Abstract
Schwannomas are benign tumors derived from the sheath of Schwann cells. Though it is common to see schwannomas in the head and neck region, auricular schwannomas are rare and only few cases have been reported. There are no distinguishing clinical findings or images; therefore, the histopathological diagnosis is mandatory. We describe a case of auricular schwannoma with clinical pictures and discuss the differential diagnoses according to histopathologic findings.
History
A 40-year-old female presented with a slow-growing left auricular mass for 12 months. She complained of progression in size with tenderness and denied bleeding, ulceration, tinnitus, aural fullness, hearing impairment, otalgia, otorrhea, vertigo, fever, and body weight loss. She denied history of trauma, operation, or systemic disease. Physical examination revealed a well-circumscribed nodule located at the left auricular concha cavum measuring 0.7 * 1 cm in size with telangiectasia and without obliteration of the external auditory canal (Figure 1). On palpation, the nodule was elastic, tender, and adherent to the underlying tissue. The external auditory canal was patent with normal appearance of the tympanic membrane. No other lesions were found on her face and neck. The well-circumscribed auricular nodule is elastic, tender, and adherent to the underlying tissue.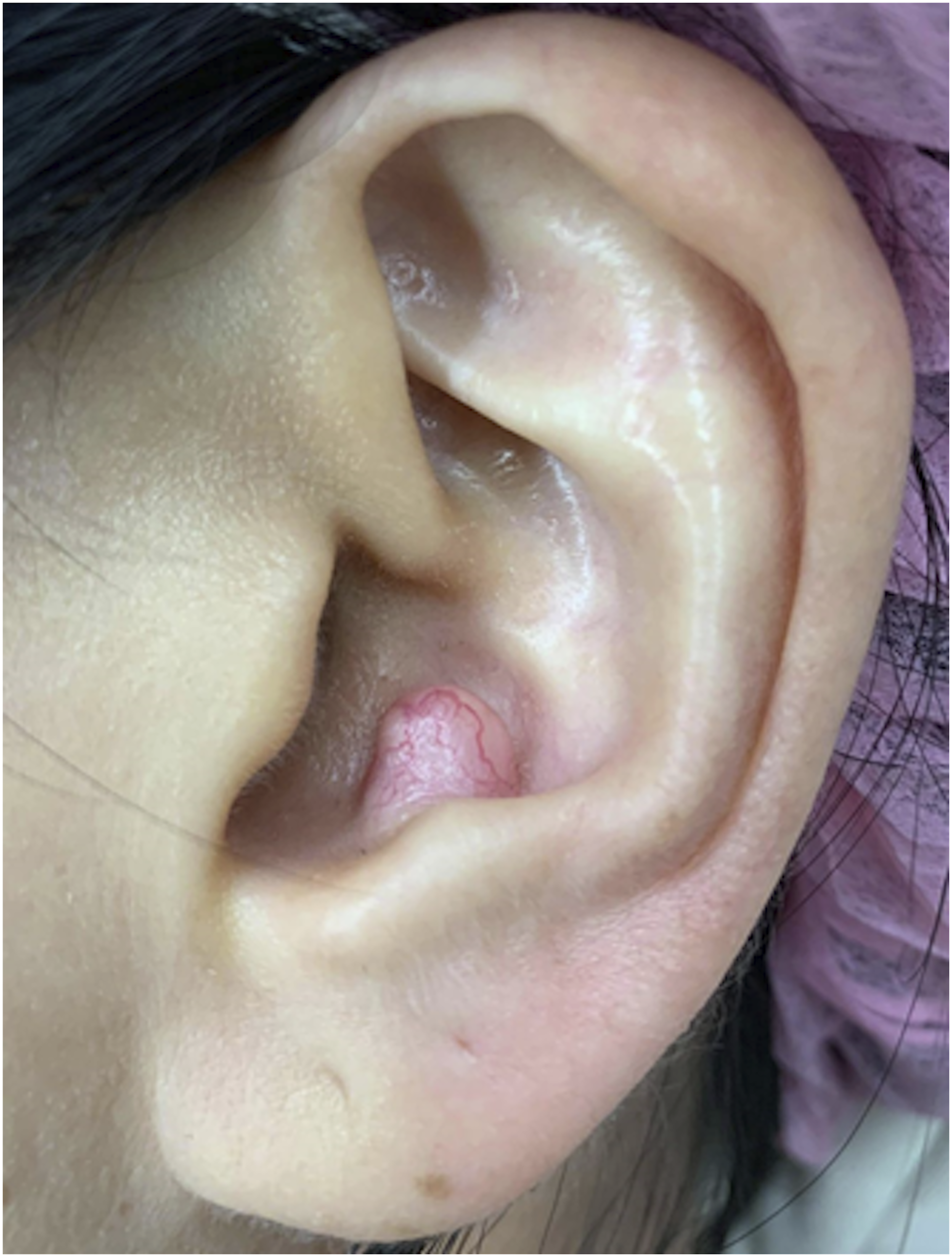
The patient underwent tumor excision under local anesthesia. Intraoperative findings showed a firm, well-encapsulated, and lobulated tumor.
Histopathological examination revealed the tumor to be composed of spindle cells with bland nuclei (Figure 2A), mostly arranged in the cellular palisading pattern as well as focally in looser hypocellular fascicles (Figure 2B and 2C). (A) Hematoxylin and eosin stain: Spindle cells arrange in fascicles with areas of nuclear palisading (×200 magnification). (B) Hematoxylin and eosin stain: Tumor showed nuclear palisading (arrow) (×400 magnification). (C) Hematoxylin and eosin stain: This tumor is composed of mostly hypercellular (Antoni-A) areas and focally (asterisk) hypocellular (Antoni-B) areas (×400 magnification). (D) Immunohistochemical stain for S-100 is positive (×200 magnification).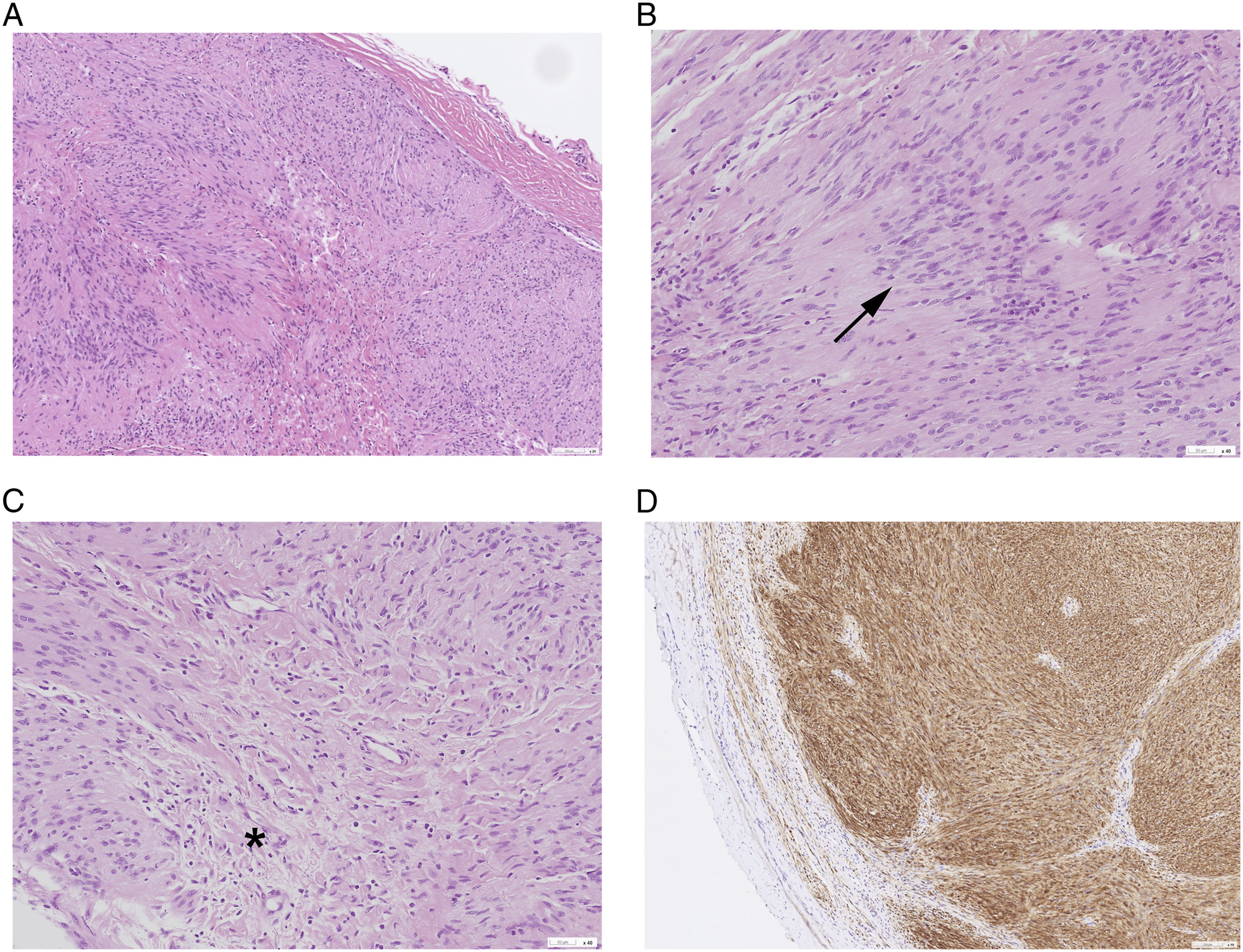
Immunohistochemical studies demonstrated diffuse and strong positivity for S-100 protein (Figure 2D) and SOX-10. CD34 immunostain was positive in a few scattered cells within the loose hypocellular areas, and EMA immunostain was faintly positive in a few cells within the peripheral capsule.
Discussion
Schwannomas generally display clinical features of benign, slow-growing and encapsulated tumors originating from the sheath of Schwann cells. 1 They are usually seen at the flexor surfaces of the extremities, neck, mediastinum, and retroperitoneum. 1,2 Schwannomas at head and neck regions account for 2540% of all schwannomas. 1,2 However, they are rare in the auricle, and the first case was reported by Fodor et al in 1977. 3 To date, only few cases of auricular schwannoma have been reported.
The auricle is innervated by the great auricular nerve, lesser occipital nerve, auriculotemporal nerve, and partly by the VIIth and Xth nerve. In the present case, we speculate that the schwannoma likely originated from the branch of great auricular nerve according to the tumor location.
Schwannomas are usually slow-growing masses without tenderness; characteristic neurogenic symptoms including pain or paresthesia are uncommon. If the tumor is near the external auditory canal and the tympanic membrane, symptoms including auricular fullness and mild conductive hearing loss may be noted. 4
Due to the tumor’s physical characteristics, the common differential diagnoses of dermoid, epidermoid cyst, basal cell carcinoma, and squamous cell carcinoma would be considered. The presence of a shiny or pearly lump with the pink scaly patch and central-ulceration would suggest basal cell carcinoma over other benign mimics. 5
The role of fine-needle aspiration (FNA) in the auricle mass is controversial. 1 We obtained a better understanding of the anatomy on the basis of the preoperative imaging study; however, it was difficult to render a definitive diagnosis since there are no distinguishing image features or criteria. According to Biswas et al, only 6% of patients with extracranial schwannoma in the head and neck region could be diagnosed preoperatively based on clinical findings, computed tomography, magnetic resonance imaging, and FNA. 1
The classic histopathologic features of schwannoma are elongated spindle cells with nuclei arranged in hypercellular Antoni-A and hypocellular Antoni-B patterns. 2,6 Antoni-A areas are composed of spindle cells arrayed in a palisading manner with formation of Verocay bodies, which consist of 2 palisading nuclei and an acellular zone. Antoni-B areas are hypocellular with loose myxedematous content.
The list of differential diagnoses for spindle cell neoplasms with similar histologic features as those of schwannomas includes neurofibroma, solitary circumscribed neuroma, and leiomyoma. 7 Neurofibromas are usually multicentric and are not encapsulated. 6 While neurofibromas are also positive for S-100, SOX-10, and CD34 immunostains, they lack Antoni-A and Antoni-B patterns and are less diffuse than schwannomas. Solitary circumscribed neuromas also have a complete, though thinner and indistinct, capsule. 7 Histologically, they display positive expression of S-100, moderate positivity to EMA immunostain, and nuclear palisading without Antoni-A and -B areas.
Schwannomas closely resemble leiomyomas for their similar spindle cell pattern; however, they are negative for smooth muscle actin and desmin immunostains, while leiomyomas are positive for both.
Pathology and immunohistochemical study remain the standard for definitive diagnosis of this entity. The treatment of auricular schwannoma is complete excision with a low recurrence rate. 1,8
In conclusion, the patient had a schwannoma at an uncommon site with nonspecific clinical presentation mimicking that of other common cutaneous lesions. This case reminds us that though schwannoma is rare in the auricle, and it should be included in the differential diagnosis. Histopathological diagnosis is mandatory after excision.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Written consent was obtained from the patient.
Ethical Approval
This study was approved by the Institutional Review Board Ethical Committee of Chang Gung Memorial Hospital. (No. 202101630B0)
