Abstract
Objectives:
Corticosteroids represent one of the mainstays of medical management of chronic rhinosinusitis (CRS) in both locally acting topical and systemic derivations. The application of topical corticosteroids is limited by a variety of factors including patient compliance, positioning, and nasal anatomy. Systemic corticosteroids confer a risk of medical complication that restricts their ability to be used repeatedly. The objective of this publication is to review the evolution of the in-office intranasal placement of corticosteroids in the management of CRS. The efficacy, outcomes, and safety of a variety of corticosteroid-containing devices meant to be placed in an office setting are reviewed.
Methods:
Pertinent literature was reviewed and summarized beginning with the earliest reports of direct intralesional injection of corticosteroids up through manufactured modern-day bioresorbable implants that contain corticosteroids.
Results:
The utilization of in-office placement of corticosteroid-containing material and implants has rapidly evolved since the concept was introduced, particularly in the last decade. Modern-day corticosteroid-eluting implants are reliably placed in the office, yield results across a range of objective and subjective outcomes, may decrease the need for revision endoscopic sinus surgery, and have a favorable safety profile.
Conclusions:
In-office placement of corticosteroid-containing stents are a viable treatment option for select patients, particularly those wishing to avoid revision surgery, and should be considered an important adjunct for treatment of refractory CRS in an otolaryngologist’s armamentarium.
Topical corticosteroids, which vary in formulation and concentration, have historically played a key role in the management of chronic rhinosinusitis (CRS). 1 Targeted delivery in the office setting, which allows for the precise application and sustained release of the medication to diseased sinonasal mucosa, has been a popular investigational topic dating back to a few decades ago. Starting with direct application, with or without a carrier medium, the concept of in-office corticosteroid placement has since undergone rapid evolution to modern day drug-eluting implants with the advent of new technology and novel devices. This review article will summarize the trends and development of the in-office intranasal placement of corticosteroid material for CRS as well as the safety, efficacy, and outcomes of such practices throughout the last few decades.
Direct intranasal, intralesional corticosteroid injection for nasal polyps (Figure 1) is a technique that can be dated back to the 1950s. Myers et al first published a case series in 1958 demonstrating its efficacy in treating nasal polyposis. 2 This technique was initially controversial as case reports of devastating ocular complications associated with intranasal corticosteroid injections began to surface, although they were more commonly associated with injections to the septum or inferior turbinate, despite its already low complication rate. 3 To illustrate this point, in over 117 000 intranasal corticosteroid injections reported in today’s literature, 19 cases of visual complications were documented (8 transient vs 11 permanent): 12 resulted from injections into the inferior turbinate, 2 from the septum, 3 nasal polyp, and 2 unspecified. 4 Marby published a set of guidelines that aimed to minimize complications associated with this technique. 5 He recommended the use of corticosteroid with small particulate size and the use of a topical vasoconstrictive agent to mitigate the risks of retrograde embolization and vaso-occlusive events. Aforementioned reports of ocular complications appear to be a result of deviation from these recommendations based on case description. 4 For example, one incident involved injection into the septum of a postoperative field (the safety of which has not been studied), 6 a second described 3 separate injections along the inferior turbinate instead of 1 directly into its head, 7 while a third described mixing the injectable corticosteroid with other solutions such as a vasoconstrictive agent. 8 In 2020, Hansen and colleagues conducted a systematic review on the efficacy, methodology, and safety of corticosteroid injections specifically into nasal polyps. 9 Five studies (2 randomized controlled trials (RCTs) and 3 retrospective studies) with a total of 386 patients and 2490 intrapolyp corticosteroid injections were identified. Doses and formulation ranged from 10 to 40 mg of triamcinolone acetonide (TA). Decreases in total polyp score (TPS), total nasal symptom score, and Lund-Mackay score were observed. Only 2 reported cases of transient visual complication were reported of the 2490 injections performed. Excluding case reports, the authors report that these 2 incidences are the only published ocular complications in larger case series specifically associated with intrapolyp injection following Mabry’s case series in 1981.
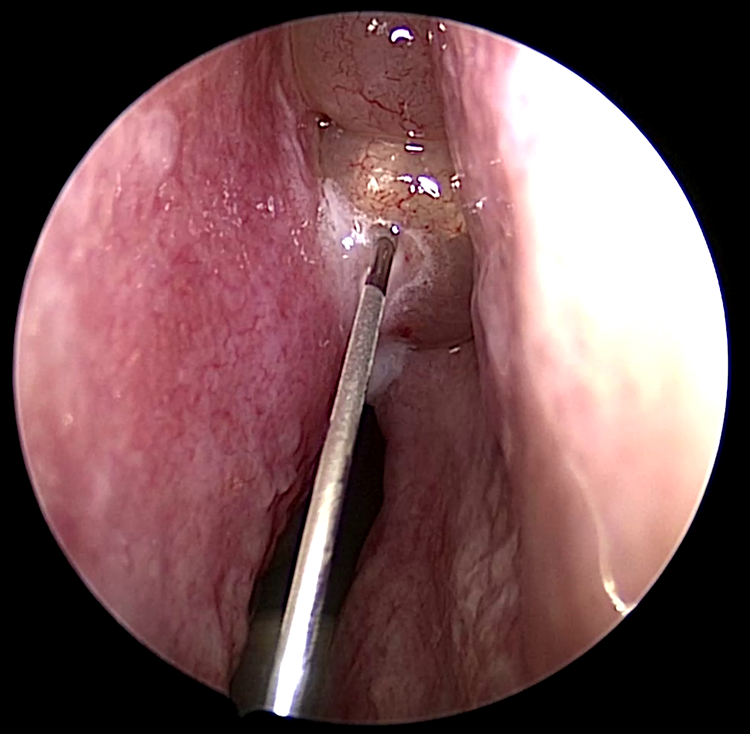
This picture demonstrates the application of intralesional injection of triamcinolone acetate (TA) into a left-sided nasal polyp. Note the blanched appearance of the polyp that has been infiltrated with TA. The process of polyp injection is relatively straightforward and frequently be performed without any anesthesia.
In 1998, Citardi and Kuhn described a case series of endoscopically guided frontal sinus beclomethasone instillation for refractory frontal recess polyposis. 10 To address the high rate of frontal sinus restenosis and polyposis recurrence following surgery, during an office visit, the investigators used a size 8 Frazier tip suction under direct endoscopic guidance to instill 1.0 to 1.25 mL of aqueous beclomethasone into the previously operated frontal sinuses of 16 patients in 31 instances. In order to maximize the time and degree that the medication would have direct contact with the diseased sinus mucosa, the authors asked the patients to stay supine for 20 minutes following the procedure, conjecturing that the recurrent polyposis and edema would serve as barriers to the egress of the medication. In a 4-month follow-up period, polyposis had completely resolved in 9 frontal sinuses, improved in 5, and remained unchanged in 10. No systemic alterations in cortisol levels were noted in the patients.
In 2002, Lavigne and coworkers published a cases series demonstrating a method of high dose topical corticosteroid delivery to the maxillary sinus via a novel intubation device (maxillary antrum sinusotomy device) deployed in the office setting, 11 an idea first conceived by Jasbi and Ritter in the treatment of pediatric sinusitis in 1977. In their double blind, placebo-controlled study, 26 patients with CRS failing medical therapy were recruited. One of the affected maxillary sinuses of every patient was intubated with a maxillary antrum sinusotomy tube, which was described as a flexible tube that allowed access to the maxillary sinus cavity with a distal anchoring mechanism. The curve of this distal end and the flanges conform to the lateral wall of the nose and extend the flexible tube under the inferior turbinate, allowing for delivery of a 3-week course of 256 µg of budesonide or placebo treatment. They noted improvement in the symptom scores in 11 of the 13 patients in the budesonide group. Maxillary sinus tissue biopsies immediately before placement and at time of follow-up show a decrease in CD-3 (P = .02) and eosinophils (P = .002), a decrease in the density of cells expressing interleukin-4 (P = .0001), and a decrease in interleukin-5 messenger RNA (P = .006) after the 3-week treatment period. It is important to note that one treatment arm patient did experience increase in insulin requirement, which is consistent with the heightened bioavailability of budesonide compared to the newer generations of corticosteroids.
In 2008, Kang and colleagues further explored another avenue of in-office high dose topical corticosteroid delivery in cases of recalcitrant CRS following endoscopic sinus surgery (ESS). 12 In a RCT, they compared the effects of topical spray and high-dose topical steroid gauze packing on the prevention of recurrent nasal polyps after ESS. Among the 32 patients with recurrent nasal polyposis following sinus surgery, 18 were placed in the control group receiving intranasal topical TA corticosteroid spray, which was administered with 2 sprays/nostril per day (55 μg/actuation) after revision ESS for 2 months (total dose, 12.3 mg). Fourteen patients were placed in the experimental group wherein a small gauze saturated with aqueous TA (40 mg/vial) mixed with 1 mL of normal saline (1:1) was applied for 10 minutes once a week for 2 months (8 applications; total dose, 320 mg) to the postoperative neocavity. Their follow-up period was 12 months. They noted a significantly lower polyp recurrence rate in the experimental arm (7.1%) compared to the control arm (44%) and did not notice major complications including systemic adrenocorticotropic hormone or cortisol fluctuations or ocular symptoms in the experimental group. They concluded that in-office placement of high dose corticosteroid-soaked material allowed for more precise and targeted medicinal delivery to the inflamed nasal mucosa to a greater degree than traditional metered-spray pump devices, as effective delivery relies on several variables such as head position and size of the sinus cavity. 13,14
Carboxymethylcellulose (CMC) foam, a plant-based polysaccharide matrix with hemostatic properties, has also been studied as an off-label corticosteroid carrier. In 2010, Pletcher and Goldberg conducted a prospective cohort study that included 8 patients with recurrent CRS following ESS (recurrence defined endoscopically and based on symptomatology). 15 They described an in-office placement of CMC foam (SINU-FOAM; Smith & Nephew Surgical Devices) that was hydrated with 8 mL of TA, 40 mg/mL, in the patients’ ethmoid cavities. Mean SNOT-20 score improved at both 1 week and 1 month after treatment (2.44 compared to 1.65, P < .05, and 2.44 compared to 1.36, P < .01, respectively). Modified perioperative sinus endoscopy score also improved at 1 week (11.8 vs 8.2, P < .001) and 1 month (11.8 vs 7.9, P < .001). No complications were reported. Given these findings, they concluded that in-office placement of corticosteroid-infused CMC foam was able to improve subjective and objective outcomes for patients with recurrent CRS following ESS.
Similarly, Chaudry and colleagues conducted a retrospective study in 2014 evaluating the use of TA impregnated CMC foam for acute exacerbations of CRS for patients who had previously undergone ESS. 16 In their study, 116 documented exacerbations in 130 patients were treated with a concoction composed of 80 mg of TA mixed with sterile water in a single application of CMC foam. The mixture was applied to the sinus cavity in the office setting under endoscopic visualization, and patients were asked to refrain from performing nasal irrigation for 48 hours following the procedure. In their mean follow-up period of 49.3 weeks, they reported a statistically significant decrease in SNOT-22 scores (preinstillation, 1.69 ± 0.86, vs postinstillation, 1.23 ± 0.8; P < .01) and a trend of decreasing oral corticosteroid use which was not reported as statistically significant. No major adverse outcomes were reported aside from 1 patient (n = 1, 0.8%) having reported an increase in blood sugar level that self-resolved without alternations to his insulin regimen. As such, they concluded that in-office application of TA/CMC appears to be a well-tolerated method for mitigating oral corticosteroid burden for patients experiencing acute exacerbations of CRS. A few limitations the authors reported include the retrospective nature of the study as well as the lack of pharmacokinetic evaluations of TA in this topical formulation.
More recently, Food and Drug Administration (FDA) approval for corticosteroid eluting implants has been granted. The mometasone furoate (MF)-eluting stent (Propel, Intersect ENT) is composed of a biodegradable polymer that slowly expands to deliver 370 µg of MF over 30 days to an operated ethmoid cavity. 17 A series of prospective blinded studies in 2011 to 2012 showed that intraoperative placement of these stents into operated ethmoid cavities resulted in statistically significant improvements in both subjective and objective outcomes. 18 -20 Although these MF implants were FDA approved for intraoperative placement in the postoperative ethmoid cavity, placement in the office gained popularity. In 2014, Matheny and colleagues 21 prospectively examined the feasibility, safety, and outcomes of in-office placement of MF implants into ethmoidectomy cavities 1 week after bilateral ESS. Placement was 100% successful, and no serious adverse events were reported. Subsequently, in 2015, a lower-profile version of the MF implant (Propel Mini, Intersect ENT) was evaluated in an off-label in-office fashion. In this 2-patient case report, 22 Janisiewicz and Lee placed the lower-profile MF implant into the frontal sinus ostia in patients undergoing in-office balloon catheter dilation of the frontal sinuses. The authors reported no adverse events, complete implant resorption by 7 to 10 weeks after implantation, and preserved ostial patency up to 11 months following implantation (Figure 2).
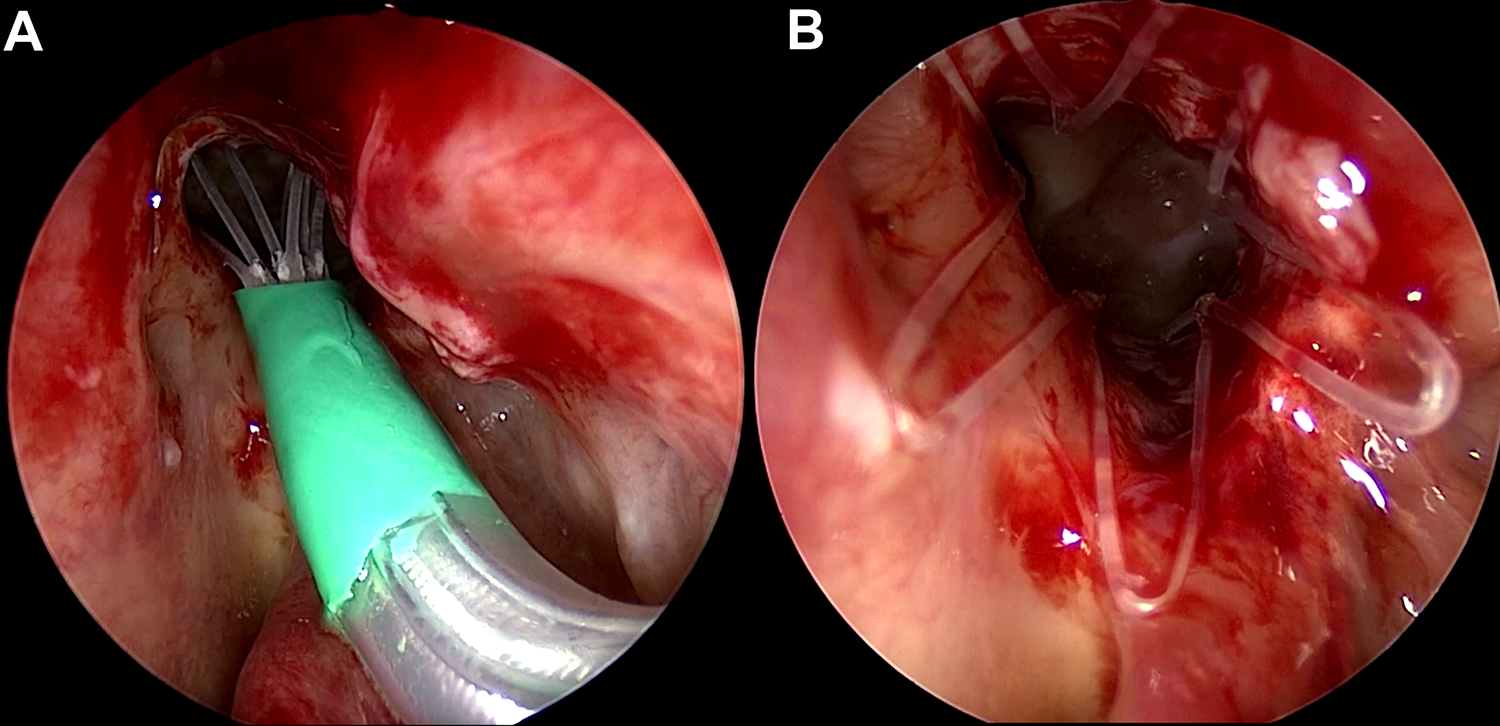
This figure demonstrates the placement of a frontal sinus mometasone furoate (MF) implant in a patient who underwent revision frontal sinusotomy in the office. The patient had a history of frontal sinus inverted papilloma and returned for follow-up with a stenotic frontal ostium, which was dilated with a balloon catheter and further revised with instruments. The MF implant was placed (A), and good mucosal apposition of the flanges (B) can be seen across the denuded surfaces.
A higher dose MF implant (known originally as the S8) was developed and evaluated for an in-office indication treatment of CRS with nasal polyps (CRSwNP). This device contained over 3.5 times as much MF (1350 μg) compared to its predecessors. The S8 conferred greater radial strength and ability to physically dilate the obstructed cavity while maintaining mucosal apposition as the polypoid tissue regressed. Lavigne and colleagues 23 initially carried out a prospective multicenter study to evaluate the efficacy and safety of this larger dose in-office implant in patients with CRSwNP. Twelve patients with CRSwNP with bilateral polyp recurrence following ESS who were candidates for revision ESS due to steroid refractoriness were enrolled. Implants were put into bilateral ethmoid cavities after obtaining local anesthesia and removed after 60 days. Endoscopic and SNOT-22 scores were obtained at regular intervals through 6 months postimplantation, at which point patients were assessed for persistent revision ESS candidacy. The implant resulted in improvement in SNOT-22 scores at 1-month (2.9-0.9, P = .001) and 6-month follow-up (1.03, P = .012). Total polyp score was reduced at 1-month (4.5-2.3, P = .008) and 6-month follow-up (2.3, P = .0008). Of 11 patients, 7 (64%) were considered to be no longer candidates for revision ESS. There were no serious adverse events for the duration of the study.
Additionally, Ow and coworkers 24 evaluated the pharmacodynamic safety profile of the S8 implant. The study design followed the protocol of Lavigne et al 23 ; however, Ow and coworkers captured data for only 30 days postimplantation. At 1-month postimplantation, there were significant improvements in TPS (4.6-2.8, P = .037), SNOT-22 (2.0-0.6, P = .01), and nasal obstruction/congestion (NO/C) score (2.2-1.2, P = .002). There were no serious adverse events, plasma concentrations of mometasone were undetectable or near-undetectable for all participants, and no significant derivations in mean morning cortisol were observed.
Subsequently, Han and coworkers conducted the first single-blinded RCT of the 1350 µg MF implants (RESOLVE) across 18 US clinical centers, 25 which enrolled 100 patients with recurrent refractory CRSwNP following ESS who were candidates for revision surgery. In all, 53 patients underwent bilateral in-office endoscopic placement of 1350 µg MF implants while 47 patients underwent bilateral sham procedures (endoscopy, insertion of delivery system without implant deployment). At standardized follow-up visits over the first 60 days postimplantation (at which point devices were removed), endoscopic scores and patient reported outcomes (PROs) were collected. Patients were followed up to 90 days postimplantation (30 days following implant removal) and reassessed. At the 90-day time point, Han and colleagues found that patients in the treatment arm demonstrated a 2-fold improvement in NO/C scores (−1.33 vs −.67, P = .13), which reached statistical significance in patients with severe polyp burden (defined as grade ≥2 bilaterally) when compared to controls (−1.4 vs −0.52, P = .025). Additionally, treated patients experienced a significant reduction in TPS (−1.0 vs −0.1, P = .0269) and endoscopic ethmoid sinus obstruction (−21.5 vs +1.3, P = .0001). Importantly, approximately 53% of treated patients (compared to 23% of control patients) were no longer considered candidates for revision ESS. In 2016, Forwith and colleagues 26 reported longer-term 6-month patient outcomes in the same group of patients reported by Han et al. 25 Forwith and colleagues found that patients receiving the 1350 µg MF implants demonstrated durable and statistically significant objective and subjective improvements. Additionally, control patients had 3.6 times higher risk of having indications for ESS at 6 months as compared to treated patients.
To further evaluate the efficacy of the 1350 µg MF in-office implants, Kern and colleagues carried out a larger multicenter double-blinded and sham-controlled RCT (RESOLVE II). 27 Three hundred adults with recurrent severe CRSwNP following ESS who were candidates for revision ESS were randomized 2:1 to receive either bilateral 1350 µg MF implants or bilateral sham procedures (as previously described), to which the patients were blinded. Implants were removed at 60 days (Figure 3) to allow for 90-day TPS to be reported by independent scorers blinded to treatment and sham assignment in light of implant removal at 60 days. Total study duration was 90 days, during which patients used MF intranasal corticosteroid sprays once daily and had regularly scheduled endoscopic and PRO measures recorded. Statistically significant reduction in TPS at 90 days as assessed by graders blinded to treatment and sham assignment was seen (−0.56 vs −0.015, P = .0073). Improvement in NO/C score at 30 days was also statistically significant (−0.80 vs −0.56, P = .0074). There was a significant reduction (39% vs 63.3%, P = .0004) in the number of patients who were no longer considered to be candidates for revision ESS when compared to the placebo group.
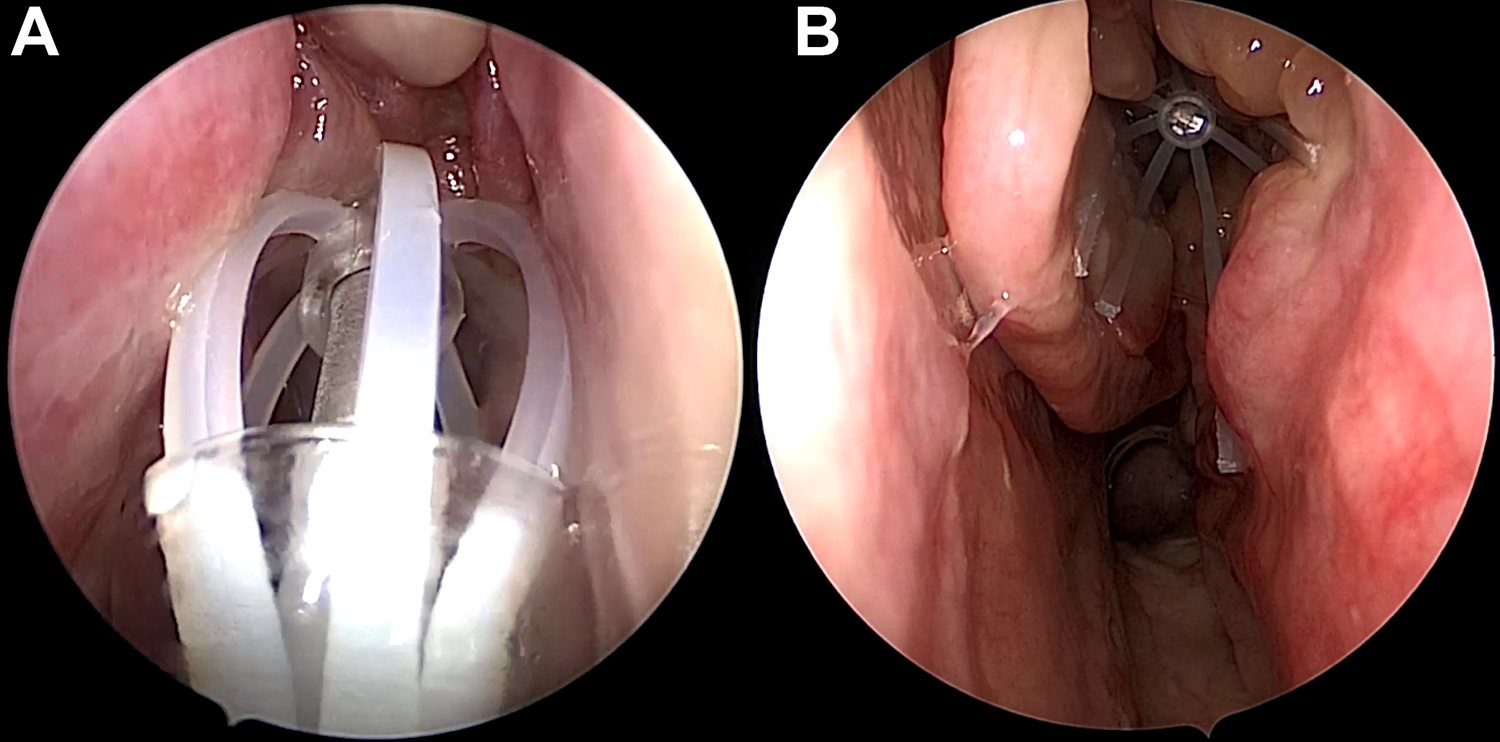
In this figure, the before-and-after effects of a high dose mometasone furoate (MF) implant can be observed. The MF implant is placed into the operated ethmoid cavity with recurrent polyps (A). The effect of the high dose MF implant at 60 days (B) demonstrates significant reduction in polyp burden and a visible ethmoid cavity.
Stolovitzky et al 28 performed a more recent pooled analysis of 90-day subjective and objective outcome measures following in-office MF placement for recurrent CRSwNP, allowing for detailed subgroup analyses. The results of this study demonstrated that the patients who were randomized to the MF sinus implant overall experienced significant improvements in NO/C score, TPS, and ethmoid sinus obstruction severity when compared to controls, and that only 41% of treatment versus 69% of control patients remained candidates for revision ESS. Subgroup analysis demonstrated that patients whose primary surgery was within 24 months and those with TPS >5 had the largest improvements on objective endoscopic outcomes and need for revision ESS. A history of prior or current smoking was associated with a smaller magnitude of both objective and subjective outcomes. Additionally, patients with moderate-to-severe allergic rhinitis (AR) reported greater reductions in subjective and objective outcomes than those with no-to-mild AR. In the 400 included patients, there was only 1 (0.4%) implant-related serious adverse event (epistaxis). This study solidified the utility of mometasone implantation in recurrent polyposis following ESS and suggested that the implants may be particularly efficacious in patients with severe polyposis, severe AR, and ESS within 24 months of implantation.
It is important to note that the preceding publications assessed safety and efficacy of MF-eluting stents specifically in groups of patients who had polyps and a history of prior surgery. Douglas and colleagues 29 recently published the results of a phase 1 open-label single-arm study evaluating the safety, tolerability, and efficacy of an MF-eluting implant (LYR-210, Lyra Therapeutics, Inc) in unoperated patients both with and without polyps who were candidates for ESS. This implant, made of a tubular biodegradable polymeric mesh- (Figure 4), is designed to fit into an unoperated middle meatus and applies even mucosal pressure over 24 weeks to deliver 2500 µg of MF. The authors found that there were no effects on serum cortisol or ocular pressure and that serum concentrations of MF were consistently at or below the lowest detectable limit. At 24 weeks, there was significant improvement in SNOT-22 scores from baseline in both polyp and nonpolyp patients.
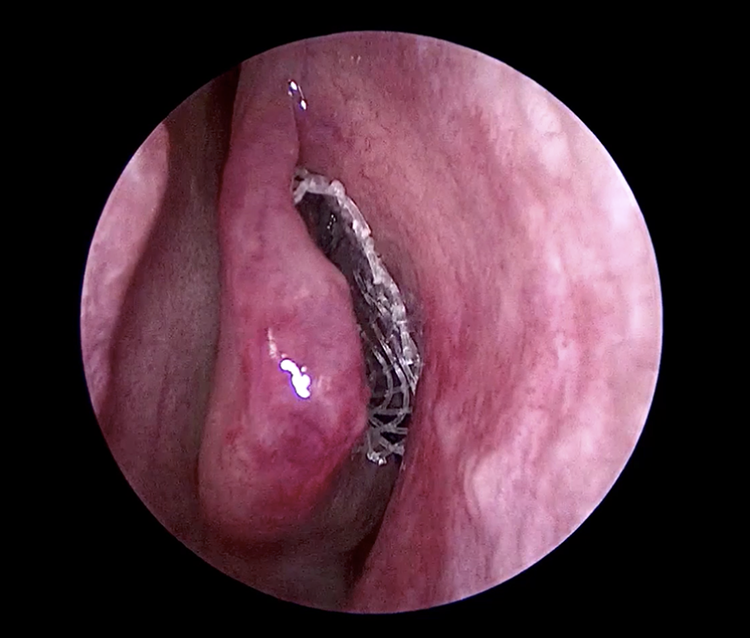
An endoscopic view of the mometasone furoate (MF) implant (LYR-210) within the unoperated ethmoid cavity. This technology is composed of a biocompatible mesh scaffold that dynamically adapts to the targeted anatomy while it elutes MF for several months to the mucosa. Figure courtesy of Richard Douglas, MD.
In summary, topical corticosteroids play a vital role in CRS treatment and disease control. Although topical steroids have the same potential side effects as do systemic steroids, the nature of application and metabolism significantly reduces the likelihood of these complications. However, use of topical steroids can be limited by patient compliance. In-office application of targeted corticosteroids has undergone a great deal of evolution in the past several decades. In-office steroid eluting devices have shown efficacy in the treatment of refractory CRS and reduction in rates of reoperation, supplementary oral steroid requirement, and overall inflammation with minimal side effects. As evident in recent clinical trials, the newest generation of drug eluting implants specifically has proven to be an attractive alternative for select patients wishing to avoid revision surgery and should be considered an important adjunct for treatment of refractory CRS in an otolaryngologist’s armamentarium.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: KCW has consultant relationships with Baxter, Acclarent, and OptiNose.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article OptiNose sponsored the publishing costs for this publication.
