Abstract
Objectives
Laryngeal squamous cell carcinoma (LSCC) is a common squamous cell carcinoma of the head and neck with no reliable diagnostic biomarkers. However, recent studies have shown that inflammation plays an essential role in tumor development, and several inflammation-based biomarkers have been shown to have prognostic value. This study aimed to investigate the auxiliary value of fibrinogen (FIB), fibrinogen degradation products (FDP), and lymphocyte/monocyte ratio (LMR) in LSCC diagnosis and prognosis.
Methods
Clinical data from 218 patients recently diagnosed with LSCC and 207 diagnosed with benign laryngeal lesions (BLLs) were retrospectively reviewed. Potential diagnostic biomarkers were evaluated using univariate and multivariate analyses; receiver operating characteristic (ROC) curve analysis was used to identify cut-off values and diagnostic efficiency. Least absolute shrinkage and selection operator (LASSO) Logistic regression analysis was used to screen for independent risk factors to construct a diagnostic nomogram. The chi-squared test and Kaplan–Meier method were performed to investigate the correlation of clinicopathological characteristics and 3-year overall survival (OS) with FIB, FDP, and LMR in patients with LSCC.
Results
FIB, FDP, and LMR levels were significantly different between the LSCC and BLL groups (P < .001), and all were independent risk factors for LSCC. The area under the ROC curve of the diagnostic nomogram was .894. Additionally, FIB, FDP, and LMR were correlated with some invasive clinicopathological features, and LMR ≥4.29 was associated with reduced OS (P = .038).
Conclusion
FIB, FDP, and LMR demonstrated potential as biomarkers for the diagnosis and prognosis of LSCC; however, further studies are needed to confirm their efficacy.
Introduction
Laryngeal squamous cell carcinoma (LSCC) is one of the most common head and neck tumors, with various factors influencing the incidence of laryngeal carcinoma. 1 Treatment options for LSCC include surgery, radiotherapy, and chemotherapy. Although therapeutic modalities have evolved, the 5-year survival rate for LSCC has not improved significantly for decades, and is related to the low percentage of early diagnoses. 2 However, there are currently no specific tumor biomarkers that could be used as an auxiliary diagnostic index; therefore, biochemical and blood biomarkers are urgently required to predict the occurrence of LSCC.
Recent studies have shown that the occurrence and development of malignant tumors are accompanied by inflammation. The lymphocyte/monocyte ratio (LMR) is one of several indexes used to evaluate the systemic inflammatory response, which, to a certain extent, can reflect the dynamic correlation between inflammatory response and tumor immunity. 3,4 Fibrinogen is a soluble protein synthesized by liver cells. Accumulating evidence has shown that FIB and FIB degradation products (FDP) are closely involved in the development of tumors and are elevated in a variety of malignancies, including colorectal, lung, pancreatic, ovarian, and gastric cancers; moreover, hyperfibrinogenemia has been shown to be associated with poor prognosis of tumors. 5 -9 However, to our knowledge, there have been no reports describing the use of inflammation-related indicators (ie, FIB, FDP, and LMR) in the diagnosis of LSCC.
In the present study, clinical data from patients with LSCC were obtained from the First Affiliated Hospital of Anhui Medical University (Anhui, China). Univariate and multivariate analyses were used to screen for inflammation indicators. Subsequently, a diagnostic nomogram was constructed to preliminarily analyze the prognostic value of the indicators.
Patients and Methods
Patients
This study was approved by the First Affiliated Hospital of Anhui Medical University Ethics Committee. Clinical data from patients with laryngeal diseases, admitted to First Affiliated Hospital of Anhui Medical University between April 2017 and October 2020, were investigated. Inclusion criteria were as follows: benign laryngeal lesion (BLL) or American Joint Committee on Cancer tumor node metastasis (TNM) stage I to IV LSCC confirmed by postoperative pathology; availability of complete clinicopathological data; and provision of written informed consent to collect medical data. Individuals who had undergone surgery, chemotherapy, and radiotherapy before admission, those in whom the disease was complicated due to other malignant tumors, hematological diseases, active inflammatory diseases (eg, autoimmune disease and infection), liver and kidney diseases, or by the long-term use of oral anticoagulant drugs and corticosteroids were excluded. All patients underwent a basic physical examination, fibrolaryngoscopy, contrast-enhanced computed tomography scan of the head, neck, chest, upper gastrointestinal radiography, electrocardiography, and laboratory examination for comprehensive evaluation.
Data Collection
Fasting venous blood was collected from patients with laryngeal diseases in the morning, within 24 h of admission. Blood samples were used for routine blood tests, as well as assessment of FIB and FDP levels. Patient prognosis was primarily assessed based on outpatient reexamination and telephone follow-up. Follow-up is ongoing in this study.
Statistical Analysis
R and various packages were used for statistical analysis. Data with a non-normal distribution are expressed as median (minimum–maximum); the Wilcoxon rank-sum test was used to compare the groups. Logistic regression analysis was used to evaluate the correlation of FIB, FDP, and LMR with LSCC. Receiver operating characteristic (ROC) curve analysis was used to evaluate the diagnostic value of FIB, FDP, and LMR. The area under the ROC curves (AUC) was compared using the Z-test. Least absolute shrinkage and selection operator Logistic regression analysis was performed to evaluate independent diagnostic factors of LSCC. The relationship between FIB, FDP, and LMR and clinicopathological features of LSCC patients was evaluated using the chi-squared test. The Kaplan–Meier method was used to compare differences in 3-year overall survival (OS) between the high- and low-level groups, and statistical significance was determined using the log-rank test. Differences with P < .05 were considered to be statistically significant.
Results
Distinguishing Diagnostic Inflammatory Response Indicators in LSCC
A detailed flow diagram is presented in Figure 1. The study population included 425 patients with laryngeal diseases, including 207 with BLL and 218 with LSCC. Clinical data of these patients are summarized in Table 1. Flow chart of this study. Patient Demographics and Clinicopathological Characteristics. Abbreviations: LSCC, laryngeal squamous cell carcinoma; BLL, benign laryngeal lesion; TNM, tumor node metastasis.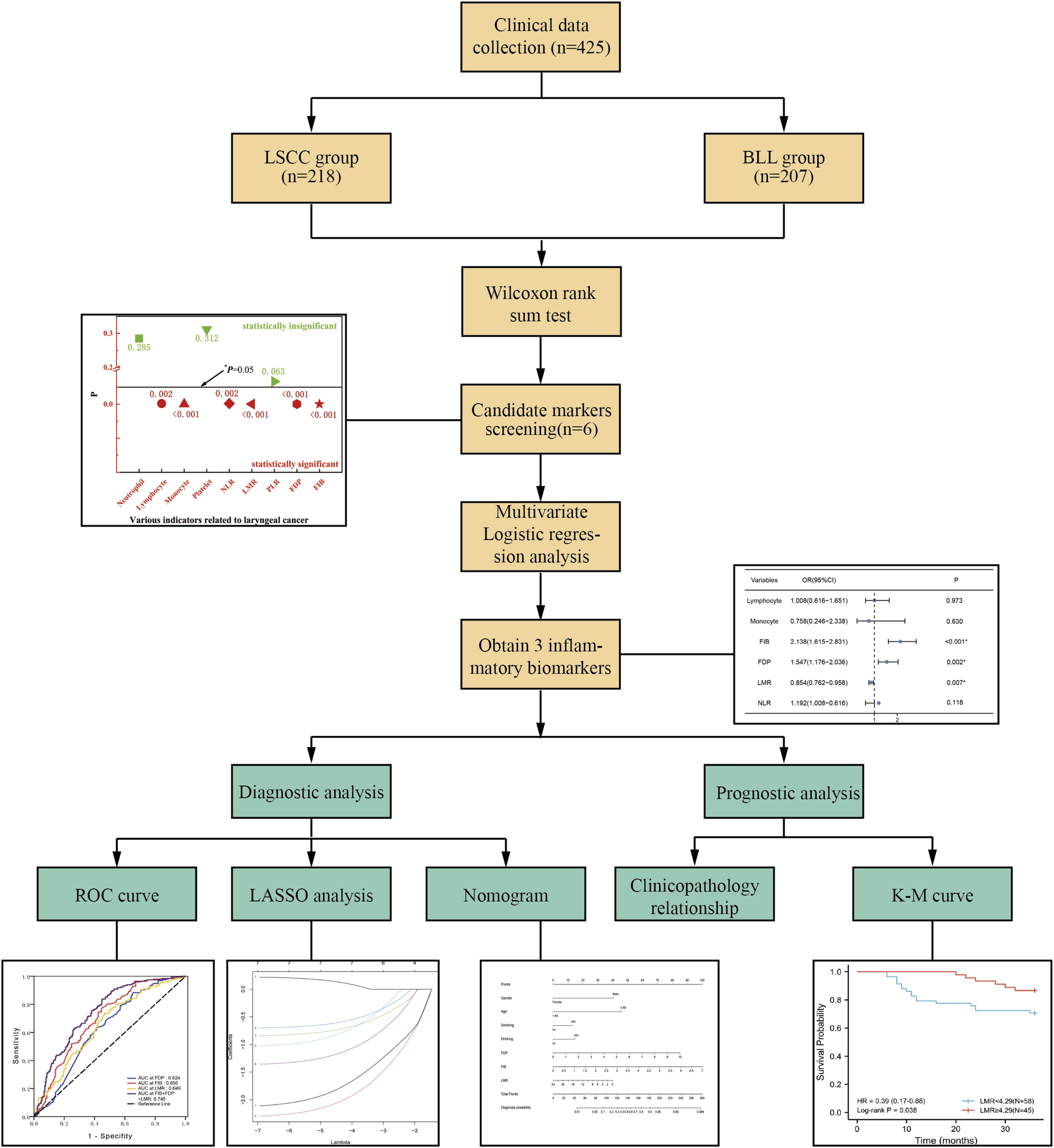
Comparison of Various Inflammatory-Related Biomarkers Between LSCC Group and BLL Group.
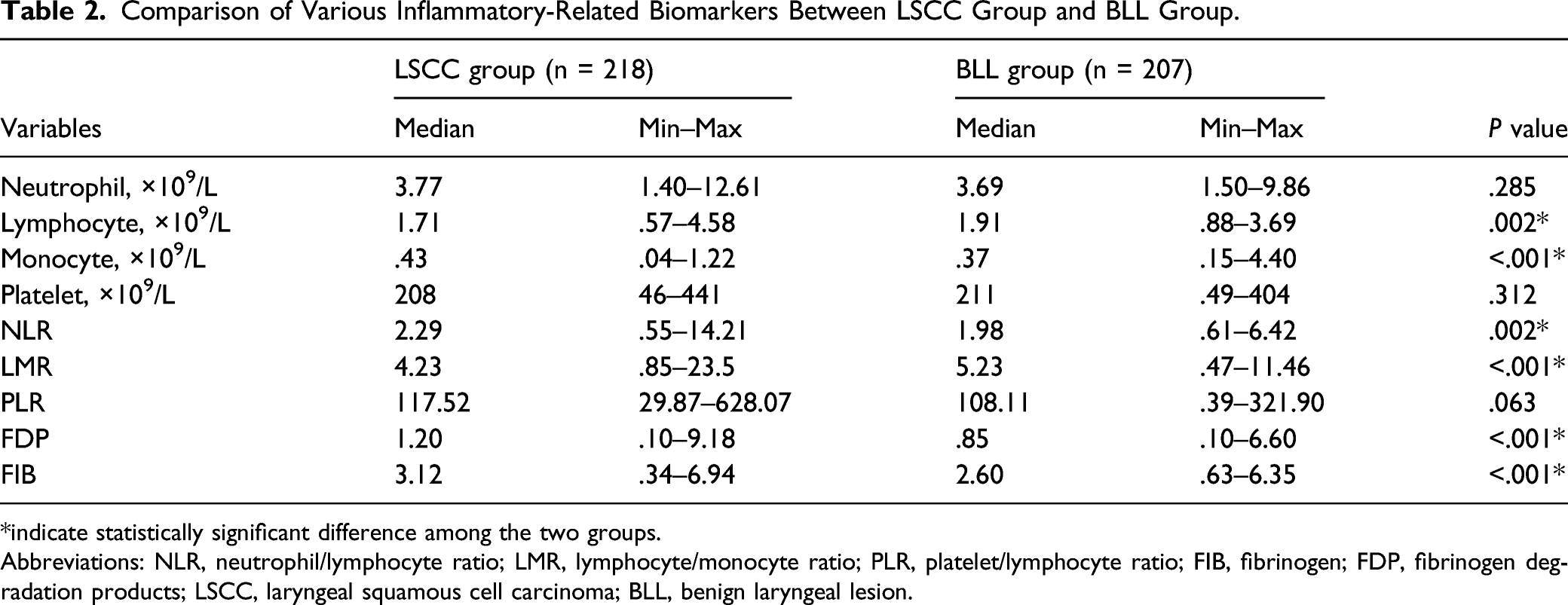
*indicate statistically significant difference among the two groups.
Abbreviations: NLR, neutrophil/lymphocyte ratio; LMR, lymphocyte/monocyte ratio; PLR, platelet/lymphocyte ratio; FIB, fibrinogen; FDP, fibrinogen degradation products; LSCC, laryngeal squamous cell carcinoma; BLL, benign laryngeal lesion.
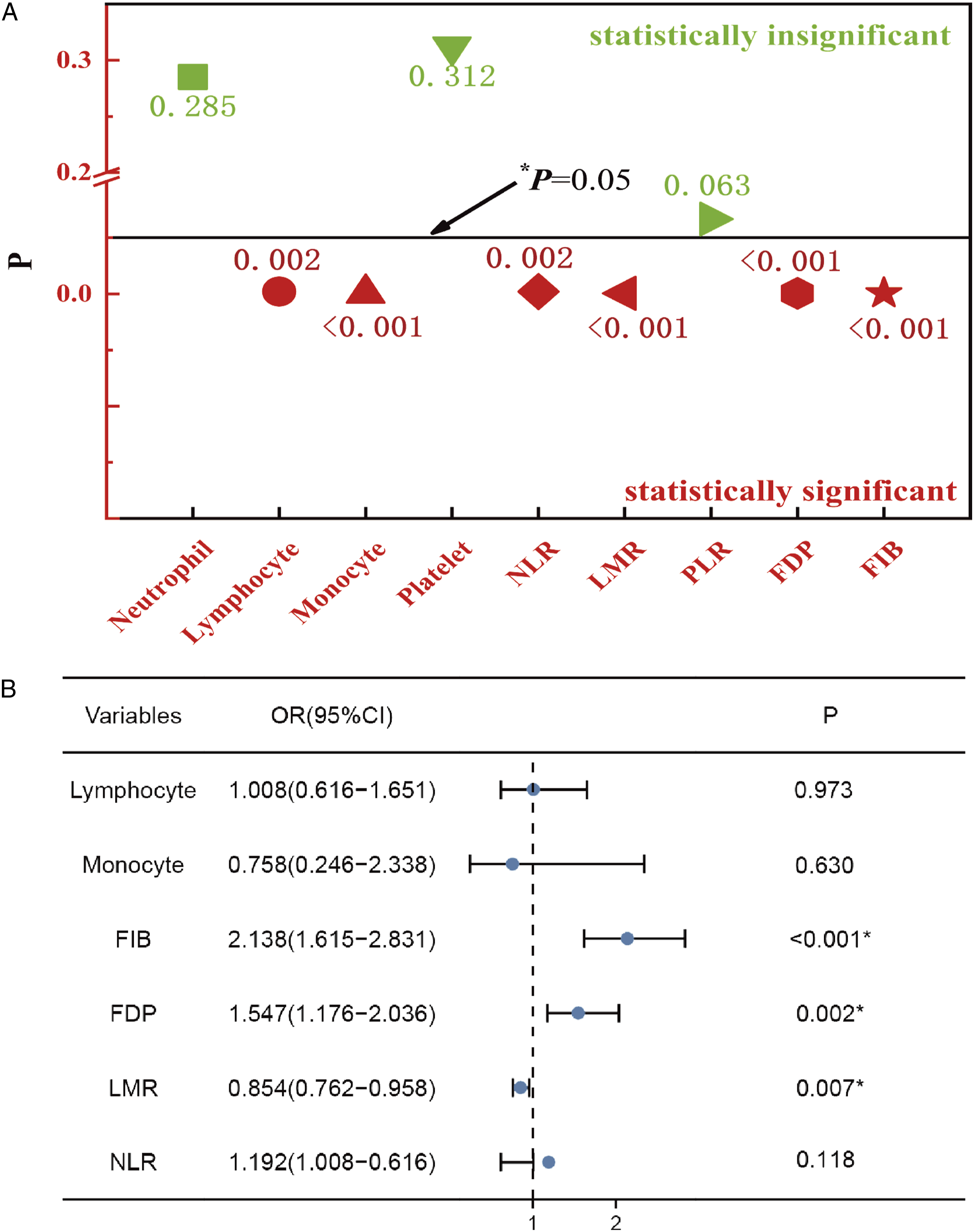
Candidate Markers screening. (A) P values for comparison of various markers between the two groups. (B) A forest map showed 3 indicators identified by Logistic regression analysis.
Efficacy of FIB, FDP, and LMR in Predicting Laryngeal Carcinoma
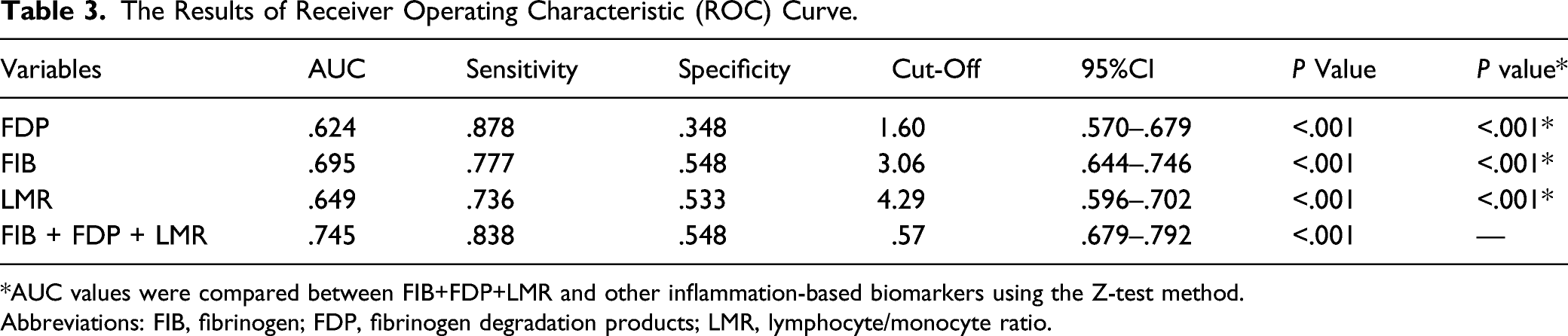
As shown in Figure 3, the predictive effect of the above independent risk factors was evaluated using ROC curve analysis, and the AUC and corresponding 95% confidence interval (CI) for FIB, FDP, and LMR in the diagnosis of LSCC were .695 (95% CI .644-.746), .624 (95% CI .570-.679), and .649 (95% CI .596-.702), respectively (P < .05) (Table 3). After combining the three indicators, diagnostic efficiency significantly improved (P < .001) (Table 3). The optimal cut-off values and corresponding sensitivity and specificity for the parameters mentioned above are listed in Table 3. ROC curve analysis for FIB, FDP, LMR, and FIB + FDP + LMR in LSCC diagnosis. Abbreviations: ROC, receiver operating characteristic; FIB, fibrinogen; FDP, fibrinogen degradation products; LMR, lymphocyte/monocyte ratio; LSCC, laryngeal squamous cell carcinoma. The Results of Receiver Operating Characteristic (ROC) Curve. *AUC values were compared between FIB+FDP+LMR and other inflammation-based biomarkers using the Z-test method. Abbreviations: FIB, fibrinogen; FDP, fibrinogen degradation products; LMR, lymphocyte/monocyte ratio.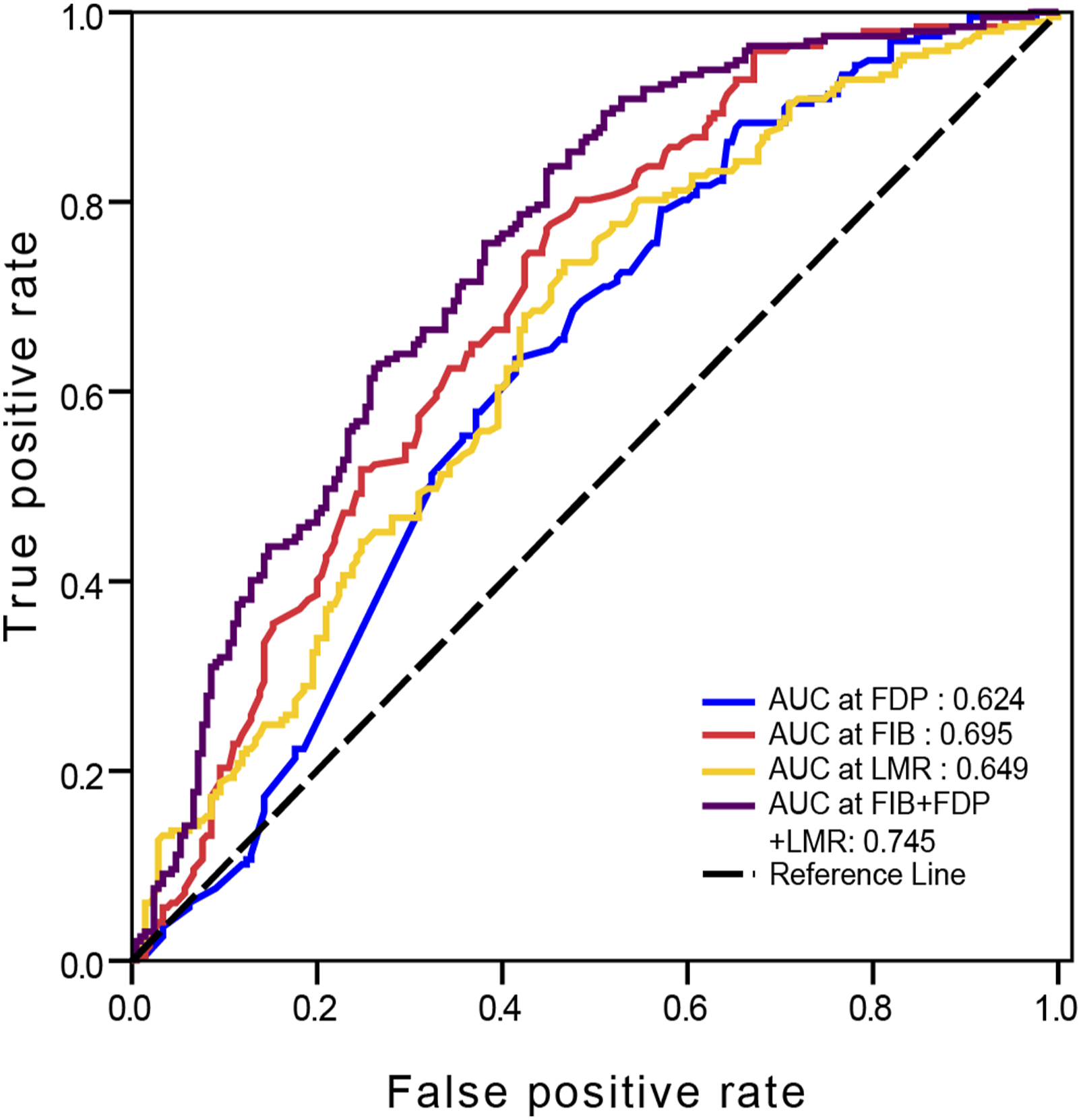
Independent Analysis and Nomogram Construction
To clarify whether the three markers were independent diagnostic factors for LSCC, the markers and other clinical features (age, sex, smoking, and drinking) were analyzed. LASSO Logistic regression analysis suggested that FIB, FDP, and LMR were significantly associated with LSCC occurrence (Figure 4A and 4B). In addition, age, sex, smoking, and drinking were confirmed to be independent factors of LSCC diagnosis (Figure 4A and 4B). A nomogram integrating the three markers and other clinical features was constructed to diagnose and assist clinicians in providing better treatment for patients with LSCC (Figure 4C). The AUC of the nomogram was .894 (Figure 4D), and a calibration curve was constructed to estimate the precision of the nomogram (Figure 4E). In addition, decision curve analysis demonstrated that the nomogram had good net benefits for clinical utility (Figure 4F). Construction and validation of diagnostic nomogram. (A) The LASSO coefficients profiles of 7 candidate diagnostic indicators. (B) Tuning parameter (λ) selection cross-validation error curve. (C) Nomogram for predicting the patients with LSCC. (D) ROC curve analysis of the nomogram. (E) Calibration curve plot for predicting LSCC. (F) DCA curves of the nomogram for LSCC diagnosis. Abbreviations: LSCC, laryngeal squamous cell carcinoma; ROC, receiver operating characteristic; LASSO, least absolute shrinkage and selection operator.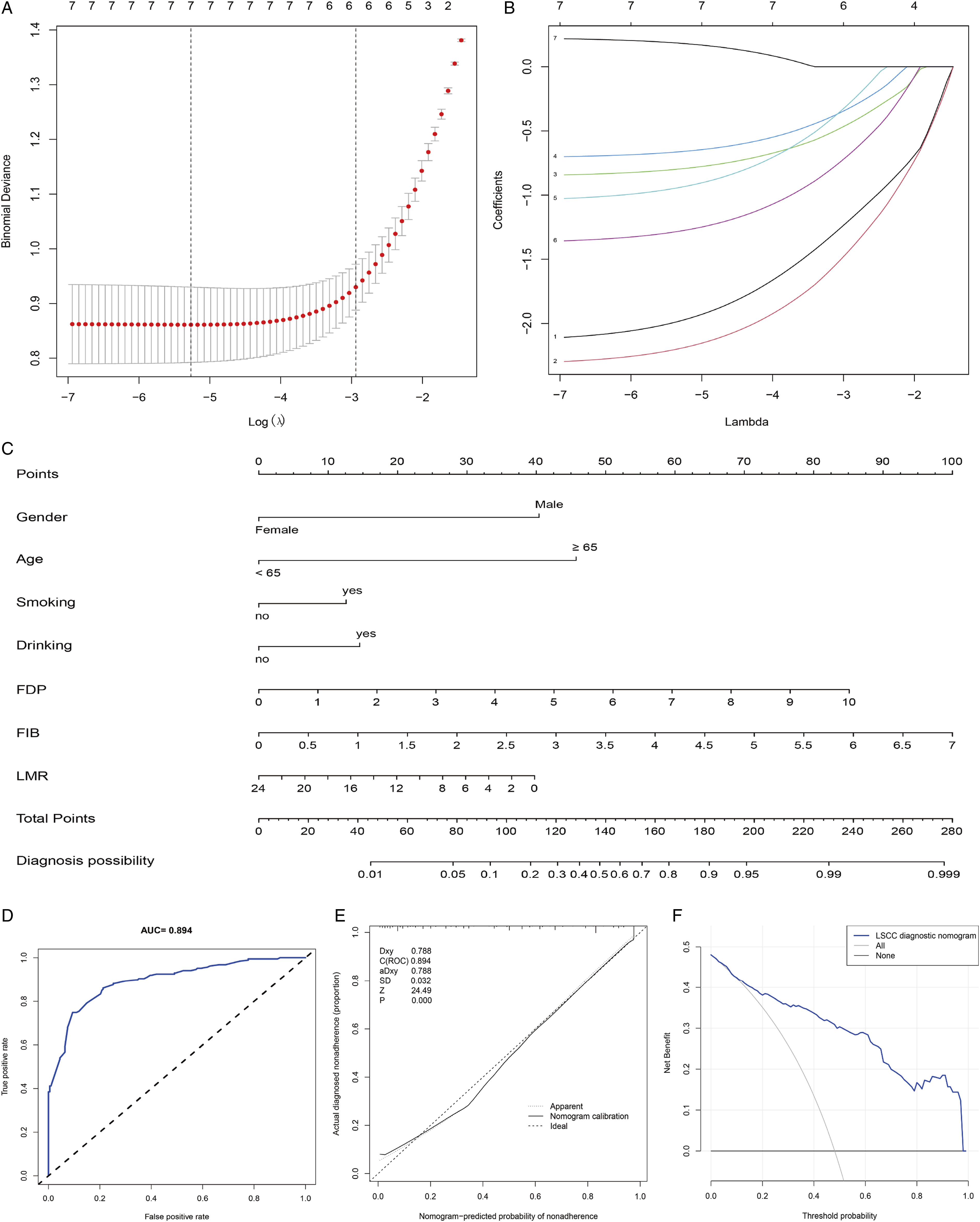
Correlation Between FIB, FDP, and LMR and Clinical Features and OS
Correlation Between Inflammation-Related Biomarkers and Clinicopathological Characteristics of LSCC Patients.
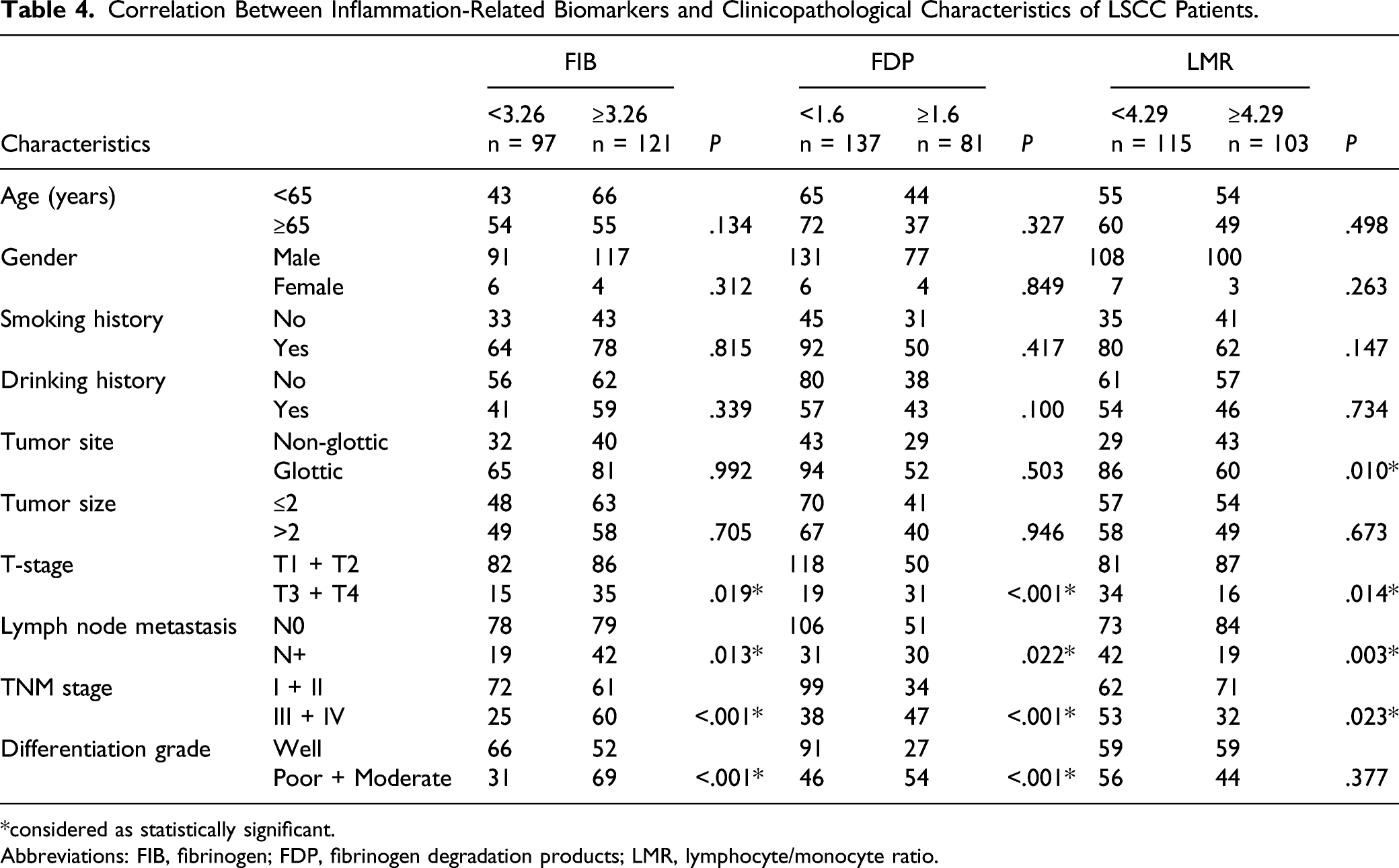
*considered as statistically significant.
Abbreviations: FIB, fibrinogen; FDP, fibrinogen degradation products; LMR, lymphocyte/monocyte ratio.
A total of 103 LSCC patients who completed the 3-year follow-up or died midway through were divided into high- and low-level groups according to the cut-off. The Kaplan–Meier curve illustrated that the patients with low LMR had a significantly shorter 3-year OS than their high-level counterparts (P = .038) (Figure 5C). Kaplan–Meier survival curves for the high- and low-level groups. (A) FDP. (B) FIB. (C) LMR. Abbreviations: FDP, fibrinogen degradation products; FIB, fibrinogen; LMR, lymphocyte/monocyte ratio.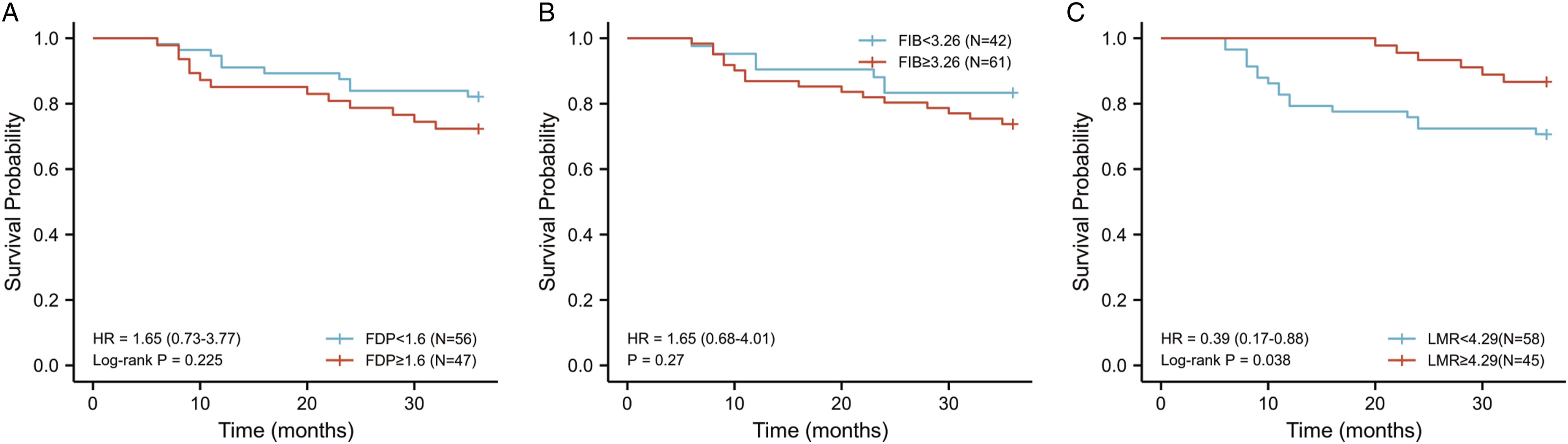
Discussion
Laryngeal squamous cell carcinoma is the most common squamous cell carcinoma of the head and neck. Despite the continuous development of therapeutic modalities, no significant improvement has been observed in 5-year survival rates of LSCC for decades, which may be explained by the difficulties in early diagnosis of laryngeal cancer. 2 An increasing number of studies have, for years, 10,11 reported that inflammation plays a key role in the occurrence, invasion, and metastasis of tumors. Serum markers associated with inflammation, such as NLR, platelet/lymphocyte ratio, and LMR, have been reported to perform well in predicting the prognosis of tumors. 3,12 However, inflammation-related indexes as diagnostic predictors of LSCC have not been developed. The results of the present study demonstrated that preoperative FIB, FDP, and LMR were independent factors affecting LSCC occurrence. Preoperative FIB, FDP, and LMR have important clinical significance because they are economical and simple tests for preliminary screening of patients with LSCC.
As one of several commonly used indicators to reflect systemic inflammatory response, LMR has been proven to be closely associated with tumor recurrence, lymph node metastasis, and prognosis. 12,13 Lymphocytes have been reported to play an important role in inhibiting tumor cell proliferation and metastasis, and lymphocytopenia is a pervasive immunosuppression. 14 Monocytes can promote tumor metastasis via the blood circulation and the tumor-monocyte-endothelial cells axis. 15 As such, LMR is a representative biomarker that may be used to comprehensively evaluate host immune homeostasis and the tumor microenvironment. 3 In the present study, LMR levels were significantly reduced in patients with LSCC, and the AUC used to predict the occurrence of LSCC was .649, which is a potential supplementary diagnostic marker. Furthermore, we determined that the best cut-off value for LMR, according to ROC curve analysis, was 4.29. Compared to LMR ≥4.29, the 3-year OS in patients with low levels was significantly reduced (P < .05). As such, it may be feasible to use LMR levels to diagnose and predict survival in patients with LSCC.
Previous studies have reported that patients with malignant tumors generally exhibit a hypercoagulable state, which may affect tumor infiltration and even metastasis. 16,17 Fibrinogen degradation products have been shown to promote endothelial cell migration, proliferation, cytokine expression and differentiation, and increase the angiogenic activity of vascular growth factors during tumor development. 18 Fibrinogen, also known as coagulation factor I, is the most common coagulation protein in plasma 19 and is an important factor involved in the coagulation cascade. In malignant tumors, FIB can bind to several growth factors, such as vascular endothelial growth factor and fibroblast growth factor, and promote the binding of these growth factors to the corresponding surface receptors of tumor cells, thereby playing a key role in promoting tumor proliferation and angiogenesis. 20,21 Tumor cells can also directly activate FIB, eventually leading to abnormal blood coagulation. Huang et al found that FIB levels in patients with cervical cancer were significantly higher than that of those with cervical intraepithelial neoplasia and healthy volunteers. 22 Results of the present study are consistent with those of the above studies. Serum FIB and FDP levels in patients with LSCC were significantly higher than those in patients with BLL. The results of multivariate logistic regression and ROC curve analysis suggested that preoperative FIB and FDP were possible auxiliary markers predicting the occurrence of LSCC.
Metastasis and recurrence are vital factors affecting the survival of patients with LSCC, and cervical lymph node metastasis is a common mode of metastasis of LSCC. Jemal et al 23 reported that the incidence of supraglottic LSCC could be as high as 65%. Lymph node staging is an important factor in surgical methods and adjuvant chemoradiotherapy selection 24 for malignant tumors. The preferred methods to clinically diagnose laryngeal cancer, with or without lymph node metastasis, are palpation and imaging. However, even among patients clinically diagnosed with non-lymph node metastases using preoperative imaging, 4%–40% still have occult metastases confirmed by postoperative histological examination(s). 25 -27 Therefore, it is necessary to develop biomarkers that can predict lymph node metastasis in LSCC. A previous study reported that hyperfibrinogenemia in patients may indicate that tumor lesions have undergone hematogenous and lymph node metastasis, or that the tumor has invaded to a deeper degree, and reduced the OS. 28,29 Similar results were observed in our study. It was found that high FDP and FIB levels and low LMR were significantly correlated with advanced T-stage (T3+T4), lymph node metastasis, and advanced TNM stage (III + IV). Therefore, FIB, FDP, and LMR levels may be used as supplementary indicators for preoperative evaluation of LSCC lymph node metastasis status and tumor stage.
In this study, three inflammation-related indicators—FDP, FIB, and LMR—were combined for joint diagnosis, and the AUC was obviously improved. Compared with a single indicator, the diagnostic efficiency was significantly improved (P < .001). Then, by combining the three inflammatory markers with other clinical factors, we constructed an easy-to-use predictive nomogram model, which is valuable for diagnosis and may be useful for early clinical treatment in LSCC patients. In summary, we conclude that FDP, FIB, and LMR were potential biomarkers for the auxiliary diagnosis of LSCC, and their significance was strengthened after they were combined. In addition, compared with electronic laryngoscope, which is not suitable for screening in large-scale populations, these blood biomarkers are convenient, inexpensive, and easily generalizable, which could be effectively used for clinical initial screening and auxiliary diagnosis of LSCC.
However, there were some limitations to our study, the first of which was its single-center retrospective design involving patients from the First Affiliated Hospital of Anhui Medical University with low heterogeneity, which may have introduced selection bias. In this study, the cut-off values for all indicators were selected using a simple statistical method, and the practical application of FDP, FIB, and LMR in the clinical diagnosis of LSCC and distinguish it from other forms of cancer remains to be further studied. Due to data limitations, 115 LSCC patients were followed up for <3 years. The survival analysis results for FIB and FDP in the present study were not statistically significant, which are inconsistent with some previous studies. This may be explained the small sample size used in the survival analysis. Hence, a larger-scale, multicenter, prospective study is needed to further determine the diagnostic and prognostic value of these factors in patients with LSCC.
Conclusions
Fibrinogen degradation products, FIB, and LMR levels in patients with LSCC were closely associated with clinicopathological characteristics including tumor occurrence, degree of primary tumor invasion, regional lymph node metastasis, and clinical staging. Currently, FDP, FIB, and LMR levels in LSCC patients cannot be used to diagnose tumors alone due to the lack of specific validation, but they can be used as supplemental markers for early screening, auxiliary diagnosis, and prognosis prediction of tumors. Construction of the nomogram significantly improved the clinical ease of use. Nevertheless, a multicenter, prospective study is needed to confirm this conclusion.
Footnotes
Author Contributions
The four authors worked together to complete the paper.Yehai Liu, as the Corresponding Authors, guided the design of the entire study and the writing of the paper.Yanxun Han and Zhiyao Ren are responsible for the collection of data, the writing of papers, design of the research, and other scattered work; they contributed equally to this work as first authors.Yuchen Liu was responsible for the collection of data and statistical analysis of the data.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Natural Science Research Project of Anhui Higher Education Institution (KJ2018ZD021).
Ethics approval
The data collection was approved by the medical ethics committees of the First Affiliated Hospital of Anhui Medical University
Informed Consent
Written informed consent was obtained for each participant according to federal and institutional guidelines.
