Abstract
Background
Exhalation delivery system with fluticasone (EDS-FLU) delivers medication high and deep in the nasal passages and has been shown to reduce nasal polyp (NP) grade, an objective measure of efficacy, and to yield clinically meaningful improvements on subjective measures of symptoms in patients with chronic rhinosinusitis with nasal polyps (CRSwNP).
Objectives
To better characterize EDS-FLU treatment, we analyzed responder rates for four outcome measures used in the EDS-FLU pivotal trials, in the overall study population as well as in subgroups of patients with or without prior sinus surgery or prior use of a standard intranasal corticosteroid spray (INS).
Methods
Data were pooled from two randomized, 24-week (16-week, double-blind + 8-week, open-label), placebo-controlled studies (NAVIGATE I and II). Results for patients receiving EDS-FLU (186 µg [n = 161] or 372 µg [n = 160]) or EDS-placebo (n = 161) twice daily during the double-blind phase are described. Responder criteria included NP grade reduction (≥1-point), 22-item Sino-Nasal Outcome Test (SNOT-22) reduction (>12-points), Patient Global Impression of Change (PGIC) (much/very much improved), and congestion score improvement (>0.5-points).
Results
More patients in the EDS-FLU group responded to each of the four responder criteria compared with EDS-placebo. More patients receiving EDS-FLU responded to ≥ 1 criterion compared with EDS-placebo at week 4 (82.7% and 60.4%, respectively) and week 16 (95.7% and 80.3%, respectively). Patients responded similarly irrespective of prior sinus surgery or prior INS use. Patient-reported outcome measures showed earlier responses than NP scores.
Conclusions
Meaningful improvements were seen across multiple response criteria with EDS-FLU, suggesting that the broad treatment effect of EDS-FLU includes objective reduction in polyp grade and improvements in several patient-reported outcomes.
Trial Registration
ClinicalTrials.gov (NAVIGATE I: NCT01622569 and NAVIGATE II: NCT01624662)
Keywords
Introduction
Chronic rhinosinusitis (CRS) with or without nasal polyps (CRSwNP and CRSsNP) is an inflammatory syndrome of the sinonasal cavity, with the defining symptoms of persistent nasal congestion/obstruction, rhinorrhea/post-nasal drip, facial pain/pressure, and/or hyposmia/anosmia. 1 In patients with CRSwNP, bilateral polyps are present in the ostiomeatal complex. 1 Chronic rhinosinusitis affects an estimated 2.1% to 13.8% of individuals in the United States,2,3 and 1.7% to 2.7% of individuals in the United States have CRSwNP. 3
Chronic rhinosinusitis is associated with substantial morbidity.4-6 Most patients report severe symptoms that affect daily functioning, including nasal congestion/obstruction, nasal drainage, facial pain/pressure, and hyposmia. 5 The impact of CRS can also extend beyond the sinonasal region to profoundly affect health-related quality of life (QoL) and functional well-being.5-7 Common extrasinus manifestations of CRS include acute infections, bodily pain, depression, fatigue, impaired workplace activity, and sleep disorders.5,7,8
Until recently, CRSwNP treatment options have been limited. Topical, nasally delivered corticosteroids, administered via sprays or nebulizer/atomization, are currently considered first-line medical therapy for the initial management of CRSwNP and are also used for post-surgical management.1,3,7 However, many patients report frustration with nasally delivered corticosteroids due to inadequate symptom relief. 5 This may be because the anatomical configuration of the nasal cavity prevents conventional delivery approaches from consistently depositing corticosteroids in the appropriate sinus regions (the middle meatus and ostiomeatal complex). 5 A short course of systemic corticosteroids is an option that achieves greater symptom control than standard corticosteroid nasal sprays.1,3,9 However, this treatment is associated with a higher rate of adverse events than nasally delivered corticosteroids (e.g., reduced serum cortisol and/or elevated blood glucose levels and gastralgia). 9
Since 2017, the treatment landscape has expanded, with several new medical options approved by the US Food and Drug Administration (FDA) to address the persistent symptoms and uncontrolled inflammation associated with nasal polyps (NP). These include the exhalation delivery system with fluticasone (EDS-FLU) for the treatment of NP 10 ; a mometasone-eluting sinus implant (SINUVA® [mometasone furoate]) for patients with NP who had previous ethmoid sinus surgery 11 ; and injectable monoclonal antibody treatments (dupilumab, omalizumab, and mepolizumab) as add-on maintenance treatment in patients with inadequately controlled CRSwNP.12-14 Exhalation delivery system with fluticasone, a “nonstandard” nasal product, uses an exhalation delivery mechanism to deposit medication high and deep in key regions affected by chronic inflammation due to CRS.15,16 This system is unique in that it utilizes a flexible mouthpiece and a sealing nosepiece to administer medication simultaneously with exhalation. Exhalation via the EDS generates positive pressure, effectively sealing the sinonasal compartments from the oropharynx, and enhances the delivery of corticosteroid to areas high and deep in the sinonasal cavities vs standard corticosteroid nasal sprays.3,15,16
Clinical trials evaluating EDS-FLU included approximately 1500 patients with CRSwNP and CRSsNP and showed broad improvement in all four defining symptoms of CRS, as well as other primary and secondary endpoints.16-19 These included two single-arm, prospective cohort studies, one of 3 months’ duration (EXHANCE-3 [N = 705]) and the other of 12 months’ duration (EXHANCE-12 [N = 223]) in patients with CRSwNP and CRSsNP,17,18 as well as two pivotal, randomized, controlled, double-blind, 24-week trials in patients with CRSwNP (NAVIGATE I [N = 323] and NAVIGATE II [N = 323]).16,19
In patients with CRS, disease burden and individual sinonasal symptoms are typically described using symptom scales, endoscopic grading systems, and/or sinus-specific QoL instruments.7,20 To better characterize responses to EDS-FLU in patients with CRSwNP, this post hoc analysis of the NAVIGATE I and II clinical trials measured responder rates for four different outcome measures at multiple time points over 16 weeks of treatment. Responders were defined as patients who met predefined criteria representing clinically meaningful change. Furthermore, we evaluated the proportion of patients meeting multiple responder criteria, and we compared responder rates for subgroups of patients with or without prior endoscopic sinus surgery (ESS) or prior intranasal steroid intranasal corticosteroid spray (INS) use.
Patients and Methods
Study Design and Patients
NAVIGATE I and II were similarly designed, 24-week, prospective, randomized, parallel-group, multicenter, EDS-placebo-controlled trials that have been previously published.16,19 Enrolled patients had at least moderate nasal congestion/obstruction over the previous 7 days and bilateral NP with a total score of ≥ 3 on a 6-point modified Lildholdt scale. Patients with sinonasal surgery in the past 6 months or more than 5 lifetime sinonasal surgeries were excluded. NAVIGATE I and II were conducted in accordance with the Declaration of Helsinki, all relevant federal regulations, and Good Clinical Practice guidelines. Independent ethics committees/institutional review boards reviewed and approved the protocols and informed consent. All patients provided written informed consent prior to participation.
Data for this post hoc analysis (N = 482) were obtained from pooled data for patients randomized to the 16-week, double-blind phase of NAVIGATE I and II and included only the EDS-FLU doses subsequently approved by the FDA (186 µg [n = 160] and 372 µg [n = 161], both administered twice daily [BID]), as well as EDS-placebo, BID (n = 161).16,19,21
Outcome Measures
The post hoc analysis categorized patients as responders or non-responders to EDS-FLU treatment based on scores for the following measures: NP grade, the 22-item Sino-Nasal Outcome Test (SNOT-22), Patient Global Impression of Change (PGIC), and congestion score. All measures were assessed at weeks 4, 8, and 16, except that PGIC was not assessed at week 8.
Nasal polyps were objectively assessed at each patient visit via nasal endoscopy using a modified Lildholdt scale. 22
Sino-Nasal Outcome Test-22 is a validated, patient-reported outcome measure commonly used in clinical and research settings to assess CRS health-related QoL. The total score can range from 0 to 110, with higher scores signifying worse QoL, and a minimum clinically important difference of 12.23,24 Patient Global Impression of Change is a patient-reported measure of change in a condition following treatment, using a 7-point Likert scale, and has been widely used in clinical trials of CRS.25-27 Finally, the degree of individual nasal congestion/obstruction—also a core outcome in CRS intervention trials 26 —was reported twice daily (morning and evening) by patients, using electronic diaries and a 4-point scale (0, none; 1, mild; 2, moderate; and 3, severe). Seven-day average instantaneous morning diary symptom scores were used in this analysis.
Responder criteria were an NP grade reduction of ≥ 1 point (on a 0-6 scale); a SNOT-22 reduction of > 12 points; a PGIC rating of much or very much improved (weeks 4 and 16 only); and a congestion score improvement of > 0.5 points.
Statistical Analysis
Responder rates were based on the number of non-missing observations at each visit week. Descriptive statistics were used to summarize study population characteristics and response to each criterion. Subgroup analyses were performed among patients with or without a history of ESS, as well as among patients with or without prior INS use (defined as use of a standard corticosteroid nasal spray within 30 days prior to study entry). A subgroup analysis was also performed for patients who failed the NP responder criterion (i.e., no NP grade reduction of ≥ 1).
Results
Baseline Characteristics
Study population baseline characteristics.
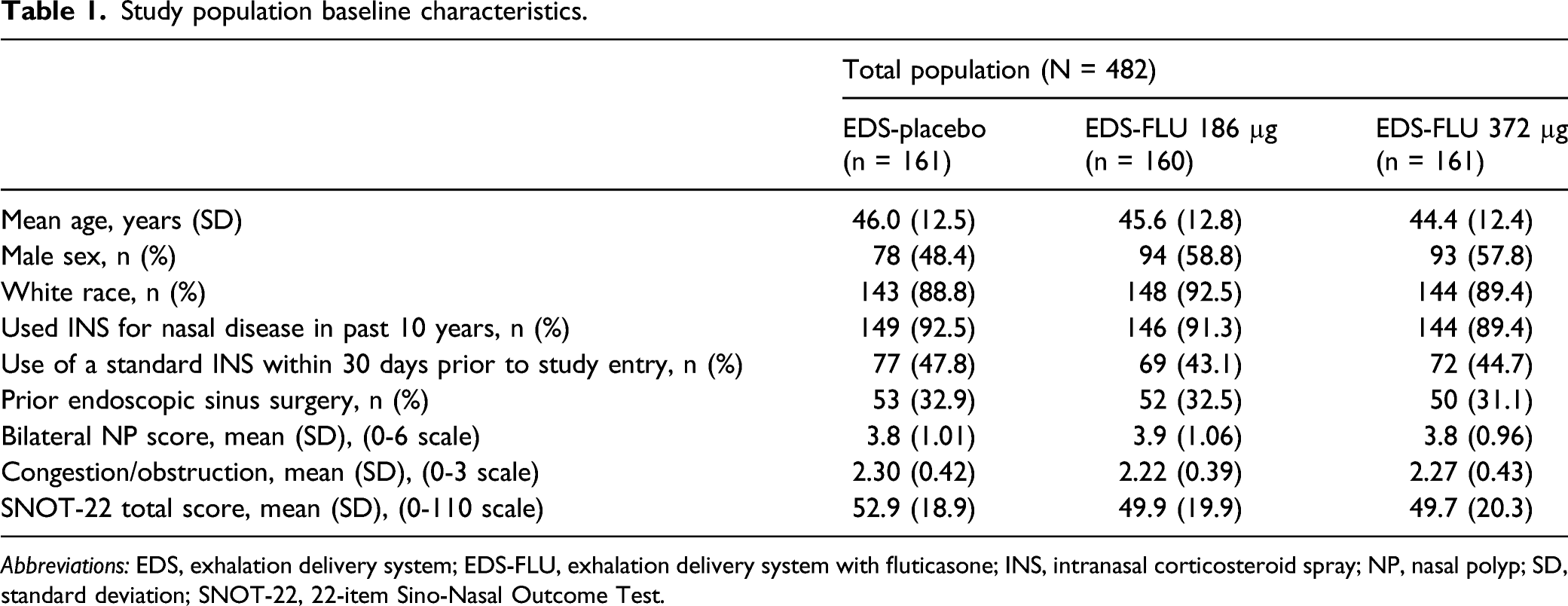
Abbreviations: EDS, exhalation delivery system; EDS-FLU, exhalation delivery system with fluticasone; INS, intranasal corticosteroid spray; NP, nasal polyp; SD, standard deviation; SNOT-22, 22-item Sino-Nasal Outcome Test.
Responder Analyses
Throughout the study, for each outcome measure, a numerically higher percentage of patients met responder criteria in the pooled EDS-FLU group than for EDS-placebo (Figure 1). A separation between groups was evident starting at week 4 and persisted through week 16. Percentage of patients meeting responder criteria by treatment group and visit. Responder criteria include: (A) nasal polyp grade reduction of ≥ 1 point; (B) SNOT-22 reduction of > 12 points; (C) PGIC rating of much or very much improved; and (D) congestion score improvement of > 0.5 points. Note that PGIC was not collected at week 8. Percentages are based on the number of non-missing observations at each visit week; number of patients (n) receiving EDS-placebo at weeks 4, 8, and 16 was 149, 145, and 136, respectively, and number of patients receiving EDS-FLU at weeks 4, 8, and 16 was 308, 301, and 294, respectively. Abbreviations: BID, twice daily; EDS, exhalation delivery system; EDS-FLU, exhalation delivery system with fluticasone; PGIC, Patient Global Impression of Change; SNOT-22, 22-item Sino-Nasal Outcome Test.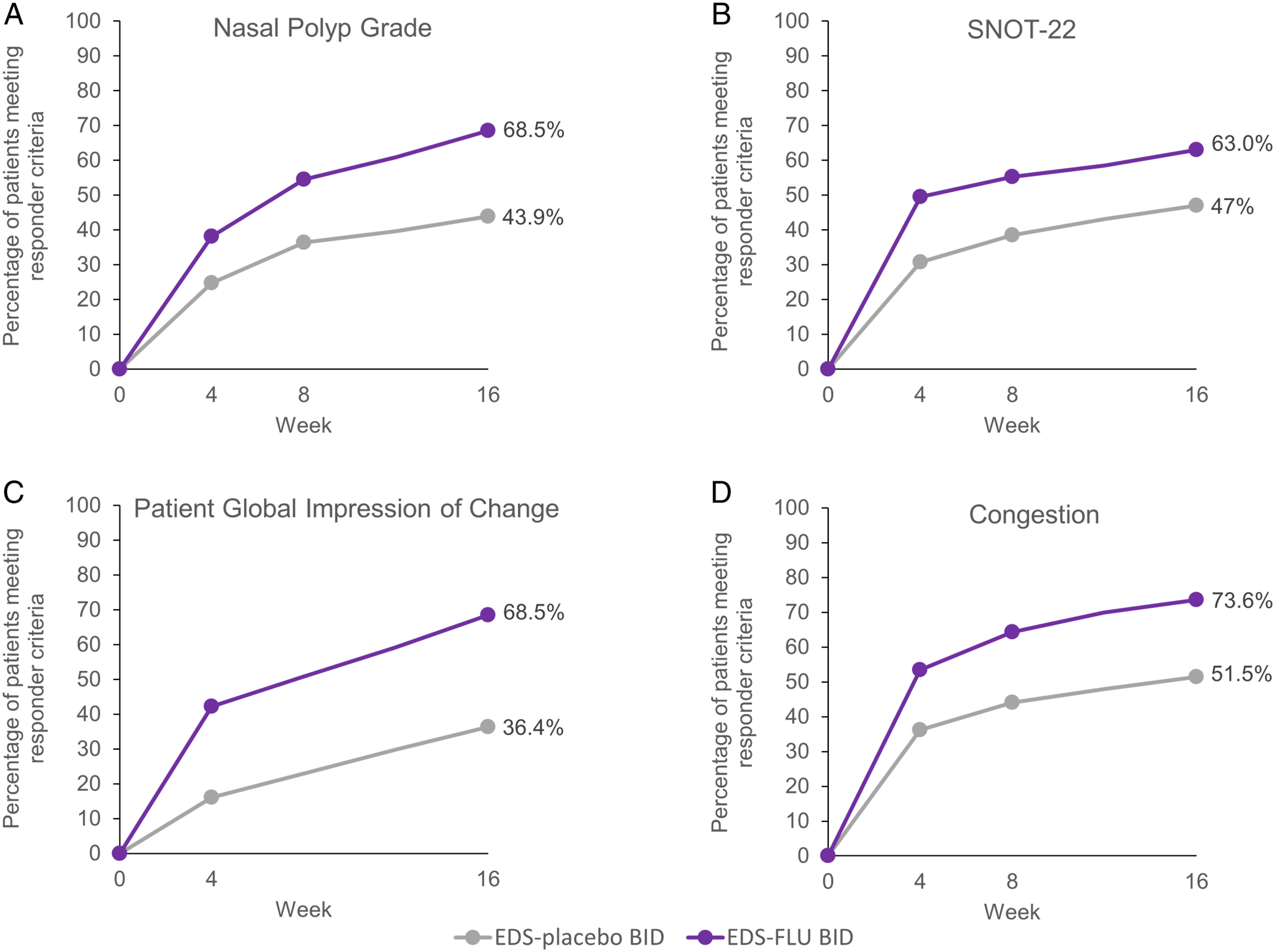
Figure 2 shows the distribution of patients in the pooled EDS-FLU group and the EDS-placebo group who responded to 0, 1, 2, 3, or all 4 responder criteria after 4 and 16 weeks of treatment. At both time points, the proportion of non-responders was greater for patients receiving EDS-placebo than EDS-FLU. At week 16, 34.4% of patients in the pooled EDS-FLU group met all 4 responder criteria compared with 9.1% in the EDS-placebo group. Number of responder criteria met, by treatment group, study weeks 4 and 16. The four responder criteria were: nasal polyp grade reduction of ≥ 1 point, SNOT-22 reduction of > 12 points, PGIC rating of much or very much improved, and congestion score improvement of > 0.5 points. Percentages are based on the number of non-missing observations at each visit week. Abbreviations: EDS, exhalation delivery system; EDS-FLU, exhalation delivery system with fluticasone; PGIC, Patient Global Impression of Change; SNOT-22, 22-item Sino-Nasal Outcome Test.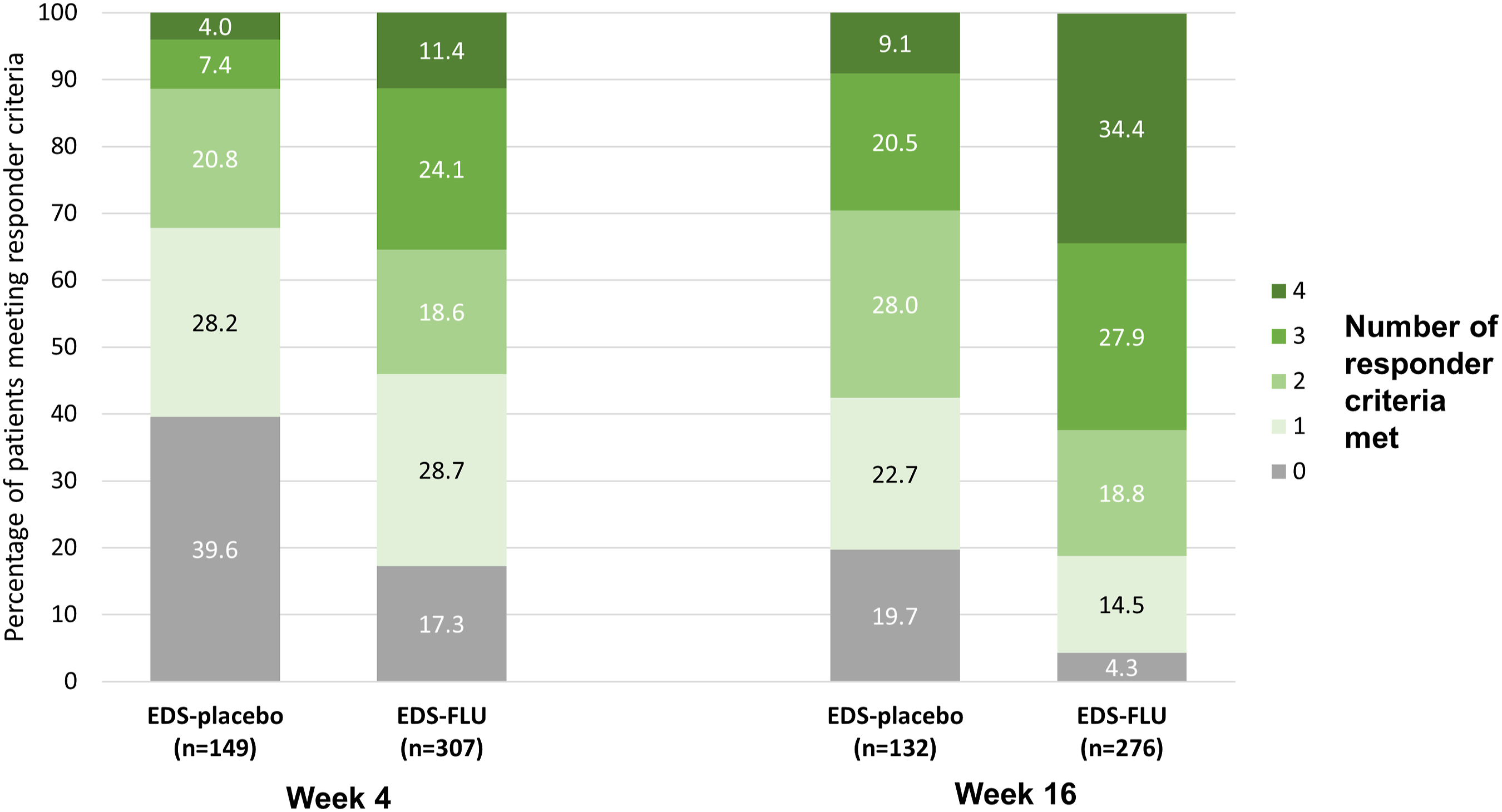
Number of responder criteria met, by treatment group and visit; full analysis set.
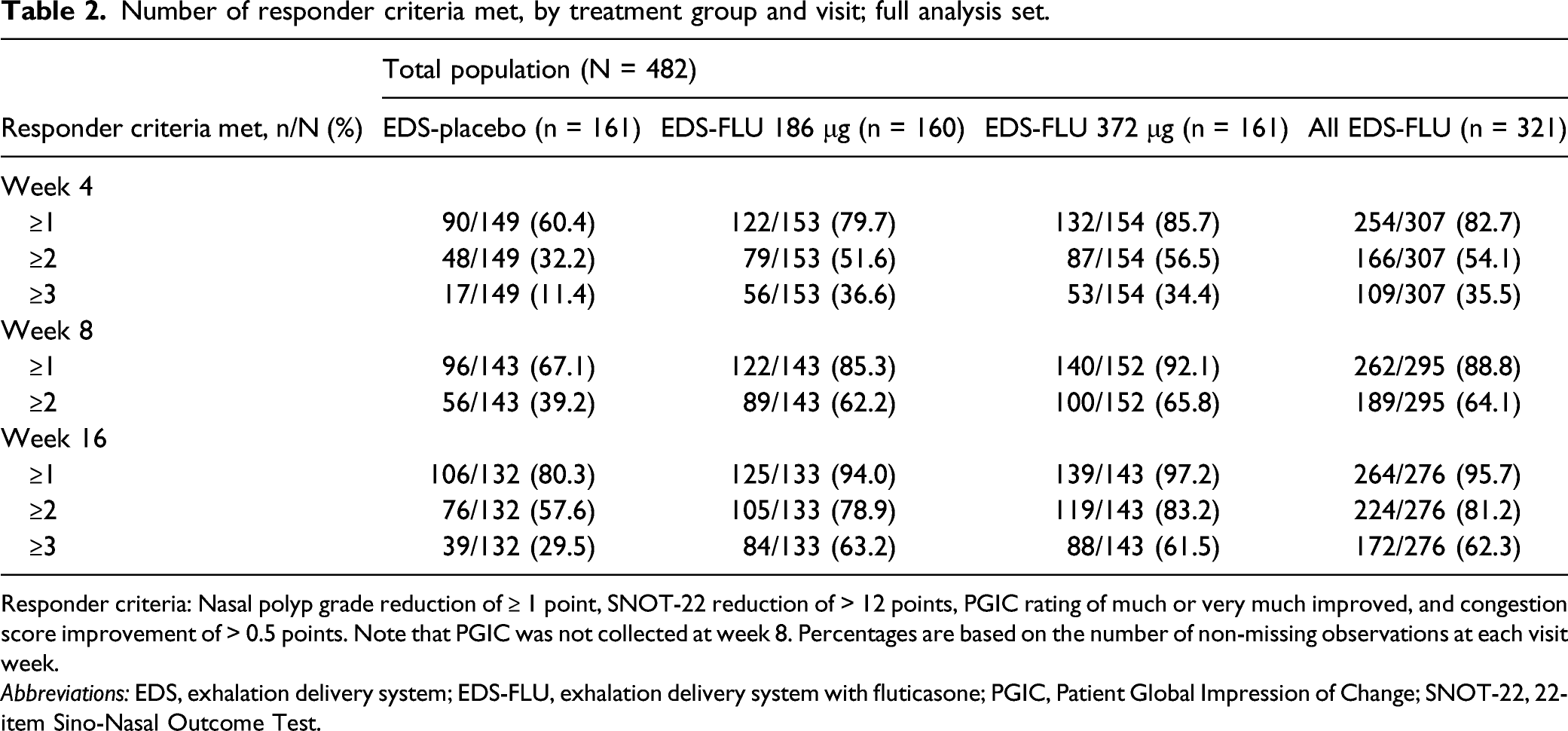
Responder criteria: Nasal polyp grade reduction of ≥ 1 point, SNOT-22 reduction of > 12 points, PGIC rating of much or very much improved, and congestion score improvement of > 0.5 points. Note that PGIC was not collected at week 8. Percentages are based on the number of non-missing observations at each visit week.
Abbreviations: EDS, exhalation delivery system; EDS-FLU, exhalation delivery system with fluticasone; PGIC, Patient Global Impression of Change; SNOT-22, 22-item Sino-Nasal Outcome Test.
Subgroup Analyses
Figure 3 shows the responder rates for each outcome measure among subgroups of patients with or without a history of ESS. For all criteria, the responder rates among EDS-FLU patients with or without a history of surgery were comparable. Percentage of patients meeting responder criteria, by treatment group and prior surgery. Responder criteria include: (A) nasal polyp grade reduction of ≥ 1 point; (B) SNOT-22 reduction of > 12 points; (C) PGIC rating of much or very much improved; and (D) congestion score improvement of > 0.5 points. Note that PGIC was not collected at week 8. Percentages are based on the number of non-missing observations at each visit week. The number of patients with a history of surgery receiving EDS-placebo at weeks 4, 8, and 16 was 50, 45, and 43, respectively, and the number of patients receiving EDS-FLU at weeks 4, 8, and 16, was 99, 95, and 88, respectively. The number of patients without a history of surgery receiving EDS-placebo at weeks 4, 8, and 16 was 99, 98, and 89, respectively, and the number of patients receiving EDS-FLU at weeks 4, 8, and 16 was 207, 199, and 187, respectively. AbbreviationsBID, twice daily; EDS, exhalation delivery system; EDS-FLU, exhalation delivery system with fluticasone; PGIC, Patient Global Impression of Change; SNOT-22, 22-item Sino-Nasal Outcome Test.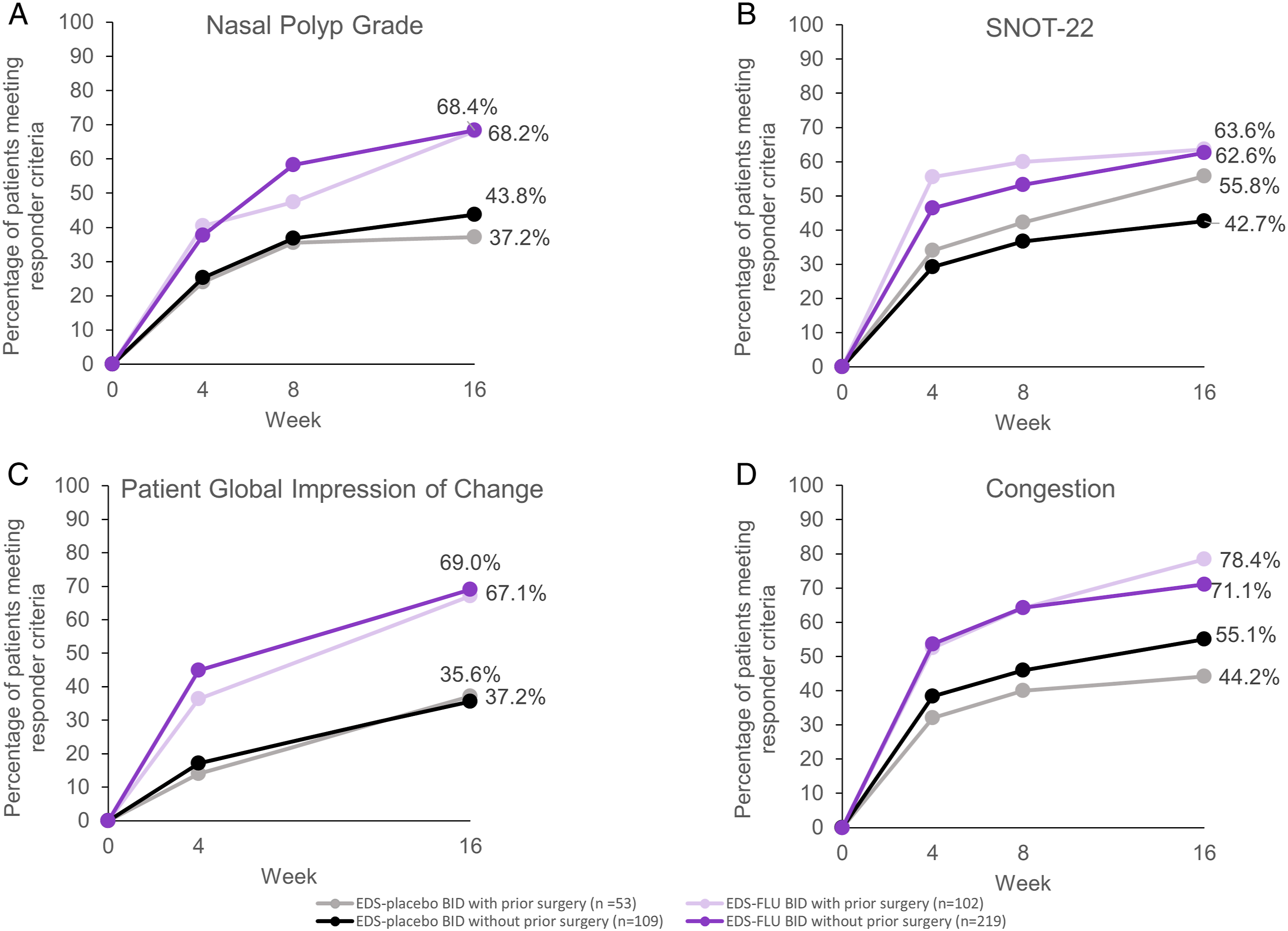
Figure 4 shows the responder rates for each outcome measure among subgroups of patients with or without prior INS use. For all criteria, the responder rates in the pooled EDS-FLU groups were numerically higher than in the EDS-placebo groups, regardless of prior INS use. In addition, for all criteria, the responder rates among EDS-FLU patients with or without prior INS use were comparable. Percentage of EDS-FLU–treated patients meeting responder criteria, by treatment group and prior INS use*. Responder criteria include: (A) nasal polyp grade reduction of ≥ 1 point; (B) SNOT-22 reduction of > 12 points; (C) PGIC rating of much or very much improved; and (D) congestion score improvement of > 0.5 points. Note that PGIC was not collected at week 8. Percentages are based on the number of non-missing observations at each visit week. The number of patients with prior INS use receiving EDS-placebo at weeks 4, 8, and 16 was 68, 65, and 58, respectively, and the number of patients receiving EDS-FLU at weeks 4, 8, and 16 was 133, 131, and 117, respectively. The number of patients without prior INS use receiving EDS-placebo at weeks 4, 8, and 16 was 81, 78, and 74, respectively, and the number of patients receiving EDS-FLU at weeks 4, 8, and 16 was 174, 164, and 159, respectively. *Use of a standard INS within 30 days prior to study entry. Abbreviations: BID, twice daily; EDS, exhalation delivery system; EDS-FLU, exhalation delivery system with fluticasone; INS, intranasal corticosteroid spray; PGIC, Patient Global Impression of Change; SNOT-22, 22-item Sino-Nasal Outcome Test.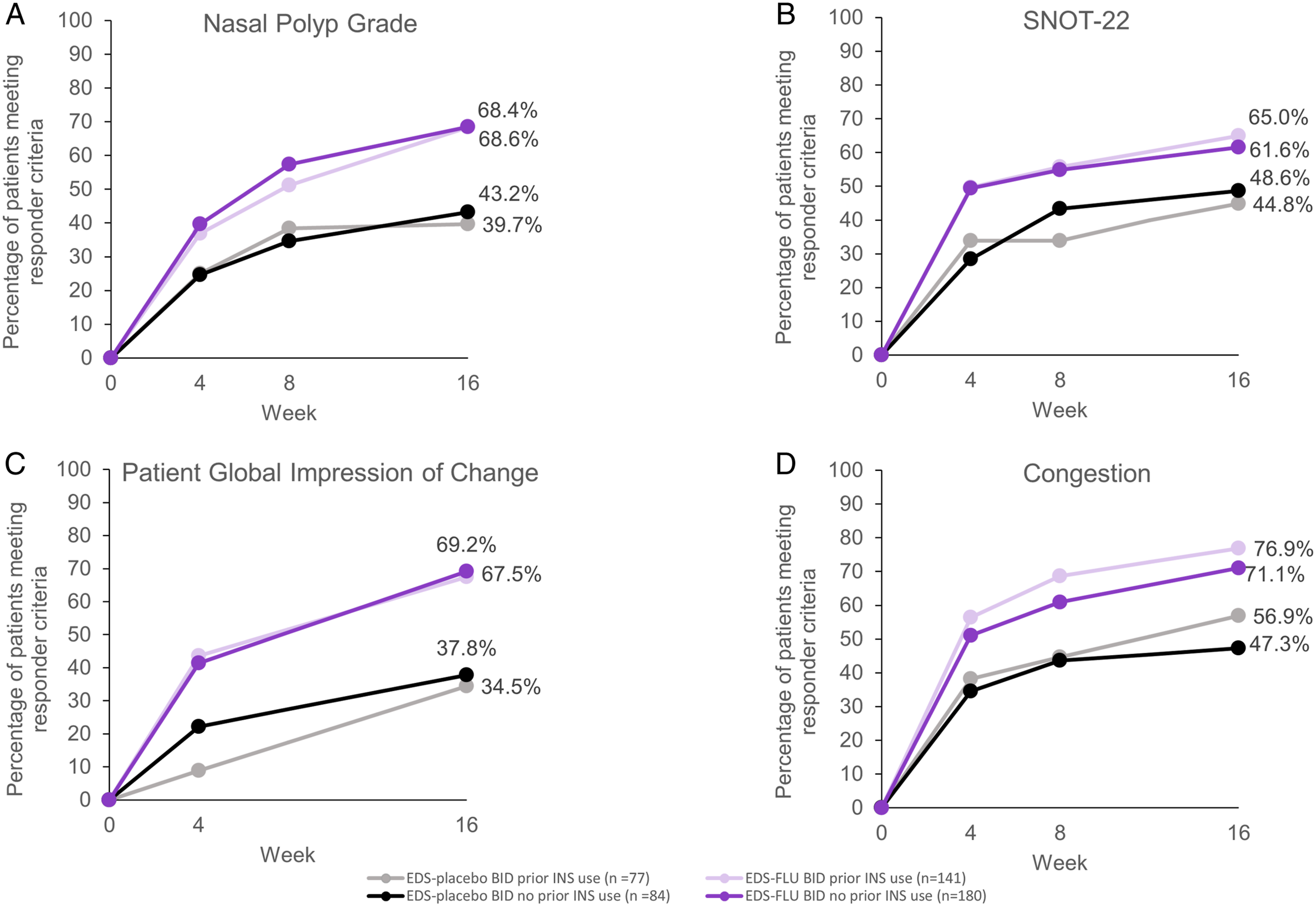
Figure 5 shows the distributions of the number of responder criteria met at week 16 in patients in the pooled EDS-FLU subgroups for patients with or without a history of ESS and with or without prior INS use. Across these four subgroups, patient response to EDS-FLU treatment was generally similar. EDS-FLU patients, number of responder criteria met at week 16, stratified by prior surgery or recent INS use*. The four responder criteria were: nasal polyp grade reduction of ≥1 point, SNOT-22 reduction of > 12 points, PGIC rating of much or very much improved, and congestion score improvement of > 0.5 points. Note that PGIC was not collected at week 8. Percentages are based on the number of non-missing observations. *Use of a standard INS within 30 days prior to study entry. Abbreviations: EDS, exhalation delivery system; EDS-FLU, exhalation delivery system with fluticasone; INS, intranasal corticosteroid spray; PGIC, Patient Global Impression of Change; SNOT-22, 22-item Sino-Nasal Outcome Test.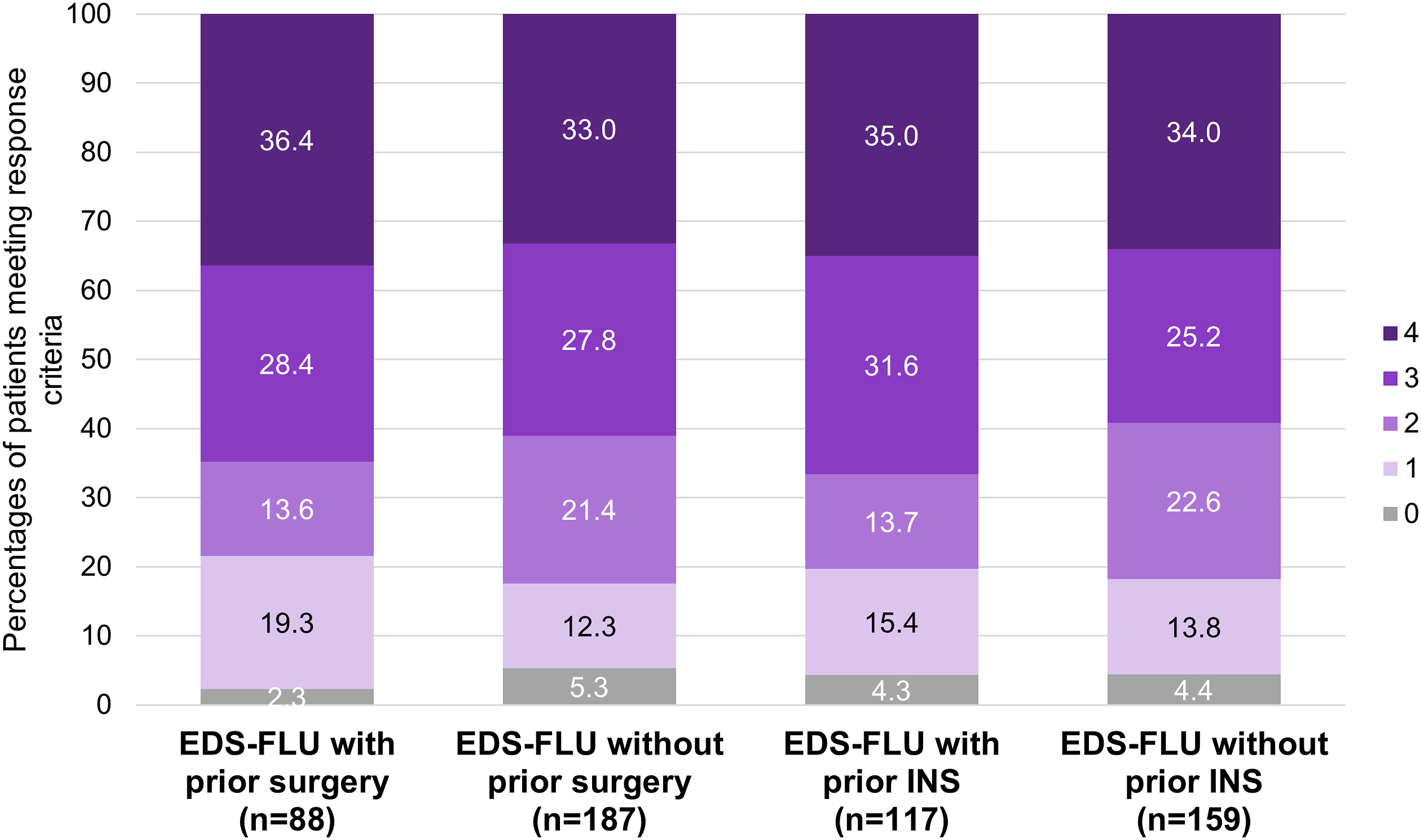
Responder rates to patient-reported outcome measures among patients who did not meet nasal polyp grade reduction responder criteria.
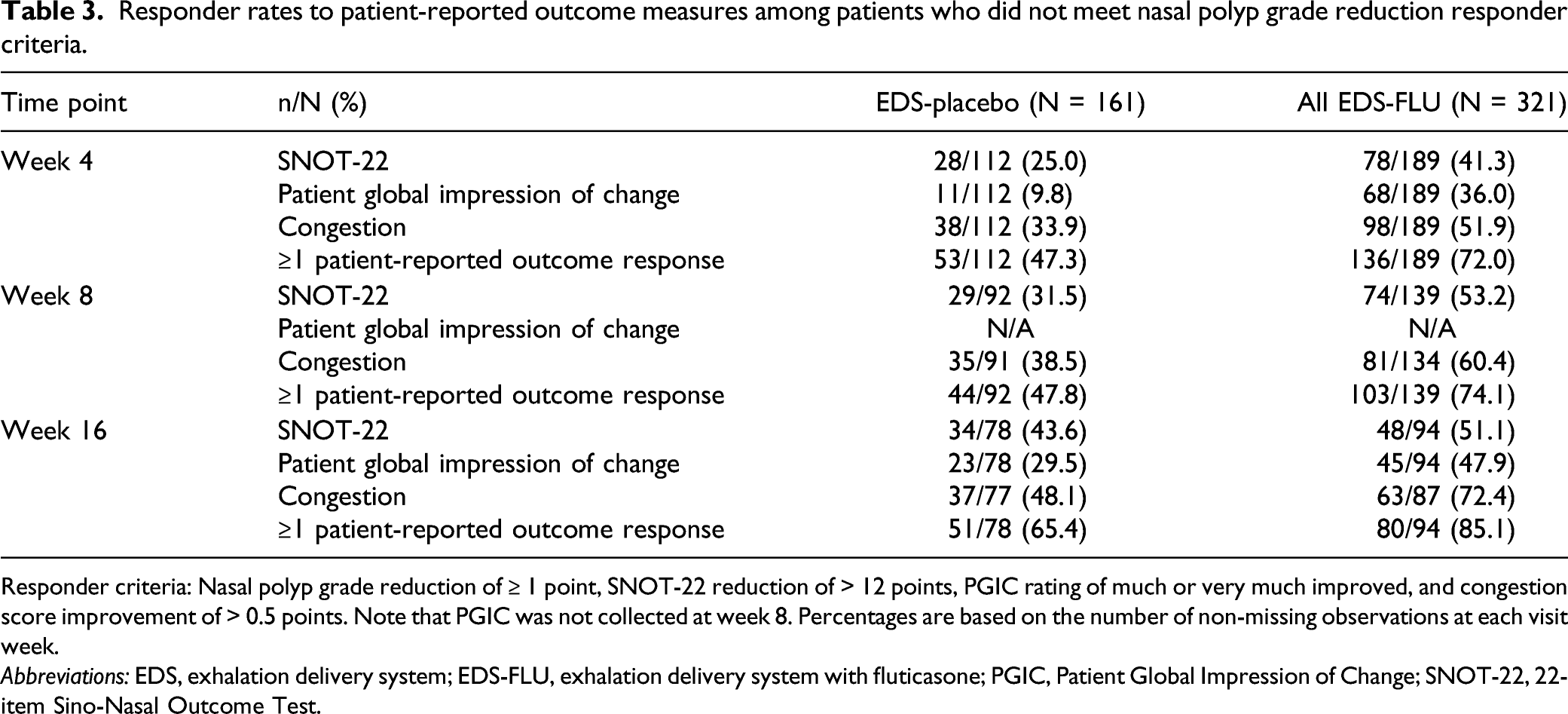
Responder criteria: Nasal polyp grade reduction of ≥ 1 point, SNOT-22 reduction of > 12 points, PGIC rating of much or very much improved, and congestion score improvement of > 0.5 points. Note that PGIC was not collected at week 8. Percentages are based on the number of non-missing observations at each visit week.
Abbreviations: EDS, exhalation delivery system; EDS-FLU, exhalation delivery system with fluticasone; PGIC, Patient Global Impression of Change; SNOT-22, 22-item Sino-Nasal Outcome Test.
Discussion
This post hoc observational analysis of the NAVIGATE I and II trials was conducted to detail the symptomatic and physiologic benefits observed over the course of 4 months (16 weeks) of treatment with EDS-FLU in patients with CRSwNP. Pooled data were obtained at several time points from two similarly designed clinical trials,16,19 and responder rates were calculated for three subjective measures and one objective measure. Analysis was conducted on the total population and across subgroups defined by prior ESS and prior INS use, to assess the robustness and generalizability of previously reported improvements with EDS-FLU compared with EDS-placebo.
At all time points, for all four outcome measures, responder rates were higher for EDS-FLU than EDS-placebo. Patient-reported outcome measures showed rapid and robust improvement, consistent with the known anti-inflammatory effects of fluticasone. 10 Additionally, EDS-FLU responder rates increased from week 4 to week 16, by 76% for NP and 59% for PGIC, indicating that there are late responders to EDS-FLU treatment for whom a 4-week trial may not be adequate to see benefits. Even among patients who did not achieve NP score reductions within 16 weeks, a majority of those treated with EDS-FLU (85.1%) met responder criteria for one or more patient-reported outcomes at study end. Responder rates with EDS-FLU were similar regardless of prior ESS or INS use.
This finding is consistent with a 2020 analysis of pooled EDS-FLU data that showed no difference in treatment outcomes between the subgroup of patients who switched to EDS-FLU from a standard INS and the overall study population. 28 Although previous publications reported mean improvements in these 4 outcome measures with EDS-FLU vs EDS-placebo,16,19 these responder criteria analyses lend further support to the notion that the great majority of patients with CRSwNP, regardless of prior medical or surgical interventions, will experience meaningful benefits with EDS-FLU.
Clinical trials of CRS treatments have traditionally used a variety of objective and subjective endpoints to assess symptomatic, physiologic, and functional burden of disease and to measure patient response to treatment. 26 Reduction in the size of NP is an important objective measure of treatment efficacy.26,29 Subjective measures of symptom improvement are also clinically meaningful because they provide clinicians with a comprehensive profile of treatment response. 29 However, each outcome measure used in CRS research has specific strengths and weaknesses, making it important to carefully select the measures used 26 and to evaluate their findings in context with each other. In this analysis, subjective symptoms among patients treated with EDS-FLU showed notable improvement within 4 weeks, but reductions in NP grade lagged for some individuals. This may reflect limitations of existing NP grading tools, which use strict categorical scales and typically measure polyps across a limited vertical plane. 30 Because of this, not all NP changes or shrinkage will be captured with existing NP scoring systems. These findings also suggest that patients who experience symptomatic improvement with EDS-FLU treatment should stay on medication even if reductions in NP size are not initially observed.
In recent years, several new treatment options have been approved for CRSwNP. In 2021, a US expert multidisciplinary committee published a stepwise treatment algorithm for patients with CRSwNP that considered all FDA-approved clinical and interventional therapies. 3 These guidelines outline available treatments, their benefits and risks, as well their ideal duration of use prior to treatment escalation. After diagnosis, a minimum of 4 weeks of treatment with a conventional INS is recommended. 3 If symptoms persist, the guidelines recommend EDS-FLU as an option, with no treatment duration currently specified. 3 Data from the current study suggest that a 4-week trial period for EDS-FLU may not capture the full response to treatment, particularly with respect to NP reduction; as the EDS device deposits medication high and deep where NPs originate, EDS-FLU provides both rapid anti-inflammatory effects and ongoing accrual of benefits as polyps slowly shrink. The current data suggest that symptomatic improvement occurs as early as 4 weeks, but continued improvement across a broad array of symptoms can be expected up to 4 months after initiation of therapy. This may inform decisions regarding the duration of EDS-FLU use prior to assessing treatment response and considering escalation of therapy.
Limitations
This is a post hoc analysis and its findings are descriptive, with no inferential statistical analysis conducted. Furthermore, these results may not be generalizable to all patients with CRSwNP because patients in this study were symptomatic, with persistent congestion of at least moderate severity, a high rate of previous treatment, and high baseline SNOT-22 scores—all factors that imply substantial disease burden. Therefore, the magnitude of treatment effect might be lower in patients with less baseline symptomatology.
Another study limitation was the sizable placebo response, which has several potential explanations. In clinical trials, EDS-FLU and EDS-placebo patients received a similar volume of solution per nostril; therefore, patients receiving EDS-placebo may have a clinical response similar to nasal irrigation. 31 Placebo response may also be specific to the EDS device: the carbon dioxide present in the exhaled breath and delivered into the nose through the device may influence inflammatory mediator and neuropeptide activity via several mechanisms of action. However, the direct relationship of these factors to long-term symptomatic relief is not known, 10 and a persistently positive treatment response in favor of EDS-FLU was observed for all patients in the current analysis.
Conclusion
In this pooled analysis of clinical trial data, meaningful improvements were observed for a majority of patients treated with EDS-FLU, based on both objective and subjective measures, and regardless of prior ESS or prior INS use. Patient-reported outcome measures showed earlier responses than NP scores, and all outcome measures improved monotonically over 16 weeks of treatment.
Footnotes
Acknowledgments
The authors thank the patients who participated in these studies and the principal study investigators. Writing support (funded by OptiNose US, Inc.) was provided by Luminology Scientific Communications (Rachel E. Speer, PhD [medical director] and Jason C. Hall, PhD [medical writer]). Luminology assisted in data analysis, writing, formatting, proofreading, copyediting, fact checking, and publication submission. All authors contributed equally to the manuscript and approved the final version, including declaration of conflicting interests and funding, for submission.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Randall A. Ow is a member of the OptiNose Speaker Bureau, and he reports consulting fees from OptiNose US, Inc., Aerin Medical, Lyra Therapeutics, Genentech, and Sanofi. Harry J. Sacks and John P. McGinnis II are employees and shareholders of OptiNose US, Inc. Mark E. Mehle is a speaker bureau member for OptiNose US, Inc., Sanofi/Regeneron, and GSK, and he reports consulting fees from OptiNose US, Inc.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: OptiNose US, Inc. (Yardley, PA, USA). The sponsor contributed to the study design, data interpretation, and writing of the report, as well as coordinating the data collection and analysis.
