Abstract
Background:
The impact of obstructive sleep apnea (OSA) on subsequent cardiovascular events in patients with acute coronary syndrome (ACS) remains inconclusive.
Aim:
Our aim was to systematically assess the relationship between preexisting OSA and adverse cardiovascular events in patients with newly diagnosed ACS by conducting a systematic review and meta-analysis.
Methods:
We systematically searched PubMed, EMBASE, and Cochrane library for studies published up to May 1, 2020, that reported any association between OSA and cardiovascular events in patients with newly diagnosed ACS. The main outcomes were a composite of all-cause or cardiovascular death, recurrent myocardial infarction, stroke, repeat revascularization, or heart failure. We conducted a pooled analysis using the random-effects model. We also performed subgroup, sensitivity, heterogeneity analysis, and the assessment of publication bias.
Results:
We identified 10 studies encompassing 3350 participants. The presence of OSA was associated with increased risk of adverse cardiovascular events in newly prognosed ACS (risk ratio [RR] 2.18, 95% confidence interval [CI]: 1.45-3.26, P < .001, I2 = 64%). Between-study heterogeneity was partially explained by a multicenter study (9 single-center studies, RR 2.33 95% CI 1.69-3.19, I2 =18%), and I2 remarkably decreased from 64% to 18%. Moreover, OSA significantly increased the incidence of repeat revascularization (8 studies) and heart failure (6 studies) in patients with newly diagnosed ACS.
Conclusion:
Patients with preexisting OSA are at greater risk of subsequent cardiovascular events after onset of ACS. Further studies should investigate the treatment of OSA in patient with ACS.
Introduction
Obstructive sleep apnea (OSA) is an increasingly widespread public health problem worldwide that affects nearly 1 in 5 adults. 1 Emerging evidence indicates that OSA can cause initiation and deterioration of atherosclerosis through activation of oxidative stress, inflammation, and sympathetic system and metabolic abnormalities, eventually leading to high morbidity and mortality in cardiovascular disease. 2 -4 Although there has been a large improvement in medication and device therapy, with a slight decline in risk of mortality, acute coronary syndrome (ACS) is one of the leading causes of morbidity in Europe. 5
The relationship between ACS with OSA is complex. On the one hand, a study by Mohananey et al 6 of 1.85 million patients with acute ST-segment elevation myocardial infarction (STEMI) showed that OSA patients had more risk factors, such as hypertension, male sex, smoking, renal failure, chronic respiratory disease, obesity, dyslipidemia, compared to non-OSA patients. However, interestingly, OSA patients have statistically lower in-hospital mortality and longer hospital stays, which may exhibit a protective effect on the heart due to ischemic preconditioning by intermittent hypoxemia. Inversely, most other observational studies have found a worse prognostic impact of OSA in patients with ACS. 7,8 Notably, a multicenter, high-quality trial published in 2019 concluded that the prevalence of cardiovascular events is not related to OSA in patients with ACS. 9
Therefore, to date, the impact of preexisting OSA on cardiovascular events in newly diagnosed ACS seems to be controversial. Additionally, to the best of our knowledge, meta-analysis assessing the association of preexisting OSA with long-term cardiovascular outcomes in newly diagnosed ACS has not yet been reported. Obstructive sleep apnea has been recommended as an emerging risk factor for coronary artery disease by American expert consensus and Europe clinical guidelines, 10,11 and its treatment with continuous positive airway pressure (CPAP) can significantly lower life-threatening cardiovascular events in observational studies, 12 including some patients with ACS. Consequently, we conducted a systematic review and meta-analysis to evaluate the impact of preexisting OSA on subsequent cardiovascular events in newly diagnosed patients with ACS.
Methods
Search Strategies
We conducted this meta-analysis according to the meta-analysis of observational studies in epidemiology guidelines. 13 We systematically searched PubMed, EMBASE, and Cochrane library for studies up to May 1, 2020, for articles evaluating the impact of OSA on any prognostic outcomes in patients with ACS, without restrictions to language of publication. We used Medical Subject Heading terms relating to “Acute Coronary Syndrome” and “Obstructive Sleep Apnea,” as well as related text words. The references of all included studies and relevant review articles were manually searched for potential full-text citations, and only conference abstracts were excluded due to a lack of peer review. Details of each search strategy are listed in Supplemental Table 1.
Study Selection and Eligibility Criteria
Two authors (Z.W. and Y. L. both cardiologists) independently judged the eligibility of articles by reviewing their titles and abstracts. Studies that described the prognostic outcomes of OSA and non-OSA (control group) in patients with ACS were included. Details of inclusion criteria are as follows: (1) study participants consisted of adults (aged ≥ 18 years) clearly diagnosed with ACS (unstable angina, ST segment elevation myocardial infarction, and non-ST segment elevation myocardial infarction); (2) diagnosis of OSA was based on Apnea-Hypopnea Index (AHI) extracted from overnight sleep study, such as polysomnography (PSG) or portable diagnostic device, within 1 month of ACS; (3) detailed clinical outcomes longer than 3 months were clearly recorded; and (4) included a comparison between OSA and non-OSA patients. Some studies comparing treated OSA with untreated OSA and to those without OSA were included in this study, only data on untreated OSA and no OSA (control) were extracted.
The exclusion criteria were as follows: (1) OSA diagnosis was not based on AHI; (2) studies that did not specify exclusion of predominantly central sleep apnea; (3) editorials, letters to the editor, conference abstracts, reviews, case reports, and animal experimental studies.
Each full-text article was assessed by 2 authors, respectively. In case of disagreement between 2 reviewers, full discussion was conducted by inviting a third reviewer to reach a consensus.
Data Extraction and Quality Assessment
Two authors independently extracted relevant data on the basis of a prespecified electronic form. Individual items consisted of the author, year of publication, design type, geographical location, method of diagnosis, the time of diagnosis after ACS onset, sample size, type of ACS, adjusted confounders, as well as mean age, gender, and numbers of each group from each included study. Discrepancy between 2 reviewers was settled either through a consensus or by consulting a third reviewer. We also contacted corresponding authors for any missing data or full-text if related information is not available by search. Quality of eligible studies was assessed using the Newcastle-Ottawa Scale (NOS) for cohort studies in the meta-analysis. 14 The elements of NOS checklist were divided into 3 domains and included selection of included subjects, comparability of cohort on the basis of the design, and ascertainment of either the exposure or the outcomes of participants. The scale extended from 0 to 9 points. Low-quality studies were defined as ≤5 points, moderate-quality studies as 6 to 7 points, and high-quality studies as 8 to 9 points.
The primary end point of concern was major adverse cardiovascular and cerebrovascular events (MACCE), which was defined as a composite of all-cause or cardiovascular death, recurrent myocardial infarction (MI), stroke, repeat revascularization, or hospitalization for heart failure. Secondary end points consisted of individual cardiovascular events. Definitions of each end point event were in accordance with guidelines during each study period. The quality of each outcome was assessed using the Grading of Recommendations Assessment, Development, and Evaluation (GRADE) system. 15
Study Design, Patient Demographics, and Procedural Characteristics.
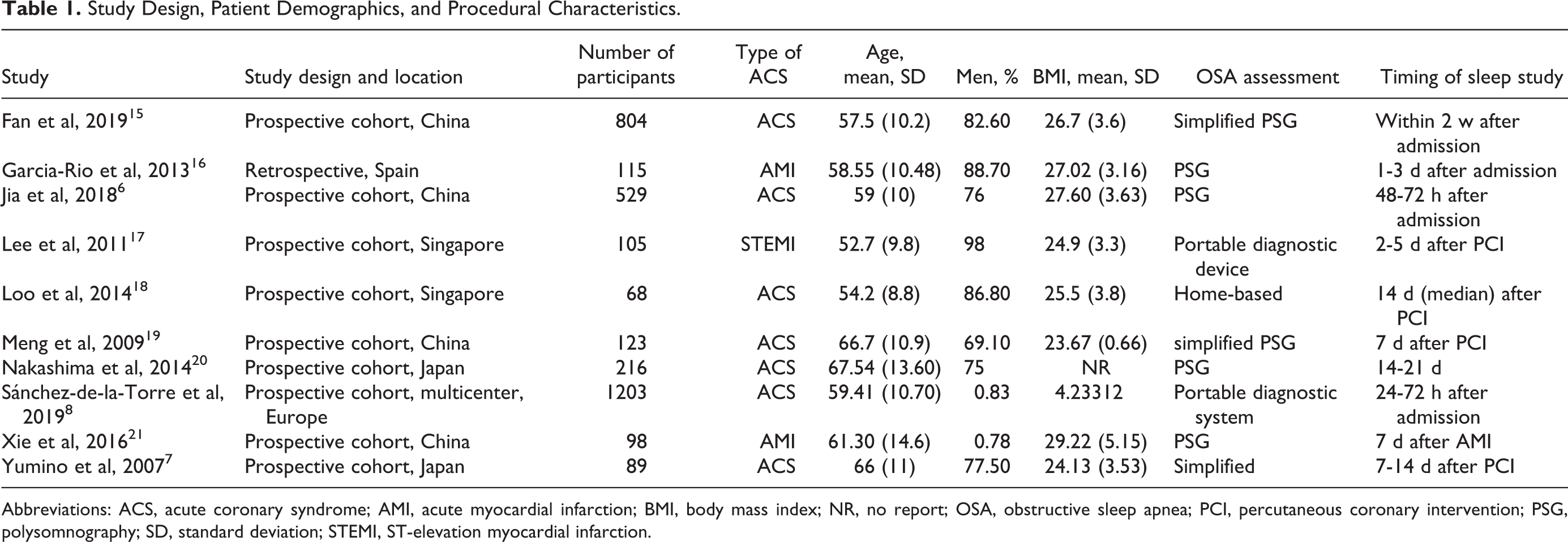
Abbreviations: ACS, acute coronary syndrome; AMI, acute myocardial infarction; BMI, body mass index; NR, no report; OSA, obstructive sleep apnea; PCI, percutaneous coronary intervention; PSG, polysomnography; SD, standard deviation; STEMI, ST-elevation myocardial infarction.
Data Synthesis and Statistical Analysis
Our objective focused on the prognostic impact of preexisting OSA on adverse cardiovascular outcomes in patients with newly diagnosed ACS. Standard meta-analysis methods were conducted. In general, multivariable-adjusted hazard ratio or risk ratio (RR) from original studies was the preferred effect estimate. Otherwise, the unadjusted RR of the outcomes of interest using crude data was calculated. Pooled RR with 95% confidence interval (CI) were calculated for both the primary and secondary outcomes by random-effects model (DerSimonian–Laird method 16 ). Heterogeneity between studies was evaluated with Cochran’s Q test (reported as χ2 and P < .10) and the I2 statistic. Definition of significant heterogeneity was referred to as I2 > 50% or P < .10. The publication bias was investigated by a funnel plot, Egger test, and the trim-and-fill method. All data analyses were performed using the Cochrane Review Manager software (version 5.3) and Stata version 11.2 (StataCorp LP). A 2-sided P < .05 was deemed significant.
Results
Study Selection and Characteristics
Our investigation for studies among the electronic databases yielded 1583 publications. After removing duplicates and screening the titles and abstracts, 36 studies remained. Subsequently, 25 studies were excluded by further full-text assessment, among which 2 studies included patients with stable coronary artery disease, 2 included patients with central sleep apnea, 2 were excluded as the diagnosis of OSA was based on non-AHI methods, and 6 were excluded as they did not report cardiovascular events or a sufficient follow-up period (>3 months). Additional exclusion criteria are presented in Figure 1. Overall, a total of 3350 subjects in 10 studies were included in this meta-analysis. 7 -9,17 -23 Detailed characteristics of each study are summarized in Tables 1 and 2. The publication date of the studies extended from 2007 to 2019. The number of participants ranged from 68 to 1203. All studies included 9 prospective cohorts as well as 1 retrospective cohort. 18 The average age of each cohort ranged from 52 to 67 years, and more males than females (81.1% vs 18.9%) were included in the studies. Participants from 7 studies had patients with ACS, 8,9,17,20 -22 whereas the other 3 studies included acute myocardial infarction (AMI) patients (2 STEMI and 1 AMI). 18,19,23 Seven studies were from Asia, 2 from Europe, and 1 from the United States.
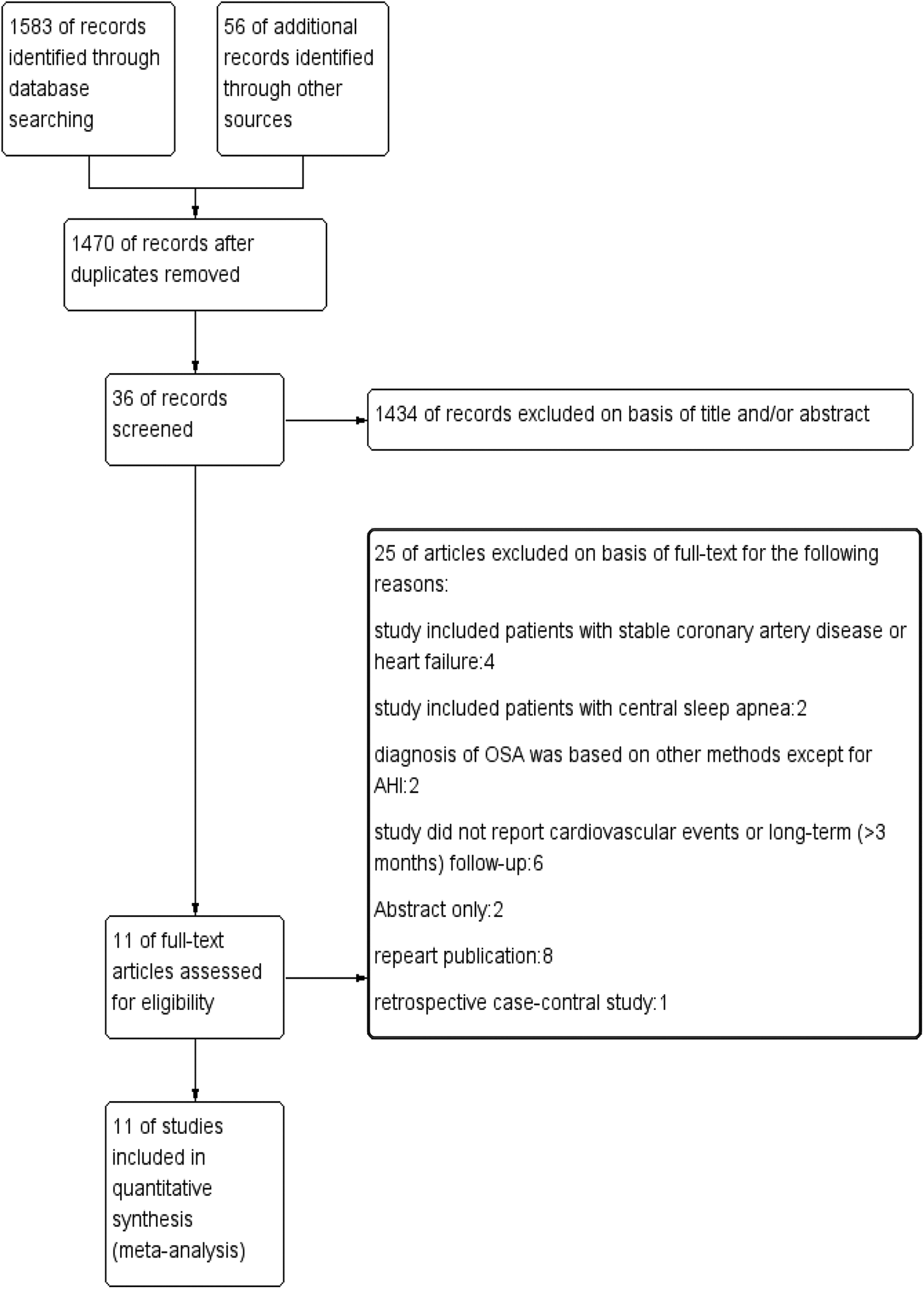
Flow chart of the study selection process for meta-analysis. OSA indicates obstructive sleep apnea.
Diagnosis of OSA is based on PSG across 4 studies and on portable sleep study across other 6 studies. Among these studies, the classification of OSA is based on standardized assessment of AHI in 5 studies, with AHI ≥ 15 as the cutoff value, 2 with AHI ≥ 30, 3 with AHI ≥ 5, and 1 with AHI ≥ 10. There were 4 studies that compared treatment of OSA with CPAP, untreated OSA and non-OSA group (control). Only untreated OSA and control groups were included in this meta-analysis. 9,18,22,23 Quality assessment using NOS score showed all included studies were moderate-high quality, with scores ranging from 6 to 9 points (Supplemental material: Table 2). The follow-up period for participants ranged from 7.5 to 78 months. The time of sleep study was 1 to 14 days after percutaneous coronary intervention or hospital admission.
Study Groups, Outcomes, Results, and Risk of Bias.
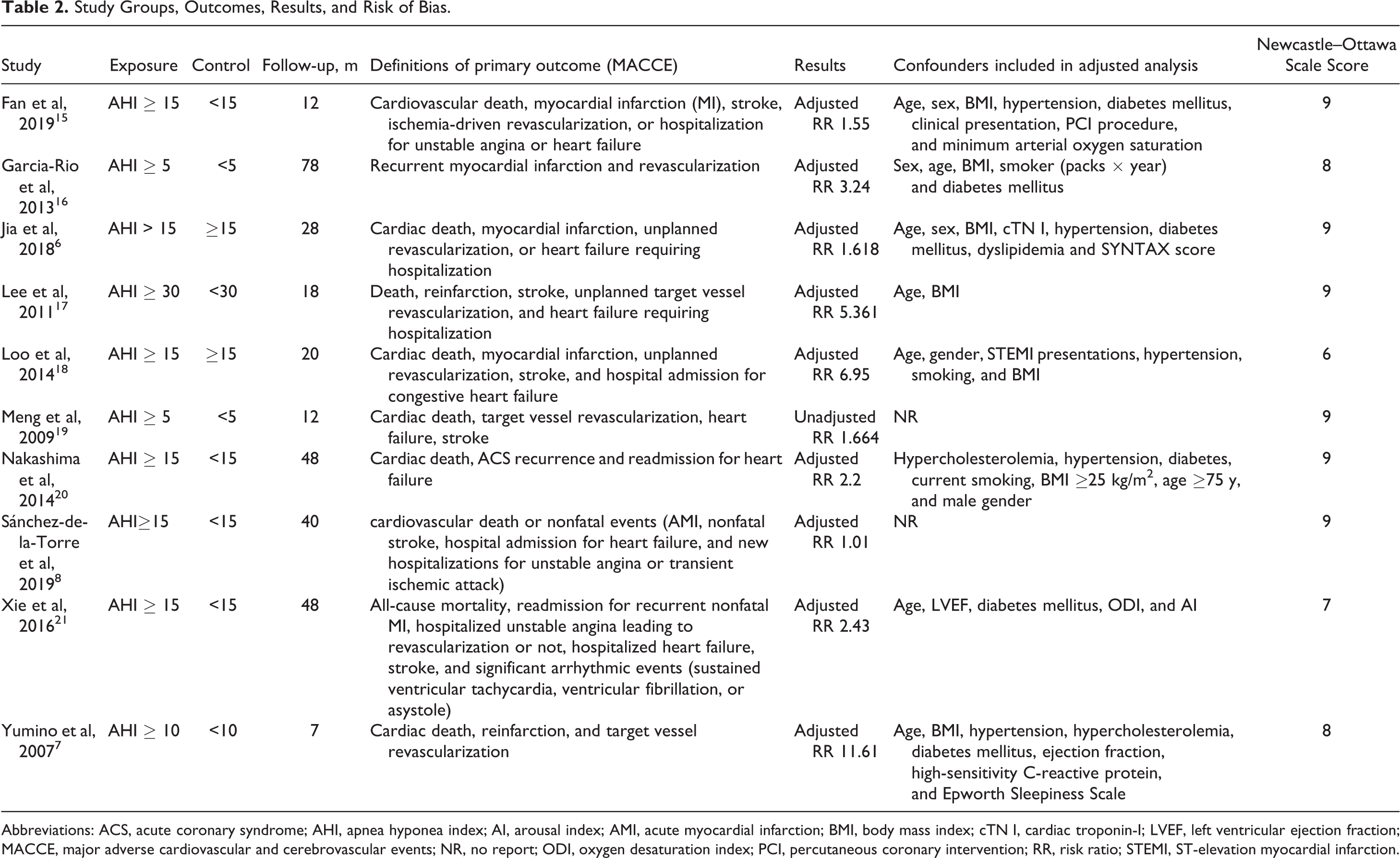
Abbreviations: ACS, acute coronary syndrome; AHI, apnea hyponea index; AI, arousal index; AMI, acute myocardial infarction; BMI, body mass index; cTN I, cardiac troponin-I; LVEF, left ventricular ejection fraction; MACCE, major adverse cardiovascular and cerebrovascular events; NR, no report; ODI, oxygen desaturation index; PCI, percutaneous coronary intervention; RR, risk ratio; STEMI, ST-elevation myocardial infarction.
Obstructive Sleep Apnea and MACCE in Patients With ACS
Overall, OSA significantly increased the risk of MACCE in patients with ACS (RR 2.18, 95% CI 1.45-3.26, P < .001; Figure 2). Evidence of significant heterogeneity across studies was observed for our primary outcome (Q statistic P = .003; I2 = 64%). Subgroup analysis according to sample size showed that the pooled RR of 3 studies with ≤100 patients (RR 4.66, 95% CI 1.74-12.53, I2 = 31%) was greater than that of 7 studies with >100 patients (RR 1.81, 95% CI 1.22-2.66, I2 = 61%). The heterogeneity did not remarkably vary across both subgroups (Supplemental Figure 1). The risk of MACCE increased in 9 studies with adjusted results (RR 2.27, 95% CI 1.46-3.54, I2 = 68%), with the exception of one study without adjustment (RR 1.66, 95% CI 0.63-4.37; Supplemental Figure 2). The pooled RR in participants based on a diagnostic method with PSG (RR 1.53, 95% CI 1.02-2.30, I2 = 75%) was greater than that of the portable sleep study (RR 2.33, 95% CI 1.17-4.60, I2 = 71%), with no significant difference in heterogeneity (Supplemental Figure 3). Interestingly, there was discrepancy in the relationship of MACCE with OSA between a large-scale, multicenter ISAACC study (RR 1.01, 95% CI 0.76-1.35) and all single-center studies (RR 2.33 95% CI 1.69-3.19). However, the risk of estimate with addition of the ISAACC study decreased (2.33-2.18), and the 95% CI became wider (1.69-3.19 to 1.45-3.26; Supplemental Figure 4).
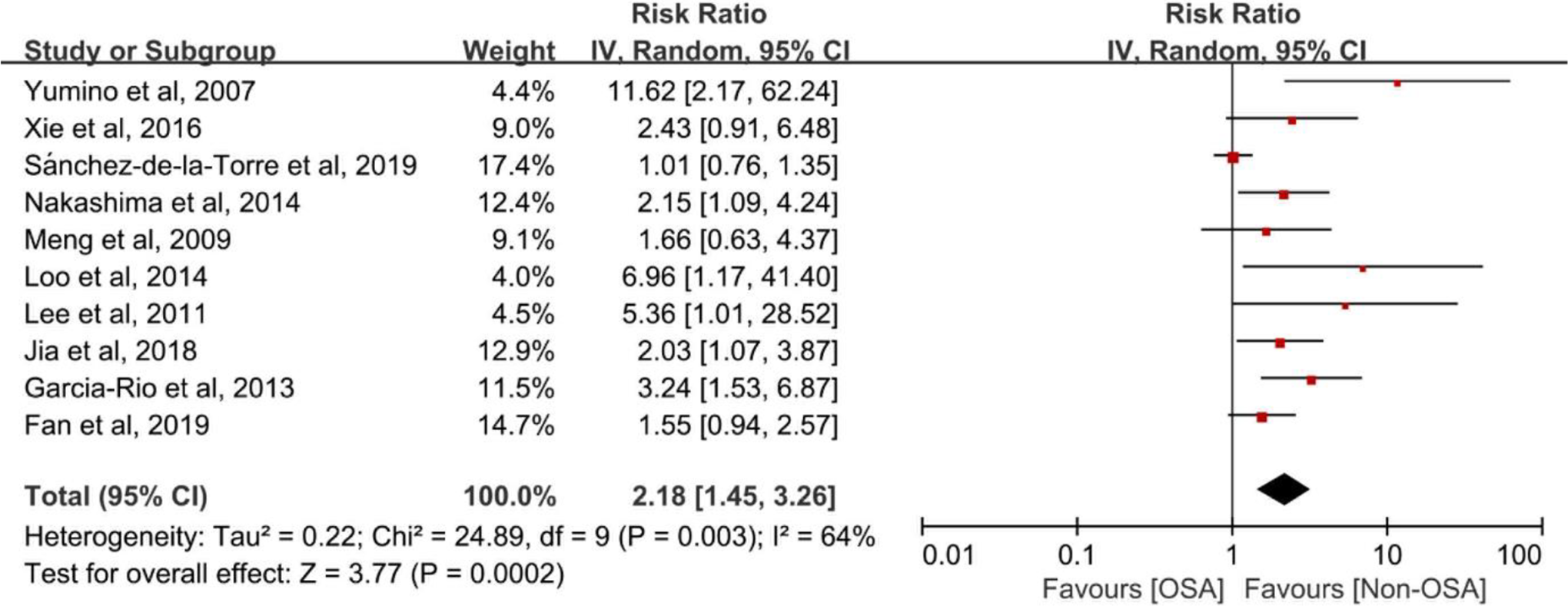
Risk estimates for MACCE. Forest plot displays the hazard ratio with 95% CI for MACCE in patients with OSA compared to control. The diamond indicates the point estimate, and the left and right end of the line indicate the 95% CI. CI indicates confidence interval, IV, inverse variance, MACCE, major adverse cardiovascular and cerebrovascular events, OSA, obstructive sleep apnea.
Sensitivity analyses were further conducted to identify the effect of variation on the quality and potential sources of heterogeneity (Table 3). We excluded a multicenter ISAACC study and found no changes in significance of effect size, while across-study heterogeneity decreased statistically (I2 from 64% to 18%). Therefore, the ISAACC trial may be the main source of across-study heterogeneity. We also evaluated the effect of excluding studies that were conducted retrospectively, with unadjusted values, cutoff value of more than AHI ≥ 15, sample size <100, follow-up duration <1 year, and 3 studies that only included AMI participants. None of the pooled results significantly influenced the size and direction of the effect of primary end point nor did they change the extent of heterogeneity. Notably, the pooled results of primary outcome should be interpreted with caution due to the low quality of included studies in the GRADE system (Supplemental Table 3).
Sensitivity Analysis for MACCE.
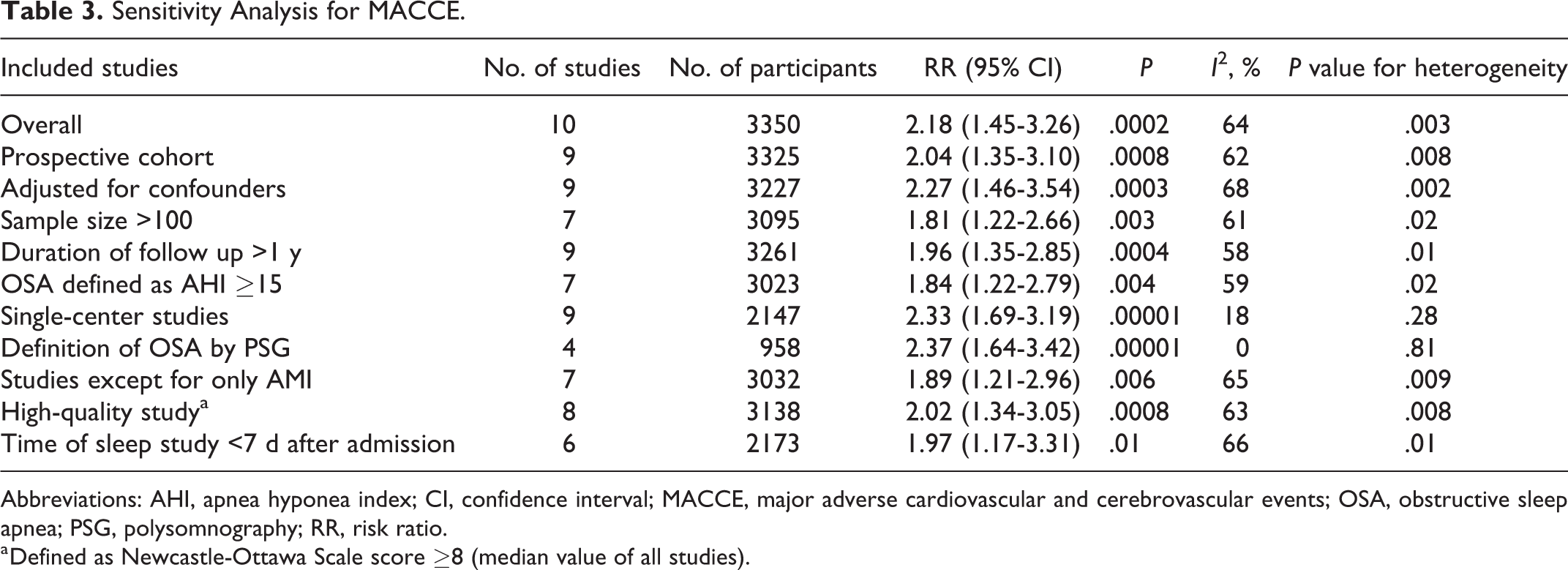
Abbreviations: AHI, apnea hyponea index; CI, confidence interval; MACCE, major adverse cardiovascular and cerebrovascular events; OSA, obstructive sleep apnea; PSG, polysomnography; RR, risk ratio.
a Defined as Newcastle-Ottawa Scale score ≥8 (median value of all studies).
There is a possibility of publication bias for outcomes of MACCE according to the asymmetry of the funnel plot (Supplemental Figure 4), which was further verified by the Egger test (P = .002). The trim-and-fill analysis was used to determine the potential size of small-study bias, the addition of hypothetical missing studies decreased the pooled RR to 1.42 (95% CI 1.07-1.88, P = .01) by the random-effects model, but still exhibited significant relationship between OSA and MACCE (Supplemental Figure 4).
Obstructive Sleep Apnea and Individual Cardiovascular Events in Patients With ACS
In studies that report outcomes of patients with repeat revascularization, OSA was found to be associated with an increased risk (8 studies with 3036 participants; RR 1.93, 95% CI 1.23-3.02, I 2 = 42%; Figure 3A) 7 -9,17 -21 in the pooled analysis, with no evidence of statistical heterogeneity. Pooled analysis including 6 studies (2519 participants) showed that OSA increased the risk of hospitalization for heart failure patients with ACS (RR 2.06, 95% CI 1.20-3.54, I2 = 0%) 9,17,19 -22 (Figure 3B). However, there were no significant differences for outcomes of recurrent MI, all-cause death and stroke between OSA and patients without OSA (Figure 4A-C). The quality of each individual outcome is shown in Supplemental Table 3.
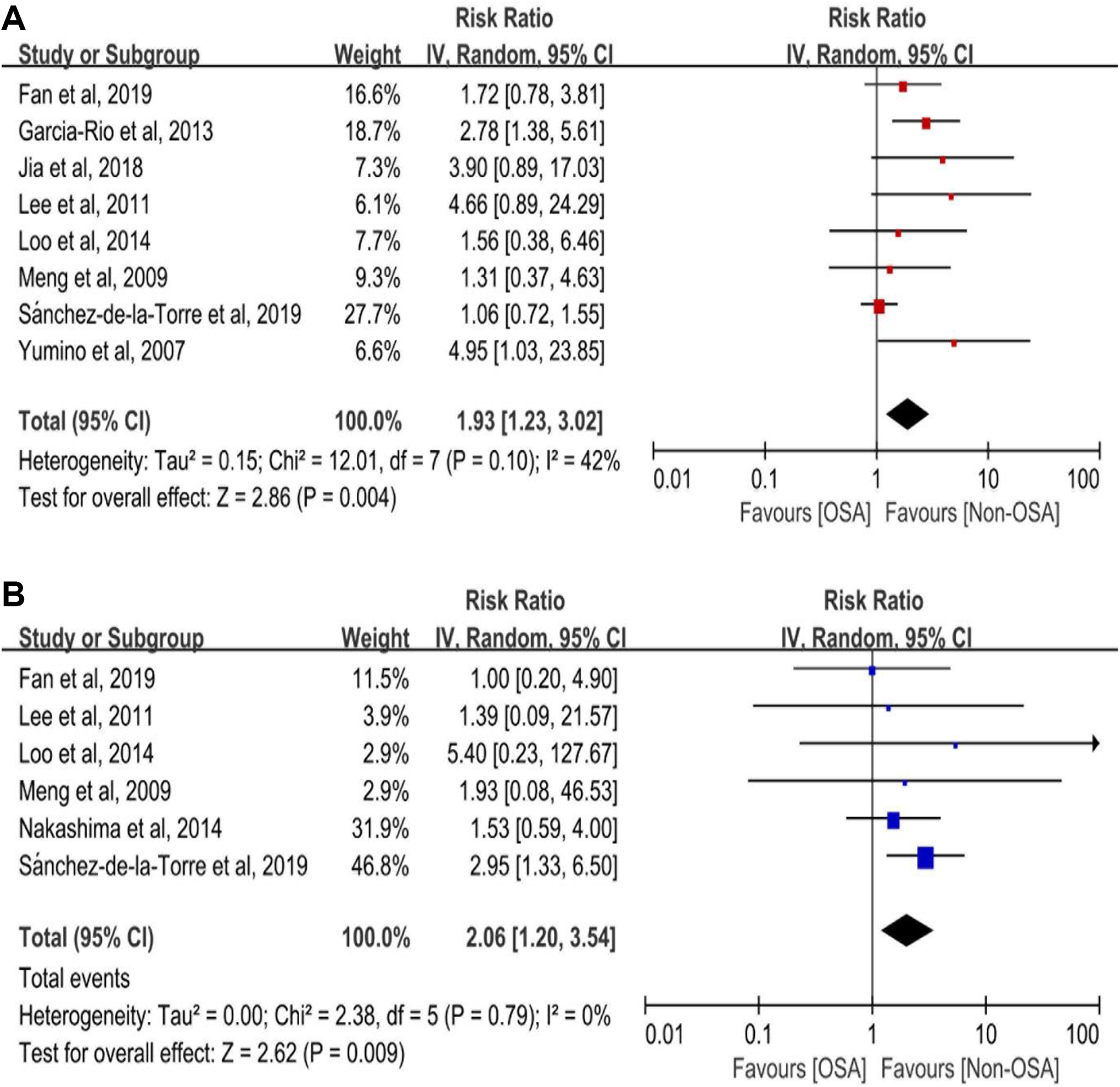
Risk estimates for repeat revascularization and hospitalization for heart failure. Forest plot displays the hazard ratio with 95% CI for repeat revascularization (A), hospitalization for heart failure (B) in patients with OSA compared to control. The diamond indicates the point estimate, and the left and right end of the line indicate the 95% CI. CI indicates confidence interval; IV, inverse variance; MACCE, major adverse cardiovascular and cerebrovascular events; OSA, obstructive sleep apnea.
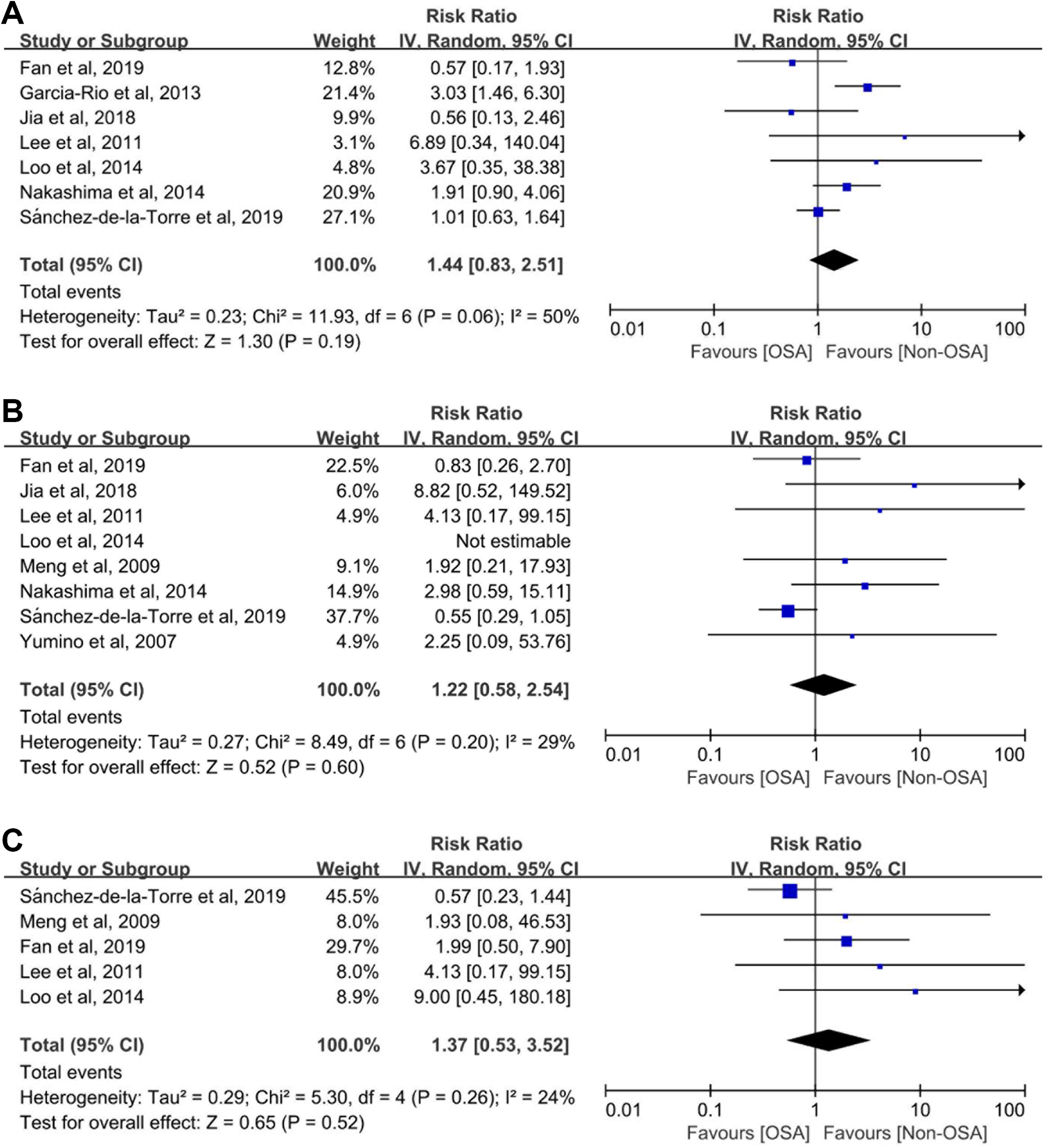
Risk estimates for recurrent myocardial infarction, all cause death and stroke. Forest plot displays the hazard ratio with 95% CI for recurrent myocardial infarction (A), all cause death (B), stroke (C) in patients with OSA compared to control. The diamond indicates the point estimate, and the left and right end of the line indicate the 95% CI. CI indicates confidence interval; IV, inverse variance; MACCE, major adverse cardiovascular and cerebrovascular events; OSA, obstructive sleep apnea.
Discussion
Results from our meta-analysis indicate that preexisting OSA is associated with a pooled 2-fold risk of recurrent MACCE in patients with newly diagnosed ACS. This finding was true despite the addition of a new large-scale ISAACC study with null findings, which demonstrates no association of OSA with increased prevalence of cardiovascular events among ACS patients without daytime sleepiness (Epworth Sleepiness Scale [EES] score ≤10). Between-study heterogeneity was largely explained by the ISAACC study, elimination of which reduced the heterogeneity from 64% to 18%. Therefore, the conclusion of this meta-analysis should be interpreted with caution. Our analysis results remain robust due to a variety of sensitivity analysis. Furthermore, OSA is significantly associated with an increased risk of repeat revascularization and hospitalization for heart failure among patients with newly diagnosed ACS.
The association of OSA with increased prevalence of cardiovascular events in patients with known stable cardiovascular disease, as well as the general population, has been confirmed in a longitudinal observational study 21 and meta-analysis. 22 However, data on patients with newly diagnosed ACS is scarce. To the best of our knowledge, the present study is the first meta-analysis evaluating the risk of OSA on adverse cardiovascular events in patients with newly diagnosed ACS. Most prior studies 6,7,16 -18,20 have shown that the presence of OSA is associated with a greater risk of cardiovascular events in patients with ACS. Nevertheless, a single newly published ISAACC study 8 in 2019, which was a large-scale, multicenter, high-quality study, contradicted this, and also found that treatment with CPAP did not significantly reduce this risk. However, it is probably premature to draw a conclusion of neutral outcomes due to the following reasons: First, the ISAACC trial only included nonsleepy OSA patients (EES ≤ 10) with a better prognosis compared to patients with excessive daytime sleepiness (EES >10), which inevitably underestimates the risk of OSA on cardiovascular outcomes. Second, period interval with 48 and 72 hours between admission to the start of the sleep study is relatively short, compared with other similar studies. Schiza et al 24 found that the high prevalence of OSA within 72 hour of ACS did not persist 6 months later, and OSA may be transient and related with the acute phase of the serious disease. So, early detection may mistakenly categorize non-OSA patients into OSA patients and underestimate the effect of OSA. Third, the definition of non-OSA is based on AHI (ie, AHI <15/h), which causes the reference group to actually include mild OSA (ie, AHI with 5-15/h). Hence, the influence of OSA on cardiovascular outcomes may be devalued in patients with ACS. Currently, all large-scale randomized clinical trials 25,26 related to CPAP treatment in nonsleepy OSA patients do not achieve the desired result. Future investigation needs to be conducted in which subsets among nonsleepy OSA patients have a worse prognosis and can benefit from CPAP treatment.
The potential mechanisms that cause OSA to increase the prevalence of cardiovascular events in patients with ACS remain complicated. Obstructive sleep apnea, which results in nocturnal intermittent hypoxic-reoxygenation cycle, can trigger endothelial dysfunction, sympathetic activation, and oxidative stress, subsequently leading to the formation and development of atherosclerotic plaques. 27 ApoE(−/−) mice exposed to intermittent hypoxic environment exhibit structural vascular remodeling of the aorta that is characterized by increased intima-media thickness. 28 Similarly, patients with OSA show a larger total atheroma volume compared to non-OSA patients through intravascular ultrasound equipment. 29 In addition, the presence of OSA increased the incidence of in-stent restenosis in patients with ACS, 8 which is similar to our meta-analysis, indicating the relationship of OSA with increased risk of repeat revascularization.
This study has several limitations that should be noted. First, we observed significant evidence of heterogeneity that is mainly derived from a large-scale, multicenter ISAACC study that recruits patients without daytime sleepiness and may underestimate the effect of OSA on cardiovascular events in patients with ACS. Second, the pooled results represent available data from observational studies that are able to describe the relationship of OSA with cardiovascular outcomes. However, they are still relatively small sample size and few events have been reported, which may influence the efficacy of this conclusion. Third, we have observed some evidence of publication bias for the primary outcome. Nevertheless, the estimate size and direction do not significantly change after performing the trim-and-fill analysis. Fourth, there are relatively few recorded individual cardiovascular events among included studies, and more data may reveal positive outcomes in secondary outcome including all-cause death and stroke. Fifth, the definition of OSA and cardiovascular events is inconsistent across studies, which is in line with most meta-analyses related to OSA, and has been resolved, at least to a certain degree, by conducting a sensitivity analysis.
Conclusion
Our meta-analysis shows that preexisting OSA is associated with increased risk of subsequent MACCE, repeat revascularization, and hospitalization for heart failure in patients with newly diagnosed ACS. Our results should be interpreted with caution, given potential heterogeneity across studies and potential publication bias. Further studies should focus on elucidating specific subgroup and severity of OSA, which can achieve benefit from CPAP treatment.
Supplemental Material
Supplemental Material, sj-pdf-1-ear-10.1177_0145561321989450 - Association of Obstructive Sleep Apnea With the Risk of Repeat Adverse Cardiovascular Events in Patients With Newly Diagnosed Acute Coronary Syndrome: A Systematic Review and Meta-Analysis
Supplemental Material, sj-pdf-1-ear-10.1177_0145561321989450 for Association of Obstructive Sleep Apnea With the Risk of Repeat Adverse Cardiovascular Events in Patients With Newly Diagnosed Acute Coronary Syndrome: A Systematic Review and Meta-Analysis by Shu-Han Yang, Yong-Sheng Xing, Zeng-Xia Wang, Yan-Bin Liu, Hong-Wei Chen, Yan-Feng Ren, Jing-Ling Chen, Sheng-Bo Li and Zhi-Fang Wang in Ear, Nose & Throat Journal
Footnotes
Authors’ Note
This article does not contain any studies with human participants performed by any of the authors. Study concept and design: Shu-han Yang, Hong-wei Cheng.
Acquisitions and analysis of data: Zeng-xia Wang, Yan-bin Liu. Statistical analysis: Yan-feng Ren, Sheng-bo Li, Jing-ling Chen. Methodology: Shu-han Yang.
Original draft of manuscript: Shu-han Yang, Zeng-xia Wang. Review and revision: Yong-sheng Xing. Supervision: Zhi-fang Wang. All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
