 18% (7 out of 40 patients) of the patients.7,12 After previous ablative head and neck surgery, including neck dissection, contralateral anastomoses were performed in 5% (7 out of 136 patients) to 61% (36 out of 50 patients).7,10,13,14 Second microvascular free flaps following initial flap reconstruction required a contralateral anastomosis in 20% up to 65% of the cases.8,11,15,16 In the presented series, the relatively lower percentage of the contralateral anastomosis can be attributed to few previously treated patients.
The reported success rate is comparable to other studies yielding rates ranging from 96% to 99%.11,13,15,17-19 Alam et al. examined patients who needed a second microvascular free flap in the head and neck region. In their opinion, the availability of recipient vessels is crucial for the flap survival rate. No flap loss occurred by choosing the recipient vessels according to the best vascular fit on either side of the neck. 8 Generally, pedicle veins with a big diameter are important for the safety of a free flap.12,17 Long vascular pedicles allow contralateral anastomoses, obliviate vein grafts and ensure safe and full head and neck mobility.8,10,12,14,17,20 There was no significant correlation between the flap survival rate or the total flap loss and any previous treatment such as neck dissection or radiation therapy.7,13
Still, there are recommendations for a contralateral microvascular anastomosis following a (radical or modified radical) neck dissection7,14 when the ipsilateral internal jugular vein is missing 21 or following radiotherapy.7,9 Previous neck dissection diminishes the selection of recipient vessels and can complicate the dissection of the recipient vessels. 7 After previous neck dissection, tissue scarring or an impaired vascular status can be expected that requires a contralateral anastomosis.13,14 Of note, in revision surgery after free flap failure, using the same recipient vessels is not recommended because of a possible vasculitis. 16 Compared to other studies, in this current study, the contralateral anastomosis was performed in 12.5% of the cases due to the consequences of a previous treatment to the neck. Accordingly, contralateral microvascular anastomosis was mostly performed simultaneously (71.9%) to extended tumor resection or a more radical neck dissection on the ipsilateral than on the contralateral side of the neck which significantly impaired the ipsilateral vascular status. Taking all aspects into consideration, a contralateral anastomosis can be as equally successfully performed as long as the recipient vessels are a good fit. This is even preferable in a scenario where the contralateral recipient vessels are superior to the ipsilateral ones.
In contrast to our study, in case of the ipsilateral internal jugular vein having been resected earlier, Head et al. favored an anastomosis on the contralateral facial artery and on the contralateral external and internal jugular vein. 14 Other groups recommend in the case of a missing ipsilateral external carotid artery or a missing ipsilateral internal or external jugular vein, among other options, a contralateral anastomosis on the external carotid artery or on the internal or external jugular vein.7,10 The ideal recipient vessel should not have been previously treated. 7 It should be free of scars and diseases and have an adequate length and caliber, comparable to the pedicle vessels. 22 If the vessels are chosen wisely, there is no difference in the success rate between the different vessels. 7 Therefore, the best match concerning comparable vein diameters, big vein diameters, long pedicules and until now untreated vessels should be considered for anastomosing the microvascular pedicle.
The RFFF was predominantly used in the presented series as it represents the workhorse flap at our institution. The advantage of this specific type of flap lies in the length and diameter of the vascular pedicle. 12 However, fibula flaps, rectus abdominis flaps,13,14,17 latissimus dorsi flaps, 13 subscapular system flaps and iliac crest flaps14,17 were also reported to be feasible options. Thus, the foremost prerequisite for successful free flap reconstruction is the experience of the performing surgeon with the respective technique.
In the literature, a revision of microvascular reconstruction between 5% and 14% has been described13,21,23 that is comparable to our study. Hanasono et al. also did not report a higher complication rate using alternative anastomoses such as the contralateral anastomosis. 10 According to Mücke et al. the complication rate was elevated after neck dissection, after radiation therapy or in case of an anastomosis with the superior thyroid vein. 23 In contrast, Head et al. did not find an increased complication rate after neck dissection. 14 Last but not least, preoperative health condition influences the perioperative complication rate.17,19,23 Compared to other studies with or without contralateral anastomoses, previously treated or not, our complication rate observed with contralateral anastomoses is similar to existing publications.
The main limitation of this study is due to the inevitable bias of the retrospective character. Furthermore, the presented results need to be interpreted in view of the predominantly used RFFF as it represents the common flap for head and neck reconstruction in the investigated cases. Of note, the decision to perform a contralateral anastomosis was left to the surgeon’s discretion. Last, the presented work does not aim to provide an algorithm for the choice of contralateral vessels as this was beyond the scope of this study. However, the total number of contralateral anastomoses performed is comparatively high and the results regarding flap success rate, flap loss and complications are comparatively good.
Conclusions
In the event of a lack of suitable vessels for ipsilateral anastomosis, anastomosis on the contralateral side of the neck is a viable and safe option, especially when using the RFFF. Anastomosing to the contralateral side of the neck does not significantly increase flap-associated complication rates and consequently does not decrease the success rate of the microvascular reconstruction.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
