Abstract
Objective
Middle ear cholesteatoma may lead to the erosion of the bony structures of the temporal bone, possibly causing intra- and extracranial complications. Surgical treatment is mandatory, and due to possible residual/recurrent disease, the use of reliable diagnostic methods is essential. Our study aimed to evaluate the reliability of non-EPI DW-MRI for the follow-up of cholesteatoma after surgery.
Methods
In a study group including 53 consecutive patients who underwent surgery for cholesteatoma at a tertiary university hospital, an imaging protocol was applied, including non-echo planar diffusion-weighted imaging magnetic resonance (MR) at 1 month after surgery and then at 6 and 12 months after surgery. Based on the combination of preoperative assessment and intraoperative findings, the study group was divided into 3 subgroups: petrous bone (PB) cholesteatoma, complicated cholesteatoma and uncomplicated cholesteatoma. PB cholesteatoma patients were treated by a subtotal petrosectomy, whereas complicated and uncomplicated cholesteatoma patients were treated either by a canal wall up procedure or a retrograde (inside-out) canal wall down technique with bone obliteration technique (BOT).
Results
The results show that patients who had positive findings on non-EPI DW-MRI scans 1 month after surgery consequently underwent revision surgery during which residual cholesteatoma was noted. All the patients who displayed negative findings on non-EPI DWI-MRI scan at 1 month after surgery did not show the presence of a lesion at the 6- and 12-month evaluations. The 6 patients who displayed residual cholesteatoma at the 1-month follow-up presented dehiscence/exposure of the facial nerve canal at the primary surgery, mostly at the level of the labyrinthine segment.
Conclusion
Non-EPI DW-MRI is a useful and reliable tool for follow-up cholesteatoma surgery, and when applied early, as was done in the protocol proposed in the present study, this tool may be used to detect the presence of residual cholesteatoma in some patients, prompting the planning of early revision surgery.
Introduction
Middle ear cholesteatoma consists of ectopic keratinized epithelial tissue that grows inside the mucosa-lined middle ear cavity and desquamates, resulting in the accumulation of keratin and epithelial debris. Middle ear cholesteatoma may lead to the erosion of the ossicles and the bony walls of the middle ear cavity, mostly by means of an inflammatory response that activates osteoclastic activity. 1 The EAONO/JOS 2017 classification identifies 4 stages of cholesteatoma based on its extension inside the middle ear and the presence of intra- and extracranial complications. 2 Facial palsy is one of the most frequent complications and is often related to an incomplete covering of the fallopian canal. In patients suffering from chronic otitis media with or without cholesteatoma, and the intraoperative incidence of facial nerve dehiscence (FND) is reported to range from 8.9% to 45.5%. 3 Approximately 80% of patients have dehiscence localized at the level of the bony tympanic segment, and the majority of them have dehiscence at the oval window tract.3,4 Due to possible serious intracranial and labyrinthine complications, surgery is an elective treatment for cholesteatoma; this approach aims to eradicate the disease while simultaneously trying to preserve middle ear anatomy and function. The canal wall down (CWD) procedure, canal wall up (CWU) procedure and bone obliterative technique (BOT) are the surgical methods presently used in the treatment of cholesteatoma. In particular, the CWD, or open technique, is also performed by limiting the lowering of the posterior bone canal wall by a retrograde or inside-out procedure. 5 CWU mastoidectomy is generally considered more likely to generate a high rate of residual and recurrent disease than a CWD procedure (from 9 to 70% vs 4 to 17%, respectively). 6 However, this latter theoretically does not provide the maintenance of a self-cleaning activity, with some social limitations, such as requiring patients to avoid water contact. The overall percentage of residual or recurrent cholesteatoma in the literature reaches almost 50% of the cases.6,7
Several radiological protocols have been proposed in recent years to detect possible residual/recurrent cholesteatoma so that revision surgery can eventually be performed. Computed tomographic (CT) scanning has been reported to be inadequate for this aim, with a sensitivity of 43% and a specificity of 48%. 8 Several studies have highlighted 2 main MRI techniques to be used for detecting primary and recurrent or residual cholesteatomas. Among them, late post-gadolinium T1-weighted sequences would be able to detect residual cholesteatomas up to 3 mm in size. 9
After surgery, both high-resolution computed tomography (HRCT) and conventional magnetic resonance imaging (MRI) sequences showed limitations in their ability to distinguish cholesteatoma from other lesions, such as cholesterol granuloma, granulation tissue, fibrosis and fluid collection. Therefore, during the past few years, new developments in MRI protocols, such as diffusion-weighted imaging (DWI), have been introduced, initially by single-shot echo planar imaging (EPI). 9 More recent publications have analysed non-echo planar imaging (non-EPI) sequences,10,11 including HASTE (half Fourier acquisition single-shot turbo spin echo; Siemens) and PROPELLER (periodically rotated overlapping parallel lines with enhanced reconstruction; general electric) sequences. These sequences can provide a higher spatial resolution than routine EPI-based DW-MRI sequences and are sensitive to cholesteatomas as small as 2 to 3 mm.12,13 These sequences, although they require longer acquisition times than EPI sequences, are less susceptible to air-bone distortions. Non-EPI DW-MRI has been shown to be highly sensitive and specific for the detection of primary cholesteatomas, with elevated positive and negative predictive values.14,15 In 2009, De Foer et al. 16 analysed the role of non–EPI-based DWI MRI for the detection of residual cholesteatomas after canal wall up mastoidectomy before planning an eventual second-look surgery and found the sensitivity, specificity, positive predictive value and negative predictive value for this technique to be 90%, 100%, 100% and 96%, respectively. A recent meta-analysis of the published literature found the overall sensitivity and specificity of non-EPI DWI-MRI in detecting cholesteatoma to be 0.94 and 0.94, respectively, lending support to the notion that these sequences may reduce or abolish the need for second-look surgery.17,18 A recent study analysed 143 patients affected by middle ear cholesteatoma who underwent 1.5-T non-EPI DWI-MRI 6 to 9 months after surgery; the patients who had hyperintense signals in the middle ear underwent revision surgery. The sensitivity and specificity of non-EPI DWI-MRI in identifying cholesteatoma in the postoperative period was 100%. 19 In most studies, non-EPI DWI-MRI was carried out between 6 and 24 months after first-stage surgery.20-22
The primary endpoint of the present study was to assess whether the adoption of early non-EPI DW-MRI 1 month after surgery should be included in the postoperative cholesteatoma protocol. The second aim was to validate the results determined by 1-month non-EPI DWI-MRI when comparing them with the 6- and 12-month findings. Finally, the third aim was to assess the correlation between the presence of intraoperative complications during the primary surgery (facial nerve involvement, lateral canal fistula and tegmen erosion) and the likelihood of detecting a residual cholesteatoma during the follow-up imaging protocol.
Methods
One hundred fifteen patients underwent surgery for cholesteatoma at a tertiary university hospital from December 2010 to January 2018. In 62 of the patients, a revision procedure was performed. Fifty-three patients (36 men and 17 women, mean age 49.6 years, ranging from 16 to 78 years) underwent primary surgery and served as the definitive study sample. These patients were divided into 3 groups according to the main localization of their disease: Petrous bone (PB) cholesteatoma (n = 8). No PB cholesteatoma (NO PB) (n = 23) with intraoperative complications/ Uncomplicated cholesteatoma, serving as the control group (n = 22) with a cholesteatoma of limited extension and without intraoperative complications.
The surgical procedures, performed by the same surgeon, included subtotal petrosectomy in the 8 PB cholesteatoma patients, CWU in 30 patients and retrograde CWD with BOT in 15 patients. For the BOT-CWD procedure, bone dust was collected while drilling on the mastoid cortex and/or the squama of the temporal bone. After cholesteatoma removal, a large piece of tragal or conchal cartilage was placed to reconstruct the posterior canal wall, supported by bone dust filling the mastoid cavity. Non-EPI DW-MRI was performed postoperatively after 1 month (P1), 6 months (P2) and 12 months (P3). P1 was included within the inpatient period of hospital stays and was therefore overseen by the hospital administration; scans at P2 and P3 were instead individually performed.
Imaging technique
MRI examinations were performed with a 1.5 T MRI scanner (Magnetom Sonata, Siemens, Erlangen, Germany) using a standard eight-channel head coil. The standard temporal bone protocol for cholesteatoma evaluation included transverse and coronal T2-weighted imaging, coronal T1-weighted imaging and coronal non-EPI DW (HASTE, half Fourier SSTSE) without contrast injection. HASTE-DW was obtained in the coronal plane with the following parameters: repetition time (TR)/echo time (TE), 2080/105 ms; 2 mm slice thickness without gap; number of excitations (NEX), 3; matrix, 192×75 and field of view (FOV) 230 mm. One b value was obtained (b = 1000 s/mm2); acquisition time: 3.05 minutes. Coronal T1-weighted imaging was performed with a TR/TE, 569/10 ms; flip angle, 150°; NEX, 3; 3 mm slice thickness, FOV, 230 mm and matrix, 384×80. Axial TSE T2-weighted imaging was obtained with TR/TE, 2200/112 ms; flip angle, 180°; NEX, 4; slice thickness, 3 mm; FOV, 230 mm and matrix, 384×70. Coronal TSE T2-weighted imaging was obtained with TR/TE, 3280/110 ms; flip angle, 180°; NEX, 4; slice thickness, 3 mm; FOV, 230 mm and matrix, 384×70.
Statistical analysis
All statistical analyses were performed using R version 3.5.3 (2019-03-11) – Great Truth Copyright © 2019 The R Foundation for Statistical Computing. The normal distribution was tested by the D’Agostino–Pearson normality test. Differences in proportions were compared by using a chi-square test, while a non-parametric Kruskal–Wallis test was used to compare medians from the 3 groups. Age, sex, dehiscent tegmen, dehiscent/involved facial nerve, dehiscent lateral semicircular canal and operating technique were included in the multiple regression analysis. The criterion for the significance level of all statistics was
Results
Demographic data of the 3 study groups.
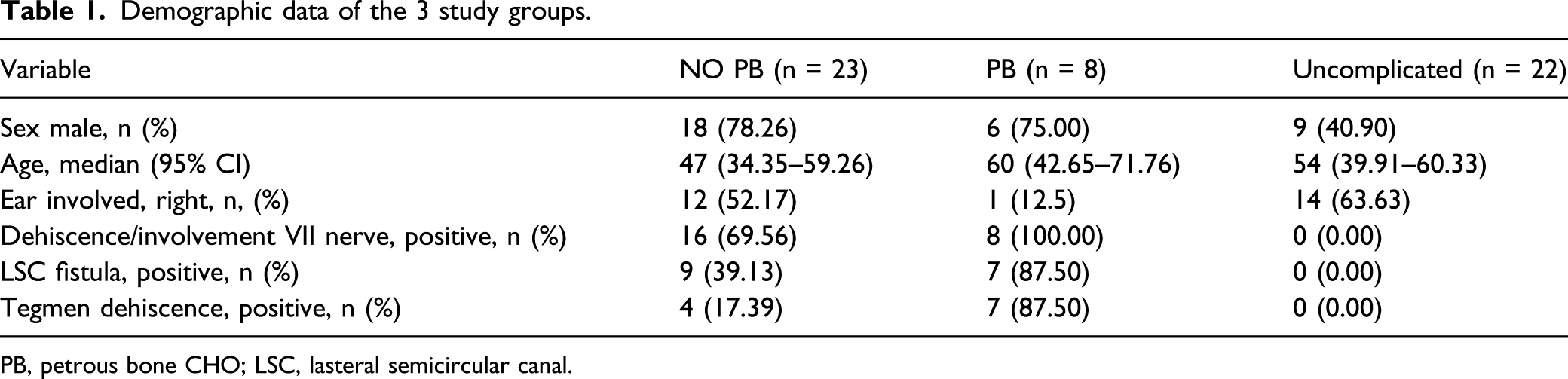
PB, petrous bone CHO; LSC, lasteral semicircular canal.
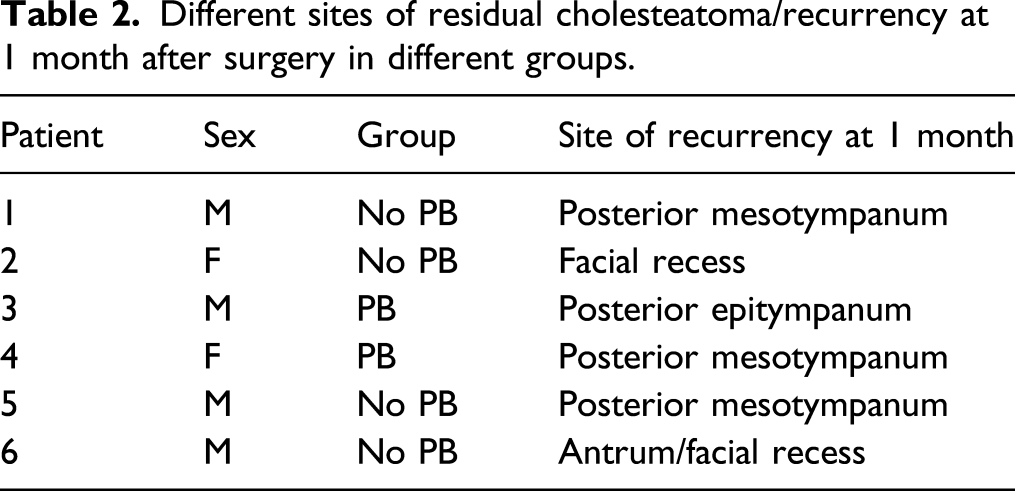
Non-EPI DW-MRI findings were positive in 2 patients in the PB group (25%), in 4 patients in the non-PB group (17.4%) and in none of the patients in the control group. Figure 1 shows MRI findings on T2 and non-EPI DWI sequences in patients with and without cholesteatoma at P1. At 1 month after surgery, we found recurrence of cholesteatoma in the posterior mesotympanum in 4 patients, at the level of facial recess in 1 patient and at the level of the antrum in 1 patient. Tegmen dehiscence was noted in 2 patients, and all 6 patients had facial canal involvement. In 4 patients, we found only inflammatory tissue at the level of the oval window region (Table 2). Hyperintense tissue on T2-weighted images (A) involves the right antral region and it is associated with bright signal on non-EPI DWI images (HASTE) (B), indicating the presence of a cholesteatoma. No pathological hyperintensity on diffusion images is evident in the second case (D), where a postoperative scar in the right mastoid is appreciable (C). Different sites of residual cholesteatoma/recurrency at 1 month after surgery in different groups.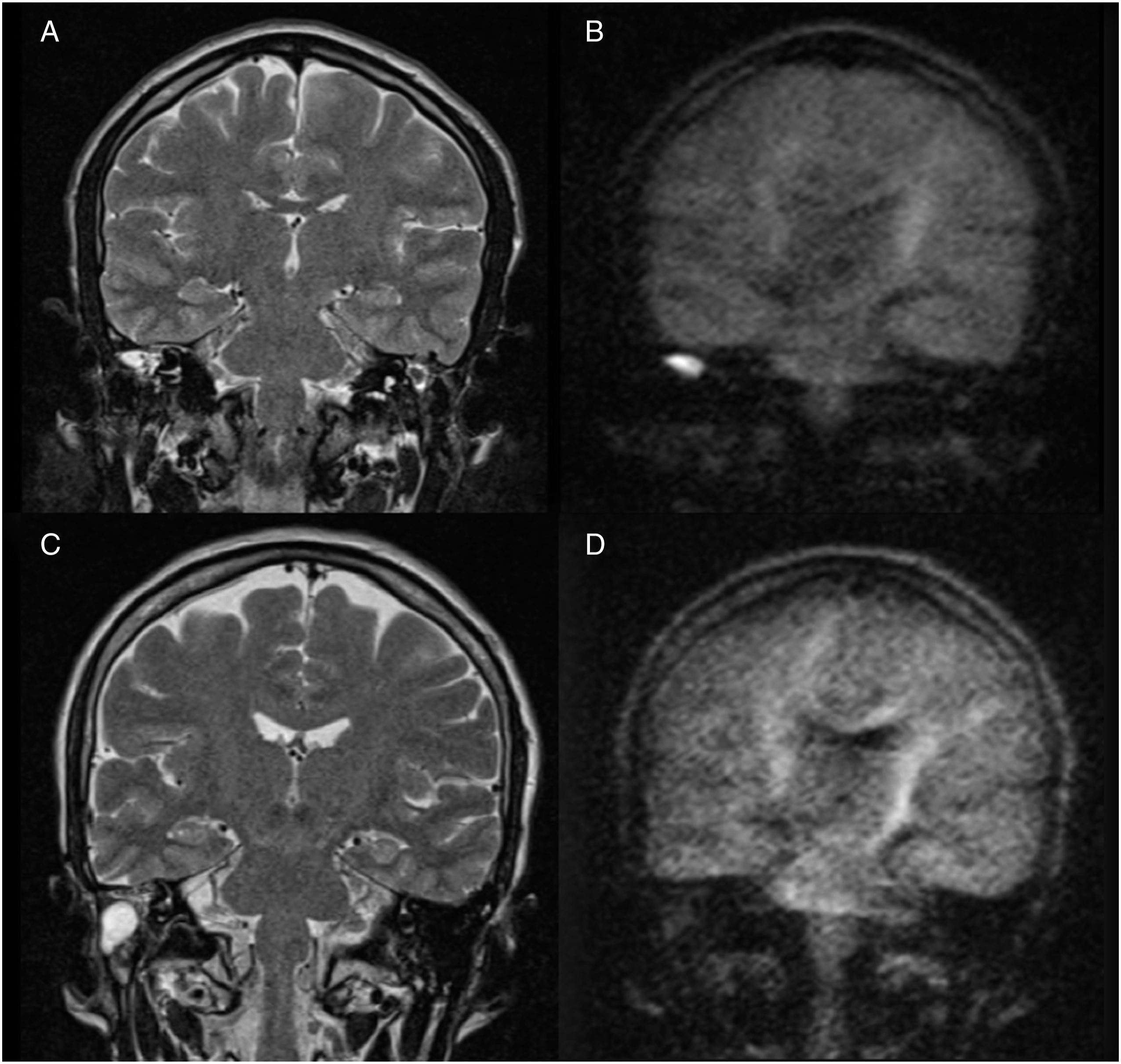
Percentage of cholesteatoma recurrence/residual after 12 months in the 3 groups. The results did not show any statistically significant difference.
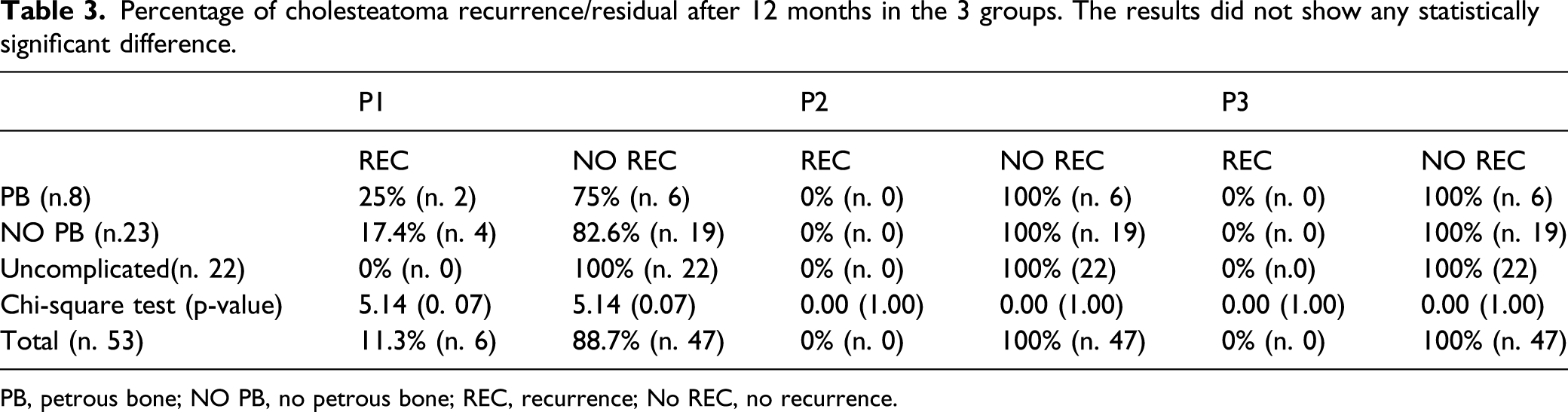
PB, petrous bone; NO PB, no petrous bone; REC, recurrence; No REC, no recurrence.
After 6 months (P2), non-EPI DW-MRI confirmed that cholesteatoma had not recurred in any of the patients who were negative at P1 (Table 3).
Twelve months after surgery, non-EPI DW-MRI confirmed the absence of cholesteatoma in all patients of the 3 examined groups (PB, non-PB and uncomplicated) (Table 3).
The comparison of the results in the 3 groups did not show any statistically significant difference; while when comparing each group individually, differences at T1 were found to be statistically significant (no PB vs uncomplicated:
Discussion
Diffusion-weighted MRI has become the most commonly used imaging technique for following up patients with residual or recurrent cholesteatoma. Two different diffusion-weighted techniques can be used: single-shot (SS) echo planar (EPI) and non-echo planar (non-EPI) DWI. Echoplanar (EPI) sequence DW-MRI has the disadvantages of showing artefacts and the inability to detect lesions smaller than 5 mm. 22 Non-EPI DW-MRI, in the form of single-shot turbo-spin echo sequences (HASTE DWI), has been shown to reduce slice thickness, to have higher resolution and to reduce artefacts in temporal bone, allowing visualization of smaller size lesions up to 2 mm in size. 23 Therefore, at the present time, HASTE DWI has become the gold standard diagnostic tool for detecting cholesteatoma, as shown in many clinical reports from the recent specific literature. 24 In their retrospective study, Dremmen et al. 25 evaluated 56 non-EPI DW-MRI patients who previously underwent an operation for cholesteatoma, confirming residual and/or recurrent cholesteatoma in 93% (25/27) of the patients. Khemani et al. 26 analysed 48 non-EPI DW-MRI patients with cholesteatoma, showing residual or recurrent disease in 38 of them, which was also confirmed during surgical revision. Another retrospective study conducted by Velthuis et al. 27 evaluated 38 patients and analysed MRI findings in relation to perioperative findings from second-look surgery. The authors found no false-positive findings, while the 4 false-negative findings were ascribed to the longer interval between DW-MRI and the second-look surgery; thus, a strict radiological surveillance protocol by 6–12 month intervals for up to 2–3 years after primary surgery is advised. The optimal timing of MRI examination after primary surgery has been the object of other clinical reports. Akkari et al. 28 evaluated the contribution of DW-MRI in comparison to that of delayed contrast-enhanced T1-weighted MRI to the postoperative follow-up of cholesteatoma patients and proposed patient evaluation by MRI scans for at least 2 years after surgery. Steens et al. 29 proposed applying a follow-up imaging protocol at 6 months after the first negative DW-MRI and prolonging it up to 4 years after surgery. Bernardeschi et al. (2019) performed a radiological follow-up with MRI and CT of patients who underwent surgery with mastoid and epitympanic obliteration of the surgical site using bioactive glass (BG) granules and found that contrast enhancement allowed differential diagnosis with cholesteatoma and was useful for the detection of other underlying temporal bone pathologies. 30 The present study aimed to offer further contributions to the timing issue and proposed a protocol that demonstrates the importance of an early postoperative imaging evaluation 1 month after surgery, with the aim of obtaining a nearly immediate picture after cholesteatoma removal. In our present clinical setting, moreover, this procedure was covered by the National Health System, since it was performed in continuation of the inpatient period, assuring at the same time the same imaging quality from the same centre. This early imaging protocol was particularly useful in PB cholesteatoma, where the anatomical preservation of an involved facial nerve was treated in a conservative manner. In all the other cases that included non-PB, with uncomplicated and complicated cases, non-EPI DW-MRI would have enabled us to visualize eventual residual disease that could have been treated with early revision surgery, thus minimizing the invasiveness of the surgical procedure. The protocol proposed in this study therefore includes serial non-EPI DW-MRI scans performed at 1, 6 and 12 months after primary cholesteatoma surgery.
Six of the 53 patients who showed positive findings in the 1-month non-EPI DW-MRI (11%) underwent a second revision surgery within 6 months that showed residual cholesteatoma in all 6 patients, with 100% specificity in the early detection of residual disease. All the patients who showed negative findings at 1 month after surgery continued to display a negative pattern at the 6- and 12-month controls. Moreover, the early detection of residual cholesteatoma has allowed us to perform minimally invasive revision surgery to completely eradicate ear disease, as assessed postoperatively with the same timing of imaging control. Subjects with a higher percentage of residual cholesteatoma at 1 month belonged to the PB group (25%) and had the highest rate of complications, namely, facial dehiscence/exposure (in 100% of the patients). In the no PB group, 17.4% of the patients had residual cholesteatoma at T1, while no patient in the uncomplicated group showed residual cholesteatoma after 1 month. When comparing the 3 groups, no significant difference was noted, while the comparison of the uncomplicated group with each of the remaining groups individually resulted in a significant difference.
An interesting finding that should be emphasized is that among the possible pathological complications (facial canal dehiscence and involvement, lateral semicircular canal fistula or tegmen dehiscence), the most negative prognostic factor for a residual cholesteatoma has been represented by the involvement of a dehiscent facial nerve in which surgical removal has been performed with a conservative approach with respect to the anatomical preservation of the nerve itself. In our experience, a percentage (25%) of PB patients were found to be positive at the 1-month control; therefore, it was included in further surgical planning.
Cholesteatoma involvement of a dehiscent/exposed facial nerve would then be more likely to lead to incomplete removal of the problematic tissue when the surgeon had previously counselled with the patient and was instructed to leave the nerve anatomically intact. It is therefore likely to deduce that for these specific patients, stricter and frequent radiological follow-up needs to be extended for several years after primary surgery.
A limitation of our study is the timing of radiological evaluation that ended 12 months postoperatively, although it is known that late recurrence can also be expected. A 2- and 5-year imaging study is currently in progress and will be part of a future report.
Conclusions
In patients with invasive cholesteatoma of the middle ear, non-EPI DW-MRI could be useful for the first early follow-up 1 month after surgery, especially in patients with facial nerve exposure caused by the disease. With this approach, it is possible to detect any residual cholesteatoma and to plan an early revision surgery, thus preventing further spread of the residual disease while minimizing the morbidity of a second surgery.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
