Abstract
Hamartomas are common in the lung, kidney, liver, spleen, and, but rare, in the sinonasal tract. Respiratory epithelial adenomatoid hamartomas (REAHs) are benign lesions common in men aged 30 to 90 years. Approximately 70% of REAHs in the head and neck region originate from the posterior nasal septum. We present an unusual case of REAH originating from the maxillary sinus and extending to the nasopharynx of an adolescent boy.
A 17-year-old boy without any salient medical history presented to our department with nasal obstruction that had persisted for 7 years as well as greenish nasal discharge, hyposmia, and a complaint of fetid smell. Sinoscopy of the osteomeatal complex (OMC) revealed bilateral mucopus and a large right polypoid tumor extending into the nasopharynx. Computed tomography of the paranasal sinuses revealed soft-tissue opacification around the right OMC, frontal sinus, ethmoid sinus, maxillary sinus, and nasopharynx. We performed bilateral endoscopic sinus surgery. REAH and chronic rhinosinusitis with nasal polyps were diagnosed on the basis of a pathology report. No evidence of recurrence was observed by 6 months after surgery, and his hyposmia, nasal obstruction, and purulent nasal discharge were alleviated considerably. Accurate diagnosis based on pathology is essential for determining the optimal treatment, which for REAH is complete surgical excision.
Introduction
Respiratory epithelial adenomatoid hamartoma (REAH) was first described in 1995 by Weinig and Heffner in a case series of 31 patients and is diagnosed on the basis of World Health Organization criteria—the proliferation of widely spaced small to medium-sized glands lined by a multilayered ciliated respiratory epithelium, often with goblet cell admixture. Such glands arise in direct continuity with the surface epithelium and invaginate downward into the submucosa.1,2 Moreover, they have a thick eosinophilic basement membrane and do not present with atypia or metaplasia.3,4
Approximately 70% of REAHs in the head and neck region originate from the posterior nasal septum. Other common sites include the nasopharynx, lateral nasal wall, olfactory cleft, and frontal, maxillary, and ethmoid sinuses; such REAHs are mostly unilateral.1,5 REAH is a benign lesion predominantly affecting men between the ages of 30 and 90 years, with a male-to-female ratio of 3:2.3,5-7 Its clinical symptoms are nonspecific and include nasal obstruction, nasal stuffiness, rhinorrhea, deviated septum, epistaxis, hyposmia, frontal headache, and chronic rhinosinusitis; these symptoms can occur for months to many years.3,7,8
Hamartomas can occur in any part of the body, originating commonly from the lung, kidney, liver, spleen, and intestine but rarely from the sinonasal tract.9,10 Hamartomas have no tendency toward unimpeded growth; their growth is self-limiting. They have no malignant potential but do not subside spontaneously.11,12
Histopathological features are the gold standard for the diagnosis of REAH. 12 Complete surgical removal of the REAH, including the tumor pedicle, is the standard treatment.6,13 No cases of recurrence have been reported after complete surgical resection, and REAHs do not need to be excised from asymptomatic patients or aggressively resected from symptomatic patients.7,12 In polypoid cases, opacification and a widened olfactory cleft (>10 mm) on computed tomography (CT) imaging should increase suspicion of REAH.3,13-15 We present an unusual case of REAH originating from the maxillary sinus and extending into the nasopharynx of an adolescent boy.
Case report
A 17-year-old boy without any medical history presented to our department with nasal obstruction that had persisted for 7 years as well as greenish nasal discharge, hyposmia, and a complaint of fetid smell. Sinoscopy of the osteomeatal complex (OMC) revealed bilateral mucopus and a large right polypoid tumor extending into the nasopharynx; a nasopharyngeal tumor was suspected. He did not have otitis media with effusion in the right ear or palpable lymphadenopathy. His EB-IgA and EBV (EA + EBNA-1) IgA levels were in the normal ranges. Paranasal sinus CT revealed soft-tissue opacification around the right OMC, frontal sinus, ethmoid sinus, maxillary sinus, and nasopharynx (Figure 1). Bilateral chronic rhinosinusitis with nasal polyps and adenoid hyperplasia was impressed after a physical examination and imaging study. Pre-OP CT scan showed soft-tissue opacification around right OMC, frontal sinus, ethmoid sinus, maxillary sinus, and nasopharynx.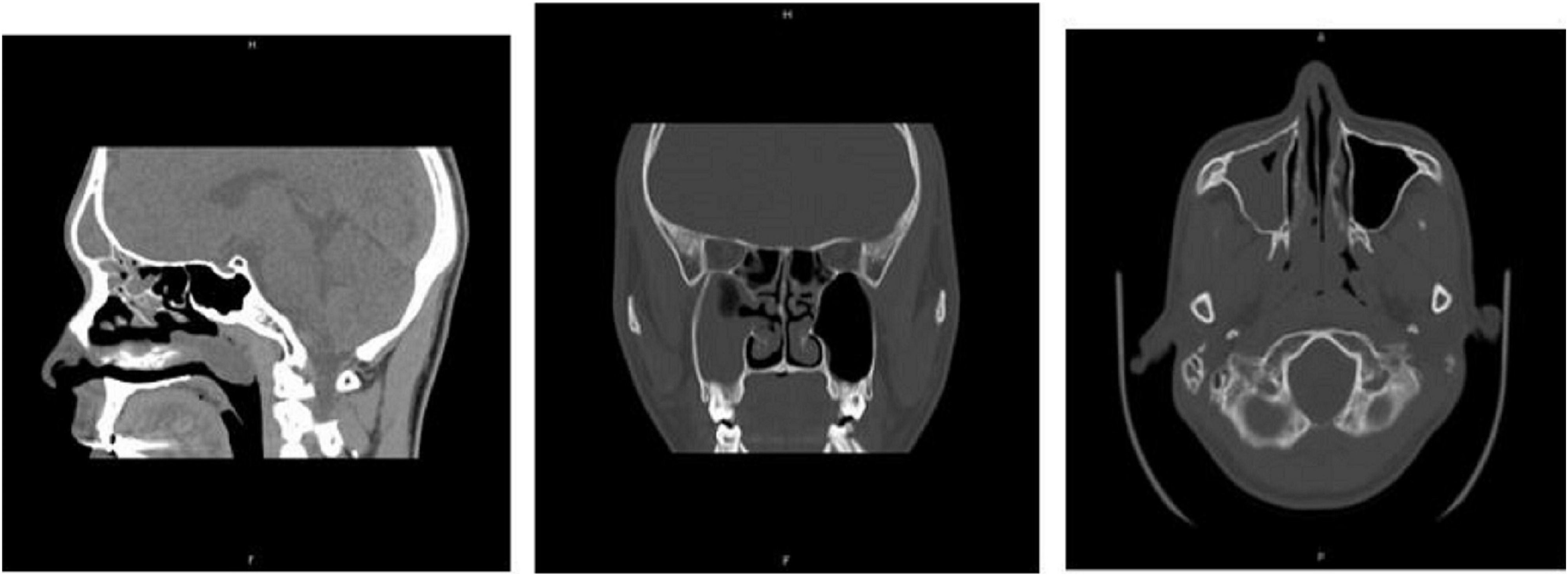
We performed bilateral endoscopic sinus surgery. No atypia or malignancy was noted in a frozen section. Histopathology of the bilateral sinonasal mucosa revealed features of chronic rhinosinusitis with inflammatory polyps. In addition, one of the polypoid lesions was composed of proliferative medium-sized glands and lined by a pseudostratified ciliated columnar epithelium with scattered mucin-secreting cells. Some of the glands were surrounded by a thick, eosinophilic basement membrane (Figure 2). Immunohistochemical examination revealed the luminal cells to be positive for CK7 and negative for CDX-2 and CK20; the basal cells were positive for p63 (Figure 3). On the basis of these features, REAH was diagnosed. (A): A low-powered photomicrograph (20x) revealed glandular proliferation arising from surface epithelium. Mixed inflammatory cell infiltrates was also noted in the stroma, including lymphoplasmacytes, neutrophils, and eosinophils. (B): A high-powered photomicrograph (200x) showed the glands lined by pseudostratified ciliated columnar epithelium admixed with some mucin-secreting cells. A characteristic feature was the presence of stromal hyalinization, and periglandular thick, eosinophilic basement membrane. Post-OP CT scan showed no evidence of residual and recurrence.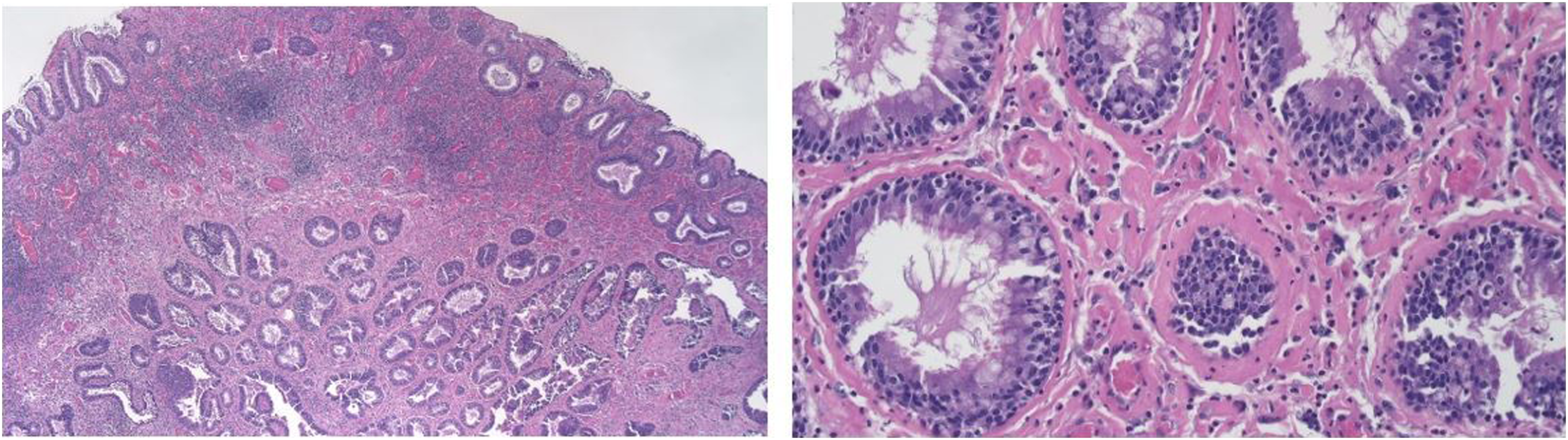
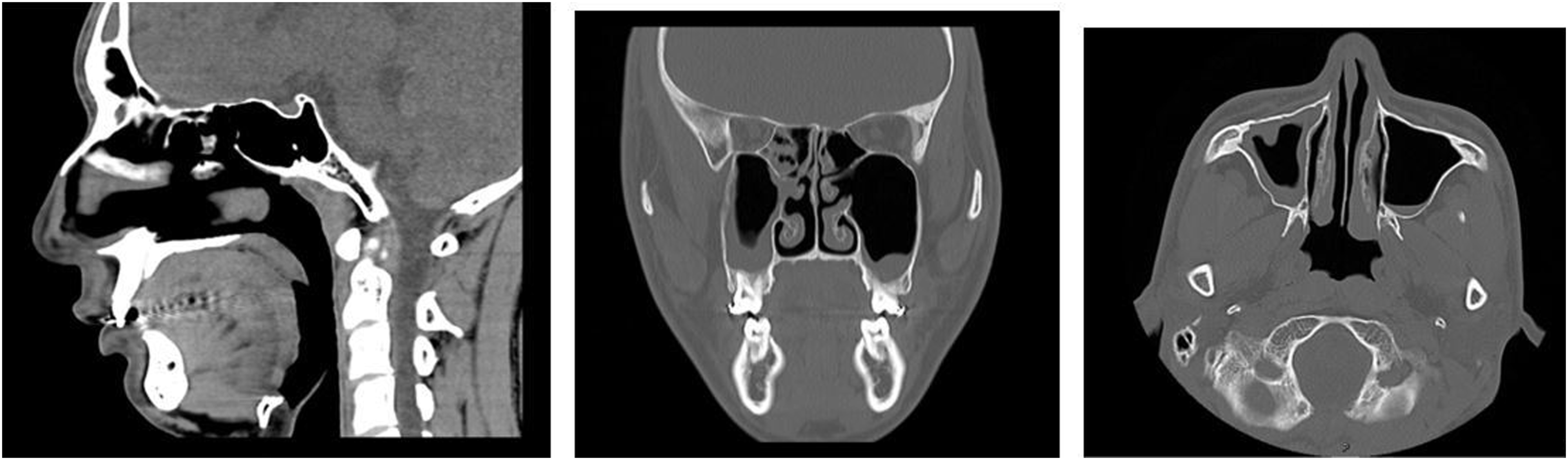
No evidence of recurrence was observed by 6 months after surgery (Figure 4). His hyposmia, nasal obstruction, and purulent nasal discharge were alleviated considerably after the operation. Immunohistochemically, the luminal cells were positive for CK7, while negative for CDX-2 and CK20. P63 expression is identified in the basal cells.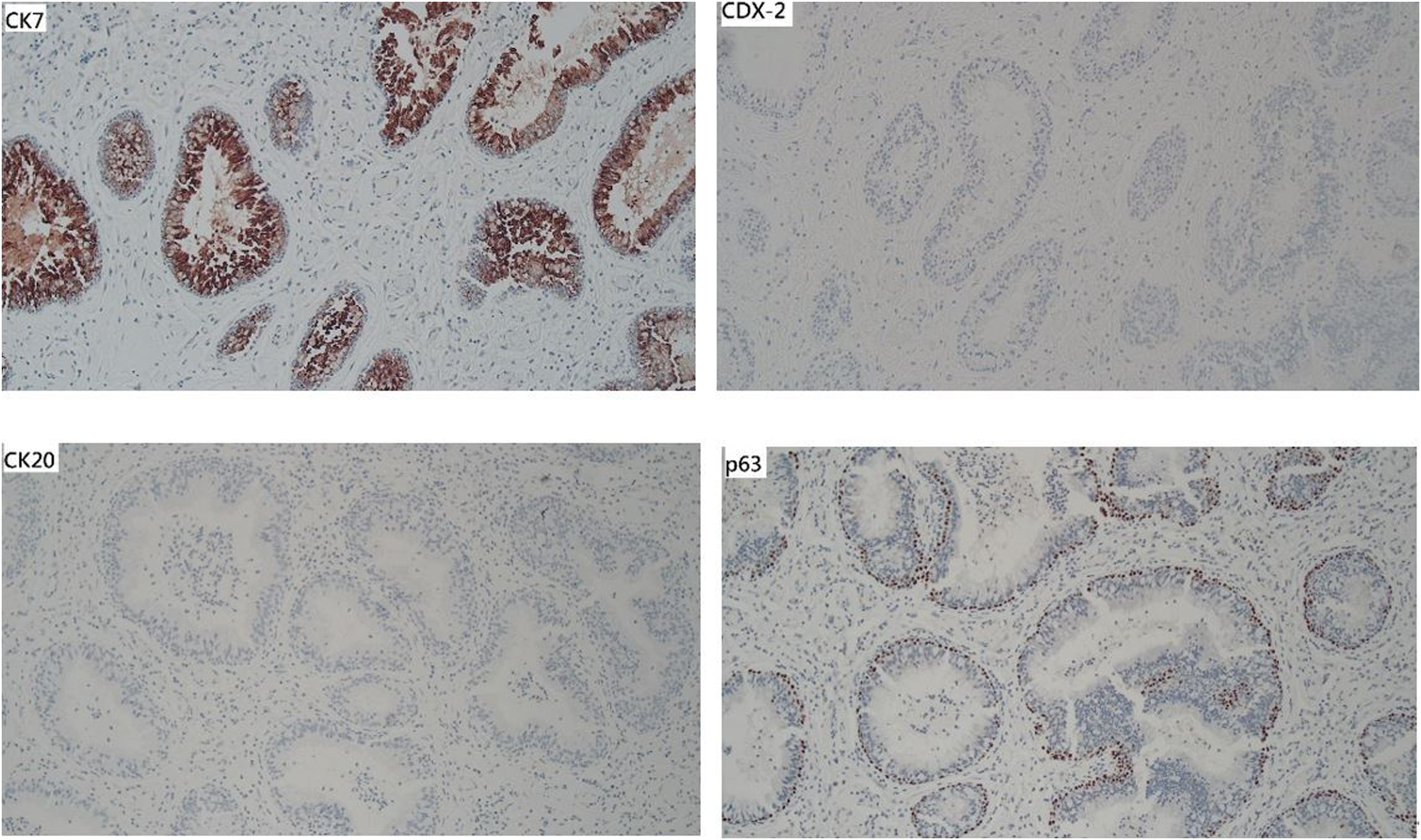
Discussion
Clinically distinguishing REAH from sinonasal malignancy or an inverted papilloma (IP) is challenging. Therefore, biopsy is required to rule out malignancy before nasal REAH diagnosis. Histopathological examination can be used to distinguish REAH from other tumors. REAH is often positive for CK7 but negative for CK20 and CDX-2. However, using immunochemistry to distinguish REAH from sinonasal nonintestinal adenocarcinoma is unreliable because such adenocarcinomas are often also positive for CK7 but negative for CK20 and CDX-2. 5 Sinonasal intestinal-type adenocarcinomas exhibit variable CK7 positivity but consistent positivity for CK20 and CDX-2. 5 However, the lesion in our case was not subjected to immunohistochemical confirmation because the characteristic features of REAH were obtained through histopathology.
Because of the patient’s age and lesion site, we did not initially consider REAH. The differential diagnoses included adenoid hypertrophy, glandular hamartoma, antrochoanal polyps, IP, sinonasal adenocarcinoma, olfactory neuroblastoma, juvenile nasopharyngeal angiofibroma (JNA), and nasopharyngeal carcinoma (NPC).
Histologically, nasal polyps do not exhibit glandular proliferation or surface epithelial invagination but typically possess a thickened epithelial basement membrane; however, this is absent in REAH. REAH usually affects the posterior nasal septum, whereas inflammatory polyps rarely exhibit septal involvement. 4 REAH presents as increased soft-tissue density in the sinuses, a presentation similar to that of nasal polyps. Olfactory cleft enlargement (>10 mm) is the most revealing finding of paranasal sinuses CT that can be used to identify REAH and differentiate it from nasal polyps. 12 The olfactory cleft spans the turbinal ethmoid walls. 3 Olfactory cleft enlargement results from compression of the middle and superior turbinate toward the exterior; however, in nasal polyps, the middle turbinate can be externally compressed against the septum. 11
REAH can be distinguished from IPs and low-grade sinonasal adenocarcinoma histologically. The main characteristic of REAH is the proliferation of round to ovular glands separated by stroma and glands distended with mucus. The basement membrane is thickened with edematous stroma without any metaplastic or atypical changes. By contrast, IPs invaginate hyperplastic squamous or respiratory epithelia with exhibit neutrophilic infiltration, and the basement membranes of IPs are thin and intact. Moreover, seromucinous glands are sparse to absent in IPs.4,12 IPs may be characterized by bone erosion, extend along mucosal surfaces, and invade adjacent structures. 8 The majority of REAHs occur in the posterior nasal septum, whereas IPs commonly originate from the lateral wall of the nasal cavity. 12
Sinonasal adenocarcinomas originate from the glandular epithelium and are characterized by a cribriform pattern of glandular growth, which can be defined as back-to-back fused tumor glands with spaces lacking intervening connective tissue. 10 Intervening stroma between ciliated glands is the most reliable feature for distinguishing REAH from low-grade sinonasal adenocarcinoma, which is characterized by malignant features and dysplastic changes. 12 The absence of a cribriform pattern and nuclear stratification and an increased mitotic rate are the other criteria used to differentiate REAHs from low-grade sinonasal adenocarcinomas. 10 A misdiagnosis of REAH as low-grade sinonasal adenocarcinoma or IP may lead to an unnecessarily extensive surgical procedure. 9
Because of our patient’s age and lesion site, we must consider several differential diagnoses. JNA occurs in adolescent boys and is a highly vascular benign neoplasm that exhibits locally aggressive and invasive behavior. JNA has both fibroblastic and vascular components and appears on CT images as an enhanced soft-tissue mass with well-marginated lobulated contours centered on the sphenopalatine foramen. Other CT findings include bone erosion and bowing of the adjacent paranasal sinus walls, orbits, and skull base. 16
Nasopharyngeal carcinoma (NPC) should also be considered. NPC accounts for less than 1% of childhood malignant tumors; the most common NPC in children is undifferentiated carcinoma, which mainly affects children aged between 10 and 19 years. On CT images, NPC appears as an asymmetric mass centered at the fossa of Rosenmüller, with aggressive features such as bone erosion, intracranial extension, and metastatic cervical lymphadenopathy. The CT characteristics of NPC are heterogeneously enhanced soft-tissue mass and associated bone erosion or sclerosis at the skull base. 16 Epstein-Barr virus(EBV) plays an essential role in transforming nasopharyngeal epithelial cells into invasive cancer. However, one study indicated no statistical difference in the EBV-encoded RNA(EBER) expression of REAH and normal mucosal tissue. 17
Conclusion
Given our patient’s age and lesion site, we initially considered diagnoses other than REAH. The differential diagnoses included adenoid hypertrophy, glandular hamartoma, antrochoanal polyps, IP, sinonasal adenocarcinoma, JNS, and NPC. Therefore, accurate diagnosis based on pathology is essential for determining the optimal treatment, which, for REAH, is complete surgical excision.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Availability of data and material data
Sharing does not apply to this article because no data sets were generated or analyzed during the study.
Ethical approval
The study was approved by the Ethics Committee of Mackay Memorial Hospital, Taipei, Taiwan (21MMHIS368e).
