Abstract
Background
Supraglottic laryngeal squamous cell carcinoma (LSCC) is the second most common type of laryngeal cancer with a poor prognosis. Current population-based estimates of the early death rate and associated factors for early death of supraglottic LSCC are lacking. The purpose of this study was to assess the early death rate and related factors for early death in patients with supraglottic LSCC.
Methods
We identified 3733 adult patients diagnosed with supraglottic LSCC between 2010 and 2017 for whom the vital status at 3 months was known from the Surveillance, Epidemiology, and End Results (SEER) database. Patients were staged according to the seventh edition of the American Joint Committee on Cancer (AJCC) tumor-node-metastasis (TNM) staging system. The early death (survival time ≤ 3 months) rate was calculated. Univariate and multivariate logistic regression analyses were performed to identify the risk factors associated with the early death rate.
Results
313 (8.38%) of the 3733 patients died within 3 months of diagnosis of supraglottic LSCC. Of these, 225 patients died from cancer-specific causes. Multivariate logistic regression analyses confirmed that advanced age, male sex, advanced T stage, advanced N stage, advanced M stage, and not undergoing treatment (surgery, radiotherapy, and chemotherapy) had significant correlations with all-cause early death in supraglottic LSCC. In addition, advanced age, advanced T stage, advanced N stage, advanced M stage, and not undergoing treatment (surgery, radiotherapy, and chemotherapy) were significantly correlated with cancer specificity in supraglottic LSCC.
Conclusion
When a tumor is newly diagnosed, we should pay close attention to sex, age, unmarried status and AJCC TNM staging to quickly detect supraglottic LSCC patients with a tendency toward early death. These findings have implications for precise prognosis prediction and individualized and personalized patient counseling and therapy.
Keywords
Background
Laryngeal cancer is a malignant tumor that originates from the epithelium or connective tissue of the larynx, which can be divided into the supraglottic larynx, glottic, and subglottic regions. It is the most prevalent type of human head and neck tumors in many countries, accounting for 1% to 2.5% of all human neoplasms, with 177,422 new cases and 94,771 deaths worldwide in 2018. 1 Histopathologically, the majority of laryngeal malignancies (approximately 95%) are squamous cell carcinoma, 2 and more than 95% of laryngeal cancer cases are identified as laryngeal squamous cell carcinoma (LSCC). Supraglottic LSCC is the second most common type of laryngeal cancer, accounting for 30% to 40% of cases of laryngeal cancer. 3 Although surgical removal or adjuvant therapy, such as radiation therapy and chemotherapy at the early stage of LSCC, can often be curative, the prognosis for LSCC has not shown any improvement in the last 30 years. 4 Compared with other laryngeal cancer subsites, supraglottic LSCC presents many distinct clinical and biological features, such as its atypical clinical symptoms in the early stage and being more likely to recur and metastasize.5,6
The incidence and mortality rates of laryngeal cancer have not remarkably decreased in recent decades. Related reports on laryngeal cancer mortality are abundant. Between 10 September 1991 and 10 September 2021, more than 1500 articles with abstracts could be identified when searching PubMed for “laryngeal neoplasms [Mesh]” and “mortality [Mesh].” However, available information on early laryngeal cancer mortality is limited, especially for supraglottic LSCC. To improve the outcome of supraglottic LSCC patients, it is of great importance to identify patients at high risk of early death based on prognostic factors and to develop treatment strategies for these patients. Therefore, the purpose of this study was to evaluate the early death rate and associated risk factors in patients with supraglottic LSCC.
Materials and methods
Study design and study population
The present research using the National Cancer Institute’s Surveillance, Epidemiology, and End Results (SEER) database was conducted for all patients with supraglottic LSCC diagnosed from 2010 to 2017. All supraglottic LSCC patients were diagnosed by histopathology, and cancer staging information for each dataset was determined using the American Joint Committee on Cancer (AJCC) Staging Manual, seventh edition. The year of diagnosis, age, sex, race, marital status, clinical stage, survival time, outcome, cause of death, total number of in situ/malignant tumors, distant metastatic sites, and treatment information, including whether the patients underwent surgery, radiation therapy, chemotherapy, or systemic therapy, were collected from the SEER 18 Registries Research Plus Date, Nov 2020 Sub (2000-2018). The SEER research data were available using SEER*Stat 8.3.9.1 (http://seer.cancer.gov//seerstat/). Patients with the following features were excluded: (1) Supraglottic LSCC was not the primary malignancy; (2) lacking information about distant metastatic sites such as bone metastasis, brain metastasis, hepatic metastasis, and pulmonary metastasis; and (3) information on age, sex, race, marital status, clinical stage, survival time, outcome, cause of death, and treatment information was unavailable. According to the latest age classification criteria of the World Health Organization (WHO), 7 age at the time of diagnosis was divided into 3 groups: (1) youths: ≤ 44 years, (2) middle-aged people: 45-59 years; and (3) elderly people: ≥ 60 years. Based on previous literature, early death was defined as an overall survival time ≤ 3 months after initial diagnosis. 8 The original SEER database contains data for 8178 supraglottic LSCC patients diagnosed from 2010 to 2017. We excluded 4445 patients and finally obtained data for 3733 supraglottic LSCC patients for this study.
Statistical analysis
A descriptive analysis was performed. Frequencies and percentages were reported for categorical variables. Comparisons of demographic and clinical characteristics were conducted using Pearson chi-square or Fisher’s exact tests as appropriate. Univariate and multivariate logistic regression analyses were used to determine potential risk factors for early death in supraglottic LSCC patients. Odds ratios (ORs) and 95% confidence intervals (95% CIs) were calculated. All factors with a P value < .1 detected in the univariate analyses were included in the multivariate analyses, and a P value < .05 was considered statistically significant in the multivariate analyses. All statistical analyses were performed using STATA version 16.0 (Stata Corp, TX, USA) and GraphPad Prism 9 (GraphPad, CA, USA).
Results
Mortality rates within 3 months following diagnosis
3-month life status and survival time of 3733 patients with supraglottic LSCC were gained from SEER database, and 313 (8.38%) died within 3 months after diagnosis.
The cause of early death for supraglottic LSCC patients
During the study, 313 early deaths were documented: 225 (71.88%) deaths from cancer-specific causes; 33 (10.54%) from heart diseases; 11 (3.51%) from chronic obstructive pulmonary disease; 5 (1.60%) from cerebrovascular diseases; 5 (1.60%) from septicemia; 3 (.96%) from pneumonia and influenza; 3 (.96%) from other infectious and parasitic diseases, including HIV; and 28 (8.95%) from all other causes (Figure 1). The cause of death in the patients with supraglottic LSCC. (A) Distribution of the incidence of early death. (B) Distribution of the incidence of non-cancer-specific early death. Abbreviation: Supraglottic LSCC, supraglottic laryngeal squamous cell carcinoma.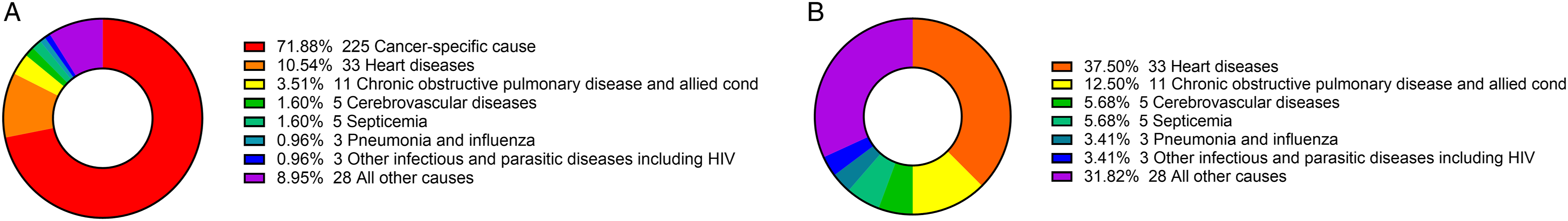
Demographic and clinical characteristics of all supraglottic LSCC patients
Demographic characteristics of supraglottic laryngeal squamous cell carcinoma patients.
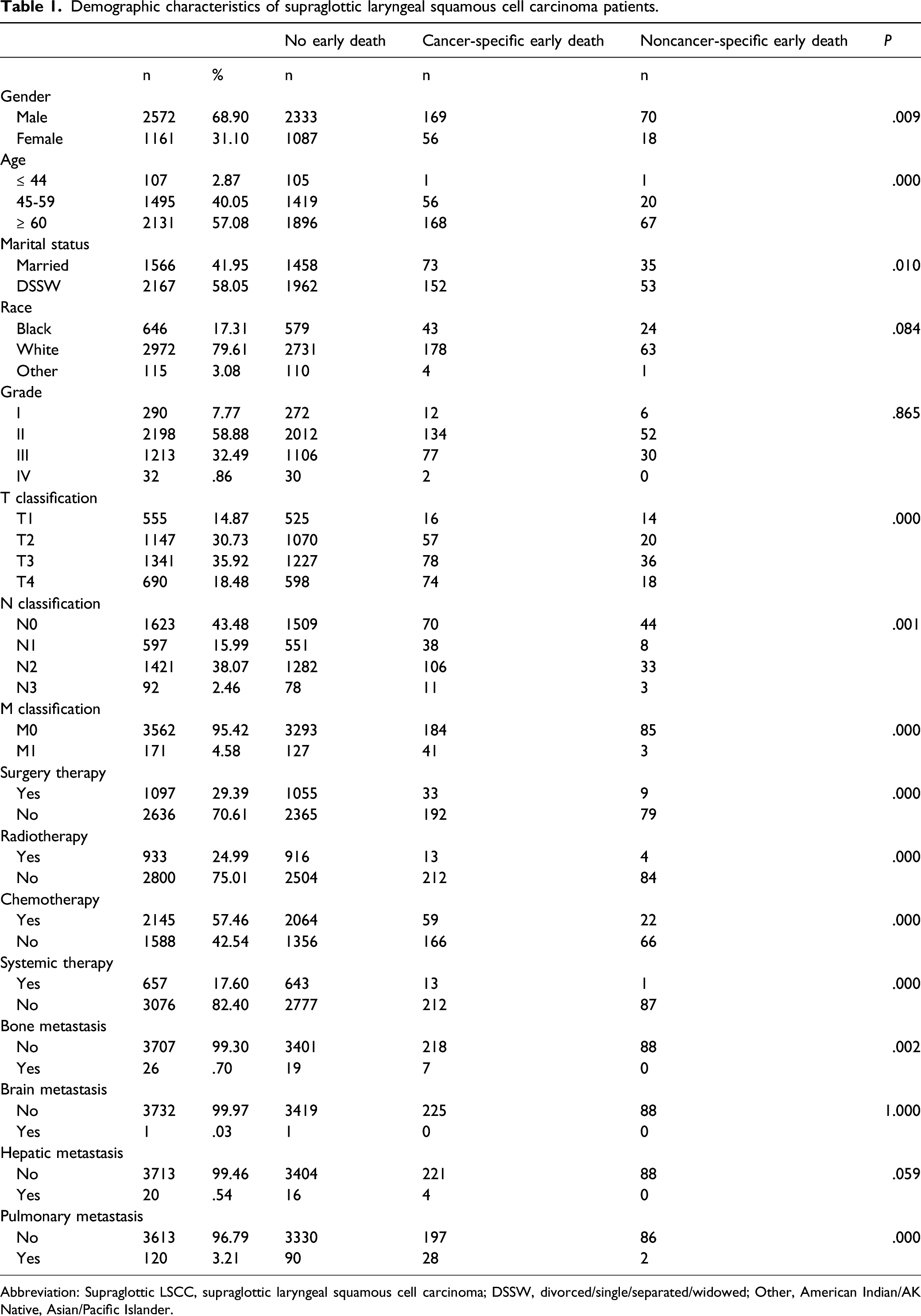
Abbreviation: Supraglottic LSCC, supraglottic laryngeal squamous cell carcinoma; DSSW, divorced/single/separated/widowed; Other, American Indian/AK Native, Asian/Pacific Islander.
Independent risk factors for early death
Univariate logistic regression analyses of risk factors for early death.
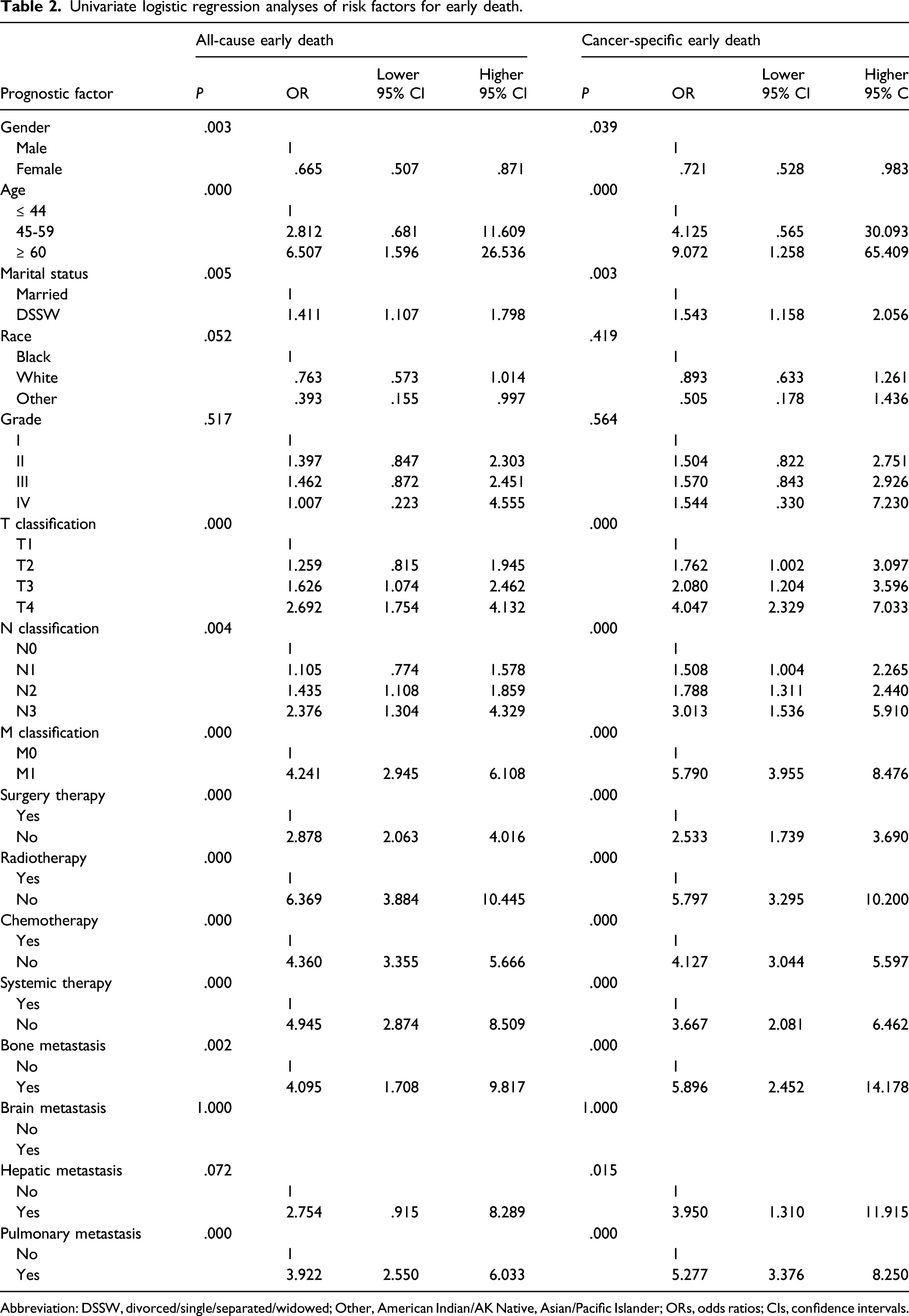
Abbreviation: DSSW, divorced/single/separated/widowed; Other, American Indian/AK Native, Asian/Pacific Islander; ORs, odds ratios; CIs, confidence intervals.
Multivariate logistic regression analyses of risk factors for early death.
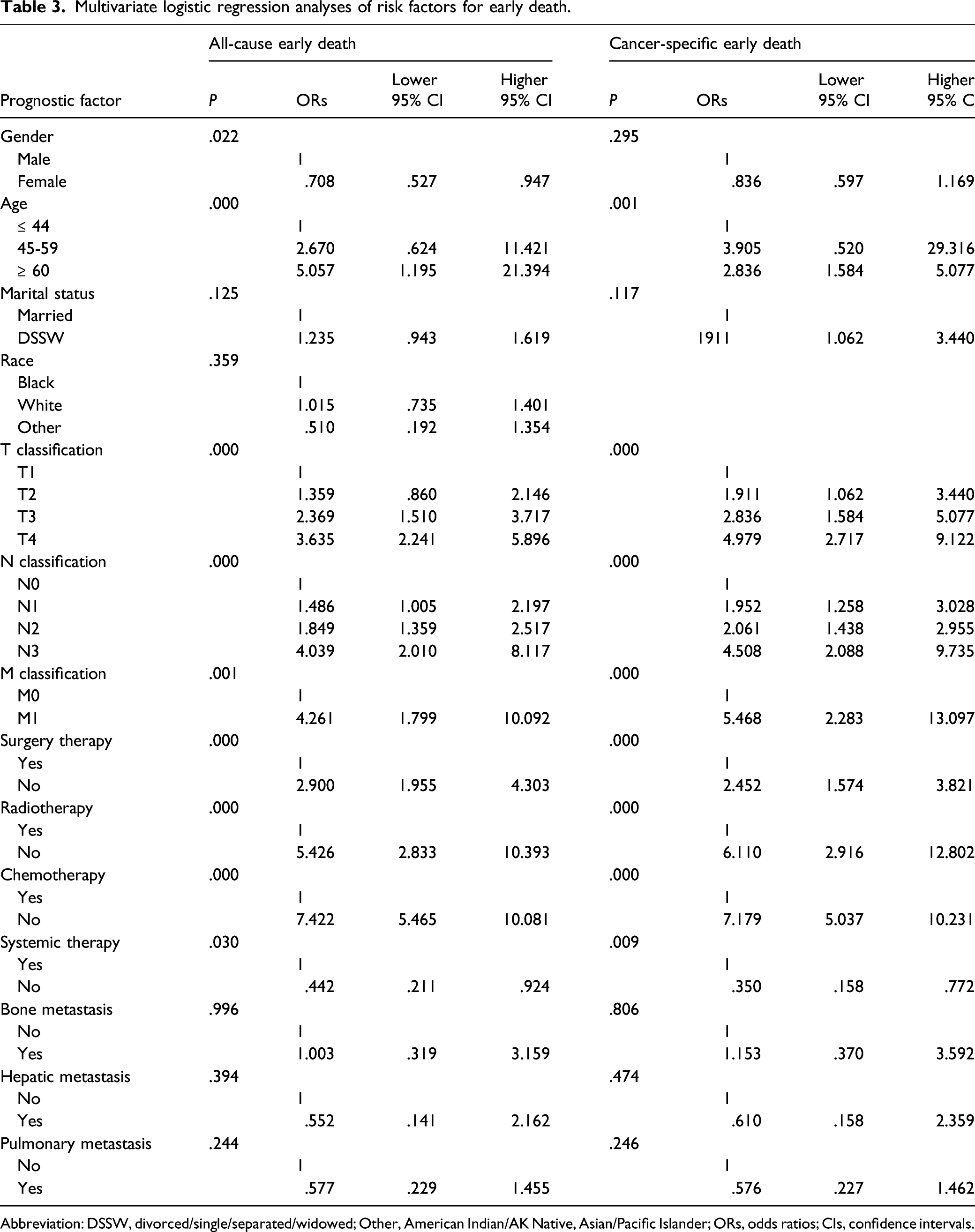
Abbreviation: DSSW, divorced/single/separated/widowed; Other, American Indian/AK Native, Asian/Pacific Islander; ORs, odds ratios; CIs, confidence intervals.
Discussion
Unfortunately, even though the overall incidence of laryngeal cancer is declining, it is one of the few oncological diseases whose 5-year survival rate has dropped from 66% to 63% over the past 40 years. 2 Supraglottic LSCC is the second most common type of laryngeal cancer, whose oncological behavior is different from that of the glottic and subglottic types, and the 5-year survival rate of patients with supraglottic LSCC is 54%, which is lower than the survival rate of glottic type.2,6 This highlights the need for further action and further research in this important area.
In this study, the 3-month mortality rates among patients with newly diagnosed supraglottic LSCC were calculated. We found that 8.38% of the supraglottic LSCC patients died within 3 months following diagnosis. Premature death is a critical and intractable problem in cancer treatment.9,10 Early death was defined as 30 days or 3 months after a new diagnosis in previous reports.9,11 Therefore, we defined early death as 3 months after newly diagnosed supraglottic LSCC in our study. To provide personalized treatment plans for patients, understanding the relationship between risk factors and premature death is critical. Therefore, based on the data of supraglottic LSCC patients in the SEER database, we analyzed the factors associated with early death and directed personalized screening for early death risk.
In this study, we found that the leading causes of early death among patients with supraglottic LSCC were cancer-specific causes, followed by heart disease, which accounted for 71.88% and 10.54% of early deaths, respectively. The AJCC TNM staging system is a widely used cancer staging system to predict prognosis and survival.12,13 However, the TNM staging system continuously lacks accuracy for the prognostic prediction of patients, as external confounding factors such as age, sex, ethnicity, marital status, and other socioeconomic factors are ignored. 14
Prior studies have reported that the smoke habit is an independent risk factor for worse specific survival in LSCC, 15 and advanced age indicates a poorer prognosis in patients with LSCC.16,17 The SEER database does not provide environmental exposure data, including smoking, so we were unable to assess smoke habit in our analyses, and our results tend to suggest that age at diagnosis is a risk factor for early death from all causes and that cancer-specific early death may also be a warning factor. There was a positive relationship between age at diagnosis of supraglottic LSCC patients and early death. The reason behind this phenomenon may likely be due to the heterogeneity of the tumors, immune status, nutritional status, and physiology of various organs. It is well known that poorer nutritional quality and immune responsiveness among elderly individuals results in increased susceptibility and severity of cancer-related complications. 18 Furthermore, the mortality of cardiac and cerebrovascular diseases likewise increased with age. 19
There is a clear consensus that laryngeal cancer occurs more commonly in men than in women.20,21 In our study, the multivariate logistic regression analyses showed that the difference was statistically significant in terms of sex (P < .05) in all-cause early death; contrary results occurred for cancer-specific early death. We argue, however, that male sex was an independent risk factor in patients with supraglottic LSCC. The possible reason for this aspect might be that male patients more commonly smoked or drank in comparison with females (smoking, 26% vs 0%; drinking, 23% vs 4%). 22
Marital status, as one of the social or psychological factors, has long been recognized as an important prognostic factor for nearly all cancers, including colorectal cancer, 23 breast cancer, 24 non-small-cell lung cancer, 25 glioblastoma multiforme, 26 chondrosarcoma, 27 and laryngeal cancer. 28 That is, the prognosis of married patients is remarkably better than that of unmarried patients in cancer patients. Is marital status at the time of diagnosis of supraglottic LSCC patients relevant to early death? In our study, the univariate logistic regression analyses indicated being divorced/single/separated/widowed (DSSW) as a risk factor for supraglottic LSCC, but the multivariate logistic regression analyses showed no statistical significance. Nevertheless, we believe that the reason why the different results were observed might be because some patients’ marital status may change during follow-up and being DSSW can be an independent risk factor for all-cause early death and cancer-specific early death in supraglottic LSCC patients. Some underlying reasons may be that marriage could improve cardiovascular, endocrine, and immune functions. 29 Moreover, romantic partnerships and marriage might enhance therapeutic interventions in patients with supraglottic LSCC within a biopsychosocial framework.
In this study, tumor characteristics, including advanced T, advanced N, and advanced M stages, were associated with a higher chance of all-cause and cancer-specific early death. Distant metastases almost invariably herald a poor prognosis with a very short overall survival, especially for head and neck cancer. 30 Possible causes include damage to vital organs and increased tumor load to lethal levels. Furthermore, when adjusted for potential confounders in multivariate logistic regression analyses of risk factors for all-cause early death, patients who underwent surgical therapy had a significant survival benefit compared to those without the surgical approach, with odds ratio (OR) = 2.900 (95% CI: 1.955-4.303); radiation therapy and chemotherapy have similar survival benefits, with OR = 5.426 (95% CI: 2.833–10.393) and OR = 7.422 (95% CI: 5.465–10.081), respectively. And, results remained similar when we analyzed several risk factors for cancer-specific early death. In short, treatments, including surgery, radiotherapy, and chemotherapy, might improve the long-term prognosis of supraglottic LSCC. This view is currently considered as a consensus. Our analysis showed that aggressive treatment targeted to tumors could reduce the early death rate. In summary, early diagnosis and effective treatment of supraglottic LSCC is the key to improving the survival rate of patients and reducing the early death rate.
Several potential limitations exist in our research. First, due to the data limitations of the SEER database, the applicability of our results is limited to America; the results may differ in other areas with different health care systems. Second, there was a lack of data, such as for comorbidities, performance status, and recent weight loss, which are potential predictors of early death. Additionally, because our data were retrospectively reviewed, future prospective studies are needed to validate our findings.
Conclusion
Our results showed that a total of 8.38% of supraglottic LSCC patients had an early death. Advanced age, male sex, unmarried status, advanced T stage, advanced N stage, distant metastasis, and not undergoing treatment are important predictors of early death in supraglottic LSCC patients. When a tumor is newly diagnosed, we should pay close attention to sex, age, unmarried status, and AJCC TNM staging to quickly detect supraglottic LSCC patients with a tendency to die early, and give patients a chance to receive systemic treatment to reduce early death, and/or give patients and their families time to plan for the future.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The present study was supported by the Discipline Leader Training Program of Health Commission (WD2019-13, Qingpu District, Shanghai, P.R. China) and the Project of Health Commission (W2021-03, Qingpu District, Shanghai, P.R. China).
Data availability
The dataset analyzed in this study is available in the SEER database.
