Abstract

Keywords
Significance Statement
Granulomatosis with polyangiitis (GPA), formerly called Wegener’s granulomatosis, is a rare autoimmune disease that commonly affects the head and neck area. Only 6% of patients complain of hearing loss as initial symptom, while 5% have facial nerve palsy (VII). Multiple cranial nerves involvement is even rarer. We present the first clinical report in the literature that combines involvement of V3, VII, VIII and IX cranial nerves, along with pseudotumoural nasopharyngeal lesion and pulmonary infiltrations.
A 74-year-old woman was referred to our Ear, Nose and Throat (ENT) department with a 3-day history of left facial nerve palsy. She also mentioned a 2-month history of cough and sharp pulses of pain at her throat and inside the mouth, that were triggered by chewing (glossopharyngeal neuralgia). She complained about left ear pain, followed by bilateral hearing impairment, mostly at the left ear one month ago. Clinical examination showed left facial nerve palsy (House Brackmann IV) and hypoesthesia at the skin below mouth ipsilaterally (Figure 1). Neck palpation was normal. Otomicroscopy revealed cloudy but intact tympanic membranes, while flexible endoscopy showed mild inflammation and swelling at the left nasopharyngeal area. Pure tone audiometry demonstrated severe bilateral sensorineural hearing loss, especially at the left ear with deafness at 125, 250, 500, 4000 and 8000 Hz. Blood test presented elevated inflammatory markers, namely c-reactive protein (CRP) = 30 mg/dL, erythrocyte sedimentation rate (ESR) = 126 mm and high white blood cell count (14.000/μL). Image showing left facial nerve palsy of the patient.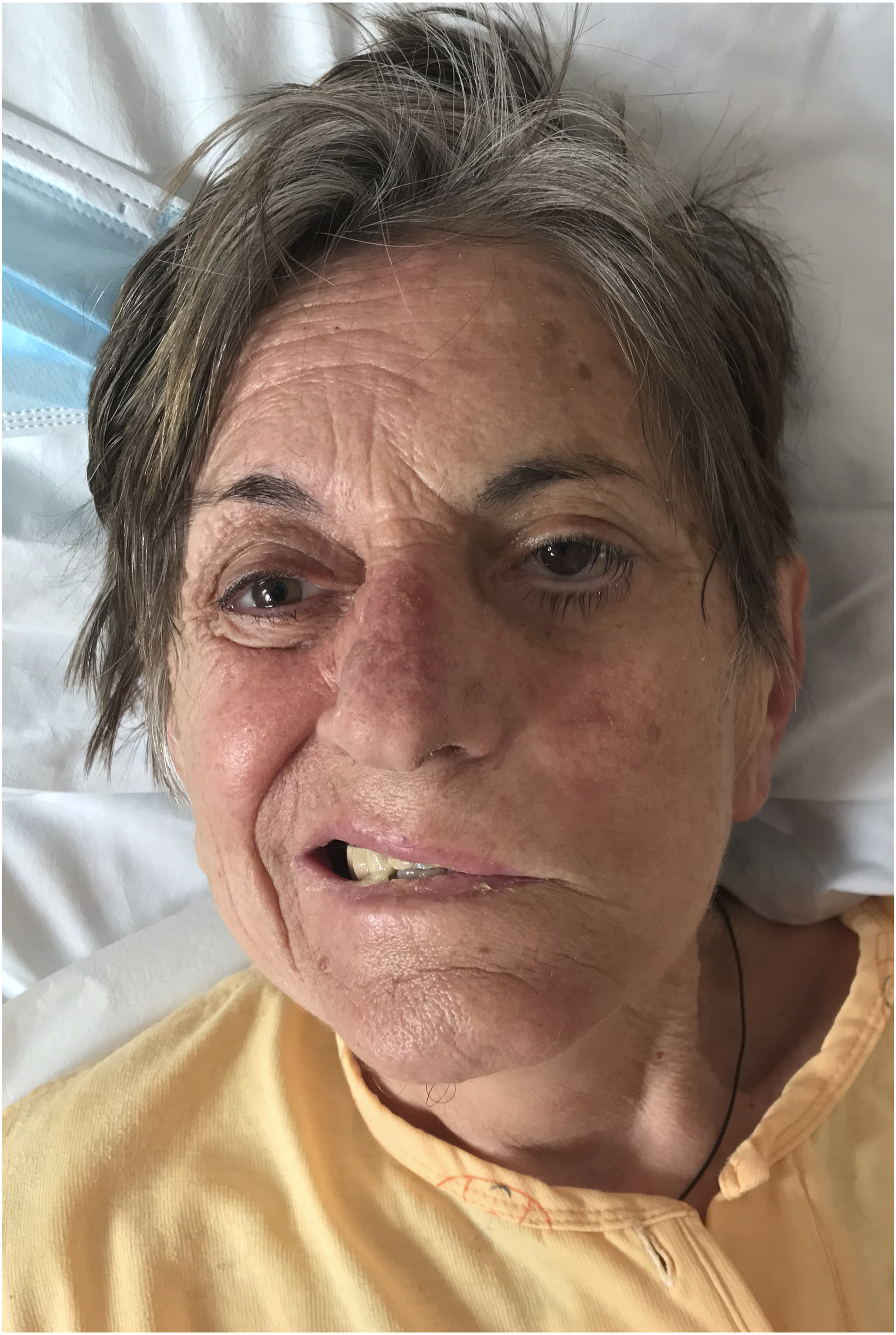
Brain, facial bones and skull base magnetic resonance imaging (MRI) and computed tomography (CT) were performed. MRI illustrated a mass at the left lateral wall of nasopharynx with extension to the lateral wall of oropharynx and parapharyngeal space (Figure 2A), perineural infiltration of the third branch of trigeminal nerve (V3) and incipient intracranial invasion through the foramen oval (Figure 2B). CT scan revealed thickening of left nasopharyngeal wall and absence of pneumatization of mastoid and tympanic cavities bilaterally. Moreover, a chest CT scan revealed a radiopaque mass measuring 4.5 × 3.3 × 2.3 cm at the right lower lobe along with scattered tuberous lesions bilaterally (Figure 2C). Magnetic resonance imaging (MRI) in axial plane showing (A) a mass at the left lateral wall of nasopharynx (blue arrows) with extension to the lateral wall of oropharynx and parapharyngeal space (T2 section); (B) perineural infiltration of the third branch of trigeminal nerve (yellow arrows) and incipient intracranial invasion through the foramen oval (Τ1 section). (C) Computed tomography (CT) in axial plane showing a radiopaque mass at the right lower lobe (green arrows) along with scattered tuberous lesions bilaterally.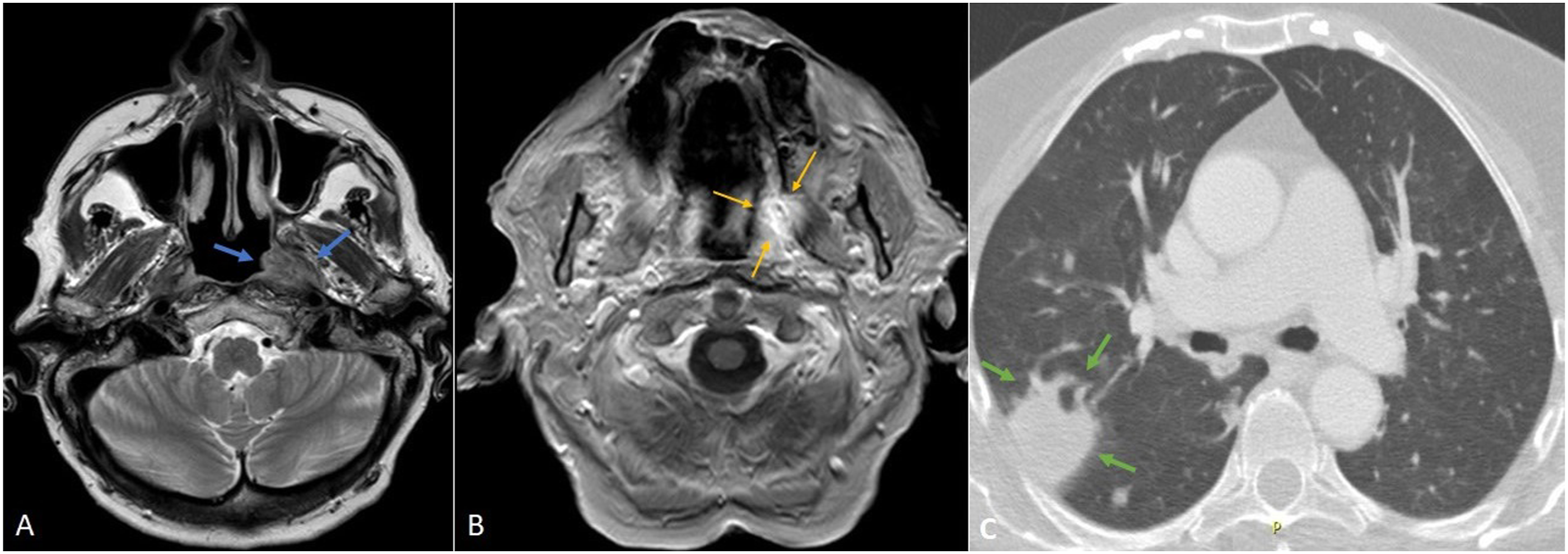
Biopsies from nasopharynx (torus tubarius and rossenmüller fossa) were taken, followed by ear tubes placement at both ears. Histopathological findings were inconclusive (Figure 3A), so CT-guided biopsy from the pulmonary mass at the right lobe was performed. Histopathology revealed necrotic granulomatous inflammation along with vasculitis (Figures 3B and 3C). Therefore, we limited our diagnoses to granulomatous diseases. Quantiferon test was negative, angiotensin-converting enzyme (ACE) normal, while cytoplasmic antineutrophil cytoplasmic antibodies (c-ANCA) were 1:20 and ANCA-proteinase 3 (PR3) 101.4 units. Diagnosis of GPA was confirmed. The patient took methylprednisone and cyclophosphamide and 10 days later blood test values normalized, with recession of neuralgia and cough. After 3 doses of cyclophosphamide VII palsy along with hearing improved prominently. She is currently in remission under regular surveillance from otolaryngologist and rheumatologist. Histopathological examination: (A) from nasopharynx showing focal inflammation inside lamina propria. Some multinucleated giant cells can be observed (H&E, ×10), (B) from lung showing locally dense inflammation consisting of lymphocytes, plasma cells, eosinophils, neutrophils and histiocytes with epitheliod-like morphology in a granuloma-resembling formation. Necrosis, in the form of nuclear debris centrally, as well as multinucleated giant cells are visible (H&E, ×20), and (C) with detailed view portraying acute inflammation of a small-sized blood vessel (red arrows) with neutrophil infiltrations through its wall and inside its lumen (H&E, ×40). H&E indicates hematoxylin and eosin.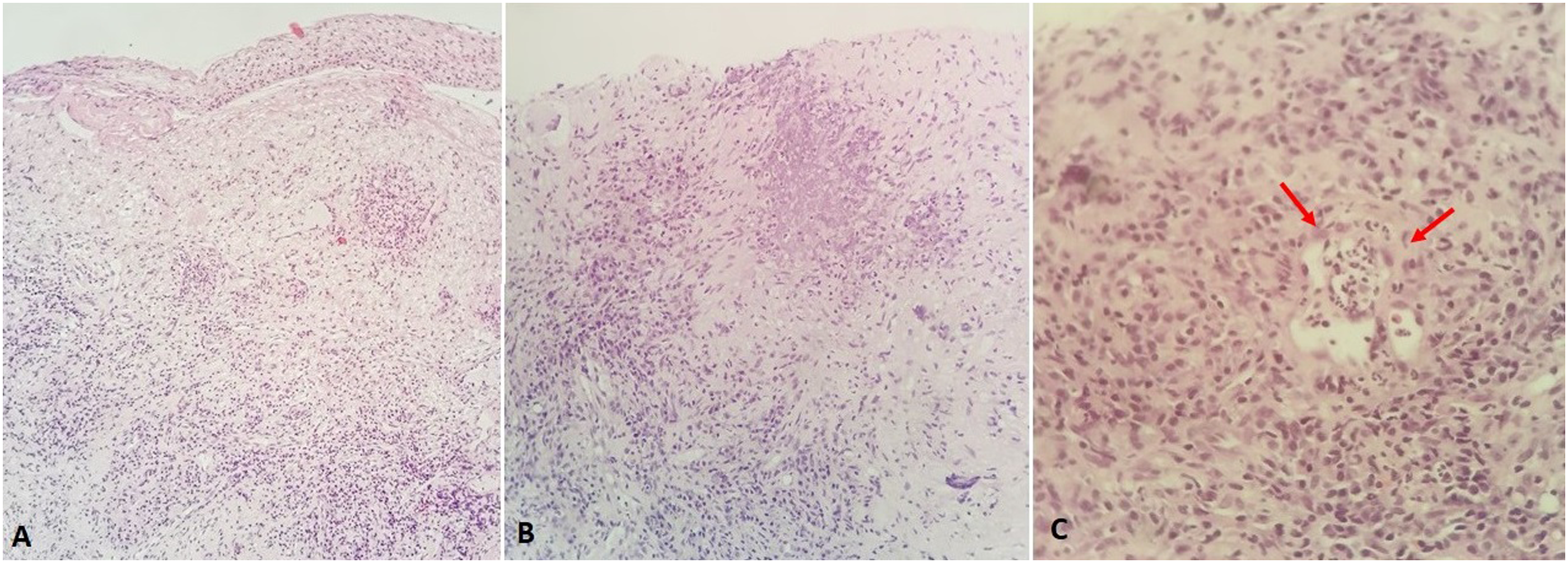
GPA is an autoimmune disease characterized by necrotizing granulomatous vasculitis of small to medium-sized vessels and mainly renal glomerulonephritis and upper and lower respiratory tract granulomas. It affects people of all ages, with prevalence of sixth to seventh decade, without sex predominance, with incidence of one to three cases per million.1,2 It reflects a broad clinical spectrum from limited to severe generalized disease. The former could involve only head and neck area, while the latter includes upper and lower respiratory tract and kidneys. 3 About 7 out of 10 cases with GPA entangle head and neck, with territorial symptoms being the initial and only that impel patients to seek help. 3 Prevailing nasal symptoms are obstruction, epistaxis, ulcerations, nasal septum perforation and saddle nose deformity. Concerning the ear, the most common symptoms are hearing loss (about 23%) due to conductive, mixed or sensorineural etiology, -much rarer as initial symptom (about 6%).4,5 From trachea, GPA can cause subglottic or tracheal stenosis, which can become life-threatening. 3 It can also cause ulcerations of the tongue and cheeks or “strawberry” gingival hyperplasia. 6 Central nervous system manifestations are rare affecting about 2-13% of patients. 7 Multiple cranial nerves dysfunction account for about 4.7%. 8 Involvement of V nerve affects about half of patients with central nervous system manifestations. 4 Facial nerve palsy is described around 5% because of necrotic angiitis of vasa nervorum, being rarer as initial symptom of GPA, while very few cases that involve IX nerve exist.4,9
Serological findings are supportive of the diagnosis and there is association with c-ANCA (ANCA-PR3). As it contains clinical manifestations involving many systems, diagnosis requires high clinical suspicion and multidisciplinary approach. All diagnostic systems agree on clinical or radiological evidence of inflammation of upper, lower respiratory tract and kidneys along with a positive biopsy for necrotic granulomatous inflammation and elevated ANCA.10,11
Differential diagnosis includes Churg-Strauss syndrome, sarcoidosis, tuberculosis, fungal or protozoal infections, lymphomas or other malignancies. 6
Treatment of choice is glucocorticoids and methotrexate for mild disease, while cyclophosphamide or rituximab is administered along with glucocorticoids for severe GPA. 12 It is worth mentioning that survival rates are 5 months mean survival for generalized untreated and 21.7 years median survival for treated GPA. 6
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Consent
We declare that written informed consent for patient information and images to be published was provided by the patient.
