Abstract
Objective
Our previous study revealed that serum amyloid A (SAA) levels in polyp tissues could serve as a biomarker for the prediction of corticosteroid insensitivity in patients with chronic rhinosinusitis with nasal polyps (CRSwNP). However, their association with disease control status in the patients after endoscopic sinus surgery remains to be assessed.
Methods
Polyp tissues and control uncinate process mucosa were collected from 48 patients with CRSwNP and 10 healthy control subjects. SAA expression was examined using immunohistochemistry and enzyme-linked immunosorbent assay. Receiver operating characteristic (ROC) curves were performed to determine the predictive value of SAA in nasal polyps. The clinical characteristics of 2 CRSwNP subtypes (SAAhigh and SAAlow) were evaluated.
Results
The SAA expression levels in polyp tissues were significantly elevated both in non-eosinophilic and eosinophilic CRSwNP as compared to the healthy controls. In patients with CRSwNP, the tissue SAA level was significantly higher in the disease-controlled patients than those of the partly controlled and uncontrolled. ROC curve analysis revealed that a cut-off value of 114.9 ng/mL for the tissue SAA level predicted the patients with disease-controlled status with 93.33% sensitivity and 63.64% specificity (AUC = .8727, P < .001). Furthermore, The SAAhigh subgroup showed higher tissue eosinophil numbers and percentage of the disease-controlled patients compared to the SAAlow subgroup.
Conclusions
Our findings suggest that measurements of SAA in polyp tissues may provide useful information for evaluating CRSwNP conditions, especially identifying the CRSwNP patients with disease-controlled status after endoscopic sinus surgery.
Introduction
Chronic rhinosinusitis (CRS) is a chronic inflammatory disease of the mucosa of the nose and paranasal sinuses. Based on the presence or absence of polyp formation, CRS is typically classified into 2 clinical phenotypes, CRS with nasal polyps (CRSwNP) and CRS without nasal polyps. 1 Although only accounting for ∼25-30% of patients with CRS, CRSwNP patients usually have greater severity of clinical disease and impairment of quality of life as well as cost burden compared to those without nasal polyps, making it more clinically challenging to rhinology clinicians.1-6 There are several treatment modalities for CRSwNP proposed by mainstay guidelines, including comprehensive medical therapy and endoscopic sinus surgery, with the main goal of achieving and maintaining clinical disease control.1,2 However, we and others have reported that over 30% of patients with CRSwNP remain uncontrolled despite current standard-of-care treatment.7-9 Evidence has emerged suggesting that it may be caused by an underlying variation of endotypes resulting in a discrepancy of clinical phenotype and disease prognosis.10-13 Thus, the need for seeking new relevant biomarkers in patients with good or poor disease control is generally acknowledged.
Serum amyloid A (SAA) is a secreted protein first characterized for its role in the acute-phase response to inflammation and therefore has been a clinical biomarker for active inflammation. 14 In addition, studies also have shown that SAA levels are remarkably elevated in patients with a broad spectrum of chronic inflammatory diseases, such as atherosclerosis, 15 allergic asthma, 16 and CRSwNP. 17 Although the biological functions of SAA in immunity and inflammation are poorly defined, previous studies have suggested that SAA participates in inflammatory responses as a chemokine of immune cells such as monocytes, neutrophils, dendritic cells, and T lymphocytes.14,18-22 Furthermore, in vitro studies showed that SAA stimulation of leukocytes resulted in increased production of proinflammatory cytokines including IL-1β, IL-6, and TNFα, 14 suggesting SAA may be involved in the inflammatory response through immune cell activation. The clinical applications of SAA have been highlighted. A series of publications have demonstrated the association of SAA with neutrophilic inflammation and Th17 immune response in airway diseases including chronic obstructive pulmonary disease (COPD), 23 allergic asthma, 24 and CRSwNP. 17 Recently, we further revealed that SAA levels in polyp tissues could serve as a biomarker for the prediction of corticosteroid insensitivity in patients with CRSwNP. 25 Since it has been well recognized that corticosteroid responsiveness is closely related to the degree of eosinophilic inflammation, whereas eosinophilic inflammation is a major phenotype associated with poorly controlled disease and polyp recurrence in patients with CRSwNP,26-28 this led us to hypothesize that SAA might be used as a potential biomarker for predicting treatment outcome in patients with CRSwNP after endoscopic sinus surgery. The aims of this study were, first, to compare SAA levels in polyp tissues in CRSwNP subjects during different conditions defined as controlled, partly controlled and uncontrolled, and second, to evaluate whether SAA levels could be a biomarker of disease control in CRSwNP.
Patients and Methods
Patients and Tissue Samples
This study was approved by the Ethics Committee of the First Affiliated Hospital of Sun Yat-sen University. Written informed consent was obtained from all participants. Patients with a diagnosis of bilateral CRSwNP based on the European position paper on rhinosinusitis and nasal polyps (EPOS 2012) 29 and performed transnasal endoscopic surgery were identified from the electronic medical database of the First Affiliated Hospital of Sun Yat-sen University between January 2015 and December 2016. All patients were under a current standard-of-care treatment and follow-up according to the Chinese and European position papers.29,30 Patients with following criteria were excluded: (1) Patients without adequate polyp specimens. (2) Patients lacked of data of baseline blood routine test, nasal endoscopy, and sinus computed tomography (CT). (3) Patients treated with systemic or intranasal corticosteroids within 3 months and 1 month before blood routine test, respectively, by cross-referencing patient’s medication history with the electronic prescription record system. (4) Patients less than 16 years. (5) Patients with a history of food allergies, allergic dermatitis, or worm infection. (6) Patients with fungal rhinosinusitis, cystic fibrosis, gastro-esophageal refiux disease, or sinonasal malignancies. Control subjects were patients undergoing optic nerve decompression because of traumatic optic neuropathy without any nasal disease. Allergic rhinitis was diagnosed according to the Allergic Rhinitis and its Impact on Asthma guideline. 31 The diagnosis of asthma was established based on the Global Initiative for Asthma guideline by a specialist physician. 32 The diagnosis of aspirin sensitivity was based on the documented history of intake of aspirin or non-steroid inciting worsening respiratory symptoms. Lund-Mackay score was performed by a trained researcher through scoring of CT images. 33
Clinical Assessment of Disease Control
Patients enrolled were followed up for the assessment of clinical disease control level based on the classification criteria of EPOS 2012 (Supplementary Table 1) by 2 senior authors (Y. S. and J. S.), 29 which has been validated by our and other groups.7-9 Briefly, CRS control status was stratified into 3 levels: controlled, partly controlled, and uncontrolled. Controlled CRS was established based on no bothersome symptoms, with healthy or almost healthy mucosa and no need for systemic medicine to control disease. Partly controlled patients experienced less than 2 of the following items: persistent nasal blockage, mucopurulent rhinorrhea/postnasal drip, facial pain, impaired smell, sleep disturbance/fatigue, disease mucosa under nasal endoscopy, and the need for a course of antibiotics/systemic corticosteroids in the last months. Uncontrolled CRS was diagnosed as 3 or more features of partly controlled CRS.
Histopathologic Evaluation
Polyp tissues and uncinate process mucosa were taken from CRSwNP patients and control subjects during endoscopic surgery, respectively. Samples were fixed in 10% formalin, embedded in paraffin, cut in 4-μm sections and stained with hematoxylin–eosin. The stained sections were observed using a microscope (Leica DM4 B; Leica, Wetzlar, Germany) by 2 independent observers in a blinded fashion to clinical data. Cell counting was performed as we previously reported. 7 Top 5 densest cellular infiltrate areas of the subepithelial layer were chosen for cell counting for each specimen under low power field (100×). Then eosinophils and total inflammatory cells were quantified in the focus of each area under high power field (400×, .072 mm2/frame, HPF). Eosinophil and total inflammatory cell count were recorded in each focus field at 400 power and reported as absolute number per HPF. Eosinophilic CRSwNP (ECRSwNP) was diagnosed when the percentage of tissue eosinophils exceeded 10% of the total inflammatory cells according to the European guideline. 2
Serum Amyloid A Immunohistochemical Staining
Immunohistochemical staining was performed using the standardized procedures as we previously described. 25 Briefly, sections were deparaffinized by serial treatment. After inhibiting the endogenous peroxidase in 3% hydrogen peroxide and with 1% bovine serum albumin, the sections were incubated overnight at 4°C in the presence of a mouse anti-human SAA monoclonal antibody (Abcam, Cambridge, MA, USA) at a dilution of 1:100 according to the manufacturer’s instructions. Each of these sections was incubated with a secondary antibody and then with horseradish peroxidase-labeled streptavidin complex (Zhongshanjinqiao, Beijing, China). Distribution of peroxidase was revealed by incubating the sections in a solution containing 3% 3,3′-diaminobenzidine tetrahydrochloride before being counterstained with hematoxylin and cover slipped. Negative control studies were performed by omitting the incubation step with the primary antibody.
Enzyme-Linked Immunosorbent Assay
Fresh polyp tissues and uncinate process mucosa samples were weighed and placed in PBS-Tween supplemented with a protease inhibitor cocktail (Keygentec, Nanjing, Jiangsu, China), and homogenized for 1 min on ice. The samples were then centrifuged at 4000 rpm for 20 min at 4°C, and the supernatants were stored at −20°C until ELISA analysis. The SAA concentrations were measured with commercially available ELISA (ab100635, Abcam) according to the manufacturer’s instructions. Detection limits were .41 ng/mL of SAA.
Statistical Analysis
GraphPad Prism 6 software (San Diego, CA, USA) was used for statistical analyses. T tests and one-way ANOVA and Tukey’s multiple comparisons post hoc tests were used for continuous variables with normal distribution. Mann–Whitney U tests were used for continuous non-normally distributed variables. χ2 Tests were used to assess differences in categorical variables, such as gender or smoking prevalence. The predictive ability of tissue SAA protein level was evaluated by receiver operating characteristic (ROC) curve. A P value less than .05 was considered as statistically significant.
Results
Characteristics of Participants
Forty-eight patients with CRSwNP and ten healthy control subjects followed for a median time of 86.4 weeks were included. The clinical characteristics of the study subjects are presented in Supplementary Table 2. Twenty-six patients and twenty-two patients were non-ECRSwNP and ECRSwNP, respectively. Age, gender, and smoking status were matched between the study groups. The comorbidity rates of allergic rhinitis and asthma in patients with ECRSwNP were higher than those in non-ECRSwNP and healthy controls. Consistent to previous reports,7,8,10,28,34 patients with ECRSwNP had significantly higher preoperative CT score, blood and tissue eosinophil numbers, and percentage of uncontrolled disease after endoscopic sinus surgery.
Association of SAA Expression With Disease Control Status in Patients With CRSwNP
We first compared the protein level of SAA in nasal tissues between patients with non-ECRSwNP, ECRSwNP, and healthy controls. By doing IHC staining, strong immunoreactivity of SAA was observed both in non-ECRSwNP and ECRSwNP. Consistent with our previous observations,17,25 the SAA immunoreactivity was mainly observed in the epithelial layer of polyp tissues. Additionally, subepithelial inflammatory cells and glands were also displayed scattered positive staining. By contrast, the SAA immunoreactivity was relatively weak in the healthy controls (Figure 1A). Consistently, the SAA protein levels in polyp tissues as evaluated by ELISA were significantly elevated both in non-ECRSwNP and ECRSwNP as compared to the healthy controls (P < .001 and .01, respectively) (Figure 1B). Increased expression of SAA in CRSwNP. (A) Representative immunohistochemical staining of SAA in non-ECRSwNP, ECRSwNP, and healthy control. Original magnification: ×400. The protein level (B) of SAA in non-ECRSwNP, ECRSwNP, and healthy control assessed by ELISA. **P < .01; ***P < .001.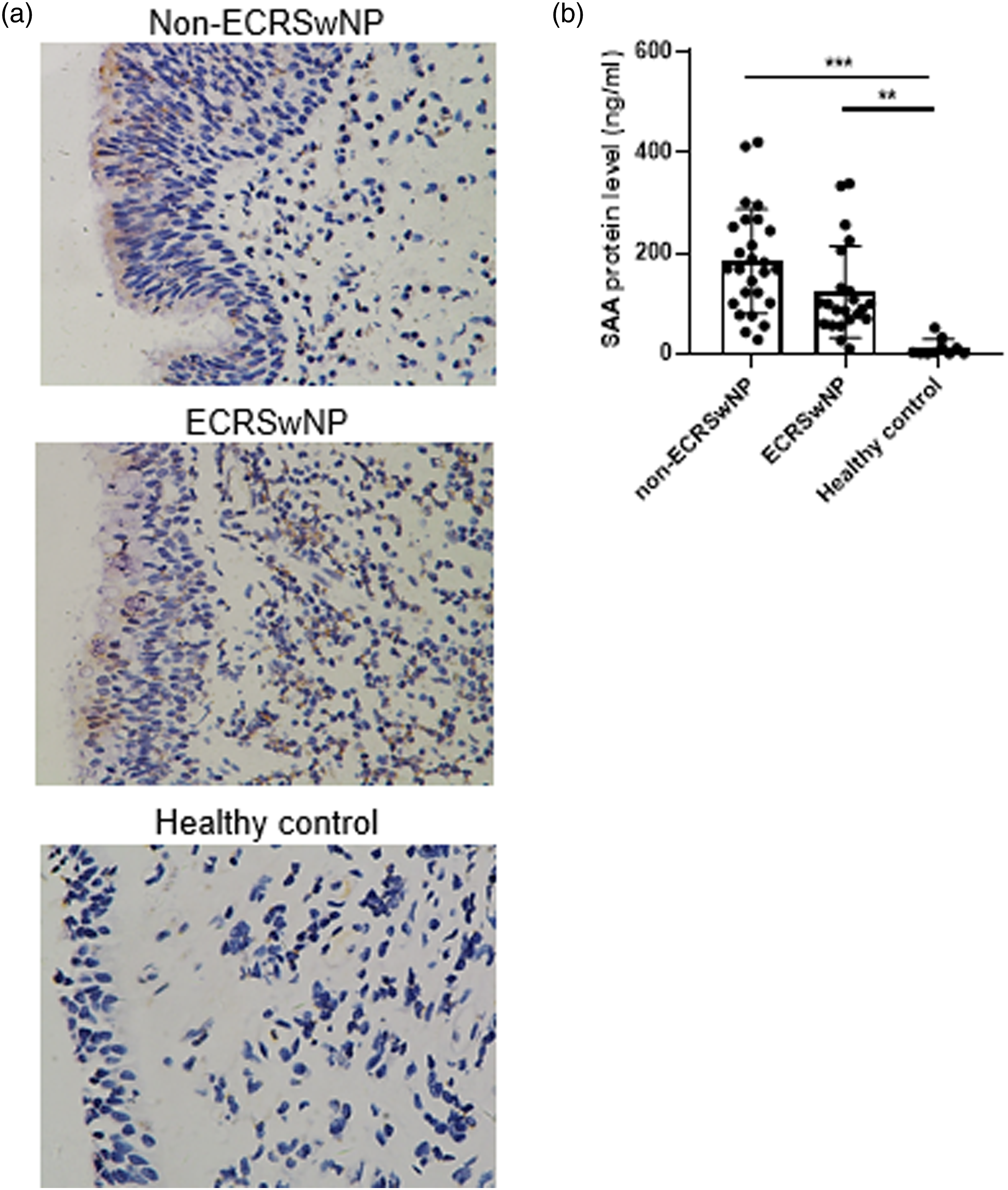
Next, we compared the expression of SAA in polyp tissues between CRSwNP patients with different disease control level. We found that the disease-controlled patients had the strongest immunoreactivity of SAA in polyp tissues (Figure 2A). ELISA analysis confirmed that the SAA level was significantly higher in polyp tissues of the disease-controlled patients than those of the partly controlled and uncontrolled (P < .001 and .01, respectively) (Figure 2B). There was no significant difference in the level of SAA in polyp tissues between the partly controlled and the uncontrolled. Differential expression of SAA in CRSwNP patients with different disease status. (A) Representative immunohistochemical staining of SAA in CRSwNP patients with controlled, partly controlled, and uncontrolled status. Original magnification: ×400. The protein level (B) of SAA in CRSwNP patients with controlled, partly controlled, and uncontrolled status assessed by ELISA. ***P < .001.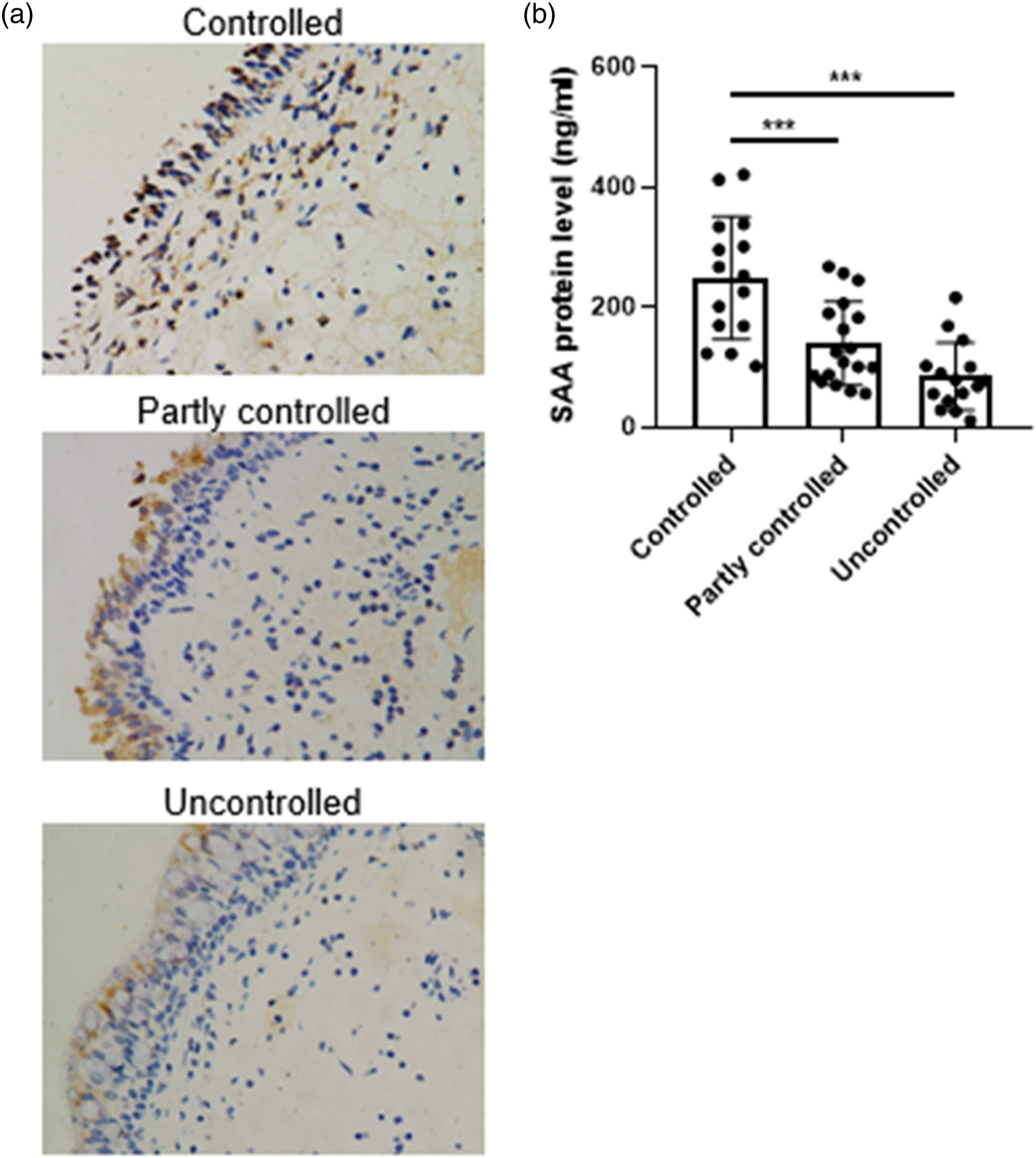
We further performed the ROC curve analysis to determine the predictive ability of tissue SAA level (Figure 3). The results showed that the area under the curve (AUC) of tissue SAA protein level is .8727 (95% CI .7736-.9719), suggesting a strong predictive value for disease-controlled status. When the tissue SAA protein level was 114.9 ng/mL or higher, the sensitivity and specificity were 93.33% and 63.64%, respectively. The ROC curve for polyp SAA protein levels. AUC of polyp SAA protein level was .8727 (95% CI .7736-.9719). ROC, receiver operating characteristic; SAA, serum amyloid A; AUC, area under the ROC curve; CI, confidence interval.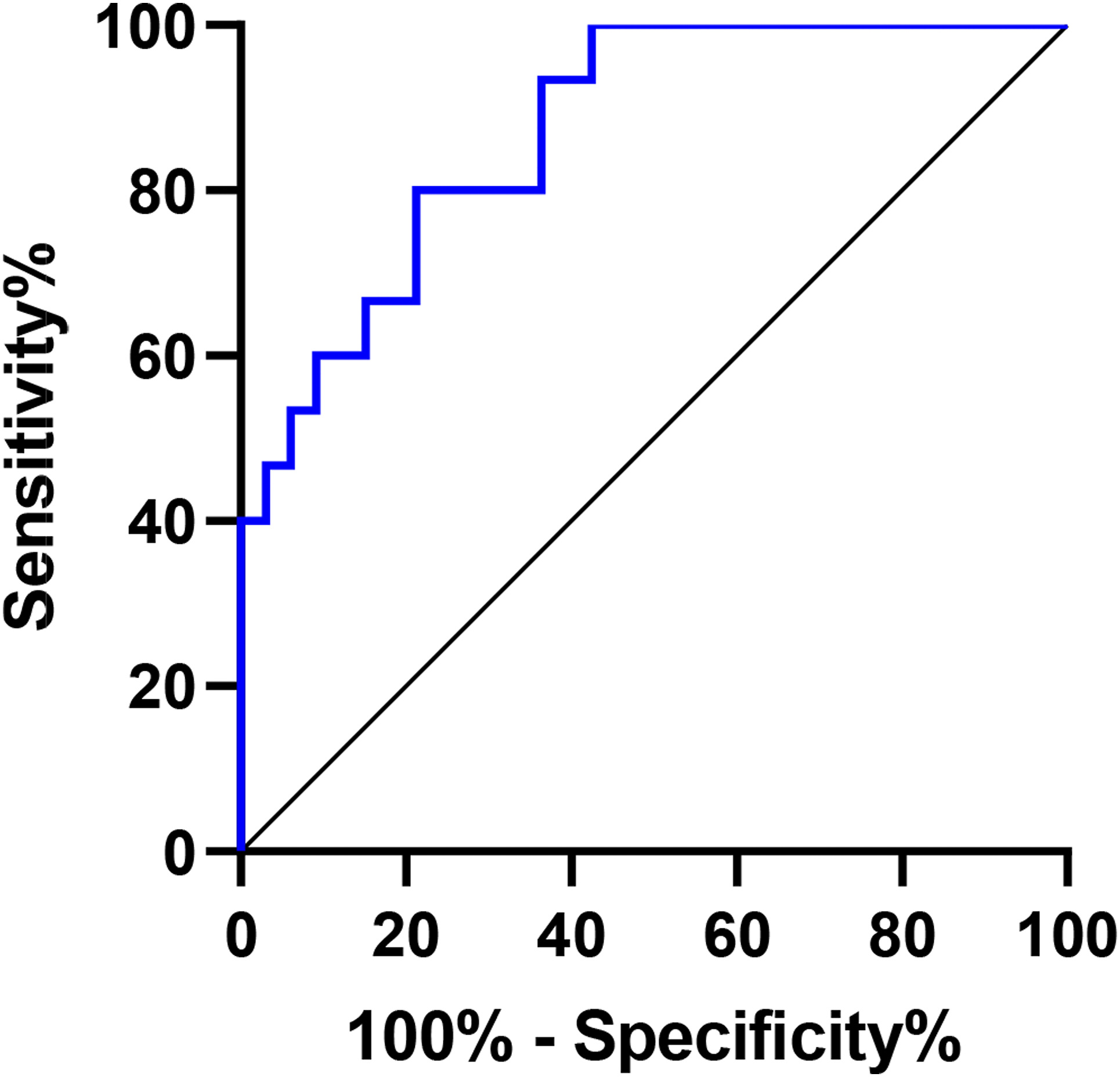
Characteristics of CRSwNP Patients With High SAA Expression
To characterize the CRSwNP patients with high SAA expression, we next subdivided the CRSwNP patients into SAAhigh and SAAlow subgroups according to the SAA cut-off value of 114.9 ng/mL in polyp tissues. As a result, there were 22 and 26 SAAhigh and SAAlow CRSwNP patients, respectively. Surprisingly, we found that the tissue eosinophil count and percentage were significantly higher in the SAAhigh CRSwNP patients than those in the SAAlow CRSwNP patients (Figure 4 and Table 1, both P < .05). No significant difference in age, gender, smoking status, percentage of allergic rhinitis and asthma, preoperative CT score, and blood eosinophil numbers were observed. Different eosinophilic numbers in the SAAhigh and SAAlow polyp tissues. HPF high power field. Comparison of Demographics, Comorbidities, and Clinical Features Between Patients in SAAhigh and SAAlow Subgroups. Abbreviations: SD, standard deviation; IQR, interquartile range; HPF, high power field; SAA, serum amyloid A.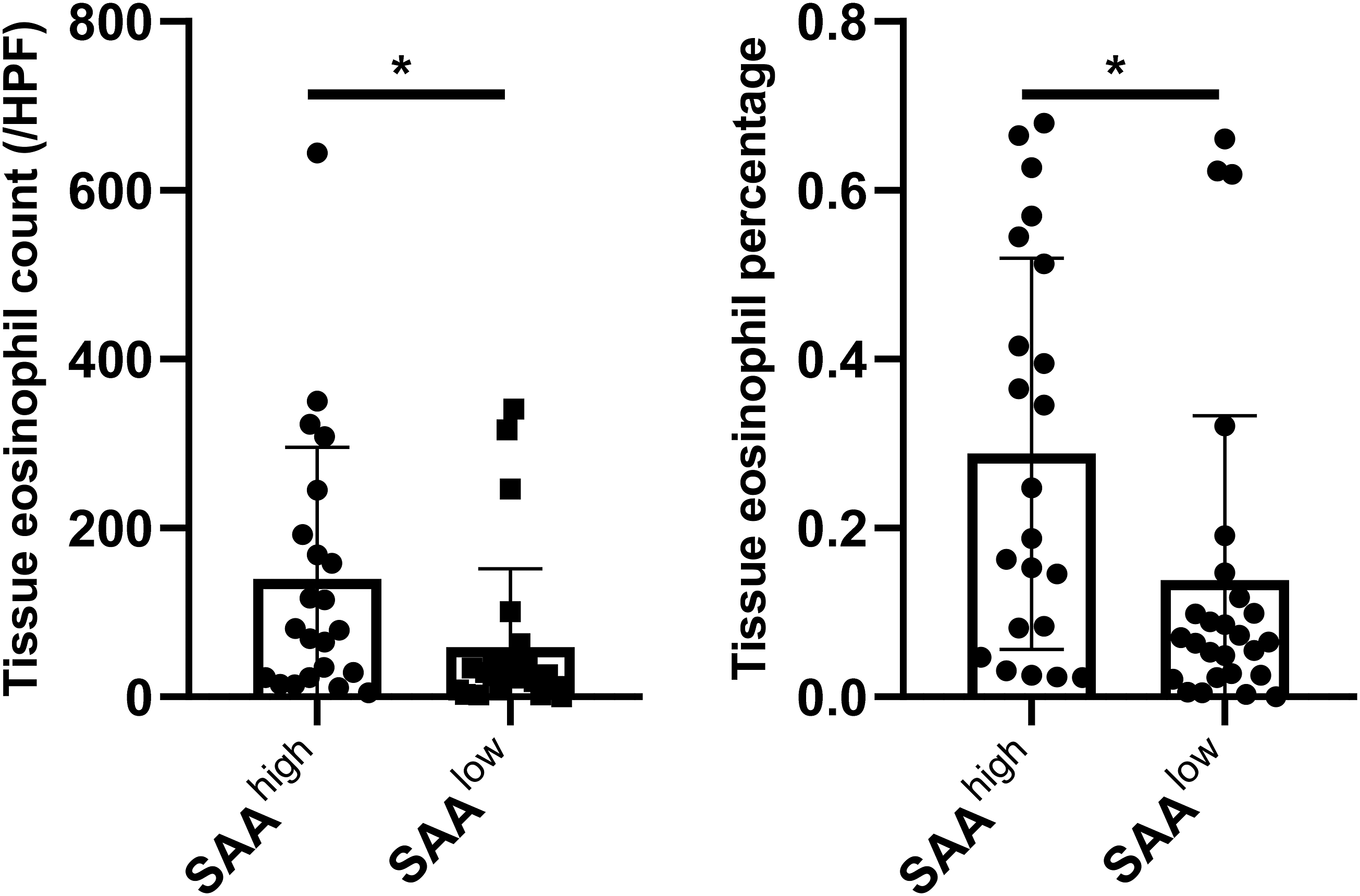
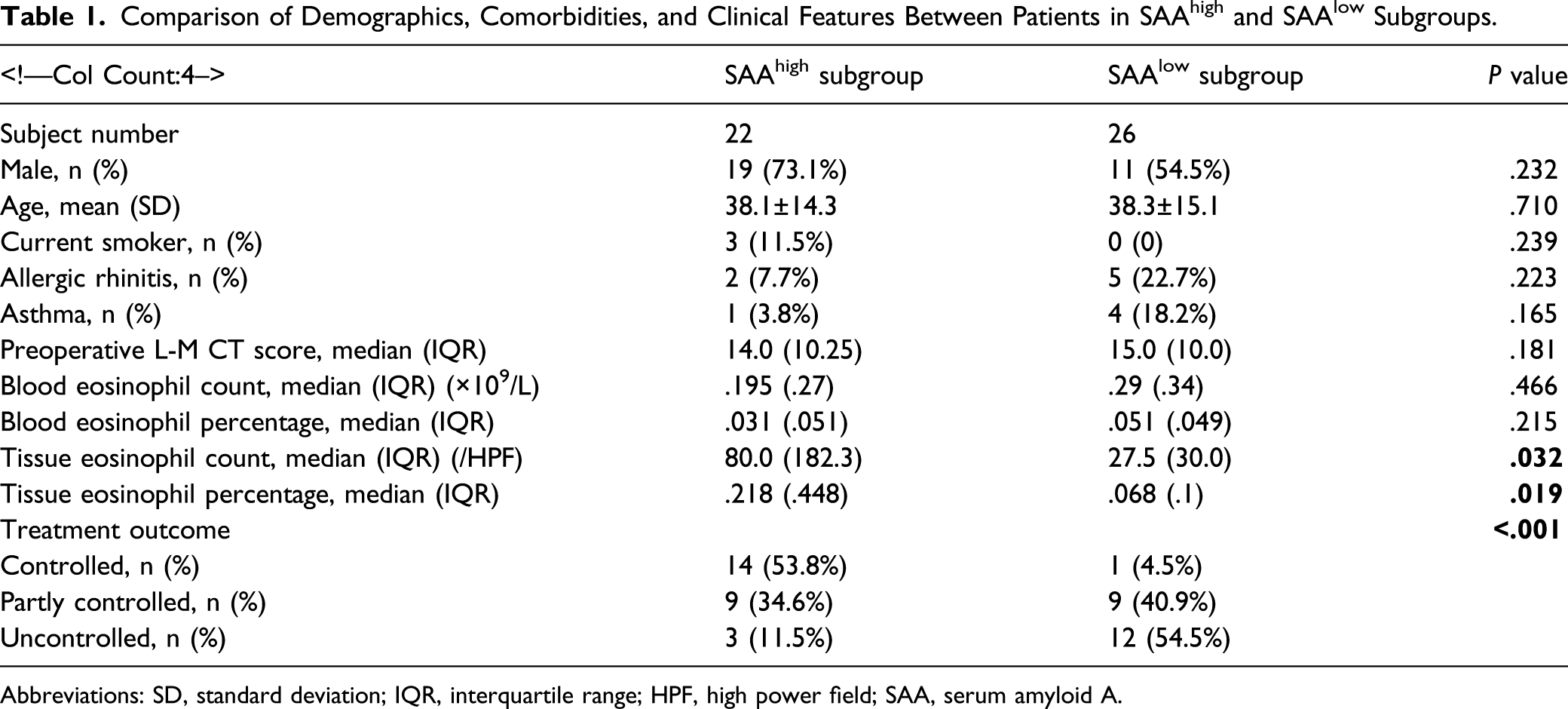
The disease-controlled patients accounted for 53.8% and 4.5%, whereas the disease-uncontrolled patients 11.5% and 54.5% of patients in SAAhigh and SAAlow subgroup, respectively (Figure 5 and Table 1). The percentages of patients in the 3 categories of disease control status were significantly different between SAAhigh and SAAlow subgroup (Table 1). Distinct treatment outcome between the SAAhigh and SAAlow CRSwNP patients. The percentages of patients in the 3 categories of disease control status were significantly different between SAAhigh and SAAlow subgroup (P<.001).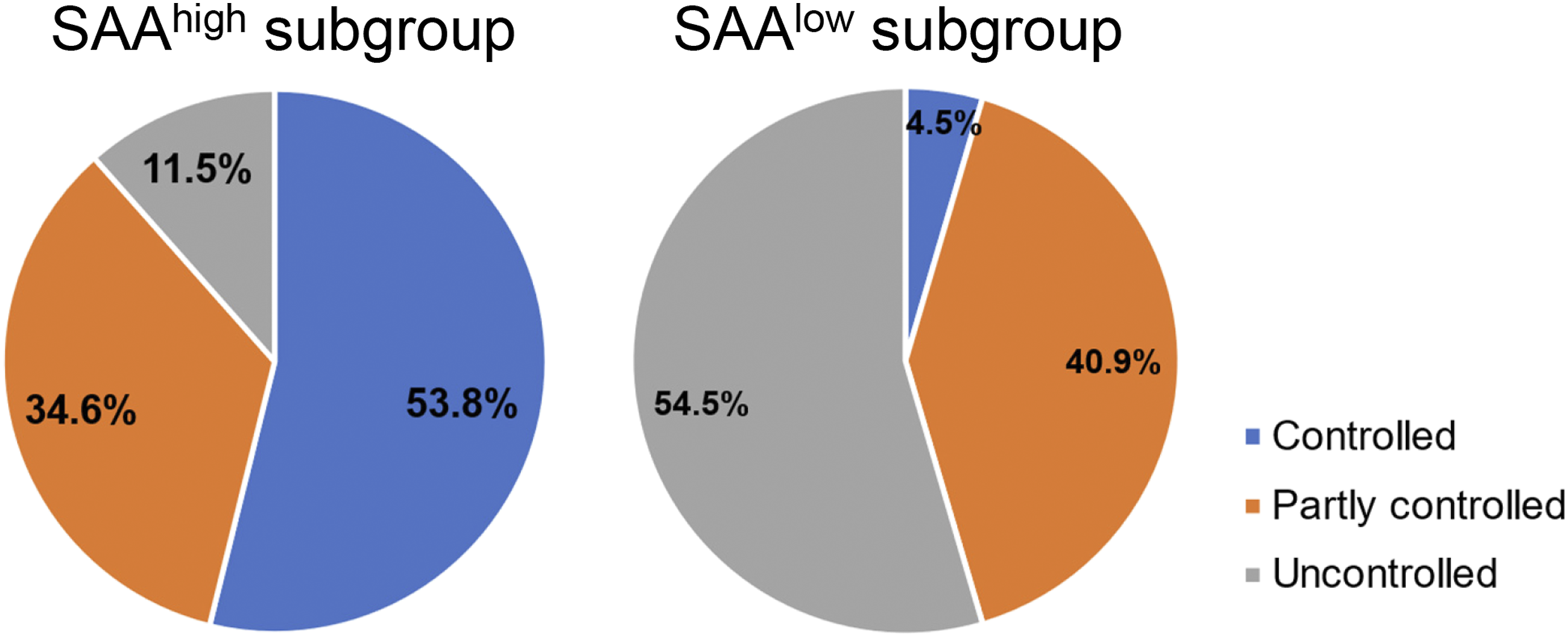
Discussion
Our previous studies reported that SAA expression is remarkably elevated in CRSwNP patients, and its level in polyp tissues has potential value in predicting systemic corticosteroid insensitivity in patients with CRSwNP.17,25 Here, we extended our previous studies and found that the tissue SAA levels were significantly higher in both non-ECRSwNP and ECRSwNP compared to the healthy subjects. In addition, the tissue SAA levels were further elevated in the patients with disease-controlled compared to those with the disease partly controlled and uncontrolled. Furthermore, ROC and AUC analysis showed that the SAA expression level was identified as a reliable predictor of the disease-controlled patients with CRSwNP. At a SAA level of 114.9 ng/mL or higher for predicting patients with disease-controlled status, the sensitivity reached 93.33%, and the specificity was 63.64%. These preliminary results suggest that the SAA level in polyp tissues could be served as a supplementary predictor for disease control assessment in patients with CRSwNP after endoscopic sinus surgery.
Chronic rhinosinusitis with nasal polyps with different pathological endotypes could display significant differences in their treatment outcome after comprehensive medical therapy and endoscopic sinus surgery. Therefore, identification of additional clinical parameters and biomarkers that could supplement the current understanding of clinical response prediction in patients with CRSwNP would greatly improve the therapy and follow-up of the disease. For this purpose, previous studies have revealed several predictors of the patients with disease uncontrolled status or polyp recurrence, including tissue and blood eosinophils,7,34 Charcot–Leyden crystals, 35 interleukin (IL)-5, 36 sinus CT score, 37 comorbid asthma. 38 However, few studies have explored factors related to the patients with good controlled status. In the present study, we showed that the SAA level in nasal polyps had a high diagnostic accuracy in identifying the patients with disease-controlled status, adding the potential clinical application value of measurement of tissue SAA for the prediction of corticosteroid insensitivity as well as supplementing the current prediction parameters. Additionally, to our knowledge, our study also provided the first evidence that increased tissue SAA levels is associated with a better prognosis in patients with CRSwNP after endoscopic sinus surgery.
During various inflammatory conditions, SAA is mainly produced in the liver and plays an important role in host defense by chemoattracting innate and adaptive immune cells, such as monocytes, neutrophils, and T lymphocytes, and inducing the production of proinflammatory cytokines, such as IL-1β, IL-8, and TNF-a, through binding to the SAA receptor formyl peptide receptor-like 1.14,20 Recent studies explored its potential value as a biomarker for translational applications. Lin et al revealed that serum SAA levels were elevated in patients with acutely exacerbated COPD and could be used as an effective index for the diagnosis and severity assessment of acutely exacerbated COPD. 39 Our group showed that SAA levels in polyp tissues could serve as a biomarker for the prediction of corticosteroid insensitivity in patients with CRSwNP. 25 In the current study, we further showed that tissue SAA levels were able to predict the CRSwNP patients with disease-controlled status after endoscopic sinus surgery with a high accuracy. Together, these studies highlight the promising application of SAA in serving as a potential diagnostic biomarker in CRSwNP.
In our previous study, we showed that tissue SAA level was positively associated with the neutrophil number in patients with CRSwNP. 25 This, together with the findings of the present study, is somewhat conflicted with recent endotype-focused studies. Delemarre et al showed that severe eosinophilic type 2 CRSwNP was frequently associated with a severe neutrophilic inflammation. 40 Furthermore, Kim et al found that tissue neutrophilia was associated with disease refractoriness in patients with CRSwNP. 41 However, similar findings were not observed in the study by Rosati et al, where neutrophilic infiltration and IL-8 expression did not correlate with the rate of polyp recurrence in CRSwNP patients. 42 The reason for these discrepancies remains unclear. However, it is possible that SAA, mainly produced by epithelial cells and acts as an upstream signal of neutrophilic inflammation, is unable to comprehensively represent the neutrophilic inflammation in CRSwNP, where reciprocal interactions between eosinophils and neutrophils play an important role in the disease persistency and severity. 43
In addition, we surprisingly found that the SAAhigh CRSwNP patients had higher numbers of tissue eosinophils than those of the SAAlow CRSwNP patients (Figure 4 and Table 1). This seems to conflict with previous observations that the patients with high eosinophil numbers tend to have higher disease recurrence rates.34,44 However, it should be noted that the tissue eosinophil counts and percentages of the SAAhigh CRSwNP patients were still significantly lower than their counterpart of the eosinophilic CRSwNP (Supplementary Table 2). Further prospective studies are needed to compare the difference in the relationship between SAA, eosinophils, and the treatment outcome in CRSwNP patients.
The present study is subjected to several limitations. The major one is the retrospective nature of the present study, in which a systematic selection bias could be introduced, thus possibly contributing to the observation that the SAAhigh CRSwNP patients had higher numbers of tissue eosinophils than those of the SAAlow CRSwNP patients. Moreover, the patients in this study were all recruited from a tertiary academic hospital and may not be representative of other medical care settings. Although we showed that measurement of tissue SAA level was able to identify the disease-controlled patients with CRSwNP, its clinical predictive value remained to be validated. In the future, a multi-center and prospective follow-up study with a larger cohort is eagerly warranted.
In conclusion, increased tissue SAA levels are associated with a better prognosis in patients with CRSwNP after endoscopic sinus surgery. Measurements of SAA in polyp tissues may provide useful information for evaluating CRSwNP conditions, especially identifying the CRSwNP patients with disease-controlled status after endoscopic sinus surgery.
Supplemental Material
Supplemental Material - Association Between Serum Amyloid A Expression and Disease Control after Endoscopic Sinus Surgery in Patients With Chronic Rhinosinusitis With Nasal Polyps
Supplemental Material for Association Between Serum Amyloid A Expression and Disease Control after Endoscopic Sinus Surgery in Patients With Chronic Rhinosinusitis With Nasal Polyps by Hangui Lu, Haiyan Liu, Kanghua Wang, Jianbo Shi and Yueqi Sun in Ear, Nose & Throat Journal
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grants from the Guangdong Basic and Applied Basic Research Foundation (2020A1515010134 and 2019A1515011029).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
