Abstract
Salivary duct carcinoma (SDC) is an uncommon but highly aggressive tumor with a poor prognosis. SDC mainly arises from the major salivary glands, typically the parotid gland. Here, we report a rare case of sinonasal SDC in a 54-year-old male patient that might have originated from the inferior turbinate. The patient presented with left nasal congestion and rhinorrhea. Following an endoscopic intervention, the histopathological examination revealed a diagnosis of SDC, characterized by the formation of solid cancer nests and central comedo-type necrosis. Given the highly aggressive nature and unfavorable prognosis of SDC, it is essential to consider it as a differential diagnosis for unilateral nasal tumors.
Introduction
Salivary duct carcinoma (SDC) is a rare subtype of malignant salivary gland tumors first reported by Kleinsasser et al. in 1968. 1 It predominantly affects older men and exhibits histopathological similarities to ductal breast carcinoma.2,3 Clinically, SDC is the most aggressive malignancy of the excretory duct system among all primary salivary gland carcinomas and has a high rate of local recurrence and distant metastasis.4,5 Here, we present an extremely rare case of SDC originating from the left inferior turbinate.
Case Report
A 54-year-old man who presented with persistent left nasal congestion and rhinorrhea for nearly five months was admitted to the outpatient clinic at the Wuhan Union Hospital in 2022. The patient reported that his nasal congestion began five months ago and was accompanied by purulent rhinorrhea and headache with no underlying cause. There were no other symptoms, such as loss of smell, vision loss, blindness, double vision, eye displacement, bloody nose, tinnitus, ear occlusion, and hearing loss. The patient received no treatment before and was admitted for formal diagnosis and treatment.
The computed tomography (CT) examination of the paranasal sinuses revealed chronic multi-sinusitis and indicated that the left maxillary sinus, ethmoid sinus, and nasal meatus were filled with soft tissue density shadows with no signs of absorption, damage, or hardening in the bone walls (Figure 1(A) and 1(B)). Thickening of the mucosa was observed in the right maxillary sinus and ethmoid sinus (Figure 1(B)). The left posterior ethmoid sinus, sphenoidal sinus, and frontal sinus were normal (Figure 1(C)-1(E)). In the axial scan, a tumor was detected in the left maxillary sinus, but there was no evidence of bone defect or invasion (Figure 1(F)). Additionally, opacification in the left anterior ethmoid sinus was observed (Figure 1(G)). There were no other abnormalities in the sinus cavity, and the choana was unaffected (Figure 1(H)). Unenhanced CT scan of the paranasal sinuses: (A) (coronal scan): There were some soft tissue density shadows in the left nasal meatus, with opacification of the left middle turbinate concha and inferior turbinate concha; (B) (coronal scan): The left maxillary sinus was infiltrated by the tumor and the left ethmoidal bulla was also affected. The mucosae of the right maxillary sinus and ethmoid sinus were partly thickened; (C)–(D) (coronal scan): The posterior ethmoid sinus and sphenoidal sinus were normal; (E) (axial scan): The mucosae in the left frontal sinus was slightly thickened; (F) (axial scan): Opacification in the left anterior ethmoid sinus, compared to the normal posterior ethmoid sinus; (G) (axial scan): The soft tissue density shadows were confined to the left maxillary sinus, with no bone defects or invasions; (H) (axial scan): The left choana was not affected by the tumor. CT: computed tomography; MTC: middle turbinate concha; ITC: inferior turbinate concha; FS: frontal sinus; MS: maxillary sinus; EB: ethmoidal bulla; AES: anterior ethmoid sinus; PES: posterior ethmoid sinus; SS: sphenoidal sinus.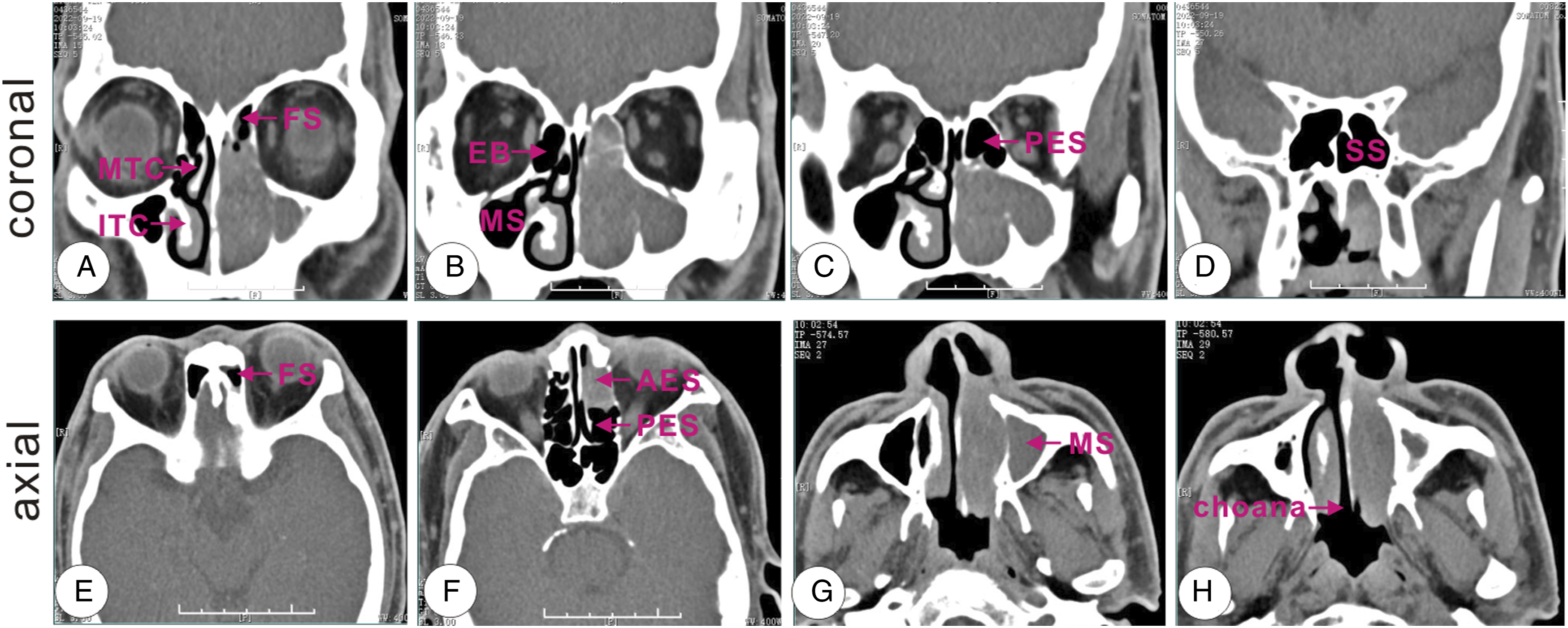
Here, an endoscopic resection of the lesions in the nasal cavity and sinuses was performed. The surgical procedure involved a combination of endoscopic maxillary sinus opening, anterior ethmoidectomy, and frontal sinus opening. Lesions resembling benign neoplasms that emerged from the inferior turbinate was detected in the left nasal cavity. Necrotic tissues were observed, and bleeding was easily induced. The left maxillary sinus was filled with a large amount of yellow and white purulent secretions. In addition, mucosal polyps were observed in the left ethmoid and frontal sinuses.
The postoperative pathology revealed a malignant tumor of the salivary gland located in the left nasal cavity, consistent with the characteristics of salivary duct carcinoma. Solid cancer nests with comedo-type necrosis beneath the attenuated sinonasal mucosa were observed at low magnification (Figure 2(A)). At higher magnification, the tumor comprised irregular nests and cords of cells with abundant cytoplasm and high-grade nuclei (Figure 2(B) and 2(C)). Results of the immunohistochemical staining of the tumor cells were as follows: PCK (+), CK7 (+), CK20 (-), TRPS1 (+), SOX10 (+), AR (multifocal weak +), HER2 (3+), GATA-3 (-), Villin (-), CDX2 (-), P40 (-), P63 (-), P16 (-), SATB2 (-), TTF-1 (-), Hepatocyte (-), PSA (-), CD56 (few +), Syn (-), S-100 (-), INI-1 (+), and BRG1 (+). Meanwhile, in situ hybridization for the detection of EBV was negative (Figure 2(D)-2(H)). (A). An overview of the sinonasal salivary duct carcinoma showed solid cancer nests with comedo-type necrosis beneath the attenuated sinonasal mucosa (original magnification×50). (B). The tumor consisted of irregular nests and cords of cells that elicited a prominent desmoplastic reaction (original magnification×200). (C). Tumor cells were characterized by abundant cytoplasm and high-grade nuclei. The nuclei were round with prominent nucleoli and brisk mitotic activity (arrow, original magnification×300). (D). The tumor cells strongly expressed CK7 (original magnification×200). Strong nuclear expressions of TRPS1 (E) and SOX10 (G) were found in tumor cells (original magnification×200). (F). Diffuse and strong membranous immunostaining for ERBB2/HER2 (original magnification×200). (H). P63 was not found in tumor cells (original magnification×200).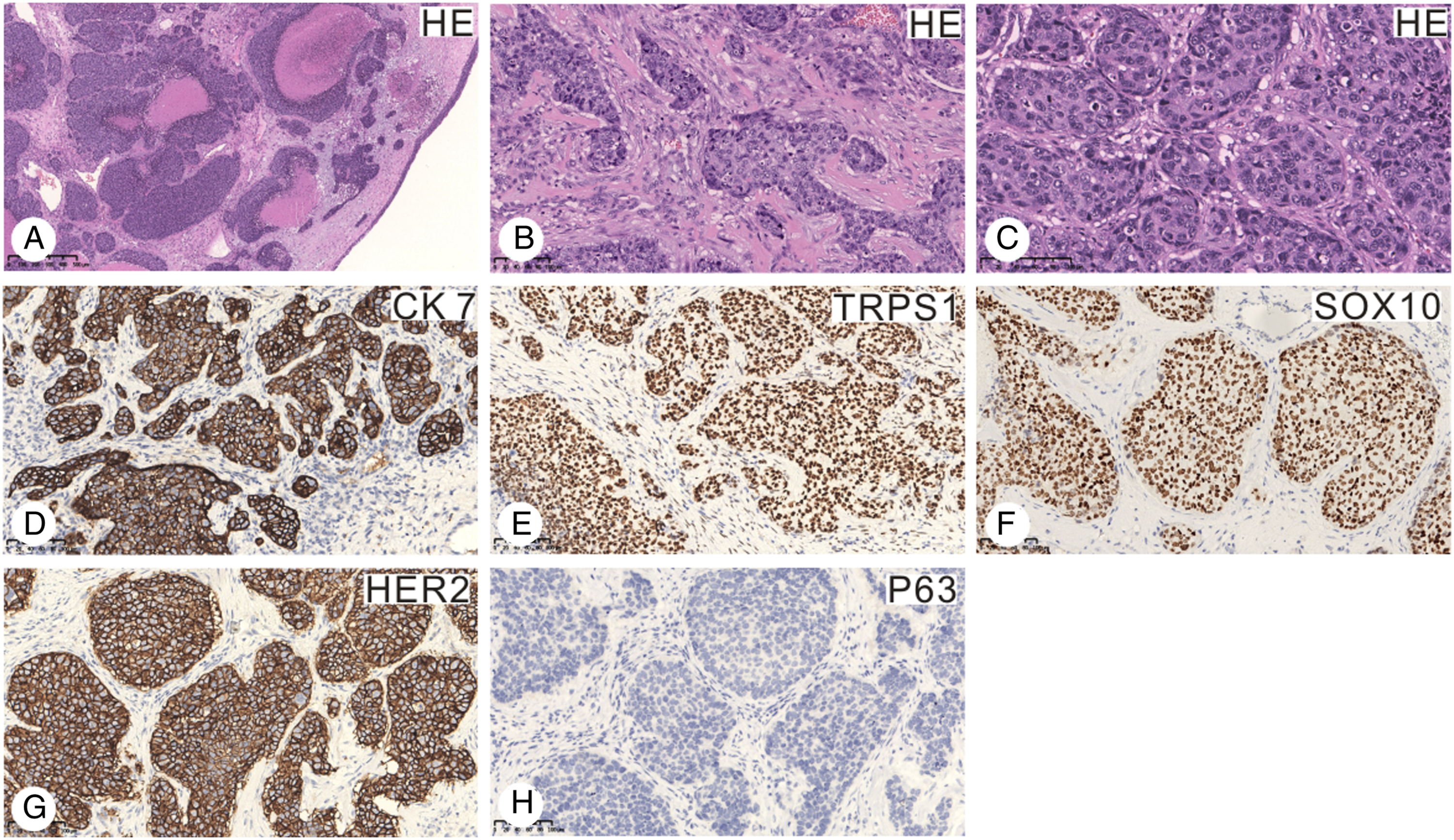
Next, the patient underwent a Positron Emission Computed Tomography (PET-CT) examination two weeks after the surgery. The results were as follows: 1. The left turbinate was absent. 2. The mucosas of the left nasal cavity and ethmoid sinus were slightly thickened with diffusely increased metabolism in both the early and delayed phases, consistent with a postoperative change. 3. There was no noticeable localized metabolic increase in the brain. (The background uptake of fluorodeoxyglucose in the brain was high, which may affect the intracranial evaluation.)
Therefore, we performed a nasal endoscopy for case tracing and further analysis. We observed postoperative changes in the left inferior concha and maxillary sinus, including the uncinate process, while the nasal septum and middle turbinate concha were unaffected (Figure 3(A)–3(C)). Nasal endoscopy following surgical intervention: (A). There were some postoperative changes in the left inferior concha, and the nasal septum was unaffected; (B). The left maxillary sinus was opened neatly and recovered from the surgery; (C). The tail of the left uncinate process was cut off, and the middle turbinate concha was not involved. NS: nasal septum; ITC: inferior turbinate concha; MS: maxillary sinus; MTC: middle turbinate concha; UP: uncinate process.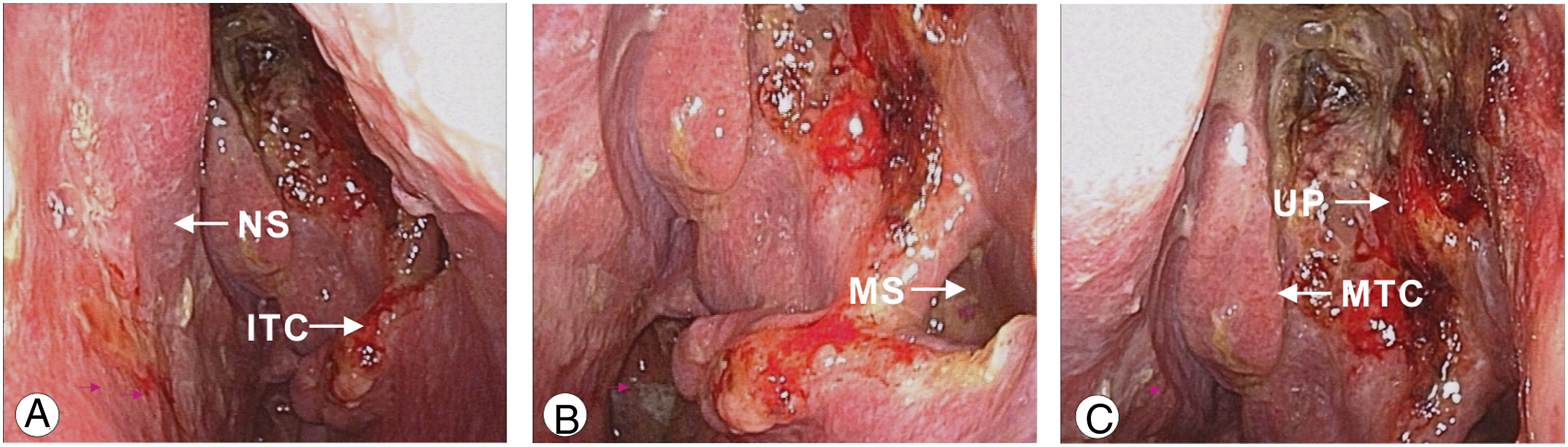
Discussion
The salivary glands are the organs responsible for secreting saliva in the mouth and consist of the major salivary glands (including the parotid gland, submaxillary gland, and sublingual gland) and the minor salivary glands located in the submucosa of the oral cavity, pharynx, nasal cavity, and maxillary sinuses. 6 Malignant salivary gland tumors are characterized by their diverse and complex structures. The main histopathological types of salivary gland tumors include adenoid cystic carcinoma, mucoepidermoid carcinoma, epithelial-myoepithelial carcinoma, and salivary duct carcinoma, to name a few. 7
Salivary duct carcinoma (SDC) is a relatively uncommon type of malignant salivary gland tumor, accounting for 10% of all cases. 8 It mainly occurs in elderly men over 50 years old. 9 SDC is most commonly located in the parotid gland (70%–80%) and infrequently in the submandibular gland, with occurrence in other salivary glands being exceptionally rare.4,10 Isolated cases of SDC in sites such as the oral and nasal cavities have been reported, albeit infrequently. 11
SDC was first reported by Kleinsasseer and his colleagues in 1968. 1 However, it was not until 1990 that SDC was first classified as a separate tumor by the World Health Organization (WHO) and listed individually from parotid gland tumors in 2017. 12
Histopathologically, SDC displays a high degree of similarity to both intraductal and infiltrating breast carcinoma with respect to both cytology and tissue landscape. The classic morphological patterns of SDC include papillary, cribriform, and solid growth patterns, with the defining features of prominent ductal proliferation and central comedo-type necrosis.13,14 Additionally, there is often infiltration of adjacent parenchymal and periglandular tissues. SDC shares similarities with prostate cancer regarding immunohistochemistry, with a high expression of the androgen receptor (AR). Overexpression of the human epidermal growth factor receptor 2 (HER2) has also been extensively reported in SDC. 9
SDC is the most aggressive tumor among all primary salivary gland carcinoma subtypes, exhibiting a high rate of local recurrence (up to 57%), distant metastasis (about 46%), and disease-related death. 6 Currently, there are no established treatment guidelines for SDC specifically, and disease management is based on the treatment of other major salivary gland malignancies. SDC has a grim prognosis, with nearly half of the patients succumbing to the disease within four to five years. 9
The occurrence of SDC in the nasal cavity and paranasal sinuses is uncommon, with limited domestic and foreign reports. Typically, unilateral nasal masses are benign, including nasal polyps, inverted papilloma, and hemangioma, while primary malignant neoplasms of the nasal cavity are rare, with squamous cell carcinoma and adenocarcinoma being the most common. Herein, our exploratory surgery revealed that the lesion was located in the left inferior turbinate. Pathological examination of the tumor revealed prominent duct formation and comedo-type necrosis, with immunohistochemical markers including androgen receptor (multifocal weak +), HER2 (3+), and P63 (-), strongly suggesting the diagnosis of SDC.
In conclusion, SDC is a rare and aggressive tumor of the salivary glands, prevalent in the sinonasal tract. Chinese cases were rarely reported in the English literature, which is why this case deserves to be noted. The presentation of SDC originating from the left inferior turbinate also highlights the need to carefully consider differential diagnoses for unilateral nasal tumors. Therefore, this case holds significant referential value for future exploration and observation in clinical practice.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest concerning the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the Natural Science Foundation of Hubei Province (2022CFB087, ZT), the Wuhan Union Hospital (F016.02004.21003.126, ZT), Open Project of Key Laboratory of Molecular Imaging (2022fzyx015, TZ) and National Natural Science Foundation of China (81771005, ZT).
