Abstract
Objective:
To observe the efficacy and safety of postoperative long-term low-dose oral administration of clarithromycin in patients with refractory chronic rhinosinusitis (RCRS), to explore the characteristics of postoperative microbiota in the nasal cavity in patients with RCRS, and to compare the differences and changes in microbiota in the nasal cavity before and after medication.
Methods:
This was a prospective, self-controlled study. Eighteen patients with RCRS who had persistent symptoms after endoscopic sinus surgery and standard therapy with normal immunoglobulin E and eosinophil level were included. Low dose (250 mg, once daily) clarithromycin was orally administrated for 12 weeks. Symptom severity and endoscopic findings were evaluated before, after 4 weeks, and 12 weeks of treatment, and nasal cavity microbiota was analyzed simultaneously.
Results:
A total of 18 patients with RCRS were enrolled and 17 patients completed the study. Four weeks after oral administration of clarithromycin, significant improvement was observed in subjective symptoms including nasal congestion, rhinorrhea, postnasal drip, and general discomfort, as well as endoscopic findings including general surgical cavity condition, rhinedema, and rhinorrhea (P < .05). After continuous treatment to the 12th week, symptoms showed significant improvement compared with baseline, and endoscopic score showed significant improvement compared with both baseline and 4 weeks after treatment. Analysis of middle nasal meatus flora revealed a significant decrease of Streptococcus pneumoniae after 12 weeks of clarithromycin treatment (P < .05), while the richness, composition, and diversity were similar before and after treatment. Patients enrolled experienced no adverse drug reaction or allergic reaction, nor clinical significant liver function impairment observed.
Conclusion:
Postoperative low-dose long-term oral administration of clarithromycin in patients with RCRS can improve the clinical symptoms and facilitate the mucosal epithelialization, with good tolerance and safety. The efficacy of clarithromycin in patients with RCRS may be related to its regulatory effect on nasal cavity microbiota.
Keywords
Chronic rhinosinusitis (CRS) is a chronic inflammatory disease involving the nasal cavity and nasal sinus, and affecting 8% of Chinese people according to a latest multicenter cross-sectional study in China. 1 The current treatment for CRS, endoscopic sinus surgery (ESS) and standard postoperative medication including antibiotic, intranasal corticosteroids, mucigogues, oral hormone, and nasal irrigation, could achieve a clinical cure rate up to 75% to 98%, 2,3 but there are still nearly 10% of patients with CRS failing to obtain the ideal clinical outcomes. 4 These patients show no obvious improvements in their symptoms and local signs after ESS and standard postoperative treatment, hence can be classified as “refractory chronic rhinosinusitis (RCRS)”. 5 -7 In patients with RCRS and part of patients with CRS who preferred nonsurgical treatment, anti-inflammatory treatment is essential to improve the efficacy.
In recent years, accumulating evidences have shown that 14-membered ring macrolides have anti-inflammatory, immunoregulatory, 8,9 and antibiofilm 10 effects. Moreover, many randomized controlled trails in China and abroad have confirmed the clinical efficacy of postoperative long-term low-dose oral administration of macrolides in patients with CRS. However, controversy remains between results from different studies. For example, one randomized controlled trial by Amali et al 11 enrolled 66 postoperative patients with CRS reported significant remissions of subjective symptoms including nasal congestion and postnasal drip after 12 weeks of azithromycin treatment. Patients with CRS enrolled in this study were not classified according to the examination of immunoglobulin E(IgE)or eosinophil level.; while another randomized controlled trial by Haxel et al 12 enrolled 67 postoperative patients with CRS only observed decrease in endoscopic score after 12 weeks of erythromycin treatment, and they did not find the statistically significant advantage between the low IgE and high IgE levels. Moreover, patients with CRS enrolled in these studies were all routinely treated with macrolides after operation, not the patients who have poor efficacy of postoperative standard therapies. Therefore, it is still necessary to further verify the efficacy of long-term low-dose macrolides in patients having RCRS with normal IgE and eosinophil level. In addition, recent studies have shown that there is colonization of commensal bacteria in human nasal cavity, 13 and the disturbance of its balance by pathogen or antibiotics may contribute to the pathogenesis and progression of CRS. 14,15 As a result, the effect of postoperative long-term low-dose 14-membered ring macrolides on the nasal commensal microbiota of patients with CRS needs to be further studied.
In order to evaluate the clinical efficacy of 14-membered ring macrolides in the treatment of RCRS, we designed this prospective self-controlled study. The study evaluated the efficacy and safety of long-term low-dose oral administration of clarithromycin in patients having RCRS with normal IgE and eosinophil level by dynamical follow-up of symptom scores, endoscopic scores, and adverse events during treatment, and analyzed its impact on nasal microbiota by comparing the 16S ribosomal DNA (rDNA) sequencing results of middle nasal meatus flora secretion before and after treatment.
Materials and Methods
Study Design and Case Selection
This study is a prospective, single-arm, single-center and self-controlled trial designed in compliance with the Declaration of Helsinki, ICH-GCP, and relevant laws and regulations of China authority. The registered ethical number for this study is TRECKY2017-030, and the Chinese clinical trial registration number is ChiCTR1800014382. Study enrolled adult patients having RCRS with normal serum IgE and eosinophil level (serum IgE < 60 KU/L, EOS% < 5.65%) treated at the Rhinology Department, Beijing Tongren Hospital, Capital Medical University during the period from June 2016 to June 2017. Patient with RCRS is defined as a patient with CRS who received ESS plus nasal polypectomy and standard postoperative treatment including antibiotic, intranasal corticosteroids, mucogogues, oral hormone (methylprednisolone, 24 mg once daily, continuous treatment for 1 week, then the dose was gradually reduced until withdrawal) and nasal irrigation, but subsequently experienced no obvious improvement or had persistent symptoms lasted for more than 1 month. Exclusion criteria: (1) patients with immunodeficiency syndromes, such as primary ciliary dyskinesia; (2) patients with bronchial asthma or concomitant severe allergic rhinitis and/or fungal sinusitis; (3) pregnant and breastfeeding women; (4) patients allergic to macrolide drugs; and (5) patients with organic diseases in heart, liver, kidney, and/or digestive tract or patients who had obvious elevated liver enzymes level and/or severe gastrointestinal adverse reactions after administration of clarithromycin. Patients who met the inclusion/exclusion criteria are well-informed of the study and signed the informed consent form before enrollment.
Treatment Schedule and Follow-Up
All the patients enrolled were orally administrated with 14-membered ring macrolides clarithromycin (Trade name: Klacid, Abbott Laboratories), 250 mg, once daily, for continuous 12 weeks on top of standard postoperative treatment such as intranasal corticosteroids, mucogogues, and nasal irrigation. Follow-ups were conducted at baseline, 4 weeks, and 12 weeks after treatment.
Clinical Data Collection
Collection of efficacy data
All the patients should complete visual analog scale (VAS) scoring and endoscopic scoring before treatment (baseline), after 4 weeks, and 12 weeks of treatment.
Collection of safety data
(1). Liver function: Liver function was examined after 4 weeks and 12 weeks of treatment. (2) Adverse drug reactions: Symptoms occurred during therapy including nausea, vomiting, diarrhea, and/or dyspepsia were collected by questionnaires at each follow-up. (3) Allergic reactions to treatment were asked and recorded at each follow-up.
Sample Collection
The secretion from the middle nasal meatus area was collected with nasopharyngeal swab through sheathing canal under the guidance of nasal endoscope, through brushing with a sheath canal. Sampling was conducted by brushing 6 cycles clockwise and 6 cycles anticlockwise. After sampling, the brush head was cutoff, placed in sterile tube, rapidly frozen in liquid nitrogen, and stored in a −80 °C refrigerator for further usage.
DNA Extraction and 16S rDNA Sequencing
CTAB Cetyltrimethylammonium Bromide method was used to extract the genomic DNA from middle nasal meatus secretion. After quantification, DNA was diluted to 1 ng/μL and 16S rDNA V4 region was amplified and purified using primers 515F and 806R. Library construction was performed using the library construction kit (TruSeq DNA PCR-Free Sample Preparation Kit). Libraries constructed were proved qualified through Qubit and quantitative polymerase chain reaction quantification, and second generation high-throughput sequencing was performed using HiSeq2500 PE250.
Sequencing Data Analysis
Based on valid data, sequences satisfying 97% consistency were clustered into Operational Taxonomic Units (OTUs) following the default, and sequences with the highest frequency within OTUs were selected as the representative sequence of OTUs and annotated. The richness, diversity, and composition of nasal flora were compared among groups.
Statistical Analysis
Quantitative data were described with (median, interquartile range) or (mean ± standard deviation). T test or nonparametric test was conducted for data analysis using SPSS 24 for Mac statistical software. A statistically significant difference was considered to be observed when P < .05, and a strong statistically significant difference was considered to be observed when P < .01.
Results
Basic Characteristics
A total of 18 patients were enrolled in this study, including 12 males and 6 females, with a median age of 42 years (age ranging from 19 to 63 years). After 12 weeks of treatment, 5 patients were lost to follow-up. Finally, the efficacy and safety data of 17 patients were integrated and included in the subsequent analysis. The basic characteristics are shown in Table 1. No patients experienced clinical significant changes in liver function after 4 and 12 weeks of clarithromycin treatment. No patients complained of discomforts such as nausea, vomiting, diarrhea, and dyspepsia; no allergic reactions to clarithromycin were observed during the study. Middle nasal meatus secretion samples before treatment, after 4 weeks, and 12 weeks of treatment were collected from 16 patients.
Basic Characteristics of Patients.
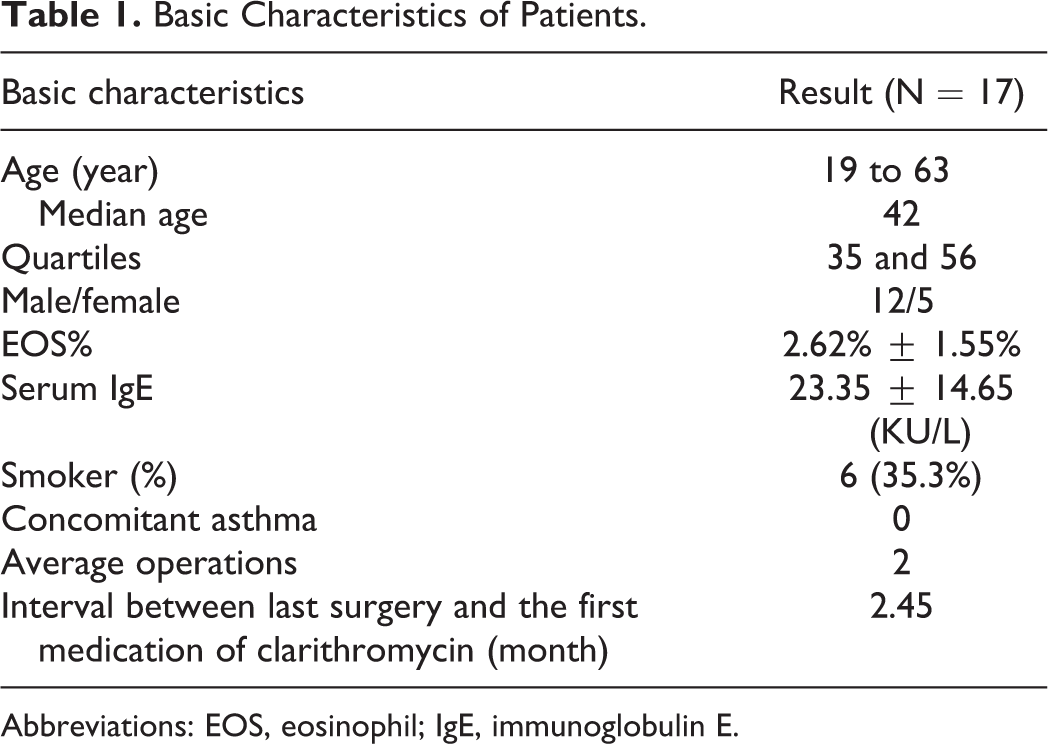
Abbreviations: EOS, eosinophil; IgE, immunoglobulin E.
Efficacy Evaluation
The VAS scoring and endoscopic scoring data showed a skewed distribution as shown in Table 2, and Friedman rank sum test was used for statistical analysis.
The VAS Scoring and Endoscopic Scoring.
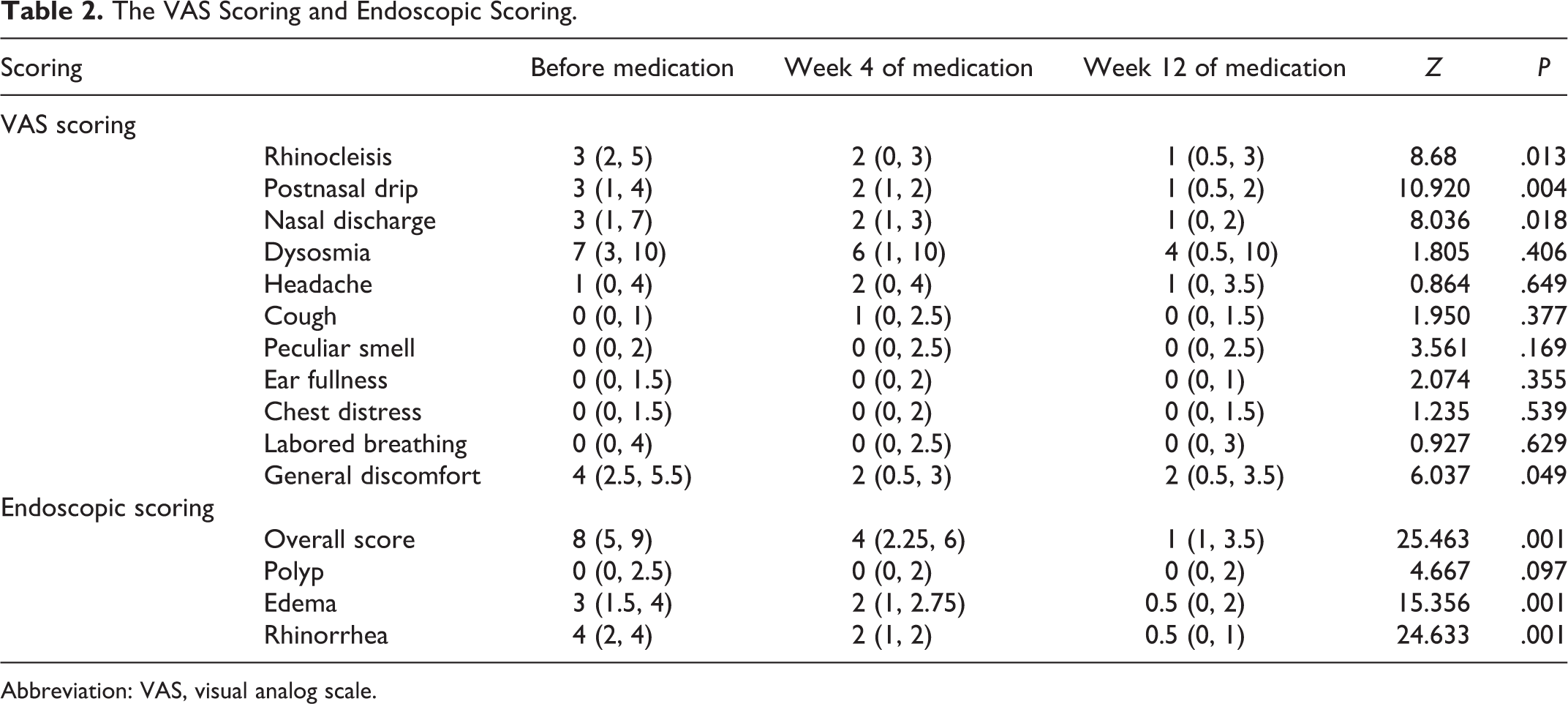
Abbreviation: VAS, visual analog scale.
Visual analog scale scoring
As shown in Figures 1 and 2, symptoms including nasal congestion, rhinorrhea, postnasal drip, and general discomfort improved significantly after 4 weeks and 12 weeks of oral administration of clarithromycin compared with baseline according to VAS scoring results. However, further significant relief of symptoms was not observed after comparison of VAS scores after 4 weeks and 12 weeks of treatment. In addition, no significant improvement of hyposmia was found after 4 weeks and 12 weeks of treatment.
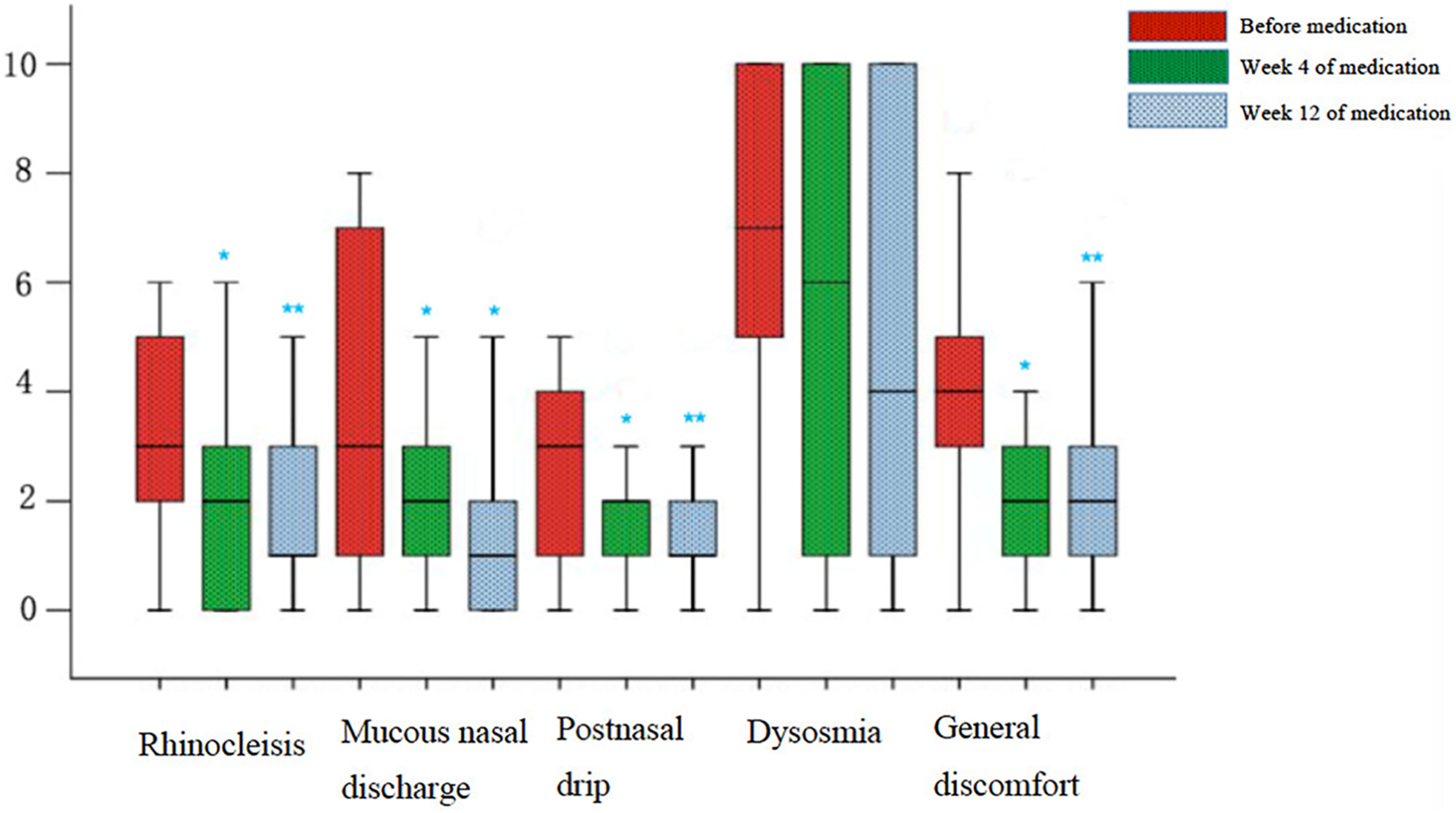
Visual analog scale (VAS) score box chart.
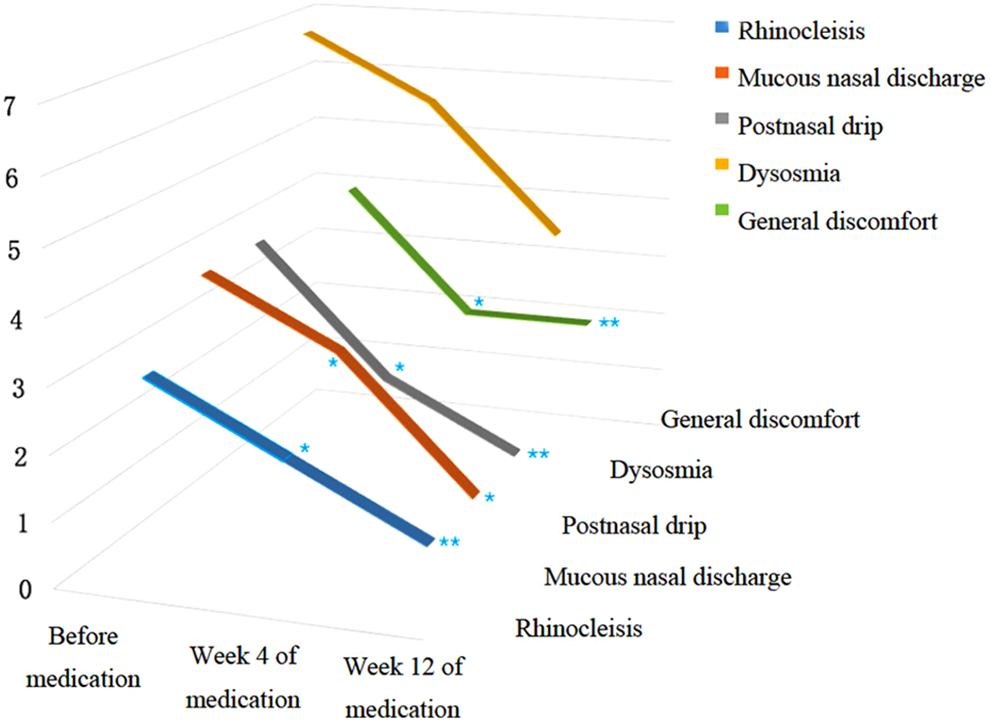
Visual analog scale (VAS) scores 3-dimensional variation trend chart.
Endoscopic scoring
Four weeks after oral administration of clarithromycin, significant improvement in endoscopic scoring including general surgical cavity condition, rhinorrhea, and edema (P < .05) was observed compared with baseline. Furthermore, when continued clarithromycin treatment till the 12th week, the endoscopic scoring in general surgical cavity condition (P < .01), rhinorrhea (P < .05), and edema (P < .01) showed significant improvement compared with baseline and the fourth week. Data mentioned above were summarized in Figures 3 and 4. Representative pictures of surgical cavity mucosa taken under endoscopy before and after clarithromycin treatment could be found in Figures 5 and 6.
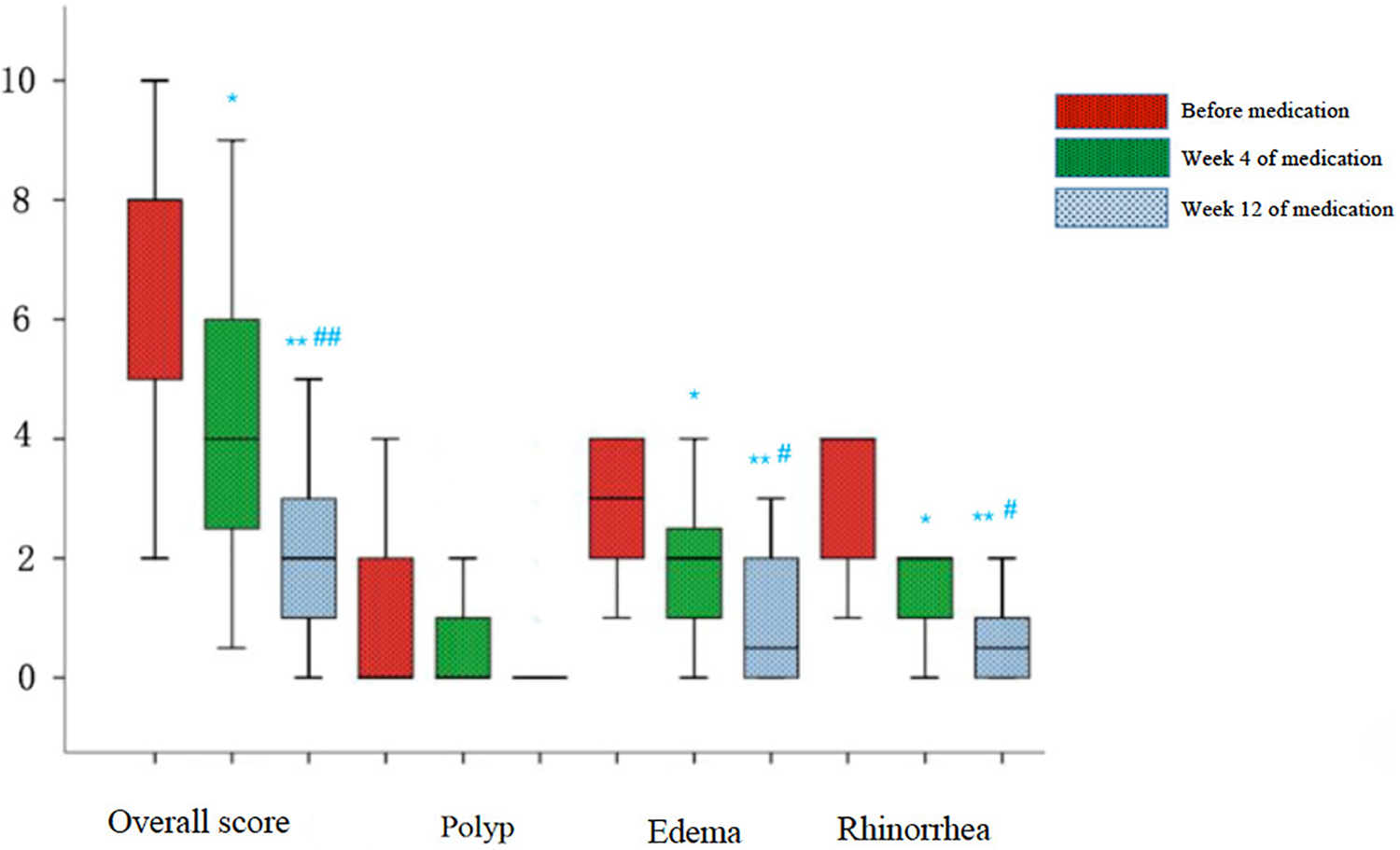
Endoscopic scores box chart.
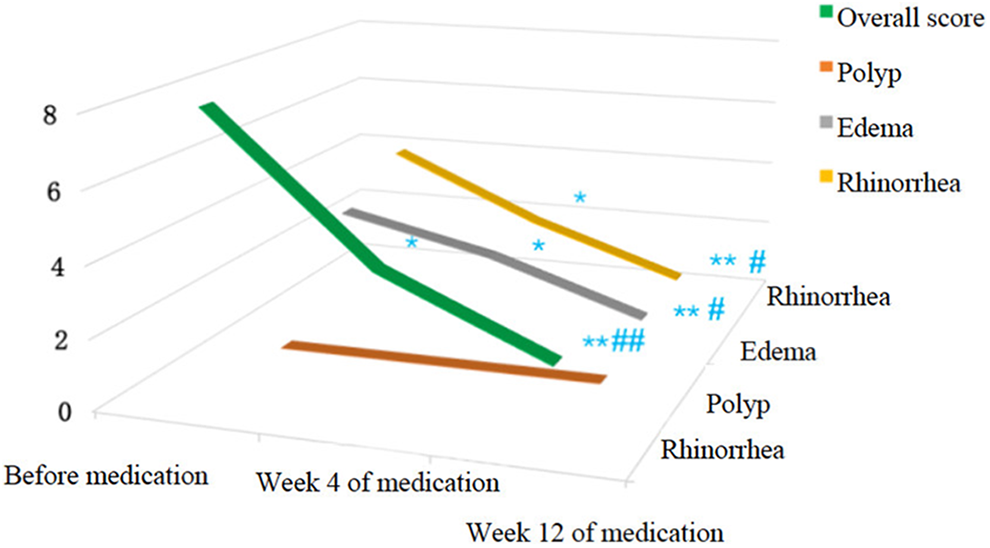
Endoscopic scores 3-dimensional variation trend chart.
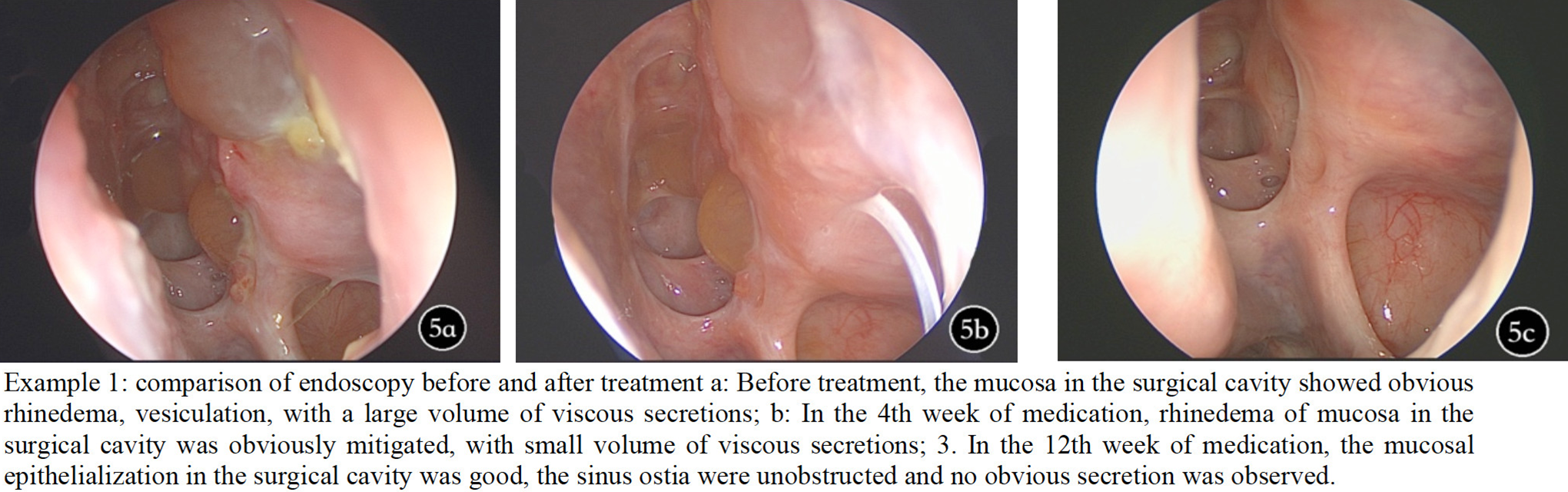
Surgical cavity mucosa before clarithromycin treatment.
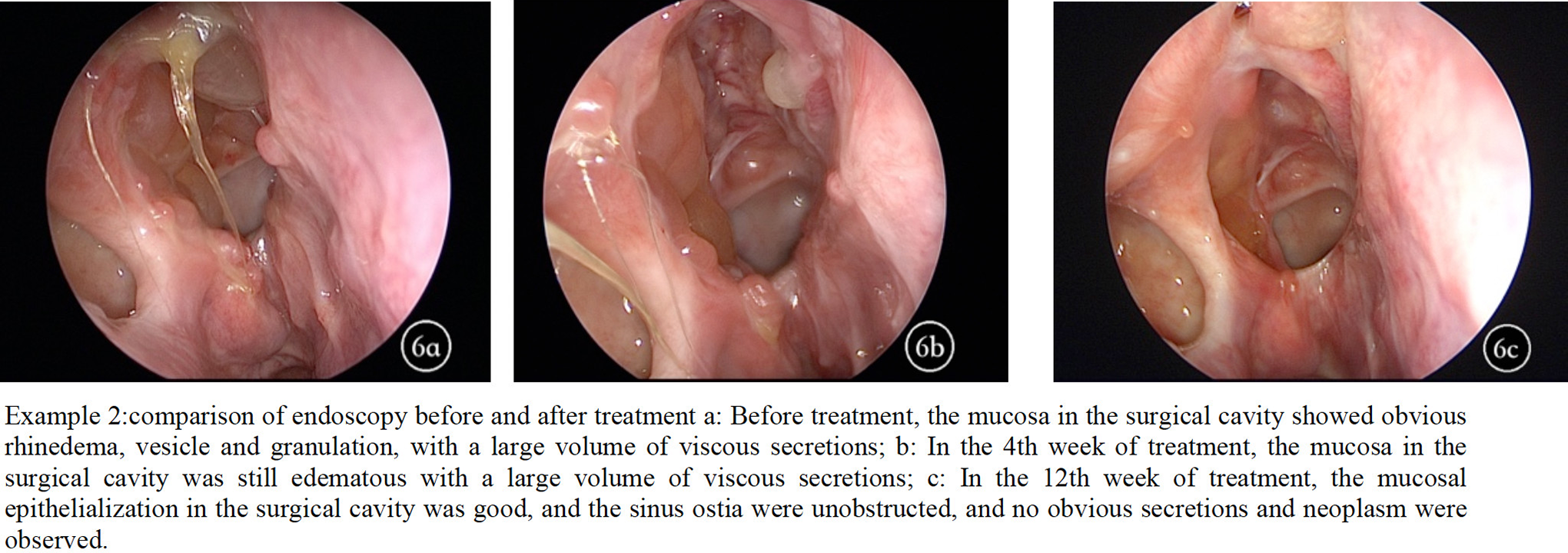
Example 2: Surgical cavity mucosa before and after clarithromycin treatment.
Flora Analysis
Nasal flora sequencing data showed a skewed distribution, so Friedman rank sum test was used for statistical analysis.
Richness
The total number of OTUs detected was 14 437 before treatment, then 14 177 after 4 weeks of treatment, and then 14 977 after 12 weeks of treatment. No statistical difference was found in the multiple comparisons among these 3 groups.
Abundance
Generally speaking, the composition of nasal flora was similar before and after clarithromycin administration. The most abundant bacteria generas before treatment, 4 weeks, and 12 weeks after treatment were all Staphylococcus (9.34% before treatment, 12.46% 4 weeks after treatment, 19.80% 12 weeks after treatment), Pseudomonas (2.93%, 2.33%, 3.20%), Streptococcus (2.26%, 1.04%, 0.25%), Corynebacterium (1.22%, 0.72%, 0.41%), Haemophilus (2.46%, 0.37%, 0.57%), Delftia (0.03%, 0.05%, 0.04%), and Fusobacterium (0.06%, 0.07%, 0.03%), with slightly differences in proportion. Among which, the proportion of Streptococcus decreased significantly after 12 weeks of treatment (P < .05), and the proportion of Haemophilus decreased significantly after 4 weeks of treatment (P < 0.05), both compared with baseline. Similarly, among the top 4 abundant bacteria species, namely Staphylococcus aureus, Pseudomonas veronii, Streptococcus pneumoniae, and Pseudomonas geniculate, only the proportion of S pneumoniae decreased significantly after 12 weeks of treatment compared with baseline (P < .05; Figures 7 and 8).
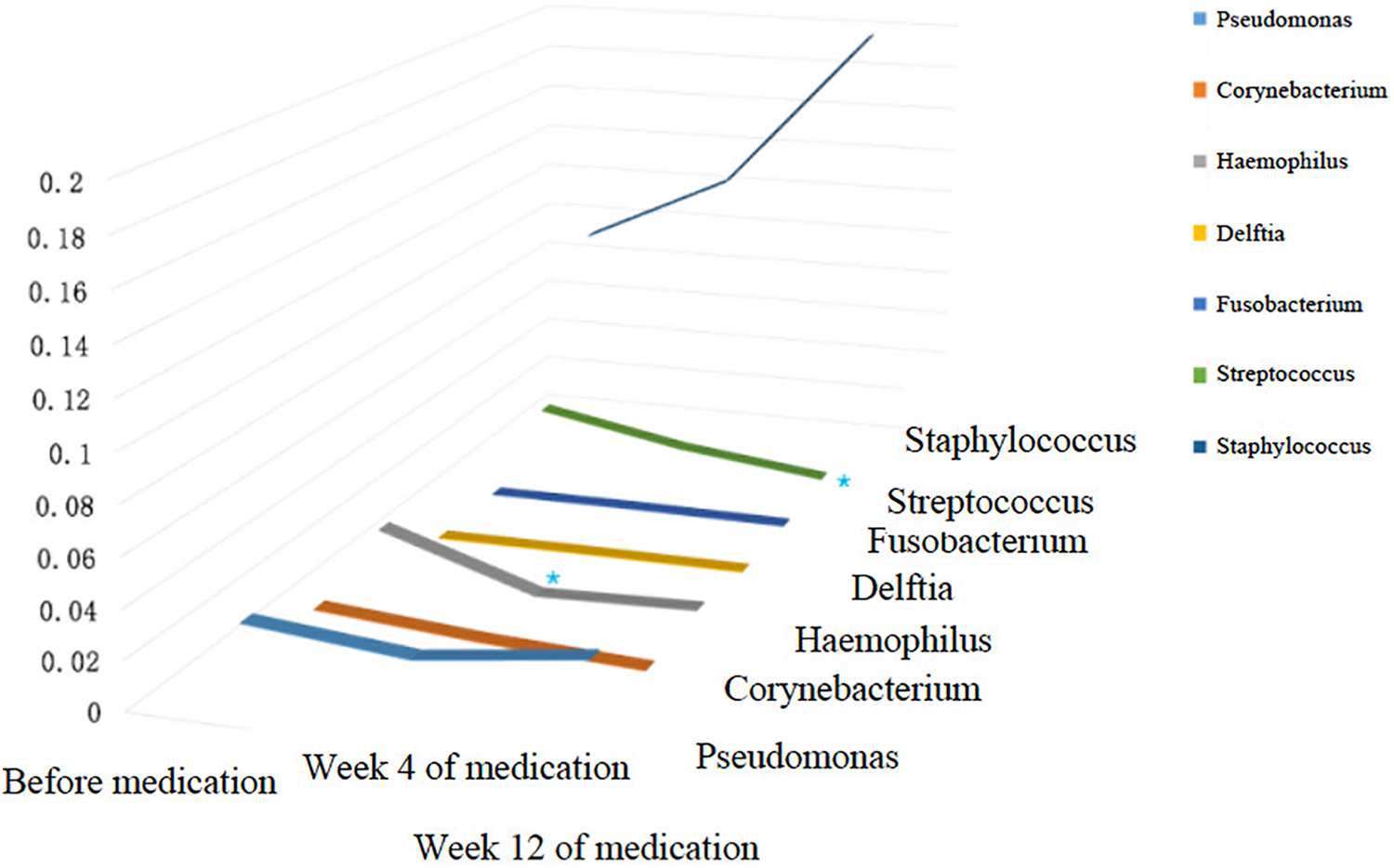
Three-dimensional variation trend chart of the top 7 generas in terms of relative abundance at genera level.
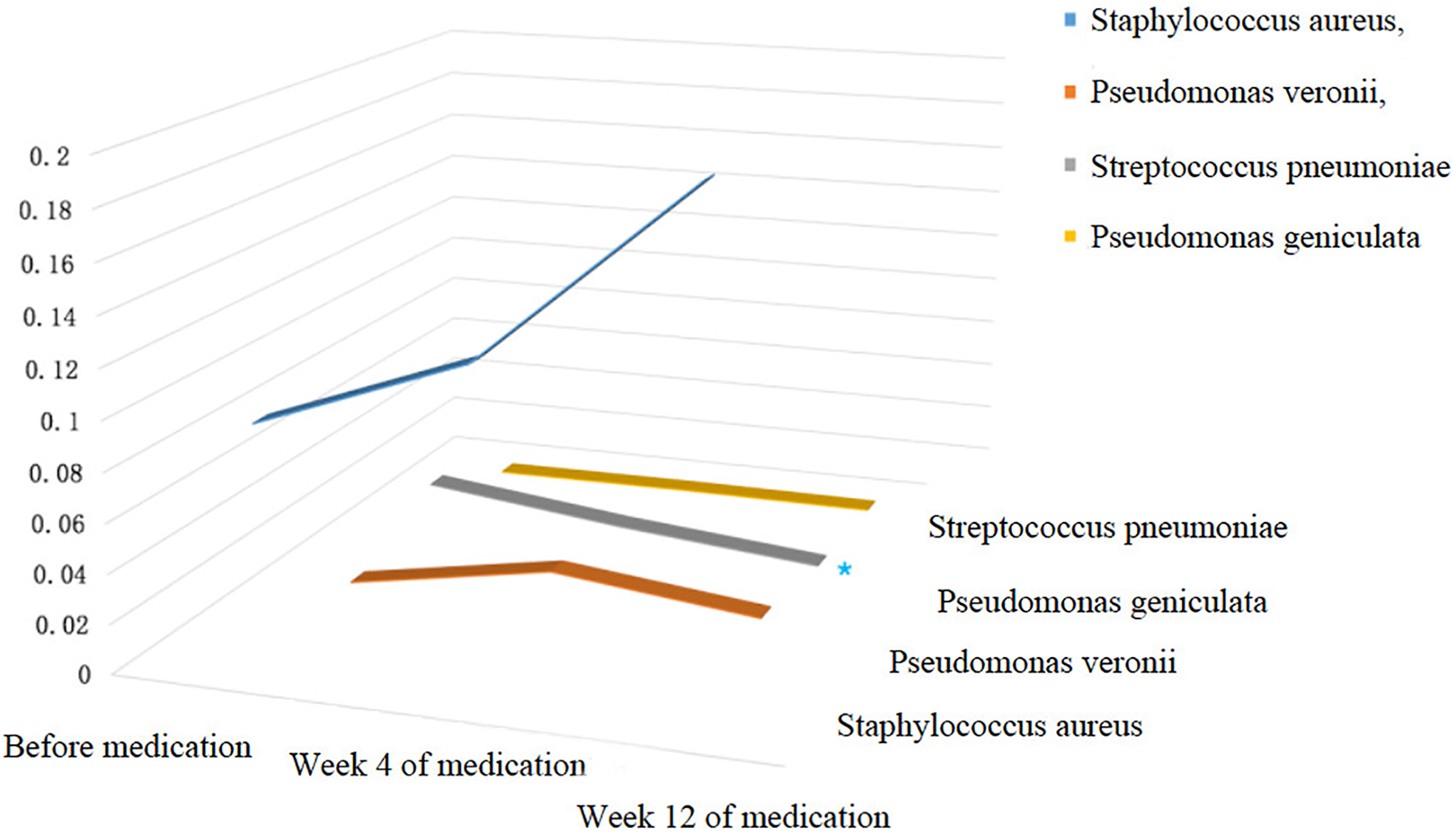
Three-dimensional variation trend chart of the top 4 strains in terms of relative abundance at species level.
Diversity
Shannon index was used to measure the diversity of the microbiota in the nasal cavity before and after treatment. As shown in Figure 9, no statistical difference was observed in the multiple comparisons among these 3 groups.
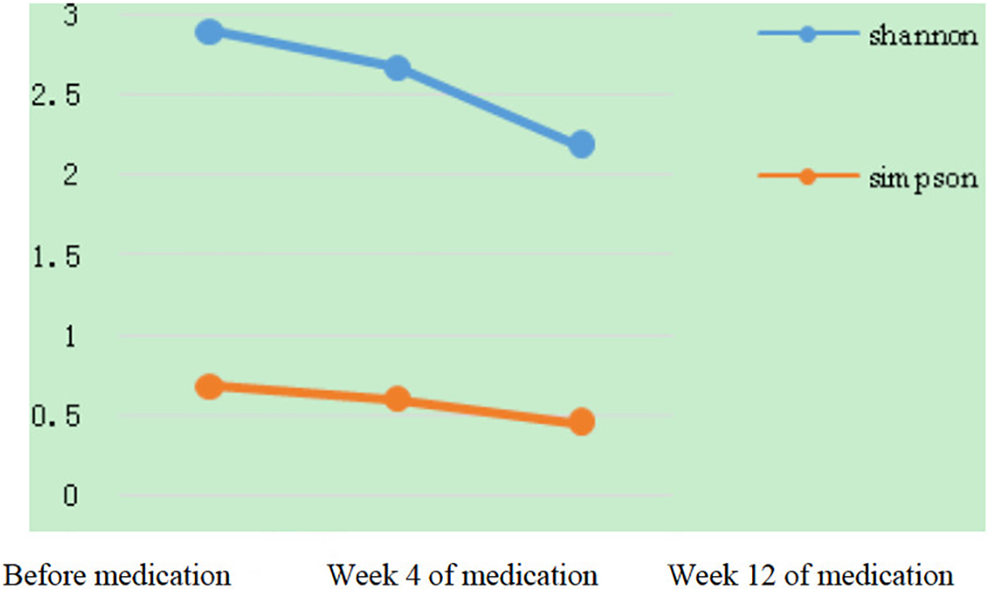
Diversity index variation trend chart.
Discussion
There are many studies on the long-term low-dose medication of macrolides for treatment of CRS, with an overall effective rate of 60% to 80%. 16 However, there are relatively few literatures analyzing the curative effects of macrolide drugs in patients with RCRS. According to the results of a self-controlled study on 13 postoperative patients with RCRS by Zuo and Xu, 17 the decreases in the scores of subjective symptoms and endoscopic scores after oral treatment with clarithromycin 250 mg/d for continuous 12 to 28 weeks had statistical significance. According to the results of a prospective open-label study on 17 postoperative patients with RCRS by Cervin et al, 18 the decreases in the scores of nasal congestion, rhinorrhea, and viscous nasal discharge among subjective symptoms and the decreases in endoscopic score after 12 weeks of oral treatment with clarithromycin 250mg/d or erythromycin ethylsuccinate 250 mg twice daily for continuous 12 weeks had statistical significance. However, in these 2 studies, esosinophils and/or IgE levels before treatment were not evaluated, and dynamic evaluation of efficacy and safety at different time points during treatment was lacking.
Macrolides are used for their immunomodulatory properties, which include the blockage of proinflammatory cytokines, inhibition of neutrophil adhesion and migration, and changes to mucus synthesis and secretion. 19 Prior studies support macrolide therapy as the most effective in nonatopic, noneosinophilic patients. In a double-blind, randomized, placebo-controlled study, Wallwork et al reported improvement in SNOT-20 score, nasal endoscopy, saccharine transit time, and interleukin 8 levels in lavage fluid in patients with CRS treated with 3 months of roxithromycin therapy, but only significantly in the low IgE subgroup. 20 Suzuki et al found a greater symptom improvement in patients with normal rather than high IgE level. 21 European Position Paper on Rhinosinusitis and Nasal Polyps 2020 also recommends macrolides as an optional treatment in patients with nontype 2 primary diffuse CRS. 22 As for eosinophils, Oakley reported low tissue and serum eosinophilia may predict a CRS phenotype suitable to a trial of long-term macrolide therapy in a case-control study. 23 In another double-blind, randomized, placebo-controlled study, Lin found that adding low-dose clarithromycin to oral corticosteroids as a post-ESS therapy showed beneficial subjective and objective outcomes in patients with chronic rhinosinusitis with nasal polyposis, especially those without tissue eosinophilia. 24 That’s the reason our study selected the patients with nonatopic, noneosinophilic RCRS with normal serum IgE. However, although this association was found, other studies reported discordant results. 12 Seresirikachorn reported that low total IgE levels in the nasal secretion but not in the serum predict macrolides response in a prospective cohort study. 25 The key point is whether systemic allergy test results could reflected the local anaphylaxis in patients with CRS. Du et al identified a relationship between type-2 inflammation biomarkers (EOS and IgE) in polyp tissue and those in blood, of which the r values were 0.423 and 0.530. 26 It is still controversial whether serum IgE and eosinophils could predict the result of macrolide therapy, additional studies are required to assess the exact role of IgE and eosinophils in serum and in nasal secretion in clinical practice.
Zheng et al 27 reported a randomized controlled trial in 43 cases for dynamic observation of the clinical efficacy of macrolide drugs. According to the results, compared with baseline, after 4 weeks of oral administration of clarithromycin, symptoms including nasal congestion, rhinorrhea, and general discomfort relieved, and endoscopic scores including edema in the surgical cavity and rhinorrhea improved. After continuous treatment till the 12th week, subjective symptoms and endoscopic scores showed further improvement. However, this study was conducted in patients with CRS without nasal polyps. In the present study, 17 patients having CRS with normal IgE and eosinophil levels who responded poorly to ESS and standard postoperative therapies were treated with long-term low-dose clarithromycin and followed up for 12 weeks. The main symptoms in enrolled patients were nasal congestion, rhinorrhea, postnasal drip, hyposmia, and edema in the surgical cavity. It was reasonable to classify these patients as RCRS from clinical perspective. After 4 weeks of oral administration of clarithromycin, subjective symptoms including nasal congestion, rhinorrhea, postnasal drip, and general discomfort significantly relieved. Endoscopic score improved in terms of general surgical cavity condition, rhinorrhea, and edema, while inflammatory signs like edema and vesiculation of surgical cavity mucosa still not fully recovered. After continued administration of clarithromycin for 12 weeks, the subjective symptoms showed no further significant improvement, indicating symptoms stabilized after 4 weeks of treatment. Noteworthily, according to endoscopic observation, surgical cavity mucosa was better epithelized, and the endoscopic scores were significantly improved compared with baseline as well as 4 weeks after treatment. These findings suggested that the subjective symptoms severity in patients with RCRS do not necessarily synchronize with mucosal inflammation and recovery status of nasal cavity. Also in this case, although subjective symptoms had been improved significantly after 4 weeks of oral clarithromycin therapy, 12 weeks of treatment could achieve more significant and more stable therapeutic effect regarding mucosal recovery and epithelialization extent.
It has been reported that biofilm formation could be found in 30% to 82% of patients with CRS 28,29 and is one of the important reasons for delayed recovery from mucosal inflammation after ESS. 30 Increased abundancy of various bacteria strains including S pneumoniae, Haemophilus influenzae, Staphylococcus, and Pseudomonas aeruginosa was reported to be related to the formation of bacterial biofilm. 31 -33 Previous studies have shown that administration of clarithromycin may have inhibitory effect on biofilms formed of S pneumoniae. 34 In this study, the proportion of S pneumoniae in the nasal flora of patients with RCRS decreased significantly after 12 weeks of clarithromycin administration (P < .05), indicating that clarithromycin may promote the improvement of clinical symptoms and recovery of surgical cavity mucosa by regulating nasal cavity microbiota and inhibiting bacterial biofilm formation. The safety of the long-term oral administration of macrolides has always been controversial. The safety concern of macrolides in most studies mainly focused on gastrointestinal reactions, allergic reactions, and liver function impairment, while there are relatively few evidence about its effect on the diversity of nasal flora. The results of this study showed that there was no significant change in the richness and diversity of nasal flora after long-term low-dose oral administration of clarithromycin for up to 12 weeks in the postoperative patients with RCRS, suggesting that long-term low-dose oral administration of clarithromycin has low risk to cause nasal flora imbalance. It is worth noting that in this study, the nasal flora of the postoperative patients with RCRS was mainly composed of Staphylococcus, Pseudomonas, Corynebacterium, Haemophilus, Delftia, Fusobacterium, and Streptococcus at the genera level, and of S aureus, P veronii, S pneumonia, and Pseudomonas geniculata at the species level, which is not fully consistent with previous literature. 35 The different composition may be related to various factors such as different races, differences in inclusion/exclusion criteria, different antibiotic exposure frequency, and so on. This also indicated the necessity to conduct similar studies in other races.
In summary, postoperative long-term low-dose oral administration of clarithromycin (250 mg/d) in patients with RCRS can significantly improve the clinical symptoms and promote mucosal epithelialization; the efficacy is more significant and more stable after 12 weeks of treatment, with good tolerability and safety. The efficacy of clarithromycin in patients with RCRS may be related to its regulatory effect on nasal flora. Due to the small sample size and short observation time in this study, long-term studies with large sample size are needed to make solid conclusions.
Footnotes
Authors’ Note
Han Chen is the first author; Bing Zhou is the corresponding author; Qian Huang, Zhenxiao Huang, and Mingjie Wang guided in writing the paper. Cheng Li, Yubin Wu, Yunxia Li, Jing Qu, and Nianci Xiao helped to collect the case. This study is a prospective, single-arm, single-center and self-controlled trial designed in compliance with the Declaration of Helsinki, ICH-GCP and relevant laws and regulations of China authority. The registered ethical number for this study is TRECKY2017-030 from Ethics Committee of Beijing Tongren Hospital, Capital Medical University. The Chinese clinical trial registration number is ChiCTR1800014382. Patients who met the inclusion/exclusion criteria are well-informed of the study and signed the informed consent form before enrollment.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: National Natural Science Foundation of China (81770977); Beijing Natural Science Foundation Program and Scientific Research Key Program of Beijing Municipal Commission of Education (KZ201410025029).
