Abstract
Orbital cellulitis is an uncommon condition with risks to sight and life. As a complication of maxillofacial injuries, the literature suggests this is only possible with fractures or direct inoculation, and there are no reports to the contrary. Here, we make the first report of a possible etiology by which orbital cellulitis developed in a 14-year-old boy even without skin breach or bony fractures; as well as a rare causative pathogen. He presented with facial abscess and progressive orbital cellulitis after blunt facial trauma, requiring functional endoscopic sinus surgery with needle aspiration of facial abscess externally. Cultures showed growth of Streptococcus constellatus/Parvimonas micra, and he received further antibiotics with full recovery.
The pathophysiology of orbital cellulitis in this patient is attributed to vascular congestion and local pressure from maxillofacial contusion and maxillary hemoantrum, with impaired paranasal sinus ventilation encouraging anaerobic bacterial growth. Further progression led to facial abscess formation and intraorbital spread with orbital cellulitis. The pediatric demographic is injury-prone, and self-reporting of symptoms can be delayed. Hence, increased suspicion of complicated injuries and orbital cellulitis may be required when managing maxillofacial contusions so that prompt treatment can be given.
Background
Orbital cellulitis is an uncommon condition that occurs with higher incidence in the pediatric population 1 due to relatively incomplete immunologic development. 2 The incidence of pediatric orbital complications from acute rhinosinusitis ranges between 3 and 7%. 3 Prompt recognition and treatment are essential as there is the potential for progression to serious morbidity, including sight loss and intracranial involvement with neurological sequelae. 1,4,5
Anatomically, the orbital compartment is kept separate from the periorbital tissues due to the lack of venolymphatic communications between the two compartments and the natural barriers represented by the bony orbit and the orbital septum. 1,6,7 In the absence of direct inoculation, orbital cellulitis usually develops from normal commensals of the respiratory tract, oral cavity, or skin that are translocated into the orbit when these usual anatomical barriers are breached. For this reason, blunt maxillofacial trauma without the presence of bony fractures or loss of skin integrity is not usually known to cause orbital cellulitis. 5,7
Here, we report our experience with a patient who developed orbital cellulitis and facial abscess after maxillofacial contusion, hypothesized to be the result of contiguous spread from a superimposed bacterial infection of an ethmoidal/maxillary hemoantrum.
Case Presentation
A 14-year-old boy presented to the Children’s Emergency with nine days of progressive facial swelling that started two days after suffering blows to the left side of his face with a hard wooden sandal. This was associated with a fever of 38 °C and thick greenish-brown rhinorrhea with occasional blood streaks. Subsequently, he developed bitemporal headache that was worse on the left, as well as left eye pain and monocular blurring of vision.
He had no previous medical history and was not on blood thinners. Prior to symptom onset, there were no upper respiratory tract symptoms or eye complaints. There was no orbital or facial swelling, external skin abrasions, lacerations, or frank epistaxis directly after the trauma was sustained.
Investigations
On examination, there was left periorbital erythema, induration of the left lateral nasal wall in the middle third, and tender swelling over the left maxillary area with smoothening of the left nasolabial fold. Anterior rhinoscopy showed grade 3 swelling and congestion of the inferior turbinates and mucopus discharge. Due to induration and medial displacement of the left lateral nasal wall, it was difficult to tell the origin of the intranasal mucopus. Intraoral examination was unremarkable.
Ophthalmological review showed normal visual acuity, color vision, and pupillary light reflexes, and no relative afferent pupillary defect (RAPD). Slit lamp and indirect ophthalmoscopy were normal. Intraocular pressures were slightly raised on the left at 21 mmHg compared to 15 mmHg on the right. Extraocular muscle function was full bilaterally but with pain on full abduction of the left globe.
Computed tomography (CT) scan of the brain, orbits, and paranasal sinuses showed complete opacification of the left maxillary sinus and ethmoid air cells, with a focal defect in the left lamina papyracea, adjacent intraorbital soft tissue stranding, and swelling of the medial rectus muscle. Significant soft tissue stranding and edema were also observed in the left maxillary, periorbital, and preseptal tissues. There were no skull vault or facial bone fractures (refer Fig. 1). Based on clinical and radiological findings, the diagnosis of orbital cellulitis with underlying maxillary/ethmoid sinusitis was established. Cuts of CT paranasal sinuses (a) sagittal through left maxillary area and maxillary sinus, showing opacification of the left ethmoid and maxillary sinuses; (b) axial at level of maxillary swelling showing significant left maxillary asymmetrical swelling and fat-stranding, with slight medial displacement of the left nasal vestibule; (c) axial at level of ethmoid sinuses; (d) coronal showing unilateral left ethmoid and maxillary sinus opacification but no fractures of the orbital rim. Filled white star—opacified maxillary sinus; unfilled star—opacified ethmoid sinuses; filled white arrow—left maxillary fat stranding; unfilled white arrow—complete left ostiomeatal unit obstruction; filled white arrowhead—small dehiscence seen in left lamina papyracea; unfilled arrowhead—fat stranding with adjacent mild edema of the medial rectus; A, anterior; I, inferior; R, right; P, posterior.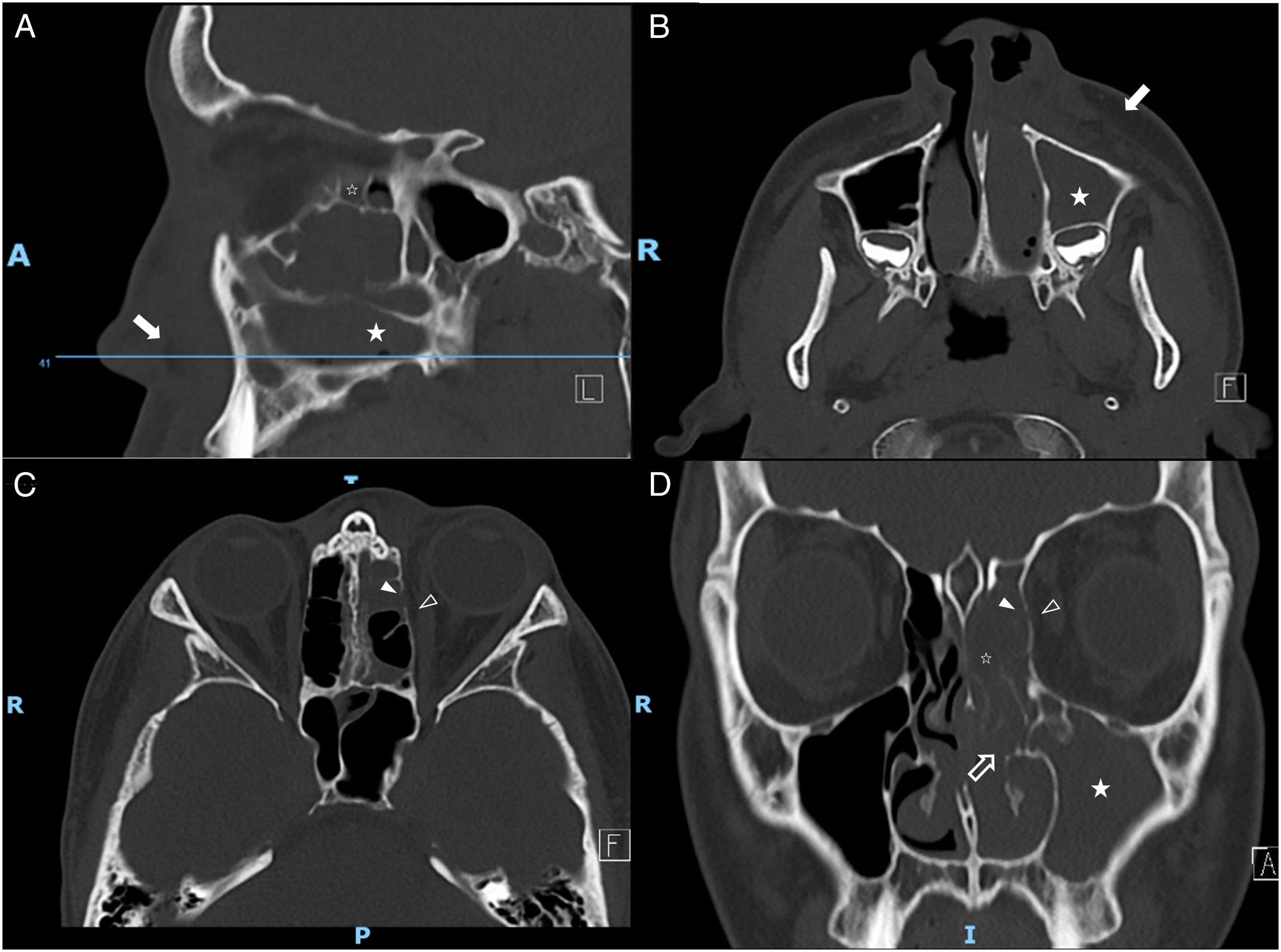
Initial blood investigations showed elevated inflammatory markers, with a C-reactive protein of 151 mg/L, elevated white cell count of 18.6 × 109/L, and an increased absolute neutrophil count of 14.1 × 109/L. Both blood cultures and aerobic culture of mucopus swabbed from the anterior nasal space revealed no bacterial growth.
Treatment
He was started on intravenous Ampicillin and Co-amoxiclav, with regular nasal douches, oxymetazoline nasal drops, and intranasal steroid spray. Timolol eye drops were also started for elevated left intraocular pressure. However, after 48 hours of intravenous antibiotics, he continued to have high fever above 39 °C and reported worsening headache, left nasal obstruction, and left eye opening. Clinically, there was progressive maxillary swelling with extension of the overlying erythema. He also developed worsening chemosis and onset of ophthalmoplegia in both superior and lateral gaze of the left globe.
He thus underwent functional endoscopic sinus surgery (FESS) on the third day of admission. Intraoperatively, there was left periorbital edema and maxillary fluctuance that was causing significant medial displacement of the left nasal vestibule. Endoscopic examination showed an edematous and everted uncinate process with small amounts of mucopus from the left medial meatus tracking posteriorly into the posterior nasal space. Left uncinectomy and medial maxillary antrostomy were performed, with copious amounts of hemopurulent discharge and organized fibrinous clots drained from the left maxillary sinus. Externally, needle aspiration of the left maxillary swelling further drained moderate amounts of hemopurulent material.
Cultures of the intraoperative material drained showed mixed pathogen growth of Streptococcus constellatus and Parvimonas micra. Fungal and tuberculosis studies were negative. Postoperatively, his antibiotic regime was changed to intravenous Co-amoxiclav and Clindamycin and continued for another week.
The patient continued to improve postoperatively, with flattening of the maxillary swelling, reformation of the nasolabial fold, decreased medialization of the nasal vestibule, and resolution of ophthalmoplegia, chemosis, and proptosis (refer Fig. 2). He was discharged after 10 days of hospitalization with a further one-week duration of oral Co-amoxiclav and Clindamycin. Subsequent review in clinic one week later showed resolution of all symptoms and facial swelling. The repeat nasoendoscopy this time showed a clean left maxillary sinus cavity without any further hematoma, bleeding or mucopus. From L–R: (a) Self-taken photograph on D9 post-injury showing mild left eyelid and maxillary swelling, (b) facial photograph on D12 (2 days after presentation) showing worsening of left maxillary and eyelid swelling with almost complete lid closure and effacement of the nasolabial and nasojugal grooves, (c) improvement in left eyelid and maxillary swelling with resolving erythema on the day of discharge.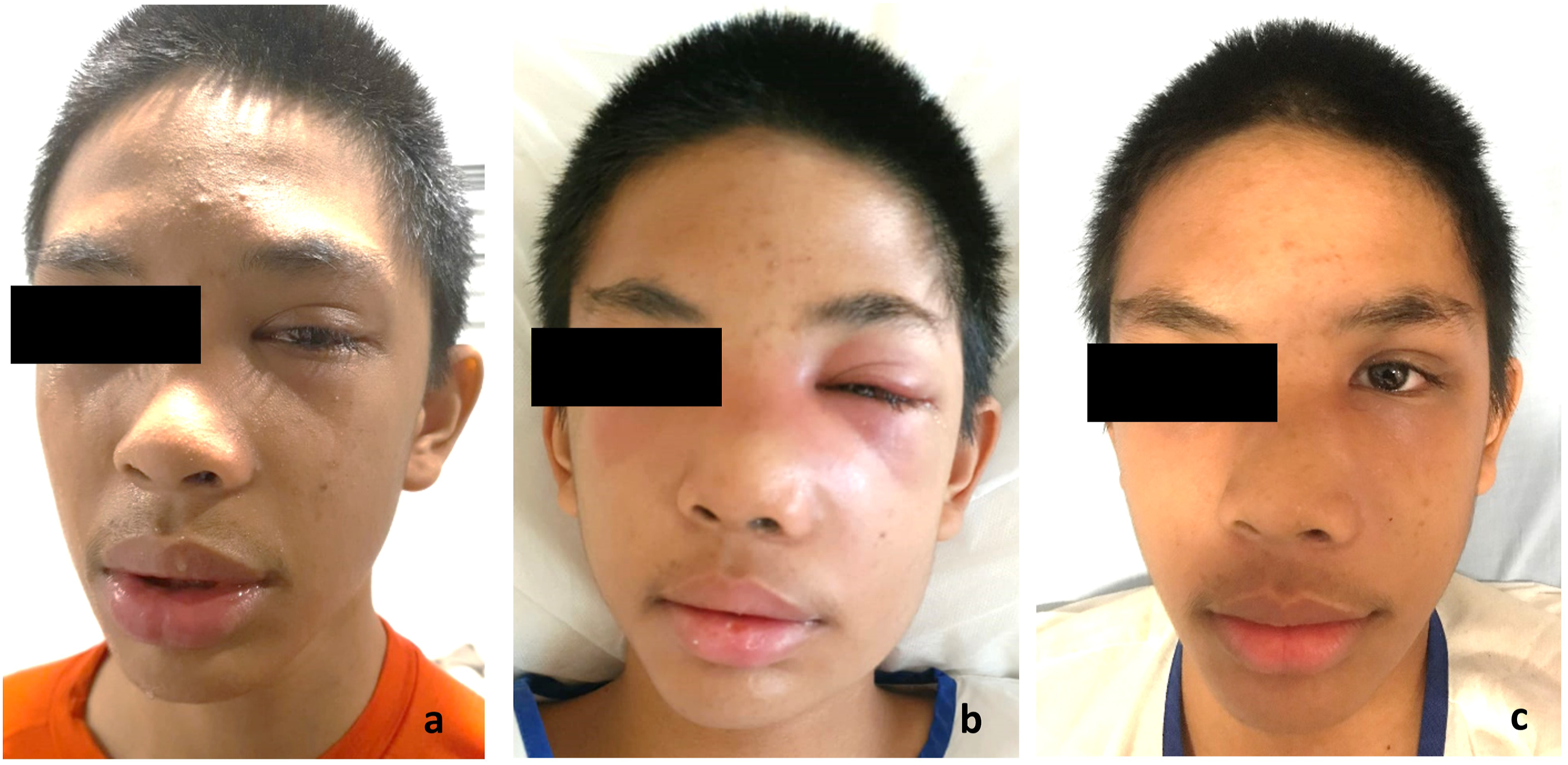
Discussion
Pathogenesis
Given the progression of our patient’s condition, we postulate that the sequence of events after trauma started with maxillofacial contusion with resultant maxillary hemoantrum. Local pressure effects and mucosal vascular congestion led to the development of left ostiomeatal unit obstruction. The inspissated blood products and impaired aeration of the paranasal sinuses created a milieu conducive for existing respiratory flora to cause secondary infection. From there, adjacent spread to the orbit from the maxillary/ethmoid sinuses occurred. Further spread to the left maxillary region caused abscess formation and medial displacement of the nasal vestibule and lateral nasal wall, exacerbating mechanical obstruction of the left maxillary sinus outflow tract.
Bacteriology
The probable pathogenesis pathway is supported by the profile of causative organisms isolated from cultures taken of the maxillary pus and blood clots. Significant growth was observed of both the anaerobe P micra and the microaerophilic S constellatus. Although both have been reported to be causative organisms for paranasal sinus infections, 6,8,9 P micra is most often associated with odontogenic causes and it is less common for P micra to result in orbital cellulitis.
Systematic reviews by Nageswaran 2006 10 and Tsirouki 2018 5 showed that bacterial isolates of orbital cellulitis mostly comprised Streptococcus pneumoniae, Haemophilus influenzae, Moraxella catarrhalis, and Staphylococcus aureus. Another study looking at the bacteriology of swabs taken from the maxillary sinus floor during maxillary sinus elevation found mostly Streptococci (45%), followed by Staphylococci (25%), and Enterobacteriaceae (25%). 11 Other studies show similar bacteriology and further report an emerging trend of methicillin-resistant S aureus (MRSA) infections. 7,10,12
However, in post-traumatic orbital cellulitis, the most common organisms isolated are S aureus and S pyogenes. 7 Furthermore, while younger children are more likely to have a single bacterial isolate, children 9 years and older typically exhibit a polymicrobial infection with anaerobe involvement, believed to be due to relative narrowing of the sinus ostia with age and consequently reduced oxygen tensions. 4,5,13,14 These microaerophilic conditions may underpin the findings of Tsirouki and Seltz, who showed that older children are more likely to have orbital cellulitis with group A streptococci, 5 and 44% of patients with positive eye secretion cultures had Streptococcus anginosus, 9 both of which comprise the microaerophilic S constellatus.
On the other hand, as an anaerobe resident to the oral, gastrointestinal, and genital tracts, the presence of Parvimonas suggests odontogenic origin must be excluded, 8 especially since it is most often isolated from lesions of apical periodontitis. 15 In the literature, reports of infective endocarditis, spinal infections, and brain abscesses involving Parvimonas show a significant proportion of odontogenic origin. In addition to clinical examination, radiological scans including orthopantomogram (OPG) or CT are commonly used to look for accompanying radiological features of common dental etiologies. However, the clinician must be aware that a negative dental evaluation does not rule out odontogenic sources, and clinical symptoms can be nonspecific or even absent. 16
In our patient, due to the presence of Parvimonas, he was referred to the oromaxillofacial surgery team to exclude odontogenic causes. Examination of his dentition and periodontal probing with cold tests revealed no dental pathology. There was no radiological evidence of periodontitis or oroantral fistula.
Appropriate Treatment
Orbital cellulitis falls into a spectrum of orbital complications of paranasal sinus infections that has been categorized by Chandler et al 17 in 1970 into 5 categories 17 to assess severity and risk of progression, with the fifth and last category being intracranial involvement with high mortality.
So far, clinical consensus places emphasis on early treatment with antibiotics that cover the most common pathogens involved in paranasal sinus infections, with some advocating MRSA cover or third-generation cephalosporins with better central nervous system (CNS) penetration to reduce the risk of intracranial dissemination of infection. 6,7,10,12 In reporting their experience in the management of pediatric orbital cellulitis in Shenzhen, Hongguang et al 18 concluded that the first 72 hours of presentation were crucial and effective treatment within this period could reduce CRP levels, the incidence of abscess development requiring surgical drainage, and overall duration of hospitalization.
Although the role for surgical intervention is evident in the presence of neurological or intraorbital complications, its role in orbital cellulitis and early subperiosteal abscesses is controversial. With varied surgical success rates 12 and reported surgical complication rates of up to 20%, 14 medical management is sometimes preferred. This is especially true for younger children under 9 years of age, who have relatively larger sinus ostia with better aeration, often manifest single microbial and simpler infections, and for whom medical therapy alone is often adequate, 2,3,5,7,19 even with small periorbital abscesses. 3,20
Surgery is nonetheless recommended when there is no improvement or when there are worsening ophthalmological findings, 10,19 as was the case with our patient. For him, surgery was done with the aim of improving paranasal sinus ventilation and evacuating trapped blood and infected material.
The learning points from the presentation of this patient include the following: (1) Clinicians must adopt a lower threshold of suspicion for orbital complications in the presence of maxillofacial trauma, as the diagnosis is confounded by a history of trauma. Delayed recognition of orbital cellulitis may result when facial and orbital swelling is wrongly attributed to the inciting injury. In this case, the mechanism of injury to the face of our patient and the timeline of the onset of signs and symptoms were important. CT imaging was an essential investigation to rule out facial fractures, assess the extent of orbital complications, and exclude neurological complications. (2) External compression of nasal anatomy by the patient’s maxillary facial abscess impeded normal sinus ventilation and further affected the effectiveness of medical treatment. This significantly limited nasoendoscopic access during FESS and made surgery difficult. External decompression by needle aspiration was required to facilitate FESS. External surgical approaches to the sinuses were also considered should the initial medial maxillary antrostomy be insufficient for the clinical resolution of orbital complications. (3) Clinicians should consider routine anaerobic coverage in antibiotic selection, particularly in susceptible age groups (over 9 years) even when the history is not suggestive of anaerobic etiology.
Conclusion
Pediatric orbital cellulitis requires early diagnosis and treatment to avoid severe morbidity. We add to the literature another possible etiology for this condition and a rare causative pathogen. The pediatric age group is a vulnerable population already at increased risk of injuries, including those to the maxillofacial region. Symptom progression may be insidious if supervision is lacking and self-reporting is inadequate. This case report illustrates that when dealing with pediatric patients with maxillofacial contusions, clinicians should keep in mind orbital cellulitis as a potential complication even in the absence of skin or bony breach.
Footnotes
Author contributions
All authors were involved in the management of the patient, including daily medical care during admission.
LMY led and coordinated clinical decision-making, including the conduct of surgery.
KWM and RA monitored the clinical response to treatment and assisted with surgery.
CWC was the lead author and major contributor to the writing of the manuscript.
All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The authors declare that they have no competing interests.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Ethical approval
The publication of this case report together with the data collection and management involved was in accordance with the guidelines of the Institutional Review Board and Ethics Committee of our institution. The need for approval was waived, but the consent to participate was nonetheless taken from the patient’s parent.
Informed consent
Written consent was obtained for the publication of the case report together with the patient’s photographs from the patient’s parent.
