Abstract
Objective:
Nearly half of parapharyngeal space (PPS) tumors present as an intraoral mass, which is diagnostically challenging. In this study, we studied whether preoperative growth patterns were associated with histopathological diagnosis for planning surgery.
Methods:
We performed a cross-sectional study in patients with PPS tumors. A simplified classification scheme based on intraoral tumor growth patterns (patterns 1 and 2) was then proposed. In pattern 1, tumors bulge submucosally to the oropharynx from the soft palate, with the center convexity above the uvula. In pattern 2, tumors bulge submucosally to the oropharynx from the lateral oropharynx wall, with the center convexity below the uvula. The association of this classification with postoperative histopathological diagnosis and surgical-related events was studied.
Results:
Twenty-two patients were enrolled in this study (12 with pattern 1, 10 with pattern 2). Of these, 91.7% (11/12) of pattern 1 tumors were salivary gland tumors (P < .001), and 90% (9/10) of pattern 2 tumors were neurogenic (P < .001). Pattern 2 tumors had fewer bleeding complications or needed external approaches when a transoral approach was chosen.
Conclusions:
This new classification of PPS tumors facilitates the prediction of salivary gland and neurogenic tumors and can improve the accuracy of preoperative radiologic diagnosis. This system will be helpful for planning surgical interventions, such as implementing transoral approaches.
Keywords
Introduction
Parapharyngeal space (PPS) tumors are rare and only represent approximately 0.5% to 1% of head and neck neoplasms. Salivary gland tumors and neurogenic tumors are the most common PPS neoplasms. 1 The preoperative diagnosis of PPS tumors is challenging due to their low occurrence rate, anatomical complexity, and histological diversity. 2 Computed tomography (CT) and/or magnetic resonance imaging (MRI) are used frequently for preoperative diagnoses. 3 The information on CT scans is unfortunately relatively limited, while MRIs generally provide more detailed information. 3 The tumor locations in the prestyloid or retrostyloid compartments delineated by imaging evaluations could improve the accuracy of presumptive diagnosis. 4 However, substantial knowledge and experience are required for radiologists to accurately provide a histological diagnosis based on imaging features of these rare tumors. Additionally, radiologic examinations are not always feasible in patients with contradictions, which makes diagnosis even more difficult.
In addition to CT and MRI, biopsy, usually by fine-needle aspiration (FNA), is another clinical method to establish a preoperative diagnosis, especially when performed under image guidance.5,6 However, the rate of nondiagnostic FNA samples could be as high as 25% to 35%.7-10 Tissue sampling also causes pain, bleeding, and damage to important neurovascular structures, which makes this technique not appropriate for certain patients. Therefore, making a definite preoperative diagnosis of PPS tumors is challenging for radiologists, cytologists, and head and neck surgeons.
The clinical signs from physical examination have important implications for the diagnosis of a PPS lesion. Large PPS tumors can protrude into the oropharynx and become visible intraorally. According to 2 systematic review articles with 2586 cases, approximately half of PPS tumors presented with an intraoral mass.1,11 Unfortunately, few published studies have described the intraoral growth patterns of PPS tumors.12-14 It was reported that parotid lesions typically displaced the soft palate and the whole tonsil, while neurogenic lesions were more likely to displace the posterior pharynx and posterior pillar of the tonsil. 12 To the best of our knowledge, the intraoral growth patterns of PPS tumors in relation to the postoperative histopathological type have not been systemically evaluated. In this study, we investigated the preoperative intraoral presentations of PPS tumors and associated them with postoperative histopathologic types. We sought to determine whether intraoral tumor growth patterns were related to the histopathologic diagnosis of PPS tumors, which has clinical implications for planning surgical strategies.
Patients and Methods
Study Design and Participant Enrollment
We performed a cross-sectional study and enrolled patients at the Department of Otolaryngology—Head and Neck Surgery, Beijing Anzhen Hospital, Capital Medical University, Beijing, China, between July 2014 and May 2020. The inclusion criterion was patients with a presumed diagnosis of PPS tumors in the presence of a large submucosal mass bulging into the oropharynx on physical examination (Figure 1). Patients with lesions obviously arising from the palate, tonsil, tongue base, or the nasopharynx, especially those demonstrating mucosal changes (such as necrosis, crusting, angiogenesis, or bleeding), were excluded (Figure 2). Patients with suspected PPS tumors that caused visible vascular and mucosal changes in the pharynx and those having previous surgery for histopathology examination were also excluded.
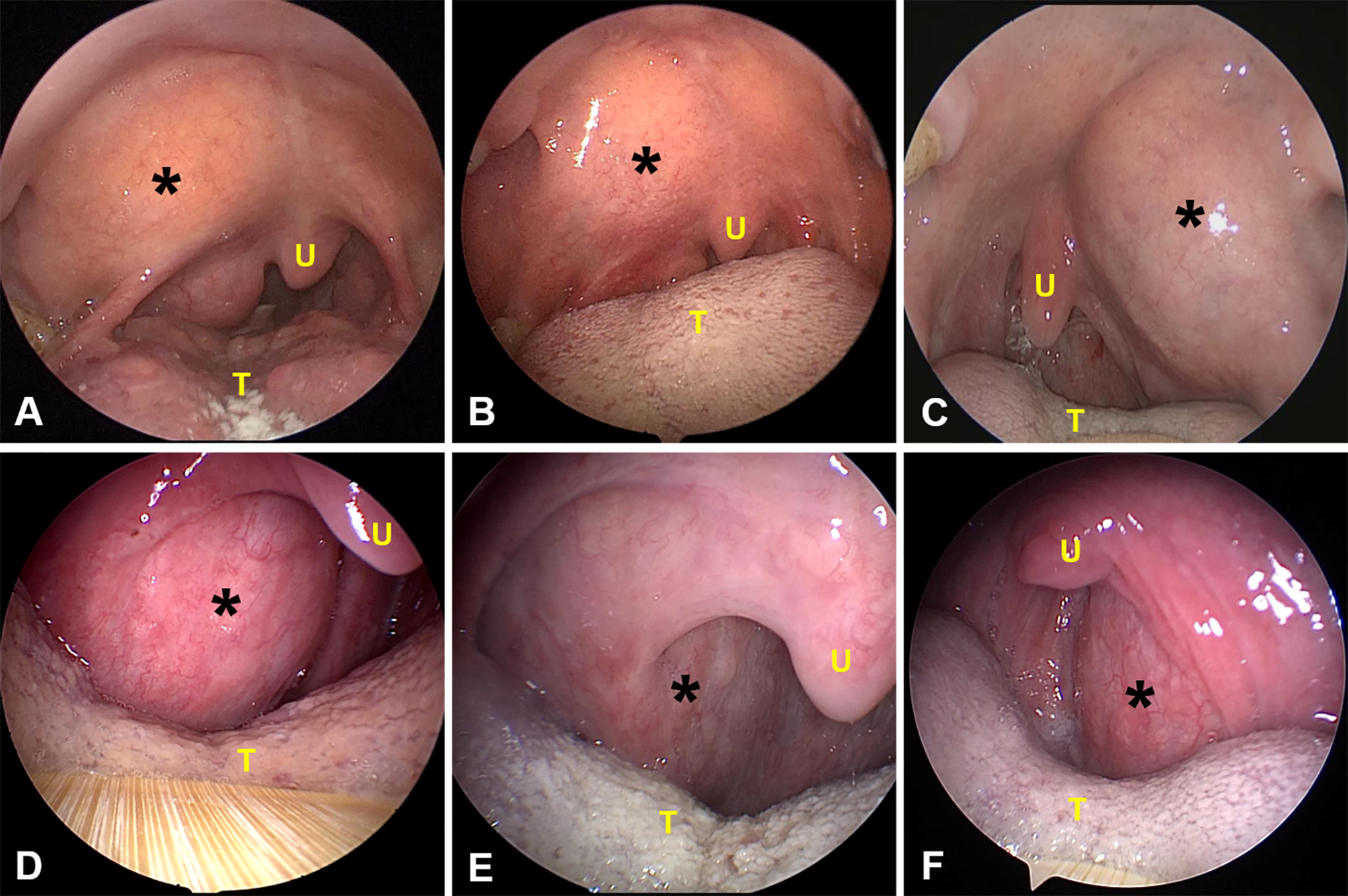
Cases that met inclusion criteria. Endoscopic imaging demonstrates submucosal bulges from the soft palate or lateral oropharyngeal wall (A-F), which implied a suspected diagnosis of parapharyngeal space tumor. The endoscopic images in (A-F) correspond to patients 12, 9, 19, 6, 11, and 13, respectively, in Table 1. Black asterisks indicate lesions. T indicates tongue; U, uvula.
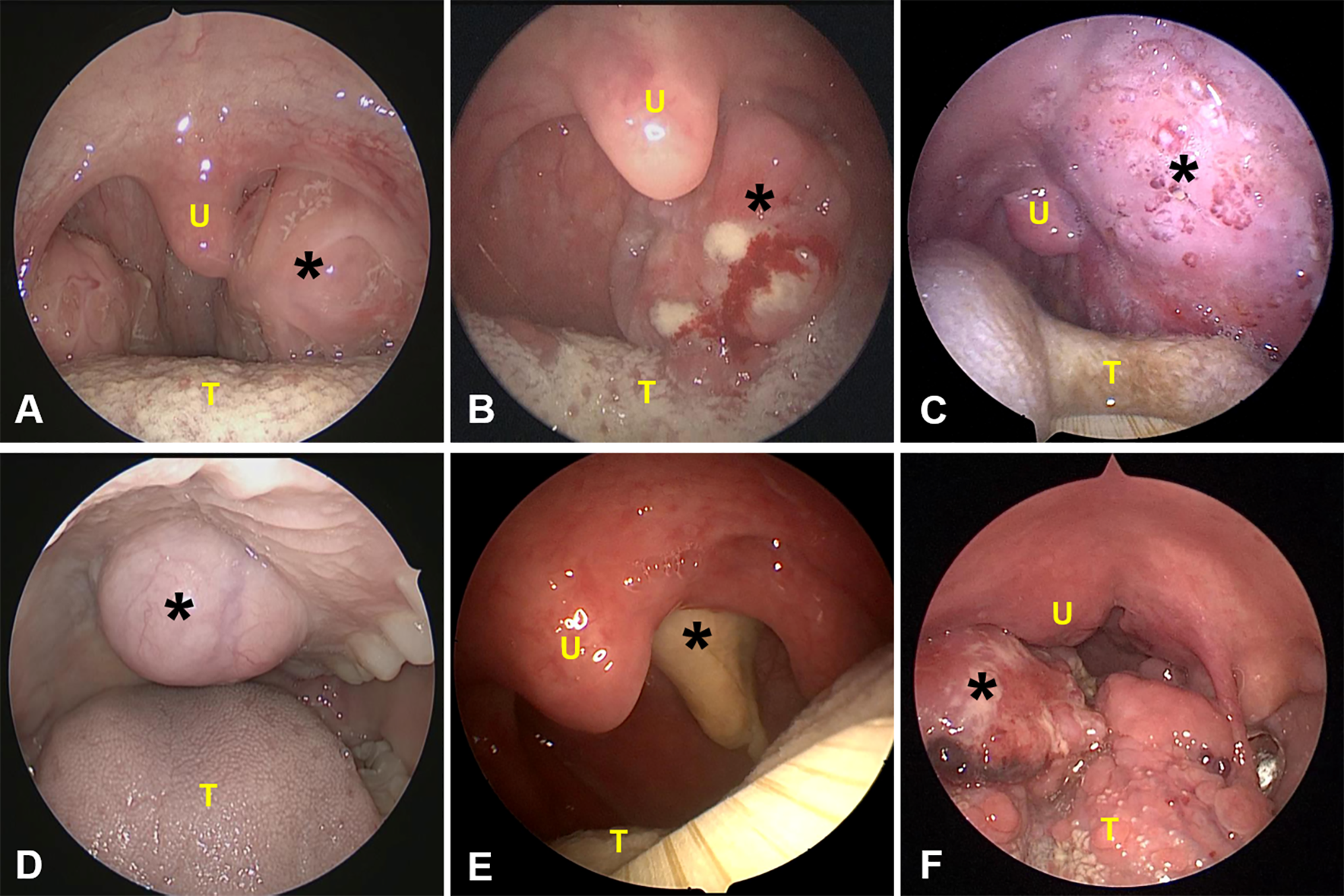
Illustration of cases that failed to meet inclusion criteria. Typical lesions arise from the palate, tonsil, tongue base, and nasopharynx, and some with mucosal changes are shown (A-F). Postoperative pathological diagnosis revealed physiological hypertrophy of the tonsil (A), tonsillar B-cell lymphoma (B), parapharyngeal space lymphangioma (C), palatal pleomorphic adenoma (D), nasopharyngeal carcinoma (E), and squamous cell carcinoma of the tongue (F). The black asterisk indicates the lesion. T indicates tongue; U, uvula.
This study was approved by the local ethics committee and followed the ethical standards of the Declaration of Helsinki. Written informed consents were obtained from all patients prior to their enrollment in this study.
Study Protocol
During the physical examination after admission, all patients underwent a comprehensive preoperative nasal and oral endoscopy. The intraoral masses were then visualized and photo-documented. We adopted a new classification proposed by the author (Y.F.) of this work. The intraoral presentation of PPS tumors was classified into 2 patterns. In pattern 1, the tumor bulged to the oropharynx from the soft palate, with the center convexity above the uvula (Figure 3). In pattern 2, the tumor bulged to the oropharynx from the lateral oropharynx wall, with the center convexity below the uvula (Figure 4). All patients received CT and/or MRI examinations before and after intraoral examination and then underwent surgery, of which the transoral approach was the most frequently used. The tumor growth patterns were then compared with the gold standard of postoperative histopathology. The clinical data of patients, including age, gender, tumor size, intraoral features, surgical approaches, and histopathologic diagnoses, were collected.
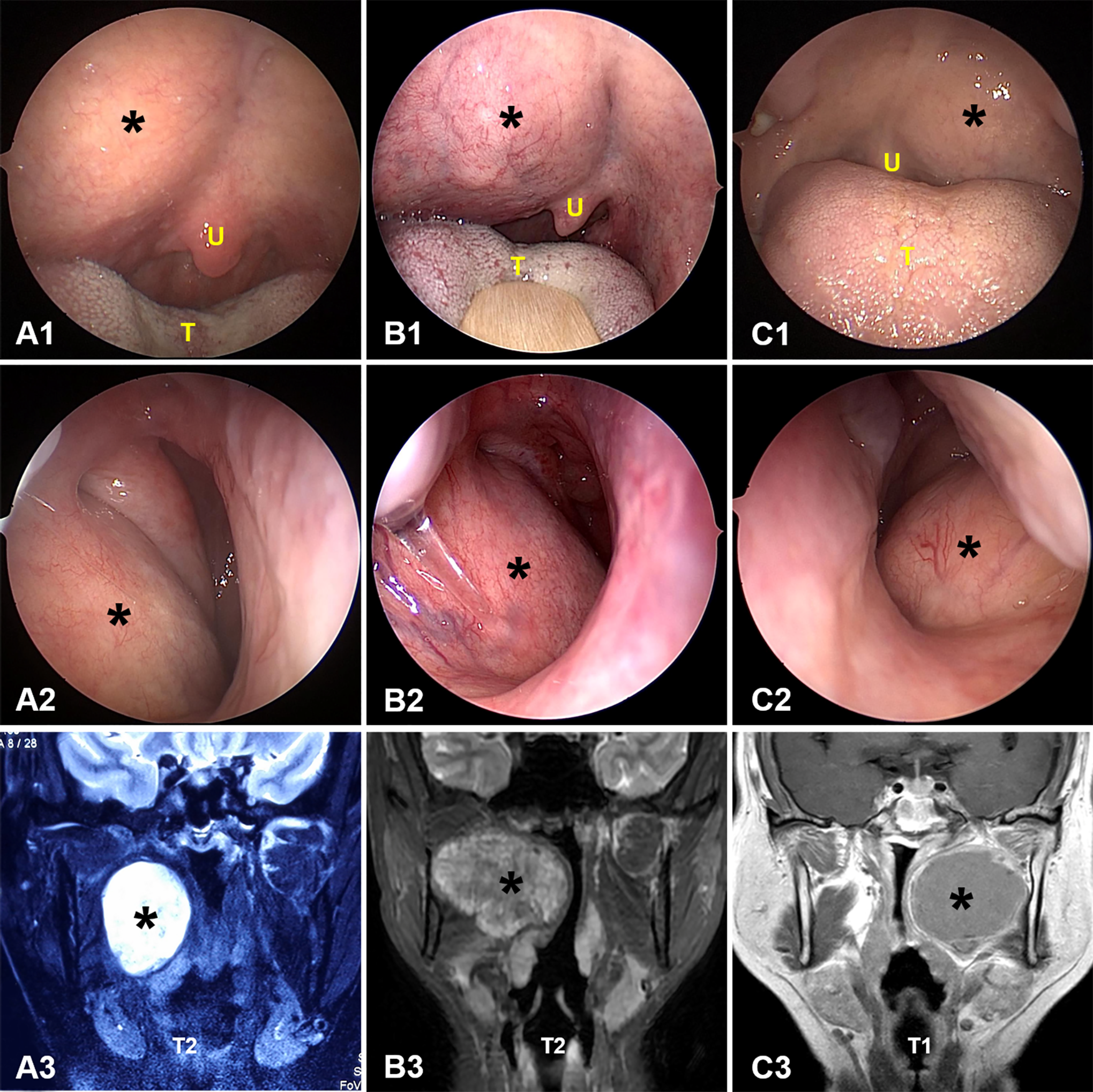
Representative pattern 1 parapharyngeal space tumors. In pattern 1, the tumor bulges into the oropharynx from the soft palate, with the center convexity above the uvula (A1-C1). The endoscopic images in (A1, B1, and C1) correspond to patients 8, 17, and 4, respectively, in Table 1. Endonasal endoscopic views of the same patients showing the tumors extending out into the nasopharynx (A2-C2). Radiologic images of the patients are also shown (A3-C3). The black asterisk indicates the lesion. T indicates tongue; U, uvula.
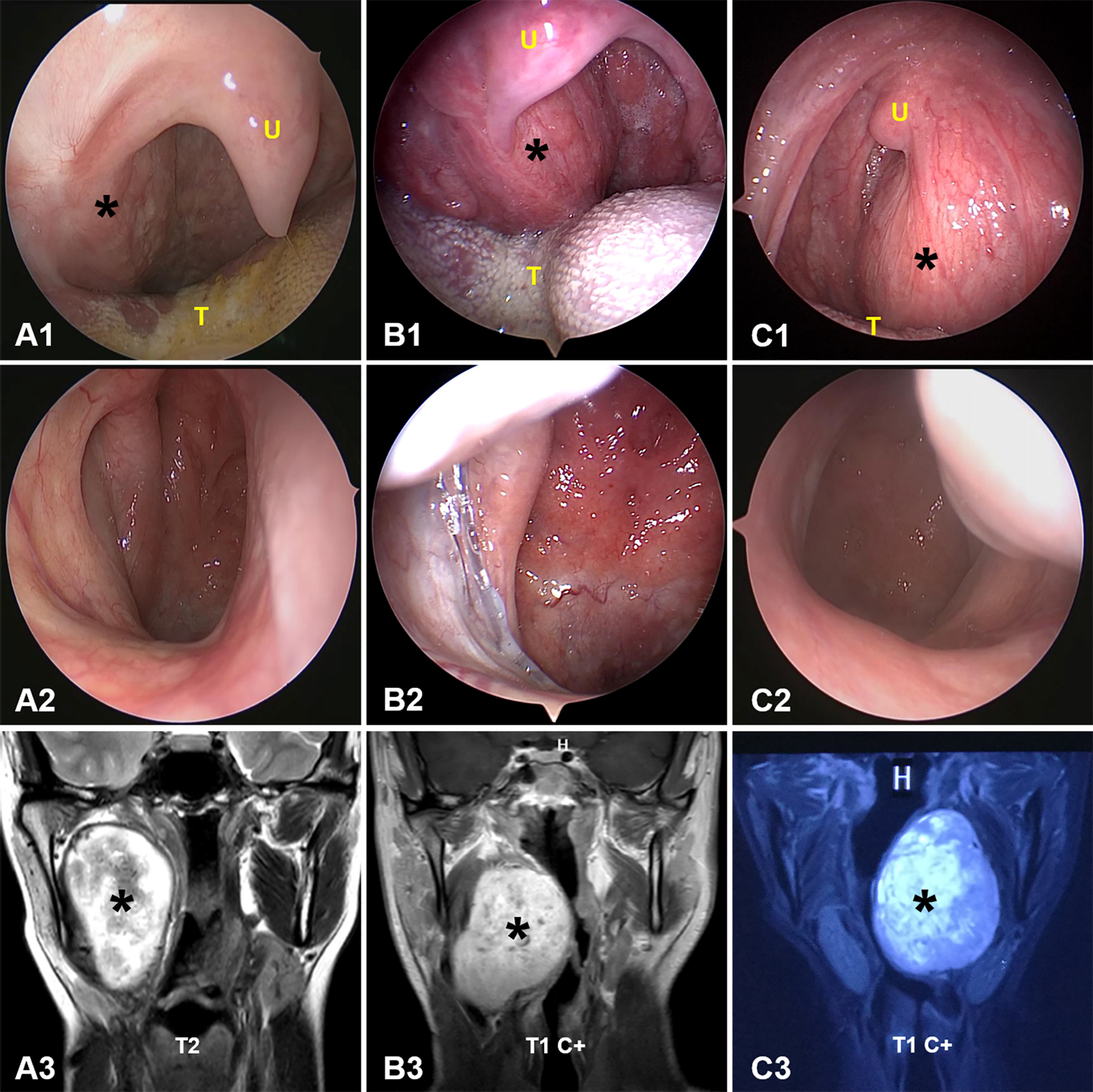
Representative pattern 2 parapharyngeal space tumors. In pattern 2 tumors, tumor bulges into the oropharynx from the lateral oropharynx wall, and the center convexity was below the uvula (A1-C1). The endoscopic images in (A1, B1, and C1) correspond to patients 18, 10, and 15, respectively, in Table 1. Endoscopic views of the nasopharynx showing no visible masses are also presented (A2-C2). Radiologic images of the patients are also shown (A3-C3). The black asterisk indicates the lesion. T indicates tongue; U, uvula.
Statistical Analysis
Continuous data are presented as mean ± SD or median with a minimal maximal range, when appropriate. Categorical data are presented as numbers and percentages. The association of the tumor growth patterns (pattern 1 or 2, according to proposed classification) and histopathological diagnosis (salivary or neurogenic tumors) was analyzed using a 2-sided Fisher exact test. Statistical analyses were performed with SPSS software for Windows, version 17.0.
Results
We enrolled 22 patients in this study, including 10 males and 12 females, and the mean age was 46.5 ± 15.7 years (range, 28-79 years). The tumor size measured by CT/MRI was 5.86 ± 1.34 cm. The postoperative histopathological investigation revealed that all the tumors were benign in nature. The diagnoses were pleomorphic adenoma (10 cases), basal cell adenoma (1 case), and hamartoma (1 case) in pattern 1 PPS tumors and schwannoma (7 cases), ganglioneuroma (2 cases), and lipoma (1 case) in pattern 2 PPS tumors. The characteristics of these patients are listed in Table 1.
Clinicopathologic Characteristics of the 22 Patients.
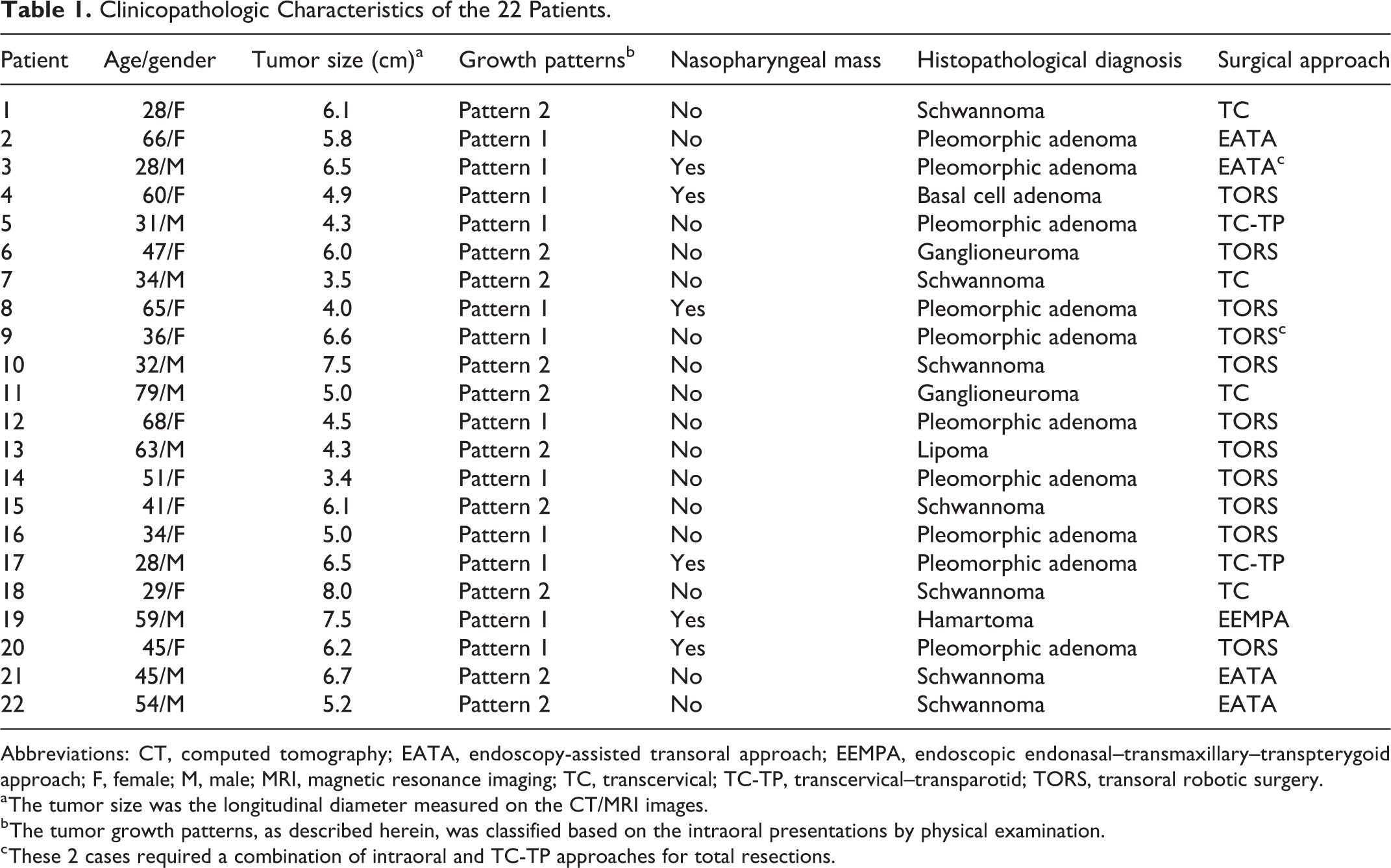
Abbreviations: CT, computed tomography; EATA, endoscopy-assisted transoral approach; EEMPA, endoscopic endonasal–transmaxillary–transpterygoid approach; F, female; M, male; MRI, magnetic resonance imaging; TC, transcervical; TC-TP, transcervical–transparotid; TORS, transoral robotic surgery.
a The tumor size was the longitudinal diameter measured on the CT/MRI images.
b The tumor growth patterns, as described herein, was classified based on the intraoral presentations by physical examination.
c These 2 cases required a combination of intraoral and TC-TP approaches for total resections.
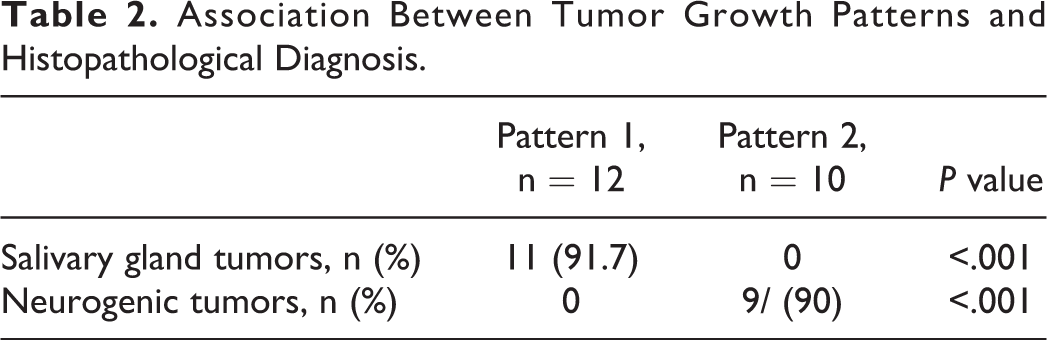
In Table 2, all 11 salivary gland tumors presented as pattern 1 tumors, accounting for 91.7% (11/12) of this group; no tumors in this group were pattern 2 tumors, which was statistically significant (P < .001). Nine neurogenic tumors presented as pattern 2 tumors, accounting for 90% (9/10) of this group, whereas none were pattern 1 tumors (P < .001). There were 6 patients observed to have a bulge in the nasopharynx region that was anterior to the orifice of the Eustachian tubes. Interestingly, salivary gland tumors were more often present in the nasopharynx (5/11) compared to neurogenic tumors (0/9; P = .038).
Association Between Tumor Growth Patterns and Histopathological Diagnosis.
In Table 1, various approaches were used, and most patients received tumor resection via the transoral approach by either the endoscopic or robotic technique. Total resections were achieved in all but 1 patient (patient 19). Of note, when a transoral approach was chosen, 1 patient with a pattern 1 tumor had major bleeding (patient 14). A combined approach with lateral access was applied for 2 patients with pattern 1 tumors (patients 3 and 9). All the pattern 2 tumors presented no severe hemorrhaging or were removed using a lateral approach when removed transorally.
Discussion
Parapharyngeal space is an inverted pyramid-shaped space extending from the skull base down to the hyoid bone.15,16 Numerous studies have described the endoscopic and microscopic anatomy of the PPS in detail.17-20 The PPS is mostly lined by adipose tissue that connects to the surrounding structures with variable tissue types. The PPS tumors are histologically diverse and have remained challenging to diagnose preoperatively. According to a systematic review of 1143 PPS tumors, up to 70 histological tumor subtypes can be identified in this anatomically complex region. 1 Radiologic examinations and FNA biopsy are the most frequently used methods for preoperative diagnosis. However, both of these examinations have limitations in some circumstances. As most PPS tumors are salivary or neurologic in origin and account for approximately 90% of all PPS neoplasms,1,2 the differentiation of these 2 categories of tumors before surgery is crucial for follow-up treatment. In this study, we proposed a simplified classification based on tumor growth patterns, which facilitated the prediction of these salivary gland and neurogenic tumors preoperatively and also helped in surgical strategy planning. This classification system can be used in coordination with radiologic evaluation without increasing preoperative investigation already commonly practiced.
Radiology examination is invaluable for suggesting a diagnosis prior to surgical intervention. 16 From the surgical point of view, imaging examinations not only provide a specific diagnosis but also greatly assist in planning the next steps for treatment. Radiologic evaluation can provide a surgeon with the location, extent, size, and vascularity of a PPS tumor. Both CT and MRI examinations provide imaging features complementary to each other. It is ideal if both modalities are performed in the same patients; however, not every patient can be a candidate for CT or MRI assessment. This is especially true for the MRI evaluation, which has the advantage of better tissue characterization but is not appropriate for patients with contraindications such as metal fillings, pacemaker implants, or cochlear implants. 3 In these conditions, additional information may be needed for accurate preoperative diagnosis.
Biopsy, typically using FNA cytology, plays an essential role in establishing a preoperative diagnosis. Fine-needle aspiration has relatively high accuracy in differentiating between benign tumors versus malignant tumors.6,21 However, the diagnostic accuracy of FNA in patients with benign PPS lesions is relatively low due to hemorrhaging, inadequate specimen collection, and other technical problems.4,7,8 Furthermore, the seeding of tumor cells and other complications should be taken into consideration when performing a transoral biopsy. 12 The use of FNA is controversial, as there are often difficulties and variability encountered when interpreting the results. 11 Some authors suggest that preoperative sampling is useful, 4 while others consider it is not always necessary, 22 and even others consider it contraindicated in diagnosing PPS tumors. 12 It is our practice that no transoral or percutaneous biopsies are performed unless the malignant nature of the PPS neoplasm is highly suspected. The FNA procedure is not a first-choice diagnostic tool for a benign PPS lesion, and other information is needed to narrow a differential diagnosis.
In addition to radiology and FNA evaluation, a physical examination may provide clues to the final diagnosis of PPS tumors. The presence of an intraoral mass has been reported as 47% or 52% in all PPS tumors in 2 systematic review articles.1,11 However, tumor presentation is usually recorded briefly as a submucosal swelling/bulge or an intraoral/oropharyngeal/pharyngeal mass, with displacement or distortion of adjacent structures.9,12-14,21,23 Such findings in intraoral examination are believed to be less informative and rarely related to histopathological types in previous reports.4,9,21 To investigate the correlation between these intraoral tumor presentations and diagnosis, the author proposed a new classification based on the intraoral tumor growth patterns and compared this classification with final histopathology. In our study with limited cases, we observed a high agreement between the tumor growth pattern and final histopathological diagnosis. The pattern 1 tumors were mainly pleomorphic adenomas, whereas the pattern 2 tumors were mainly schwannomas (Table 1). In general, salivary gland tumors accounted for 91.7% of pattern 1 tumors, but not pattern 2 tumors (P < .001), whereas neurogenic tumors accounted for 90% of pattern 2 tumors, but not pattern 1 tumors (P < .001; Table 2). As these 2 patterns are clear and easy to distinguish, we consider this new, simplified classification based on intraoral examination (either under a headlamp, a microscope, or an endoscope) to be useful for diagnosing a PPS tumor. In conjunction with radiologic assessments, this tumor classification can further improve the accuracy of the preoperative diagnosis of PPS lesions. This classification system can be applied in coordination with radiology to differentiate most common benign tumors (salivary and neurogenic tumors), thus preventing unnecessary biopsy before surgery.
Surgery is the primary treatment modality for patients with PPS tumors. 11 Multiple surgical approaches have been proposed to treat PPS lesions,15,24 and the choice of approaches is mainly dependent on a surgeon’s experience. In our experience, pattern 1 and 2 tumors can be removed through various approaches, and the transoral approach by endoscopic or robotic techniques was effective and safe for both patterns in experienced hands (Table 1). In this cohort, total resections were achieved in all but 1 patient (patient 19). When a transoral approach was chosen, a difference was observed for pattern 1 and pattern 2 tumors. Although pattern 1 tumors are easy to access intraorally, they are technically challenging to be removed via a transoral approach, especially salivary gland tumors originating in the deep lobe of the parotid gland. The risk of major bleeding and the need for a combined open approach should be taken into consideration in the transoral resectioning of pattern 1 tumors. These findings have implications for a surgeon in choosing a surgical approach, especially via a transoral route, which has become popular in the management of PPS tumors.14,25-28 In addition, the treatment strategy for pleomorphic adenoma or schwannoma, such as approach selection, extracapsular/intracapsular dissection, and surgical-related neurovascular morbidity, is clearly different.23,24,29 As pattern 1 was closely related to pleomorphic adenoma and pattern 2 to schwannoma, this new classification helps in generating a detailed preoperative plan with regard to the options of approaches, possible complications, and expected outcomes. We consider that the classification of tumors, as we described above, is valuable for planning surgical strategies.
This study was limited by its small sample size and single institution. Moreover, this classification system cannot be applied to PPS tumors that do not bulge into the oropharynx. Further investigations from multiple centers with larger numbers of cases are warranted.
Conclusions
The growth patterns of PPS tumors based on our classification system facilitated the prediction of salivary gland tumors and neurogenic tumors. This classification system allowed consolidation of radiologic diagnosis and prevents unnecessary biopsy for most common PPS tumors before surgical intervention. Additionally, this classification aided in planning surgical strategies, especially in executing a transoral approach.
Footnotes
Acknowledgments
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
