Abstract
Patients with inherited coagulopathies—including thrombophilias—undergoing microvascular reconstruction face elevated thrombotic risk, yet optimal perioperative strategies remain undefined. This retrospective case series evaluates perioperative outcomes in patients with confirmed inherited coagulopathies who underwent head and neck free flap reconstruction at a single tertiary academic center between April 2014 and October 2024. Fifteen patients met the inclusion criteria (53.3% female; median age 59 years). The most common coagulopathies were sickle cell trait (40.0%) and Factor V Leiden (26.7%). All patients received preoperative anticoagulation: 60.0% received subcutaneous (SC) unfractionated heparin (UFH), 33.3% enoxaparin, and 6.7% fondaparinux. Intraoperative anticoagulation was used in 20.0% of cases, primarily enoxaparin. Postoperative anticoagulation included SC UFH or low-molecular-weight heparin in 86.7% of patients, with 33.3% also receiving aspirin and 20.0% transitioning to direct oral anticoagulants. Two patients (13.3%) experienced flap loss due to thrombosis; neither received intraoperative anticoagulation. No major bleeding events occurred. Transfusions were required in 40.0% postoperatively and 20.0% intraoperatively. These findings underscore the need for targeted screening, multidisciplinary coordination, and individualized anticoagulation regimens to mitigate thrombotic complications in this high-risk population. Further prospective studies are warranted to develop standardized perioperative protocols and optimize microsurgical outcomes in patients with inherited coagulopathies, including hypercoagulable states.
Keywords
Introduction
Free tissue transfer is central to head and neck (H&N) reconstruction, offering functional and esthetic benefits. 1 Thrombotic events—arterial or venous—are the dominant cause of flap loss, accounting for up to 85% of all failures despite overall failure rates of only 2% to 10%.1-4 Predictors for thrombotic complications include diabetes, prior neck surgery, radiotherapy, and smoking.2,5
Hypercoagulable states are recognized contributors to flap failure. 6 Inherited conditions (eg, Factor V Leiden, prothrombin G20210A, and protein C/S or antithrombin III deficiency) affect 5% to 8% of Western populations, while acquired coagulopathies (eg, malignancy-associated hypercoagulability, cirrhosis) are even more prevalent in surgical cohorts.6-9 Their propensity for peri-anastomotic thrombosis renders patients a high-risk group during free-flap reconstruction.
Despite risks, data on H&N microvascular outcomes in this population are limited, and perioperative management lacks consensus. This case series describes perioperative courses and outcomes in patients with inherited coagulopathies—including thrombophilias—undergoing H&N free flaps, aiming to inform risk stratification and guide evidence-based perioperative protocols for this high-risk cohort.
Materials and Methods
Data Collection
Following Institutional Review Board approval (#24-0785), electronic health records were queried using Informatics for Integrating Biology and the Bedside (i2b2) to identify eligible patients. Two reviewers (M.M. and G.A.) performed a chart review.
Cohort Definition
Inclusion criteria were patients aged ≥18 years who underwent a H&N free flap between April 2014 and October 2024, with a diagnosed inherited coagulopathy. The corresponding Current Procedural Terminology (CPT) codes for H&N reconstruction and International Classification of Diseases (ICD-9)/-10 codes for the coagulopathies were input into the query (Supplementary Table 1). Exclusion criteria included non-H&N reconstruction, incomplete documentation, or <90 days of follow-up.
Outcomes
The primary outcome was flap-related thrombosis occurring intraoperatively or postoperatively and confirmed by clinical signs or intraoperative findings during take-back. Secondary outcomes included complications within 90 days postoperatively. These encompassed flap complications (eg, total or partial flap loss, infection, hematoma) and medical complications. Complete flap loss was defined as full necrosis of the flap requiring debridement or removal. Partial flap loss was defined as necrosis involving ≥20% of the flap surface area, based on clinical judgment in operative notes, provider assessments, and photographic records. We also recorded transfusion events (intra/postoperative).
Demographics and comorbidities were collected, including age, sex, body mass index (BMI), smoking history, American Society of Anesthesiologists (ASA) class, and thrombotic risk factors (alcohol abuse, contraceptive use, and immobility). Immobility was defined as an Eastern Cooperative Oncology Group score of ≥3. Each patient’s coagulopathy subtype was documented.
Surgical details included indication, flap type, operative and ischemia time, and blood loss. Anticoagulation regimens were documented, including timing, agent, and adjunctive antiplatelet use.
Statistical Methods
This study was designed as a descriptive case series with no inferential statistical analyses. Continuous variables such as age, BMI, operative time, ischemia time, and estimated blood loss were reported as individual values. Categorical variables—including patient sex, ASA class, coagulopathy subtype, flap type, anticoagulation regimen, and postoperative complications—were presented as case-level data. Given the small, heterogeneous sample, no comparative analyses were performed. Data were tabulated in Microsoft Excel.
Results
The cohort (n = 15) included 8 females, 7 males; median age 59 (range, 27-76) and BMI 26.3 kg/m² (range, 19.6-38.6). Common comorbidities included hypertension (60%), cardiac disease (33%), prior thromboembolic events (27%), chronic kidney disease (20%), diabetes mellitus, and chronic obstructive pulmonary disease (13% each; Table 1). Four were former smokers, and 1 was an active smoker. None had documented alcohol abuse or immobility. One patient (case #6) was taking oral contraceptive pills. The most common coagulopathies included sickle cell trait (n = 6, 40%) and Factor V Leiden (n = 4, 27%; Figure 1). One patient carried both sickle cell trait and heterozygous Factor V Leiden. Three cases were undiagnosed preoperatively.
Demographic and Comorbidity.
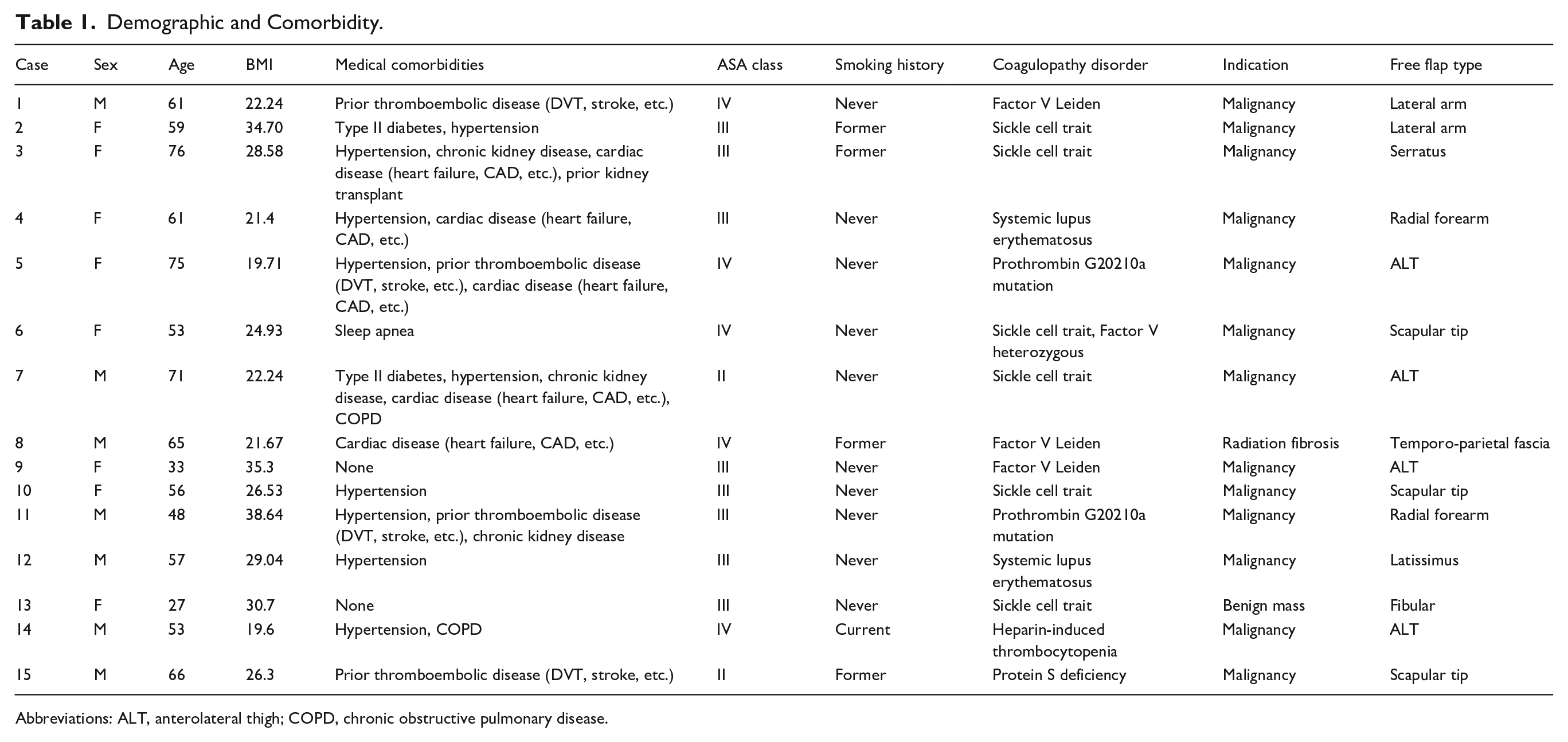
Abbreviations: ALT, anterolateral thigh; COPD, chronic obstructive pulmonary disease.
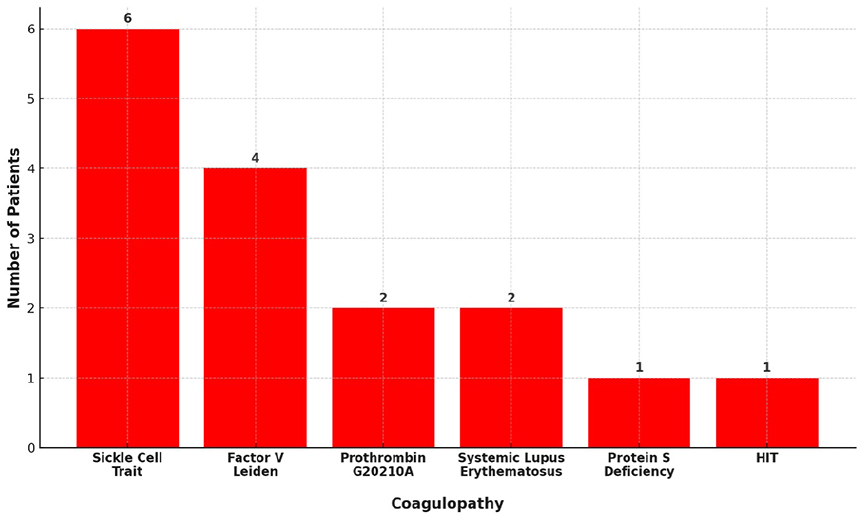
Distribution of coagulopathy diagnoses.
Most reconstructions (93%) were oncologic, with 1 case addressing radiation fibrosis. Free flap types included anterolateral thigh (ALT, n = 4), scapular tip (n = 3), radial forearm (n = 2), lateral arm (n = 2), and single cases of serratus, latissimus dorsi, fibular, and temporoparietal fascia flaps (Table 1). Operative time ranged from 256 to 930 minutes, with available ischemia times from 52 to 216 minutes. Blood loss ranged from 50 to 800 mL (Table 2).
Surgical, Coagulation, and Complication Data.
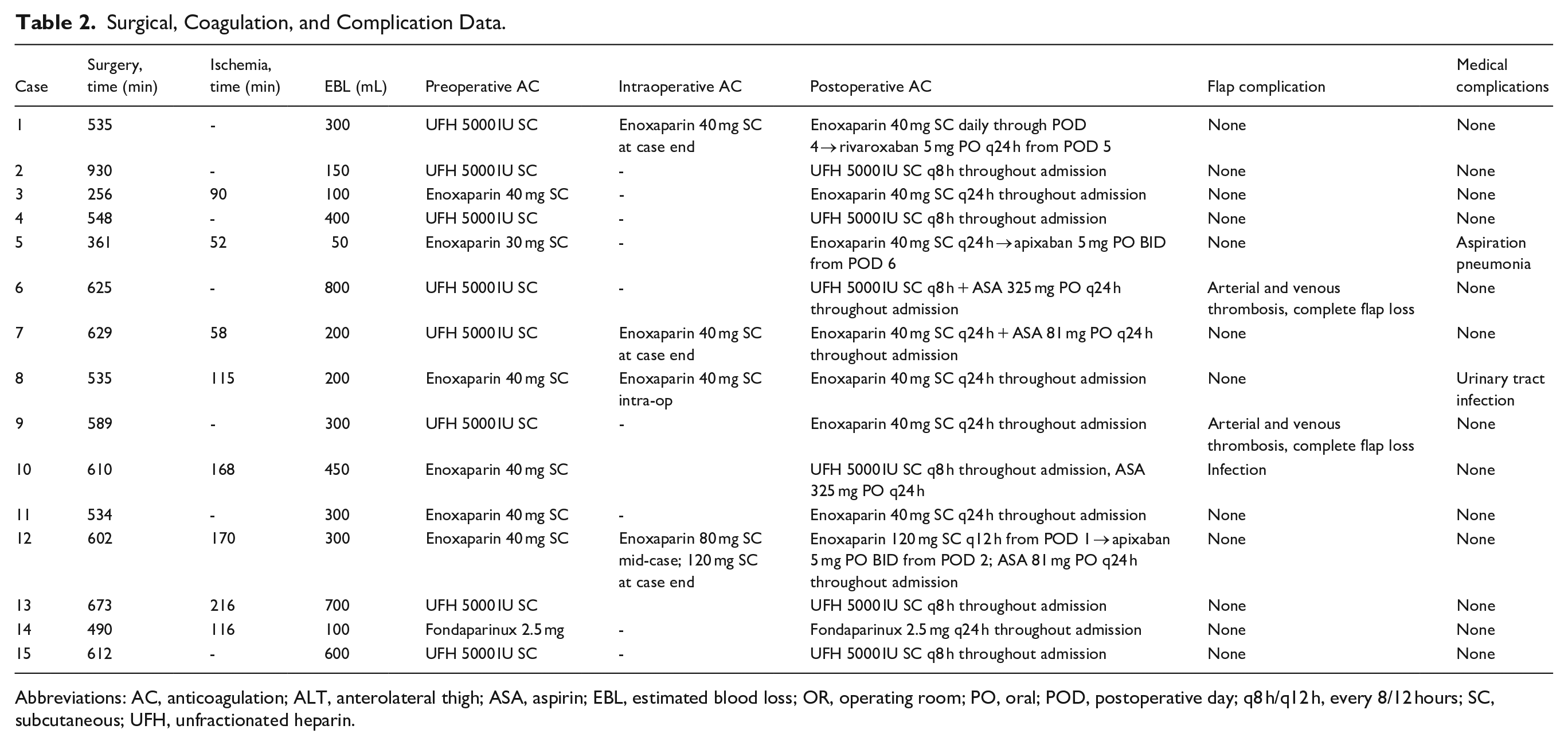
Abbreviations: AC, anticoagulation; ALT, anterolateral thigh; ASA, aspirin; EBL, estimated blood loss; OR, operating room; PO, oral; POD, postoperative day; q8 h/q12 h, every 8/12 hours; SC, subcutaneous; UFH, unfractionated heparin.
Perioperative anticoagulation regimens varied across patients (Figure 2). Preoperatively, patients most commonly received either subcutaneous (SC) heparin (5000 units) or enoxaparin (30-40 mg). One patient received fondaparinux 2.5 mg due to heparin-induced thrombocytopenia. Four patients (26.7%) received additional intraoperative anticoagulation beyond heparinized saline flushes. Three patients received enoxaparin at case completion; 1 received it intraoperatively at mid-case and again at completion. Postoperatively, 3 patients (20.0%) transitioned to direct oral anticoagulants within a week. Three patients (20%) received aspirin, ranging from 81 to 325 mg daily. Heparin infusion protocols (5000 units every 8 hours) were employed in over one-third of cases (Table 2).
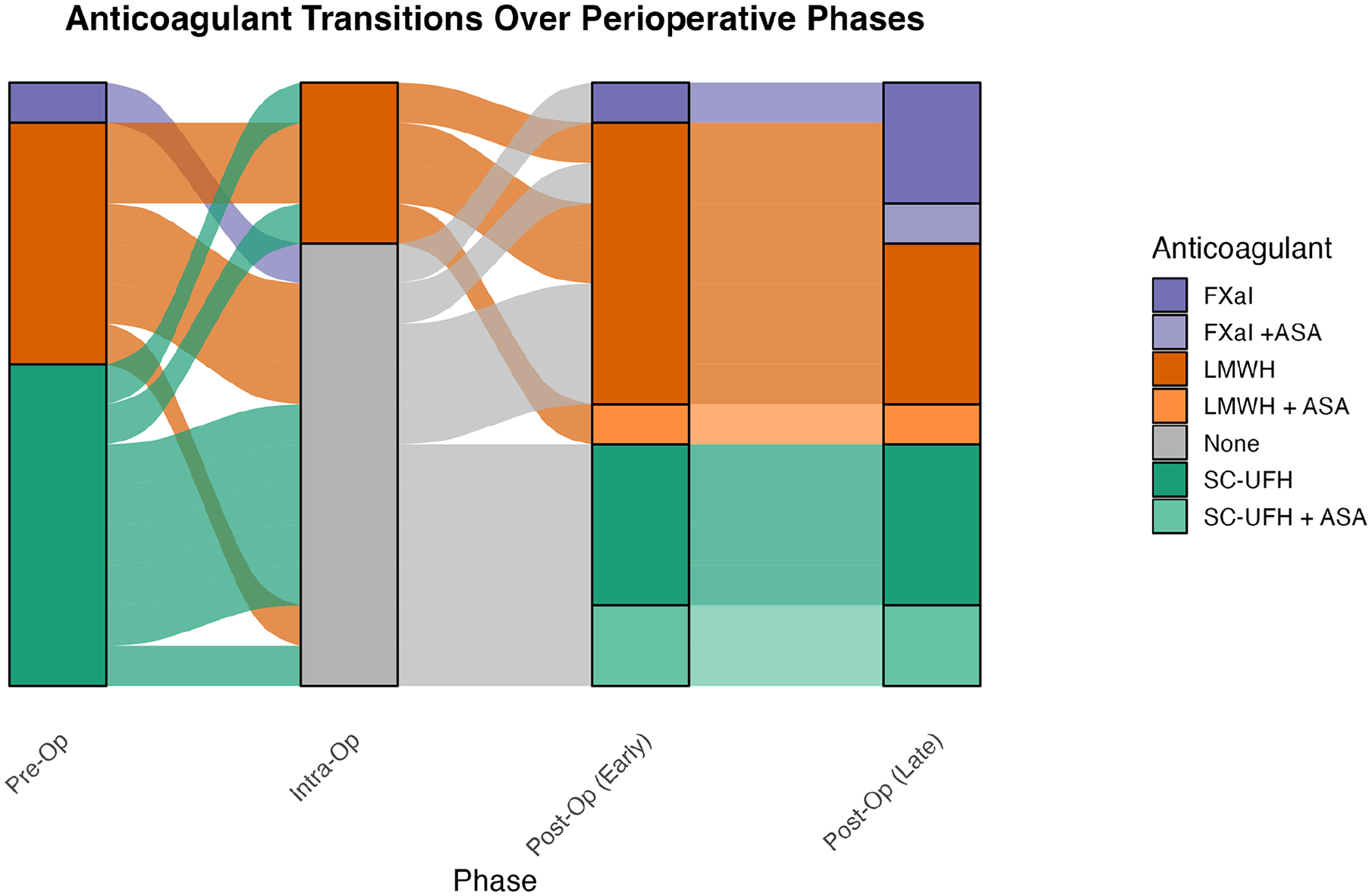
Anticoagulant transitions over perioperative phases.
The 2 patients (13.3%) who experienced vascular compromise also experienced complete flap loss due to both arterial and venous thrombosis. One of these patients was heterozygous for Factor V Leiden and had sickle cell trait, and the other had Factor V Leiden. Both received 5000 units of SC heparin preoperatively, and 1 patient continued this every 8 hours throughout admission, while the other received 40 mg of daily enoxaparin. In both, postoperative thrombosis caused flap loss despite revascularization efforts. In the first case, the patient developed flap compromise on postoperative day 3 (POD 3), prompting re-exploration, which revealed both arterial and venous thrombosis. The arterial anastomosis was revised from the facial to the superior thyroid artery, and the venous anastomosis was reestablished. However, extensive thrombosis of the jugular venous system precluded outflow, necessitating flap excision. A contralateral ALT flap was performed on POD 4, but partial flap necrosis required 2 subsequent returns to the operating room: first for local flap coverage and later for a medial sural artery perforator flap.
In the second case, flap compromise was noted on POD 6. Intraoperative findings again included arterial and venous clots. Arterial inflow was restored with intravenous (IV) unfractionated heparin (UFH), local tissue plasminogen activator injection, and thrombectomy. Despite these interventions, venous outflow could now be re-established, and the flap was sacrificed. The patient subsequently underwent a successful scapular-tip free flap reconstruction 1 year later.
The overall flap success rate was 80.0%. Postoperative blood transfusions were required in 6 patients (40.0%), and 4 patients (26.7%) received intraoperative transfusions. The median intraoperative transfusion volume was 425 mL (range, 250-1200 mL), and the median postoperative volume was 649 mL (range, 278-1300 mL). One patient (Case #13) required 1200 mL intraoperatively; 2 (Cases #9, #10) required >1100 mL postoperatively. Two patients (13.3%) experienced non-flap-related complications: 1 urinary tract infection and 1 case of aspiration pneumonia (Table 2).
Discussion
Despite high success rates, H&N reconstruction remains susceptible to failure—particularly with vascular compromise. 10 Coagulopathies, especially inherited hypercoagulable disorders, are increasingly recognized contributors to flap failure across all types of autologous reconstruction.6,11,12 In a review of 1355 patients undergoing free flaps, 3.03% had hypercoagulable conditions, with flap loss rate exceeding 15%—much higher than the typical 1% to 2%.1,13-15 Despite this elevated risk, no standardized protocol exists for screening or anticoagulation in patients with inherited coagulopathies. 16
In our series of 15 patients with documented inherited coagulopathies—including thrombophilias and other disorders such as lupus and heparin-induced thrombocytopenia—the flap failure rate was 13.3%. In both failures, re-exploration revealed arterial and venous thrombosis, despite anticoagulation. Salvage was unsuccessful, requiring secondary reconstruction with alternative free flaps, which succeeded. Notably, both patients with flap loss had Factor V Leiden mutations, and 1 had concurrent sickle cell trait—emphasizing the heightened thrombotic risk in patients with multiple prothrombotic factors. The 0% flap salvage rate observed in our cohort aligns with published literature demonstrating low salvage rates in inherited hypercoagulable states, particularly Factor V Leiden or multiple prothrombotic risk factors.13,17 While timely re-exploration remains essential, intraoperative strategies such as systemic heparinization and complete mechanical thrombus removal have shown preliminary benefit. However, even these measures prove insufficient in patients with inherited coagulopathies. 18
Patients with H&N malignancies often present with compounding thrombotic risk factors, including advanced age, malignancy-associated hypercoagulability, history of tobacco/alcohol use, immobility, and prior radiation. 19 These baseline risks are exacerbated by coexisting inherited coagulopathies. Although the Enhanced Recovery After Surgery Society acknowledges the importance of thromboprophylaxis in free flap reconstruction, their 2017 guidelines note no pharmacologic regimen conclusively prevents microvascular thrombosis. 20 While society guidelines provide recommendations for systemic Deep Vein Thrombosis/ Pulmonary Embolism (DVT/PE) prophylaxis, they do not address microvascular anastomotic thrombosis. 21 Amongst agents, low-molecular-weight heparin (LMWH) is favored for its safety profile. 19 A recent cohort study found no significant difference in flap compromise between patients receiving IV-UFH, SC-UFH, or SC-UFH combined with aspirin, suggesting that SC-UFH alone may be a safe, pragmatic option. 22 However, the optimal timing, intensity, and duration of anticoagulation remain debated. Meta-analyses suggest routine postoperative anticoagulation may not reduce thrombosis or improve flap survival, yet it increases bleeding risk.23,24
There is no standardized anticoagulation protocol at our institution for free flap patients without coagulopathies. For oncologic reconstructions, patients typically receive 40 mg enoxaparin preoperatively, as most have a Caprini score ≥5. Postoperative regimens vary; some continue enoxaparin alone, while others add low-dose aspirin (81 mg daily). In our cohort, most patients received preoperative SC UFH. Intraoperative anticoagulation was selectively administered, often as enoxaparin at case completion. Postoperatively, >75% received SC-UFH or LMWH, and approximately one-third were treated with adjunctive aspirin. Importantly, no patients experienced clinically significant bleeding complications. Among the 2 patients with flap failure, neither received intraoperative anticoagulation, and postoperative regimens differed (one transitioned to LMWH, the other continued SC UFH).
Tailored anticoagulation strategies are crucial in patients with inherited coagulopathies undergoing microsurgical reconstruction. In H&N reconstruction, Faber et al described 3 cases of flap failure in undiagnosed Factor V Leiden patients undergoing H&N reconstruction. Implementation of a revised protocol—including preoperative enoxaparin (0.5 mg/kg daily), intraoperative IV heparin (1500 IU), and postoperative heparin infusion titrated to an activated Partial Thromboplastin Time (aPTT) of 45 seconds, followed by enoxaparin BID—resulted in successful salvage in all subsequent reconstructions. 17 In a broader analysis of various flap types, Wang et al found no outcome differences among 4 anticoagulation strategies, reinforcing that multidisciplinary planning may outweigh specific regimen choice. 13
Preoperative identification of inherited coagulopathies is essential to minimize thrombotic risk.13,25 In our case series, among the 3 patients with previously undiagnosed coagulopathies, 2 experienced flap failure, underscoring the potential consequences of unrecognized hypercoagulable states. By contrast, among patients with known Factor V Leiden mutations, a structured screening protocol—including targeted questionnaires, coagulation panels, confirmatory genetic testing, and hematology referral—cut flap loss rates 10-fold. 26
At our institution, preoperative screening is typically initiated by the anesthesia team and includes assessment of personal and family history of thromboembolic events. In lower extremity free flaps, Deldar et al employed a risk-stratified algorithm incorporating both clinical and laboratory-based coagulopathy work-up to guide anticoagulation, significantly reducing flap loss rates. 27 These findings suggest that similar stratification strategies may be applied in H&N reconstruction to balance thrombotic and bleeding risks.
Hematology consultation and individualized anticoagulation regimens based on comprehensive coagulopathy work-ups have shown improved reconstructive outcomes.13,28 However, the cost-effectiveness of routine screening remains a concern given the low prevalence of hereditary coagulopathies. 8 Even when risk is identified during preoperative evaluation, insufficient time often prevents confirmatory testing or hematology input before surgery. Targeted, risk-stratified screening protocols may offer a more practical balance between clinical benefit and cost, but optimizing their integration into preoperative workflows will be essential to maximize impact.
This study is one of the few focusing on H&N microsurgery outcomes in patients with inherited coagulopathies, offering insight into perioperative management. However, this study is limited by its retrospective, single-institution design and relatively small sample size, which may limit generalizability. The heterogeneity in anticoagulation regimens and timing reflects real-world variability but may confound comparisons. Larger, prospective studies are needed to better define optimal screening and anticoagulation protocols.
Collectively, this case series highlights the deleterious impact of inherited coagulopathies—particularly hypercoagulable disorders—on free flap viability in H&N reconstruction and underscores the need for evidence-based protocols. Early identification, multidisciplinary coordination, and individualized perioperative anticoagulation regimens are essential to optimize outcomes in this high-risk patient population.
Supplemental Material
sj-docx-1-ear-10.1177_01455613251356340 – Supplemental material for Head and Neck Free-Flap Reconstruction in Patients With Pre-Existing Coagulopathies: A Case Series
Supplemental material, sj-docx-1-ear-10.1177_01455613251356340 for Head and Neck Free-Flap Reconstruction in Patients With Pre-Existing Coagulopathies: A Case Series by Grace Anne Longfellow, Makayla Matthews, Gabrielle Adams, Ezer H. Benaim, Trevor Hackman and Christopher Blake Sullivan in Ear, Nose & Throat Journal
Footnotes
Ethical Considerations
This study was reviewed and approved by the Institutional Review Board at the University of North Carolina at Chapel Hill (IRB #24-0785). All procedures were conducted in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments.
Consent to Participate
Informed consent was waived due to the retrospective nature of the study and the use of de-identified data.
Author Contributions
G.A.L. and M.M. contributed equally to study design, data analysis, and manuscript preparation. G.A., E.H.B., T.H., and C.B.S. provided critical revisions. All authors reviewed and approved the final manuscript.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Level of Evidence
4
Data Availability Statement
Data are available from the corresponding author upon reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
