Abstract
Background:
Previous studies revealed that the prothrombotic factors in patients with obstructive sleep apnea (OSA) remain controversial.
Aim/Objective:
The aim of the systematic review is to elucidate the relationship between prothrombotic factors and OSA.
Materials and Methods:
This systematic review was performed under the Preferred Reporting Items for Systematic Reviews and Meta-Analysis guidelines. The literature we investigated was extracted from 4 main medical databases (PubMed, Web of Science, Cochrane Library, and Chinese databases) as of February 2020. We used significant weighted mean differences (SMDs) with 95% CIs from random-effects model.
Results:
A total of 15 studies comprising 2190 patients were available for the meta-analysis. The pooled results showed that the levels of fibrinogen (SMD = 0.95, 95% CI = 0.26 to 1.65, P = .000), vascular endothelial growth factor (SMD = 0.37, 95% CI = −0.90 to 1.63, P = .000), and plasminogen activator inhibitor 1 (SMD = 0.61, 95% CI = 0.29 to 0.92, P = .040) increased in patients with OSA. There were no statistical differences between groups in terms of
Conclusions and Significance:
This meta-analysis indicated the relationship between prothrombotic factors in OSA hypopnea. Obstructive sleep apnea–related effects may underline the importance of considering the dysfunction of the hemostatic system. The prothrombotic factors in OSA can influence making a choice of appropriate therapy.
Introduction
Obstructive sleep apnea (OSA) is a common sleep disorder, which was characterized by the repetitive collapse of the upper airway during sleep, resulting in sleep fragmentation and periods of hypoxia. It is reported that the epidemiology of OSA is conservatively estimated to be 3% among women and 10% among men aged 30 to 49 years and 9% among women and 17% among men aged 50 to 70 years. 1 The major clinical manifestations of OSA were snoring, daytime sleepiness, frequent episodes of sleep fragmentation, intermittent hypoxia, hypercapnia, swings in the intrathoracic pressure, and increased sympathetic activity.2,3 There are growing evidences suggesting that OSA is a chronic, low-grade inflammatory condition,4-6 and inflammatory cytokines such as C-reactive protein, tumor necrosis factor α, interleukin 6 (IL-6), and interferon γ were higher in OSA. 7
Besides increased oxidative stress and activation and propagation of inflammatory pathways in OSA, several studies also have shown a change in blood coagulation in OSA.8,9 Repeated episodes of nocturnal hypoxia and endothelial injury may promote changes in hemostasis, such as platelet activation, fibrinogen, and clotting factors, which result in a prothrombotic state.10-14 The changes in prothrombotic factors may damage vascular endothelial dysfunction, disturb the coagulation system, and lead to the development of metabolic dysfunction, atherosclerosis, and cardiovascular diseases.15-18
However, there remains some uncertainty about the consistency of the prothrombotic factors in OSA since reports on unchanged prothrombotic markers do also exist.8,10 Therefore, the present study aims to assess several hemostatic parameters in patients with OSA on which a systematic review with meta-analysis was subsequently performed.
Materials and Methods
Search Strategy
We searched previous studies on PubMed, Web of Science, Cochrane Library, WANFANG (Chinese database), VIP (Chinese Database), and Chinese National Knowledge Infrastructure (by February 2020). Keywords and search strategy were as follows: Obstructive Sleep Apneas OR Obstructive Sleep Apnea Syndrome OR Obstructive Sleep Apnea OR OSAHS OR OSAS OR Sleep Apnea Hypopnea Syndrome OR Upper Airway Resistance Sleep Apnea Syndrome OR sleep-disordered breathing combined with prothrombotic OR thrombophilias OR thrombin OR throm* OR clotting OR hemostatic OR hemostasis OR hemosta* OR coagulability OR procoagulant OR hypercoagul* OR coagul*. We collected data from fully published articles written in English and Chinese. The meta-analysis strictly complies with Preferred Reporting Items for Systematic Reviews and Meta-Analysis statement. 19
Study Selection
The articles were reviewed by 2 reviewers independently. Any disagreements were resolved by discussion.
Inclusion and Exclusion Criteria of the Literature
The inclusion criteria were as follows: (1) the study must have included the individuals with OSA and the other consisting of individuals without OSA, (2) the individuals with OSA must have undergone polysomnographic monitoring to document the apnea index, and (3) data on both OSA and controls were based on means and SDs or medians and ranges.
The exclusion criteria were as follows: (1) the participants who have complications such as severe cardiovascular diseases, stroke, metabolism syndrome, cerebral infarction, hematological diseases, and various acute infectious diseases and (2) editorials, letters to the editor, conference abstracts, reviews, case reports, and animal experimental studies.
Data Extraction
The information was retrieved from each included study: first author’s name, year of publication, design of the study, sample size, mean age, study population, numbers of each apnea–hypopnea index (AHI) level, and Newcastle-Ottawa Scale (NOS) scores. Two authors independently evaluated all the final included studies according to the inclusion criteria described above and any disagreements were discussed.
Quality Assessment
The methodological quality of the included studies was evaluated using the NOS by 2 authors independently. Any disagreements were resolved via consensus or third reviewer when necessary. Scores ranged from 0 to 9 were allocated to each study: patient selection, comparability of the groups, and assessment of outcomes of interest. Relevant details were shown in Table 1.
Characteristics of Included Studies in the Meta-Analysis.
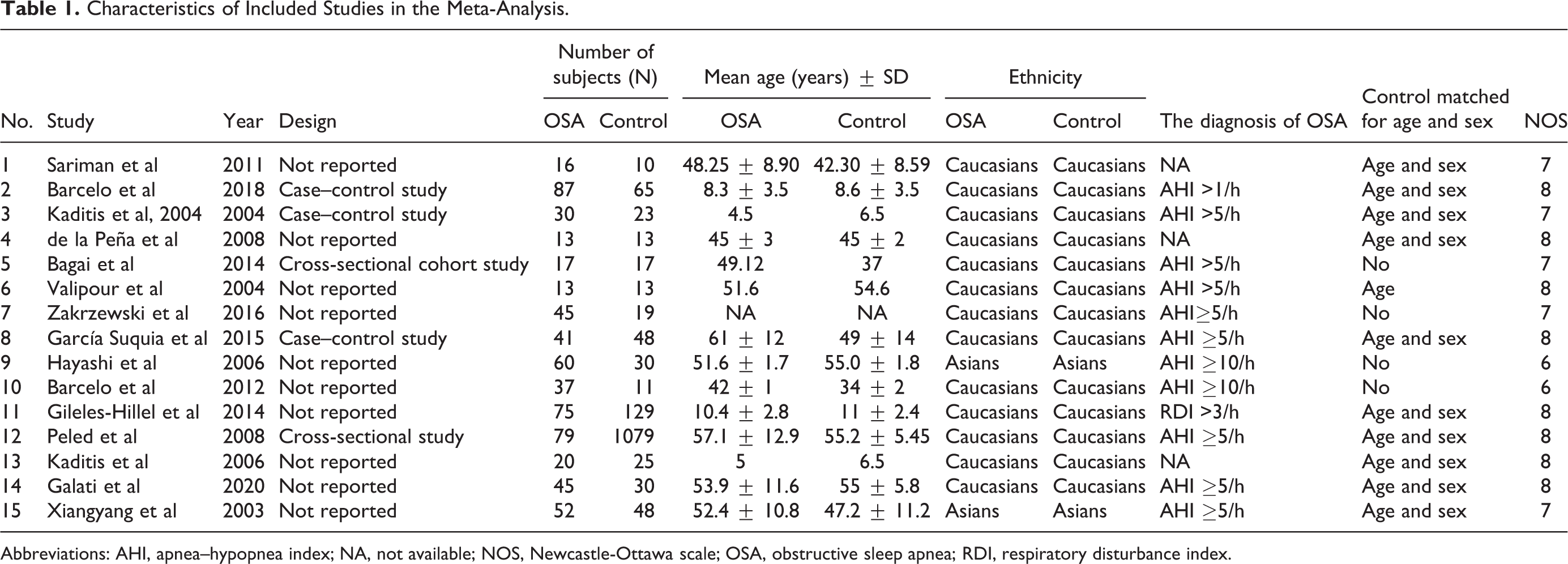
Abbreviations: AHI, apnea–hypopnea index; NA, not available; NOS, Newcastle-Ottawa scale; OSA, obstructive sleep apnea; RDI, respiratory disturbance index.
Statistical Analysis
Weighted standardized mean difference (SMD) with 95% CI was used for the meta-analysis to compare continuous variables. The I2 test was performed to quantify the heterogeneity of SMDs across the studies. Heterogeneity was defined as low, moderate, and high according to an I2 of 25% to 49%, 50% to 74%, and 75% to 100%. If a study only reported median (Q25-Q75), then the data were converted to mean and SD according to the available documents. A random-effects model was used because of the expected variability in the studies. Subgroup analysis or sensitivity analysis would be conducted according to the methodological and clinical heterogeneity. The statistical analysis was performed with STATA (12.0). A P value of <.05 was considered to be statistically significant.
Results
Search Result
We obtained a total of 726 relevant articles from the database. After removing duplicates and reviewing by titles and abstracts, 682 articles were excluded. The remaining 44 articles were full text reviewed for eligibility. Twenty-nine articles couldn’t be extracted and the data are based on means and standard deviations. Finally, a total of 15 articles met the inclusion criteria and were pooled for this meta-analysis. A flow diagram of the study selection process is presented in Figure 1.
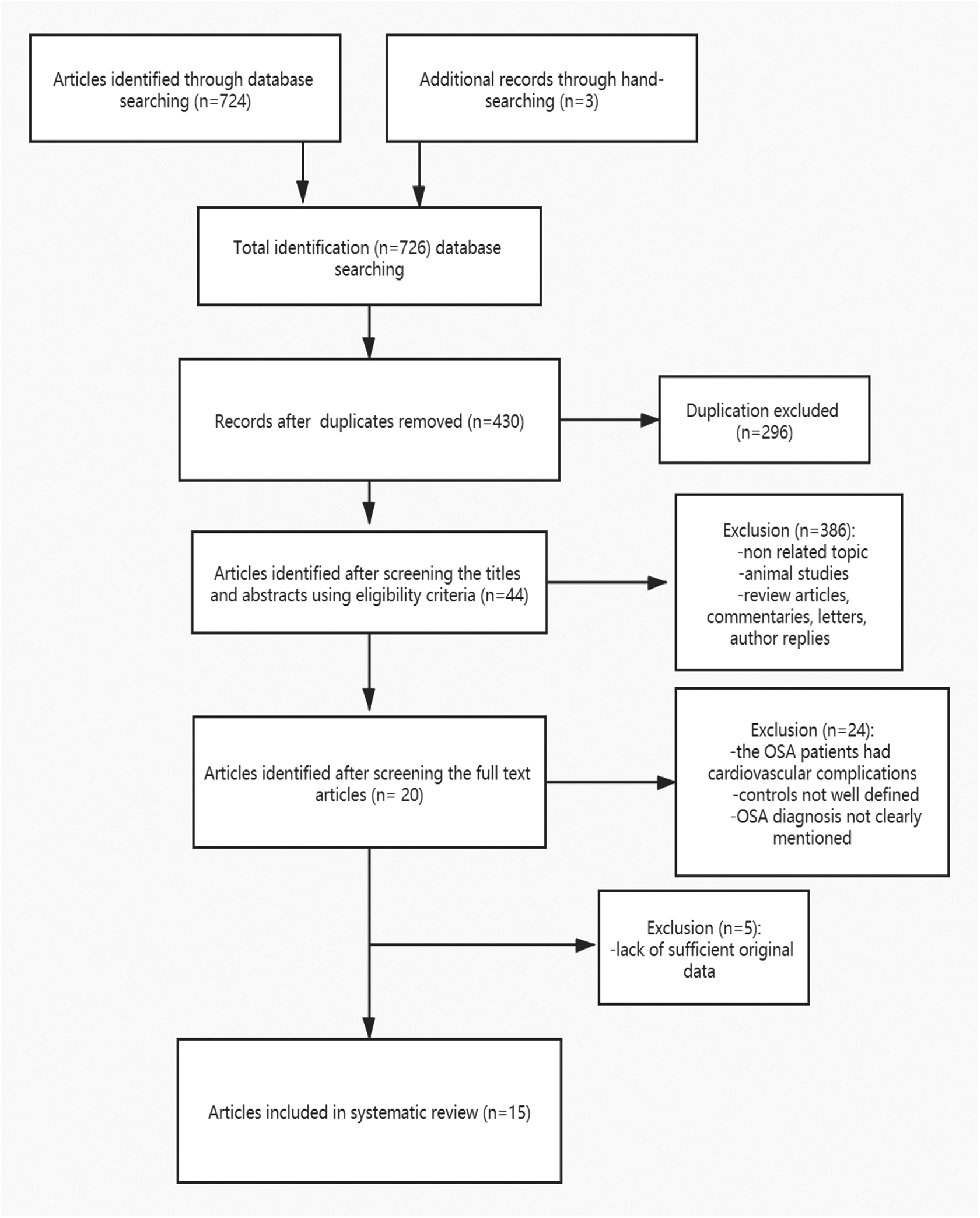
Flowchart of studies assessed for eligibility at each screening stage.
Characteristics of the Eligible Studies
We included 15 studies containing 2190 participants7,14,20-32 for our systematic review and meta-analysis (Table 2). The information of the first author’s name, year of publication, design of the study, sample size, mean age, study population, numbers of each AHI level, and NOS score of each study are shown in Table 1. Age- and sex-matched controls were used in 10 studies.
Summary of the Selected Studies of the Association Between OSA and Prothrombotic Factors.

Abbreviations: BH, bronchial hyperreactivity; DCs, circulating dendritic cell; EPCs, endothelial progenitor cells; PAI-1, plasminogen activator inhibitor 1; SDB, sleep-disordered breathing; t-PA, tissue-type plasminogen activator; VEGF, vascular endothelial growth factor.
Outcomes of Forest Plots and Meta-Analysis
We conducted 5 forest plots in relation to the following outcomes (fibrinogen, vascular endothelial growth factor [VEGF], plasminogen activator inhibitor 1 [PAI-1],
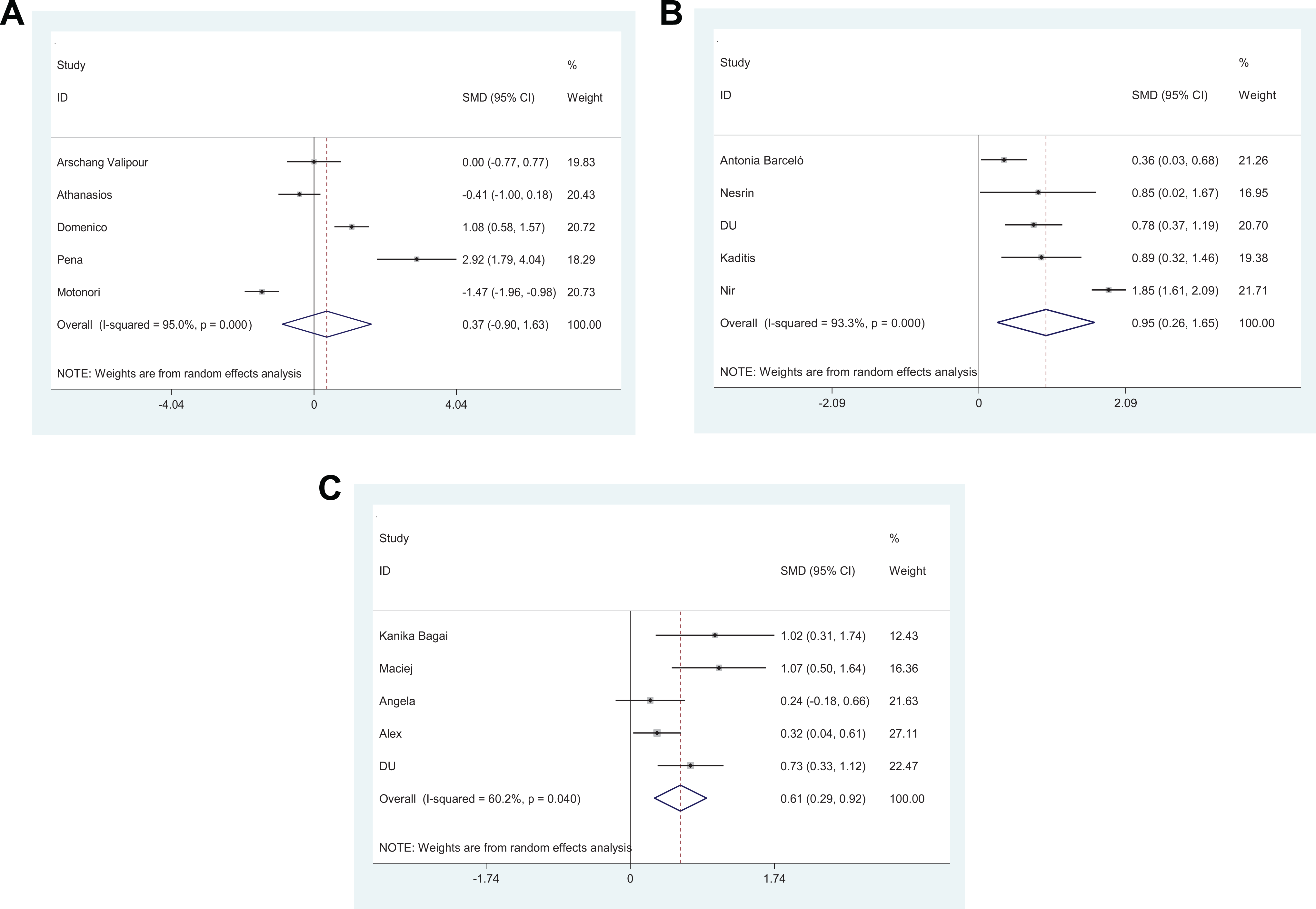
Forest plots of studies on prothrombotic factors for patients with obstructive sleep apnea (OSA) versus controls. A, Vascular endothelial growth factor (VEGF). B, Fibrinogen. C, Plasminogen activator inhibitor 1 (PAI-1).
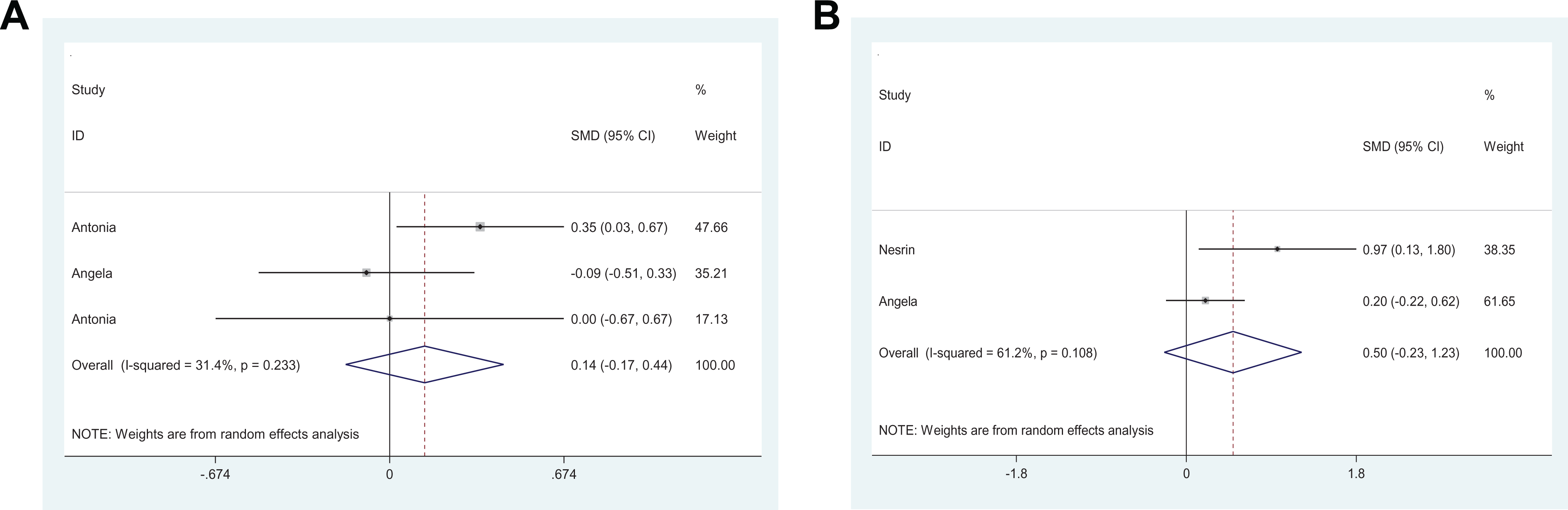
Forest plots of studies on prothrombotic factors for patients with obstructive sleep apnea (OSA) versus controls. A, Platelet counts. B,
Subgroup Analysis
Considering the high heterogeneity of the fibrinogen (I2 = 93.3%), PAI-1 (I2 = 60.2%), and VEGF (I2 = 95%), subgroup analysis was conducted according to the specimen types (plasma or serum) and age.
As for fibrinogen, subgroup differences in specimen types and age were observed. The SMD of plasma was 1.12 (95% CI = 0.45-1.80; I2 = 88.8%; P = .000), while the value of serum was 0.36 (95% CI = 0.03-0.68). When subgrouping based on age, the SMD of child was 0.57 (95% CI = 0.06-1.08; I2 = 60.3%; P = .112), while the value was 1.19 (95% CI = 0.36-2.03; I2 = 91.1%; P = .000) for adult (Figure 4).
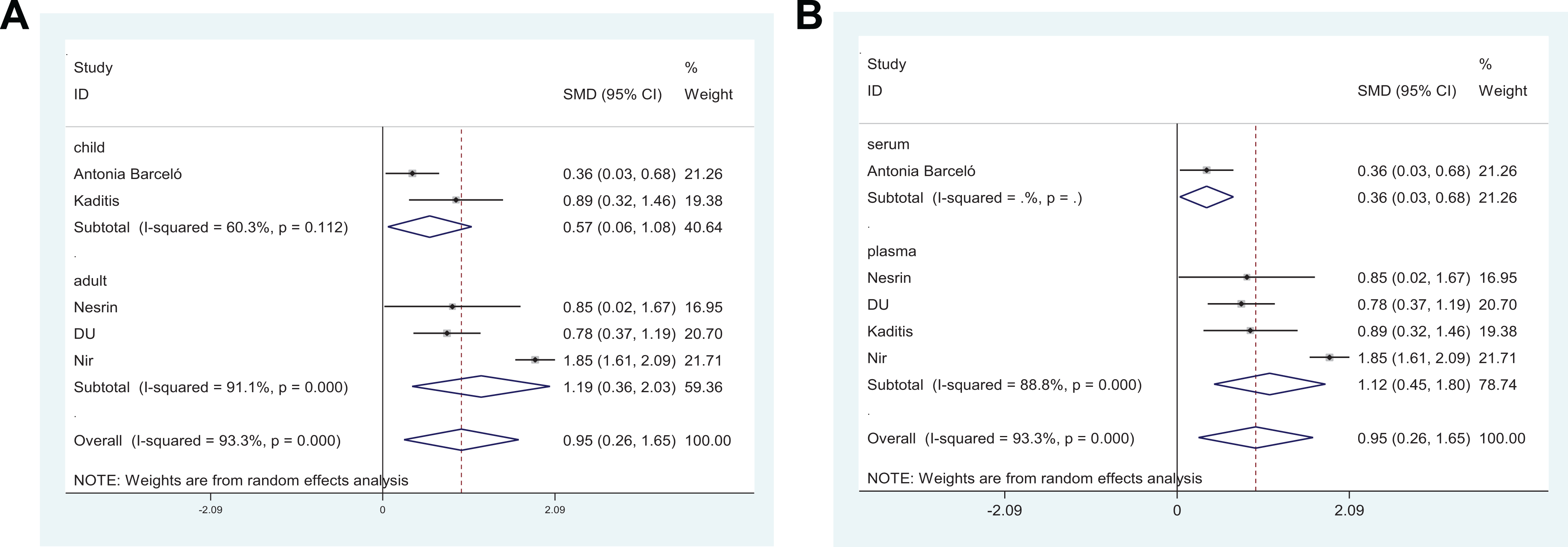
Subgroup analysis of fibrinogen levels in patients with obstructive sleep apnea (OSA) and controls. A, Age. B, Sample type.
In the subgroup analysis of VEGF based on specimen types, the SMD of plasma was 0.18 (95% CI = −1.25 to 1.62; I2 = 94.2%; P = .000), while the value of serum was 1.08 (95% CI = 0.58 to 1.57). Subgroup analyses by age showed no significant difference (Figure 5).

Subgroup analysis of vascular endothelial growth factor (VEGF) levels in patients with obstructive sleep apnea (OSA) and controls. A, Age. B, Sample type.
According to PAI-1, the specimen types were plasma. Therefore, we conducted a subgroup analysis based on age. No differences were observed in the PAI-1 level when performing subgroup analyses based on age (Figure 6).
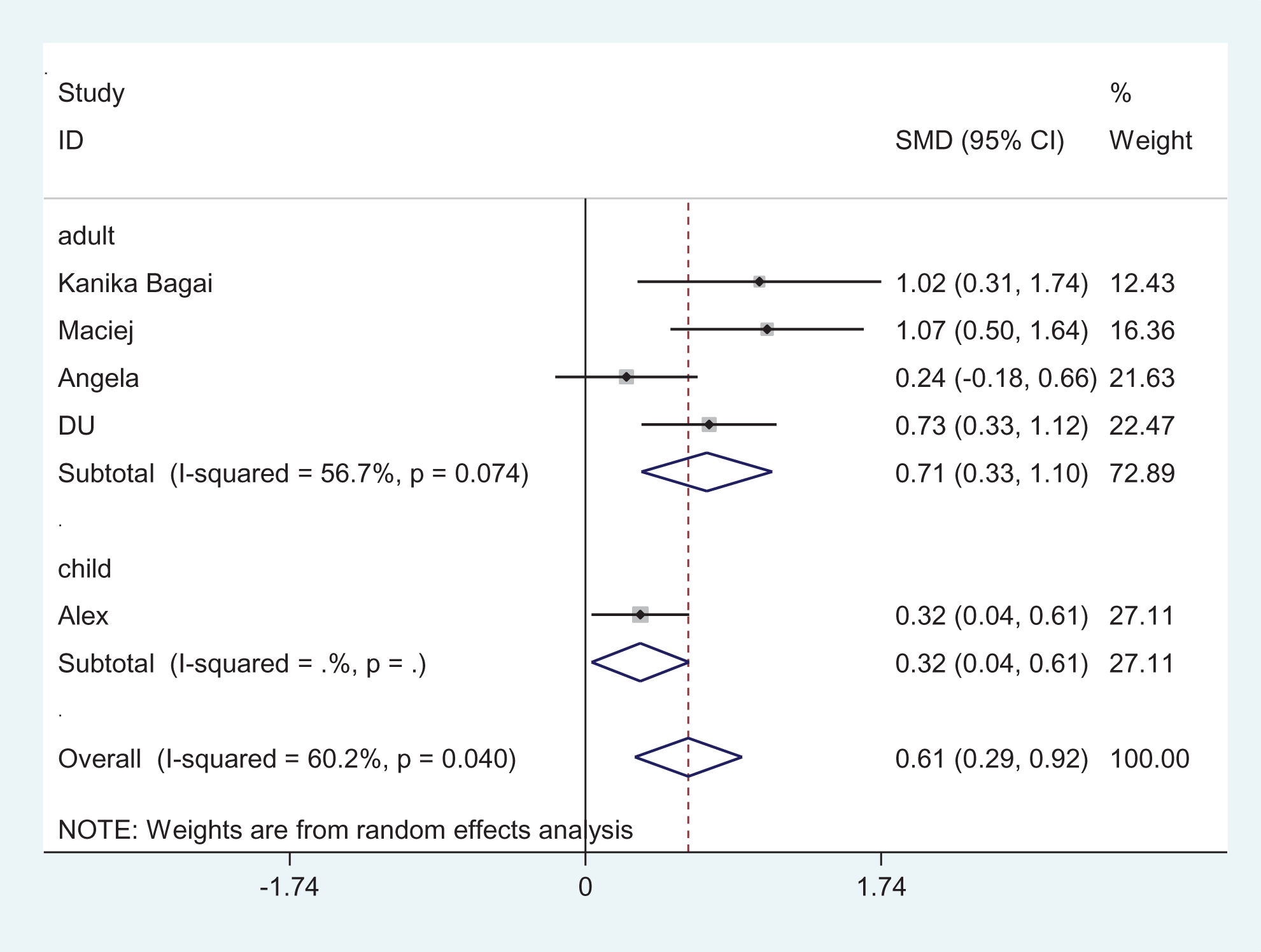
Subgroup analysis of plasminogen activator inhibitor 1 (PAI-1) levels in patients with obstructive sleep apnea (OSA) and controls (age).
Sensitivity Analysis
The stability of the results was assessed by the sensitivity analysis. The sensitivity analysis showed that removing any individual study, the pooled meta-analysis result did not fluctuate from statistical significance to nonstatistical significance.
Discussion
By screening the literature according to uniform criteria, we mainly focused on these 5 categories: fibrinogen, VEGF, PAI-1,
Fibrinogen is a 340-kDa hexameric plasma glycoprotein synthesized by the liver, which is the major structural component of a clot. 33 It is converted by thrombin to fibrin and subsequently to a fibrin-based blood clot when the vascular endothelial cells are damaged. Fibrin can impair plasminogen activation by binding of inhibitors; mediate blood platelet, tissue fibroblast proliferation, and capillary tube formation; and thereby function to promote tissue revascularization. 34
Our results showed that patients with OSA had significantly higher plasma fibrinogen levels compared with controls (SMD = 0.95, 95% CI = 0.26-1.65, P = .000). Chin et al reported that the plasma fibrinogen and hematocrit levels in the morning were significantly higher than on the previous afternoon before nCPAP treatment, 35 while increases in the plasma fibrinogen concentration and the blood viscosity in the morning disappeared after nCPAP treatment. von Känel et al claimed that patients with OSA had elevated plasma fibrinogen levels as compared to controls, 16 and average minimal oxygen saturation was found as an independent predictor of fibrinogen levels. Omer et al indicated that higher fibrinogen to albumin ratio may be predictive of inflammation in patients with moderate-to-severe sleep apnea. 36 These previous studies could imply that intermittent hypoxia experienced by OSA patients can induce endothelial injury and this in turn may initiate the increase in fibrinogen levels.
Vascular endothelial growth factor, a cytokine with potent angiogenic properties, can induce proliferation of endothelial cells and modulate thrombogenicity. 37 It may contribute to the process of cardiovascular diseases 38 and may also influence the prognosis of cancer. 39 Lavie et al have found higher VEGF levels in patients with OSA than controls and a reduction in VEGF concentrations with continuous positive airway pressure (CPAP) therapy in a subgroup of patients. 40 A meta-analysis has analyzed the differences in VEGF levels between OSA and controls, indicating that blood VEGF was significantly higher in patients with OSA, especially in those aged over 50 years and AHI ≥30 events/hour, 41 but this meta-analysis didn’t exclude the patients who had cardiovascular complications. Our meta-analysis included 5 relevant studies involving 262 patients, revealing that the level of VEGF was higher in OSA (SMD = 0.37, 95% CI = −0.9 to 1.63, P = .000), which was consistent with the previous studies. Chronic IH is the pivotal physiological characteristic of OSA. The increased expression of VEGF gene occurs following hypoxia via the mediation of hypoxia-inducible factor (HIF). 42 In addition to hypoxia-associated stimuli for the VEGF, several pro-inflammatory cytokines such as IL-6 and TNF-α can also potentate VEGF production. 43 As mentioned above, OSA is a low-grade inflammatory condition. The pro-inflammatory factors via OSA may induce the mechanism for the induction of VEGF.
Plasminogen activator inhibitor 1, a principal physiologic inhibitor of the body’s fibrinolytic system, acts as a suicide substrate for tissue-type plasminogen activator (t-PA). In controls, PAI-1 circulates briefly in plasma through an active form (activity); only a fraction of the secreted active PAI-1 has the chance to react with plasma t-PA to form inert covalent complexes. 26 Increases in PAI-1 in plasma are believed to promote pathophysiology of endothelial dysfunction and atherothrombosis. 44
Numerous original studies confirmed that high levels of blood PAI-1 are observed in OSA population,25-27 which were consistent with our results (SMD = 0.61, 95% CI = 0.29-0.92, P = .040). von Känel et al evaluated the concentration of PAI-1 in the blood serum of 44 patients with OSA. It was reported that AHI was the major predictor positively correlated with PAI-1 independent of others and PAI-1 levels decreased significantly after a 2-week treatment with CPAP. 16 The underlying mechanism responsible for the increased PAI-1 levels in patients with OSA could be due to intermittent hypoxia. Several research studies concluded that HIF-1α transcription factors could drive transcription of PAI-1.45,46
Because of the high heterogeneity, we conducted the subgroup analyses in terms of specimen types and age. According to the fibrinogen, we speculated the possible sources of heterogeneity were specimen types and age. No differences were observed in VEGF levels when performing subgroup analyses based on age, and the possible sources of heterogeneity were specimen types. The heterogeneity of each subgroup decreased but was not obvious after subgroup analyses. As for the PAI-1, we didn’t find the possible heterogeneity due to small samples. It requires further large-scale study to classify whether specimen and age may influence these prothrombotic factors among patients with OSA.
Our meta-analysis showed that no statistical differences were observed between groups in terms of
Platelets are circulating, anucleate, disc-shaped cells, whose main role is to maintain the integrity of blood vessels through adequate hemostasis. 50 Varol et al have found that the mean platelet volume values of patients with severe OSA were significantly higher than those of the controls.51,52 Regulation of platelet size is multifactorial; studies have reported that increases in platelet volume are often associated with decreases in platelet count,50,51 and hypoxia is likely to decrease the platelet by regulating mean platelet volume. However, several studies indicated that platelet count and volume may be regulated by independent mechanisms,53,54 which need to be further studied.
The conflicting results in
As we can see, hypoxia plays an important role in the prothrombotic factors with OSA. Intermittent hypoxia from OSA could modulate the levels of a number of mechanisms such as oxidative stress, nitric oxide activity, or other pathophysiological processes influencing the mobilization of vascular endothelial progenitor cells, 24 which promote the prothrombotic factors. Meanwhile, repetitive arousals from sleep could augment sympathetic activity, tilt the balance toward a heightened pro-inflammatory state,7,23 and further increase the hypercoagulability.
The study of this relationship showed the levels of fibrinogen, VEGF, and PAI-1 increased in patients with OSA. Elevated prothrombotic factors may due to chronic hypoxemia in patients with OSA and indirectly correlated with cardiovascular complications. A procoagulant state could contribute to the functional disruption of the endothelium and induce atherosclerotic plaques. 55 To study this relationship can further understand the clinical significance of changes in blood function, help to the prevention of OSA-related cardiovascular diseases.
Limitations
It has to be pointed out that this study is hardly free of limitations. First, the number of included studies and their sample size were relatively small. Large databases were warranted to be able to pool individuals. Second, although we have excluded patients with a prior history of severe cardiovascular diseases and hematological diseases, the possibility of an occult disease that would have contributed to the increased propensity of coagulation cannot be completely ruled out. Clinical diversity may bring about statistical heterogeneity. Third, notwithstanding 11 countries mainly distributing in Asia and Europe were covered in the 15 studies; the ethnic groups in our study were mostly Caucasians, and the result may more suitable for Caucasians. Although mainly focused on the Caucasian population, the included literatures were screened according to uniform criteria, which are objective. Fourth, according to our including literatures, hypoxia level and oxygen saturation of patients with OSA are not specified by the time spent <90% of O2 saturation and the severity of OSA is not specified by the AHI due to the variability in the criteria that are used for diagnoses, which may bring potential sources of heterogeneity. Future research should validate these sources of heterogeneity in a larger data set. Finally, meta-analysis is a retrospective research, which contains the methodological deficiencies of the included studies.
Conclusion
Taken together, our meta-analysis indicated that OSA-related effects may contribute to an enhanced coagulability. Increased levels of fibrinogen, PAI-1, and VEGF were the most significantly affected variables in patients with OSA. Further studies are needed to identify this relationship and the potential mechanism for developing cardiovascular diseases.
Footnotes
Authors’ Note
This study was a meta-analysis and did not concern with human participants performed by any of the authors.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Capital Funds for Health Improvement and Research (2018-1-2091), Pediatric Medical Coordinated Development Center of Beijing Municipal Administration (XTZD20180101), Beijing Natural Science Foundation (7194262), National Natural Science Foundation of China (81970900), and Beijing Hospitals Authority’ Ascent Plan (DFL20191201).
