Abstract
Oncocytoma arising from the parotid gland and extending into the parapharyngeal space (PPS) has not been previously reported. A 64-year-old woman presented with a large slowly growing mass extending from the parotid to the submandibular area, expanding medially to displace the pharynx across the midline. Core-needle biopsy revealed an oncocytoma in the PPS measuring 120 × 88 × 60 mm in size. Although the tumor was of an unprecedentedly large size and extended into multiple spaces, it could be removed via a cervical–parotid approach without osteotomy. The resected tumor was again diagnosed as oncocytoma. A postoperative complication was weakness of the ipsilateral facial nerve, which almost completely resolved in 6 months. No recurrence has been noted on 1 y follow-up. We were able to resect an extremely large oncocytoma arising from the parotid gland without osteotomy.
Introduction
Tumors derived from the parapharyngeal space (PPS) are rare and account for only .5% of all head and neck neoplasms. 1-3 The majority of PPS tumors are benign. Malignant tumors account for 12.7%-20%. 3,4 Their proximity to important nerves and blood vessels, such as the lower cranial nerves and internal carotid artery, restricts the surgical field, making surgery difficult and prone to postoperative complications. 5
Oncocytoma is a rare benign epithelial tumor that occurs in the salivary glands, kidneys, and thyroid gland. 6 It is reported to account for <1.5% of major salivary gland tumors. 7-9 This tumor is reported to rarely undergo malignant transformation 10 and is treated by resection. It is well known that salivary gland tumors such as pleomorphic adenoma (PA) frequently occur in the prestyloid compartment of the PPS. 3 However, our previous case series of 120 PPS tumors 4,11 and several systematic reviews, including a summary of 1293 cases of PPS tumor, 12 included no cases of oncocytoma. Since only 2 cases have been reported 13,14 and no details of the surgery were given, its characteristics remain unclear.
Here, we report surgery performed on a giant PPS oncocytoma measuring 120 × 88 × 60 mm and extending from the deep lobe of the parotid gland to the PPS, masticatory muscle space, and posterior cervical space. Osteotomy was unnecessary. This is the first detailed report of surgery for an oncocytoma in the PPS and is the largest PPS tumor reported to date.
Report
A 64-year-old woman presented to our institute complaining of a gradually enlarging painless mass extending from the parotid to the submandibular region over 10 y. She had no other symptoms. Her past medical history was unremarkable. Clinical examination revealed an elastic hard mass extending from the region of the parotid to the submandibular area with swelling at the left lateral pharyngeal wall. Magnetic resonance imaging (MRI) showed a mass measuring 120 × 88 × 60 mm in size in the deep lobe of the left parotid gland extending into the left PPS. The mass was a lobulated solid lesion with well-defined borders which extended from the outer base of the skull to the hyoid bone, displacing the masticator space anteromedially, the lateral pharyngeal wall medially, and the internal carotid artery (ICA) posteriorly. The tumor displayed slight hyperintensity on T1-weighted images and hypointensity on T2-weighted images. (Figure 1). Fine-needle aspiration cytology (FNAC) showed cells with eosinophilic granules in the cytoplasm, suggesting oncocytoma or Warthin’s tumor; however, a definitive diagnosis could not be made. The histopathological diagnosis of a core-needle biopsy (CNB) was oncocytoma. Since the lesion was large and extended widely, we selected the cervical–parotid approach as our primary method, with a mandibular swing approach with mandibulotomy and subtemporal fossa approach with zygomatic transection if needed. A skin incision was made from the preauricular area to below the mandible, and a superficial parotidectomy was performed to attain an adequate surgical field and preserve the facial nerve. The deep lobe of the parotid gland with the tumor was then separated from the facial nerve (Figure 2A). The submandibular gland was separated from the tumor, and the operative field was widened by cutting the stylomandibular ligament. This allowed the tumor to be easily detached from surrounding tissues. After detaching it from the medial pterygoid muscle, the tumor was detached from the skull base with blunt dissection, and the other parts of the tumor were naturally delivered and removed (Figure 2B and 2C). A large dead space was filled with a free anterior lateral thigh flap that was largely de-epithelialized with attached muscle (Fig. 2D). Closed drains were placed. A small part of the transplanted flap was exposed on the surface for blood supply monitoring, and the wound was closed. Tracheostomy was performed at the end of the operation. The time to remove the tumor was 187 minutes. Total operating time was 609 minutes and total hemorrhage volume was 718 mL. On postoperative day (POD) 5, blood flow to the flap was impaired, and it was removed by reoperation. Oral intake, closure of the tracheostomy, and discharge occurred on POD 10, 14, and 21, respectively. Preoperative MRI (A): axial T1-weighted image, (B): axial T2-weighted image, (C): coronal contrast-enhanced T1-weighted image. The large tumor arises from the deep lobe of the left parotid gland (black arrow) and extends into the prestyloid compartment of the left PPS through the stylomandibular tunnel (black arrowhead). The left ICA is displaced posteriorly (white arrow). Tumor lobulations displace the masticatory space anteromedially and displace the pharynx across the midline (white arrowhead). (A): The deep lobe of the parotid gland with the tumor is separated from the facial nerve (arrow). (B) and (C): The tumor (arrow) was easily detached from surrounding tissues and delivered out. (D): The large dead space was filled with a free flap from the thigh that was largely de-epithelialized (arrow) with attached muscle. The flap was later removed due to ischemia. (E): Macroscopically, the tumor was a nodular, circumscribed lesion measuring 120 × 70 × 40 mm in size. (F) and (G): Microscopic findings revealed an encapsulated tumor surrounded by a thin fibrous capsule. The tumor cells consisted of homogeneous eosinophilic cytoplasm and showed no atypia (hematoxylin/eosin stain, B: × 40 and C: × 400).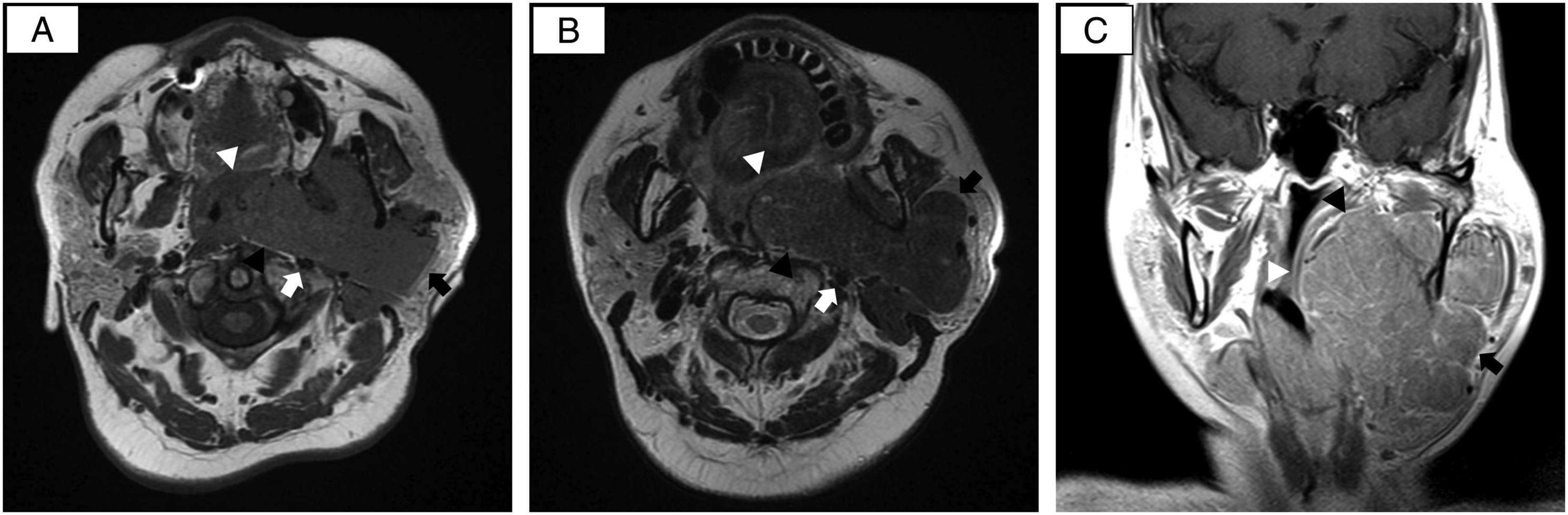
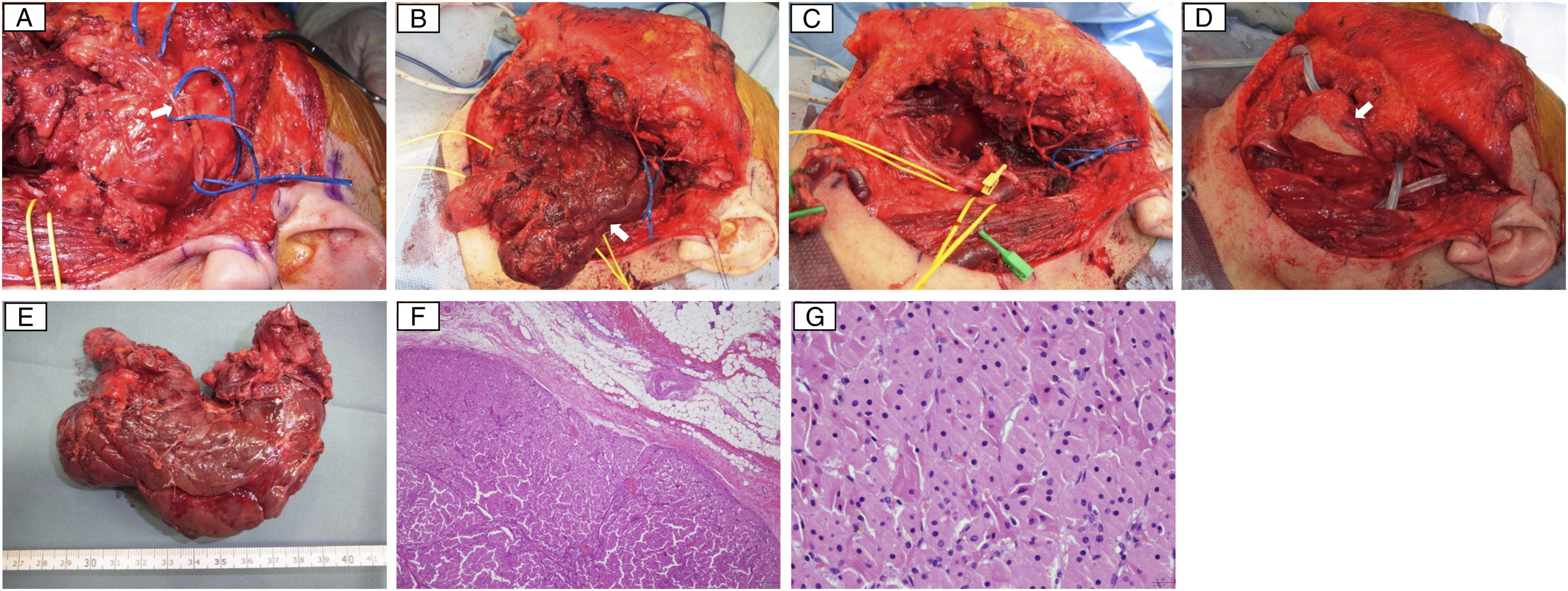
The resected tumor was 120 × 70 × 40 mm in size (Figure 2E), pathologically diagnosed as oncocytoma, and had negative margins (Figure 2F and 2G). The postoperative course was complicated by a facial palsy, which improved to only slight weakness of the descending branch 6 months after surgery. No recurrence has been noted on 1 y follow-up.
Discussion
In this report, we described the removal of a very large oncocytoma in the PPS. We suggest that oncocytomas are easy to separate from the surrounding tissue and that it may be possible to remove them without the need for osteotomy, even if extremely large. To our knowledge, this is the first report to describe surgical details in the removal of an oncocytoma in the PPS.
Preoperative diagnostic imaging revealed a PPS tumor arising from the prestyloid compartment, which was thought to have originated from the deep lobe of the parotid gland, and the ICA was displaced posteriorly. Tumor lobules extended into the masticatory muscle space and the posterior cervical space. This pattern of development is uncommon in PPS tumors of other histological types and may be ascribable to the soft, lobulated characteristics of oncocytoma.
Fine-needle aspiration cytology does not give a definitive histological diagnosis, and with this large size and extension pattern, and with bone transection such as mandibulotomy required for high-grade cancers, a preoperative CNB was performed to be certain of the histology. According to our report on FNAC for PPS tumors, 4 histology based on FNAC has an accuracy rate of 95.2% for benign and malignant tumors, and most cases of PA are correctly identified by histological diagnosis. For tumors other than PA, however, FNAC does not usually provide a diagnosis. It has been suggested that CNB is useful when the histological diagnosis indicates a surgical strategy.
A cervical–parotid surgical approach is recommended for lesions that extend to the base of the skull. 3 Mandibulotomy is recommended to establish an adequate surgical field for larger lesions. 15 In our present case, the tumor not only occupied a part of the deep lobe of the parotid gland but also extended to the vicinity of the skull base, so a cervical–parotid approach was used to preserve the facial nerve and obtain a wide surgical field. To ensure safe surgery on this unusually large tumor, we planned for the possibility of mandibulotomy and zygoma transection in the case that we encountered difficulty with the cervical–parotid approach. However, since the tumor was soft and easily detached, blind blunt dissection, including detachment from the skull base, was performed safely.
Facial palsy was an adverse event. This was most likely due to the complex detachment of all branches of facial nerve from the parotid gland out to the periphery, done both to obtain an adequate surgical field and to ensure nerve preservation.
Since the tumor was of unprecedented size and the dead space left behind was large, a de-epithelialized free flap and muscle was used to fill the defect. However, flap blood flow insufficiency occurred and the flap was removed by reoperation on the fifth postoperative day. In general, flap blood flow insufficiency occurs early after surgery and rarely occurs as late as on the fifth postoperative day. 16 The cause was considered to be gradual compression of the single penetrating branch flowing into the flap by the surrounding tissue. Unlike the dead space created by resection with wide surgical margins in malignant tumors, removal of benign tumors does not result in as much tissue loss. The space may therefore be naturally filled by the surrounding tissue with indwelling closed drainage. Although dead space is generally known to cause delayed wound healing and infections, our case suggests that the defect created by extracapsular resection of benign tumors does not always require tissue transplantation. Indeed, the dead space in our case was filled with surrounding tissue and healed naturally.
Conclusion
In conclusion, this giant oncocytoma occurred in the PPS was a unique finding. A fuller understanding of the overall significance of our approach requires accumulation of further cases; nevertheless, we suggest that oncocytomas are easy to separate from the surrounding tissue and may be excised without osteotomy, and that even large dead spaces may fill with surrounding tissue.
Footnotes
Acknowledgments
The authors would like to show their greatest appreciation to Associate Professor Ryutaro Ukisu for his support in diagnostic imaging. The authors thank Libby Cone MD, MA and Guy Harris DO from DMC Corp. (www.dmed.co.jp <![]() >) for editing drafts of this manuscript.
>) for editing drafts of this manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
