Abstract
Objectives:
Otitis media with effusion (OME) is characterized by the presence of fluid in the middle ear without the presentation of signs or symptoms of acute ear infection. The point prevalence of OME reaches as high as 60% in children younger than 2 years of age. We used the National Health Insurance Research Database (NHIRD) to investigate the use of medication in children with OME before receiving ventilation tube insertion (VTI).
Methods:
Data of pediatric patients (age ≤ 12 years) who had OME and received VTI from January 1, 2011, to December 30, 2012, were retrieved from the Taiwan NHIRD. We surveyed the use of 4 medications to understand whether health care providers achieved the standards of medication use recommended by clinical practice guidelines.
Results:
This study examines the factors affecting the use of medication for pediatric OME. Overall, according to the study’s operational definitions, the use of systemic antibiotics was most common (59.9%), followed by systemic antihistamines (23.4%), systemic steroids (8.8%), and intranasal steroids (9.6%). Systemic antibiotics use was associated with 12 factors. Ten of the 12 factors increase the use of systemic antibiotics, including namely age (age > 2 years), comorbidities, teaching hospital, and community hospital. In contrast, namely catastrophic illness and watchful waiting are the 2 factors that decrease systemic antibiotics use. For the use of systemic antihistamines, systemic steroids, and intranasal steroids were related to 6, 5, and 2 factors, respectively.
Conclusions:
The rate of drug use differs from the rate of use recommended by commonly used clinical practice guidelines. We found that the higher the number of factors that influenced the patients’ drug use, the higher the rate of drug use. According to these results, drafting a treatment guideline for OME patients in accordance with current clinical practices in Taiwan is highly recommended.
Keywords
Introduction
Otitis media with effusion (OME) is characterized by the presence of fluid in the middle ear without the presentation of signs or symptoms of acute ear infection. It differs from acute otitis media (AOM), which is defined based on the presence of middle ear infection accompanied with symptoms of fever, ear pain, and active inflammation. Both OME and AOM are characterized by the presence of fluid in the middle ear; however, in OME, the fluid is not actively infected, and pain may be absent or minimal. 1 Although OME usually resolves spontaneously, it occurs frequently after AOM. Moreover, the 3-month resolution rate is estimated to be approximately 75% among children with OME when OME follows an episode of AOM.
The point prevalence of OME reaches as high as 60% in children younger than 2 years of age. 1,2 In some cases, the middle ear fluid may persist from weeks to months. 3 Because OME usually occurs during a child’s critical developmental phase of speech, language, and classroom learning, the potential sequelae of hearing loss and other developmental problems must be clarified, discussed, and managed by clinicians and parents. Chronic OME, defined as OME that persists for longer than 3 months, 4 is an indication for surgical approaches such as ventilation tube insertion (VTI).
The high prevalence and potential sequelae necessitate the development of a standardized treatment for OME. Since 1994, the American Academy of Otolaryngology—Head and Neck Surgery Foundation, the American Academy of Pediatrics, and the American Academy of Family Physicians have collaborated to release a guideline for the care of children with OME who received VTI. According to both the 1994 5 and 2004 6 guidelines, the task force does not support the routine use of systemic antibiotics, systemic or intranasal steroids, antihistamines, or decongestants as an intervention for OME. The 2016 guideline, 1 an updated version of the 2004 guideline, further provides evidence-based, strong recommendations to clinicians that antibiotics, steroids, antihistamines, or decongestants should not be used for OME treatment.
Since the inception of the National Healthcare Insurance (NHI) system in Taiwan in 1995, the NHI program has served as the single payer Insurance system for almost all health care expenditures in Taiwan, 7,8 which means that in order to claim payment, health care practitioners and organizations must submit to the NHI information regarding all services they have provided. Therefore, we used the National Health Insurance Research Database (NHIRD) to investigate the following: (1) medication (systemic antibiotics, systemic antihistamines, systemic steroids, or intranasal steroids) use in children with OME before receipt of VTI, (2) factors affecting the scale of the aforementioned drugs’ use, and (3) methods to improve the quality of care in terms of adherence to guideline recommendation against the use of medications in children with OME.
Methods
Study Design and Data Collection
The data of pediatric patients (age ≤ 12 years) who had OME (International Classification of Diseases, Ninth Revision code 381.1 or 381.2) and received VTI (procedure code 84015B) from January 1, 2011, to December 30, 2012, were retrieved from the Taiwan NHIRD. Medication use was evaluated in terms of adherence to clinical practice guidelines. Clinical practice guidelines strongly recommend against the use of systemic antibiotics, systemic antihistamines, systemic steroids, and intranasal steroids by clinicians for treating OME (Table 1). We used operational definitions of medication use to perform the analysis: systemic antibiotics should not be used for more than 2 weeks within 90 days before VTI receipt and systemic antihistamines, systemic steroids, and intranasal steroids should not be used for more than 45 days within 90 days before VTI receipt. We surveyed 4 medications to understand whether health care providers achieved the standards of medication use recommended by clinical practice guidelines.
Medication Use to be Examined and Their Definitions.
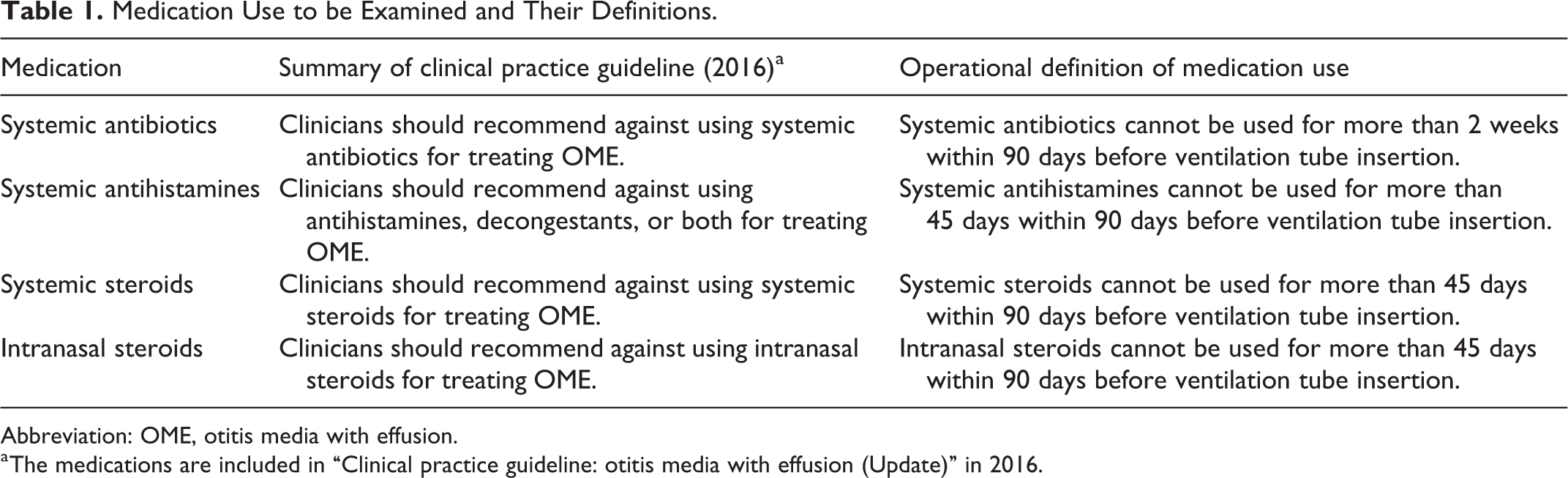
Abbreviation: OME, otitis media with effusion.
a The medications are included in “Clinical practice guideline: otitis media with effusion (Update)” in 2016.
We investigated whether patient characteristics (eg, age and sex); watchful waiting; comorbidities such as acute paranasal sinusitis (APS), chronic paranasal sinusitis (CPS), acute bronchitis, pneumonia, bronchopneumonia, nasal allergy, asthma, and other infections; catastrophic illness (according to the Registry of Catastrophic Illness Patients Database of the NHIRD, such as rheumatoid arthritis, multiple sclerosis, and transplants); hospital level (medical center, regional, or community hospitals); and geographical area (Taipei, North, Middle, South, Kaohsiung/Pingtung, and East of Taiwan) are relevant predictors that may influence the use of systemic antibiotics, systemic antihistamines, systemic steroids, and intranasal steroids in children with OME before VTI receipt. This study was used by the NHIRD, and the data collection of the patients does not involve patient privacy and personal information, the informed consent is exempted. This study was approved by the ethic committee (IRB No: CGH-P103011).
Statistical Analysis
The numbers and percentages of patients who met the definition of medication use are presented. By using the χ2 test, we compared various factors that might affect the rate of adherence to clinical practice guidelines. A multivariate regression model was used to assess the dependent variable, defined as the number of patients who received the treatment consistent with the operational definition in this study, to what extent are influenced by independent variables such as patient characteristics, hospital level, and demographic area. Odds ratios and 95% CIs were calculated. Significance levels were set at α = .05.
Results
A total of 1159 pediatric patients were identified, of whom 706 (60.9%) were male, and 453 (39.1%) were female; the mean age was 5.7 ± 2.5 years; and 923 (79.6%) patients also had nasal allergy. Table 2 presents the characteristics of the study population.
Demographics of Study Population.a
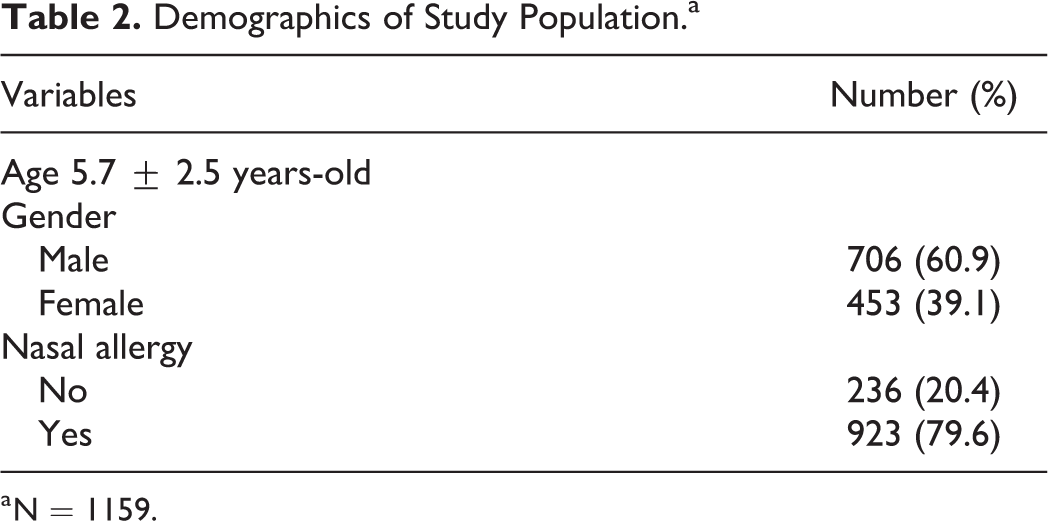
a N = 1159.
Among 1159 patients, 694 (59.9%), 271 (23.4%), 102 (8.8%), and 111 (9.6%) patients received qualified treatments not recommended by clinical practice guidelines for the use of systemic antibiotics, systemic antihistamines, systemic steroids, and intranasal steroids, according to the study’s operational definitions. The rate of adherence to systemic steroid use was the highest (91.2%), and that of adherence to systemic antibiotics use was the lowest (40.1%), as shown in Figure 1.
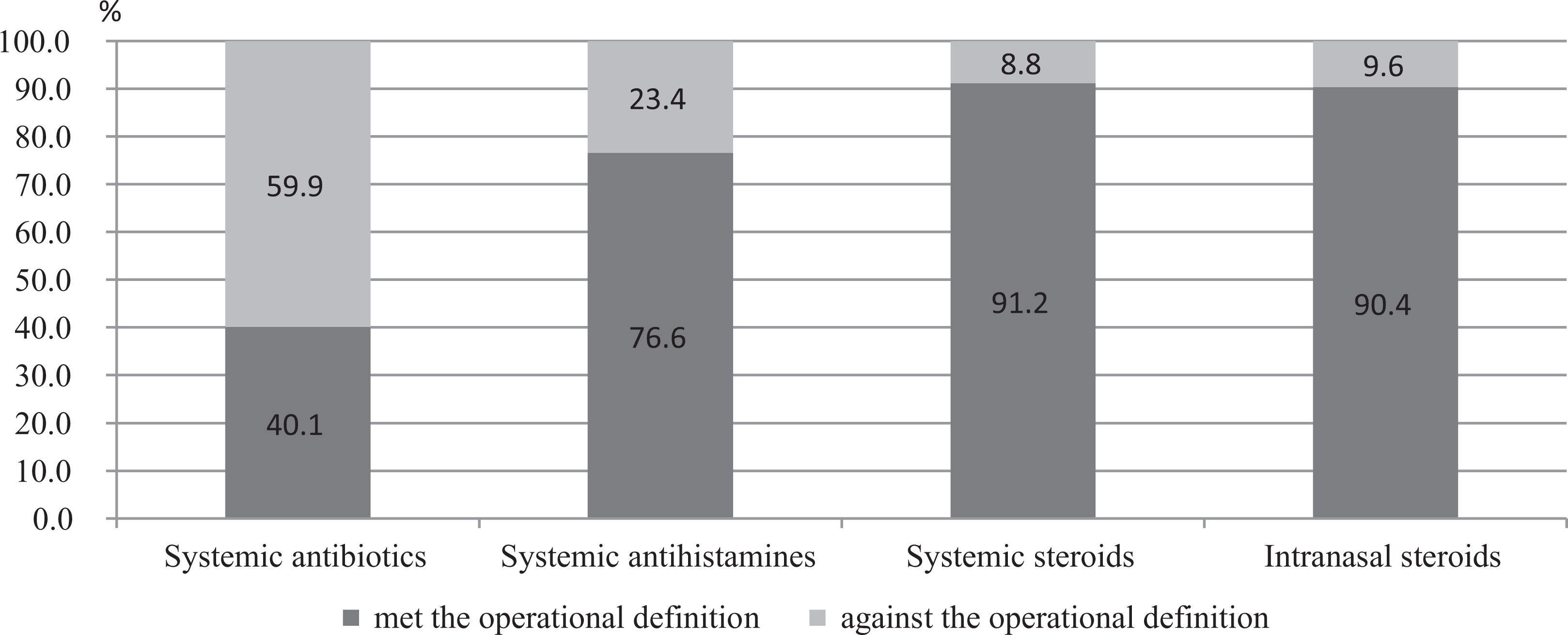
National medication use and indicators.
Factors that might influence the use of systemic antibiotics, systemic antihistamines, systemic steroids, and intranasal steroids were further analyzed, and the results are presented in Table 3. No significant difference in the use of medication comprising the 4 drug classes was observed between male and female patients.
OME Medication Indicators Comparative Analysis Before VTI (N = 1159) Based on Patient Characteristics.a
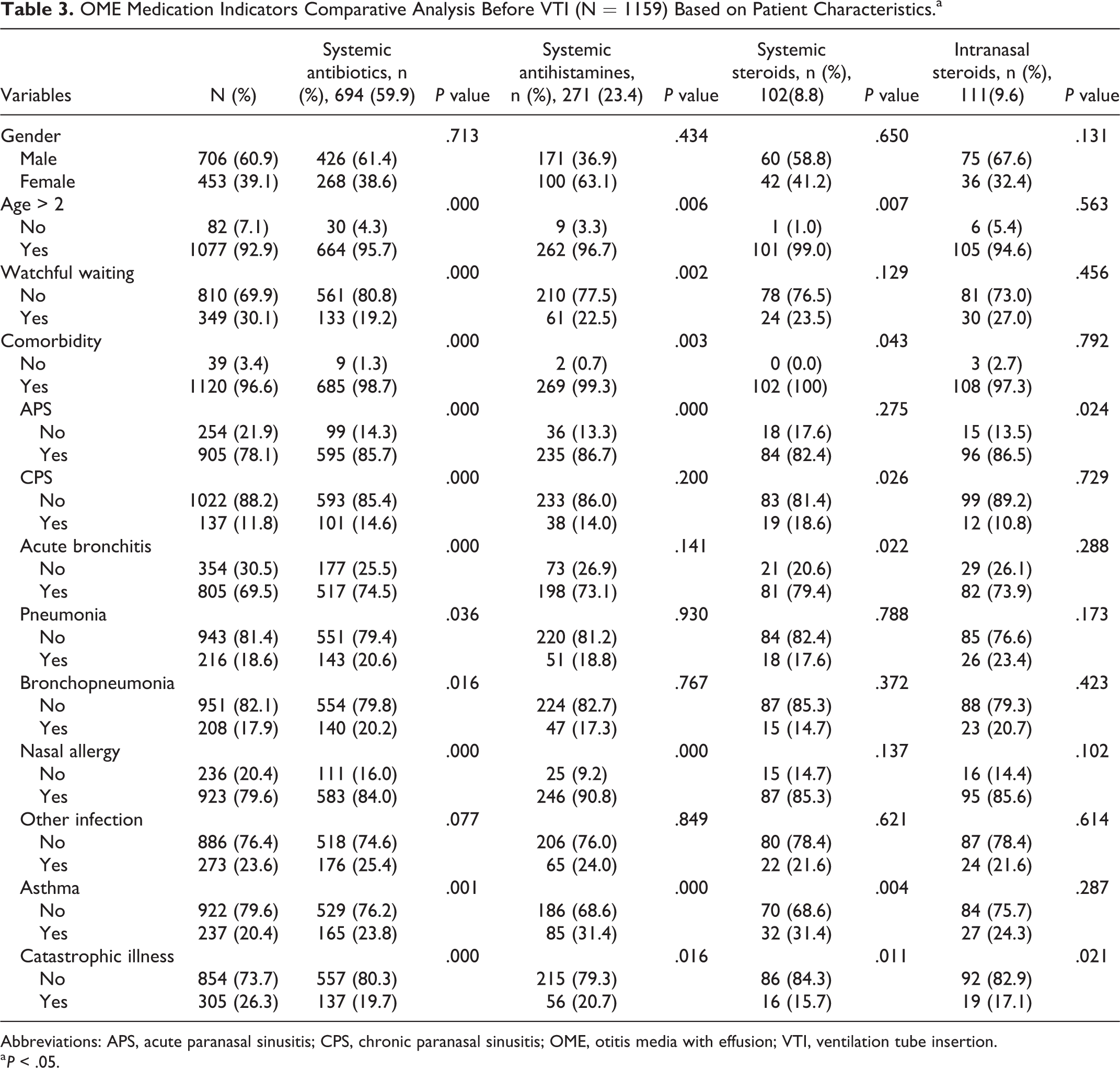
Abbreviations: APS, acute paranasal sinusitis; CPS, chronic paranasal sinusitis; OME, otitis media with effusion; VTI, ventilation tube insertion.
a P < .05.
There are 10 factors that significantly influenced the use of systemic antibiotics, namely age (age > 2 years), comorbidities (APS, CPS, acute bronchitis, pneumonia, bronchopneumonia, nasal allergy, and asthma), catastrophic illness, and watchful waiting (P < .05). The 6 factors that significantly influenced systemic antihistamines use were age (age > 2 years), comorbidities (APS, nasal allergy, and asthma), catastrophic illness, and watchful waiting (P < .05). Systemic steroids use was significantly impacted by 5 factors, namely age (age > 2 years), 3 of 8 comorbidities (CPS, acute bronchitis, and asthma), and catastrophic illness (P < .05). Only 2 factors, comorbidities (APS) and catastrophic illness, significantly influenced the use of intranasal steroids (P < .05).
Regarding the administrative level and location of hospitals where patients with OME visited, the use of only one drug class (systemic antibiotics) was significantly influenced (P < .05) by factors such as teaching hospital and geographical area (Table 4).
OME Medication Indicators Comparative Analysis Before VTI (N = 1159) Based on Hospital Characteristics.a
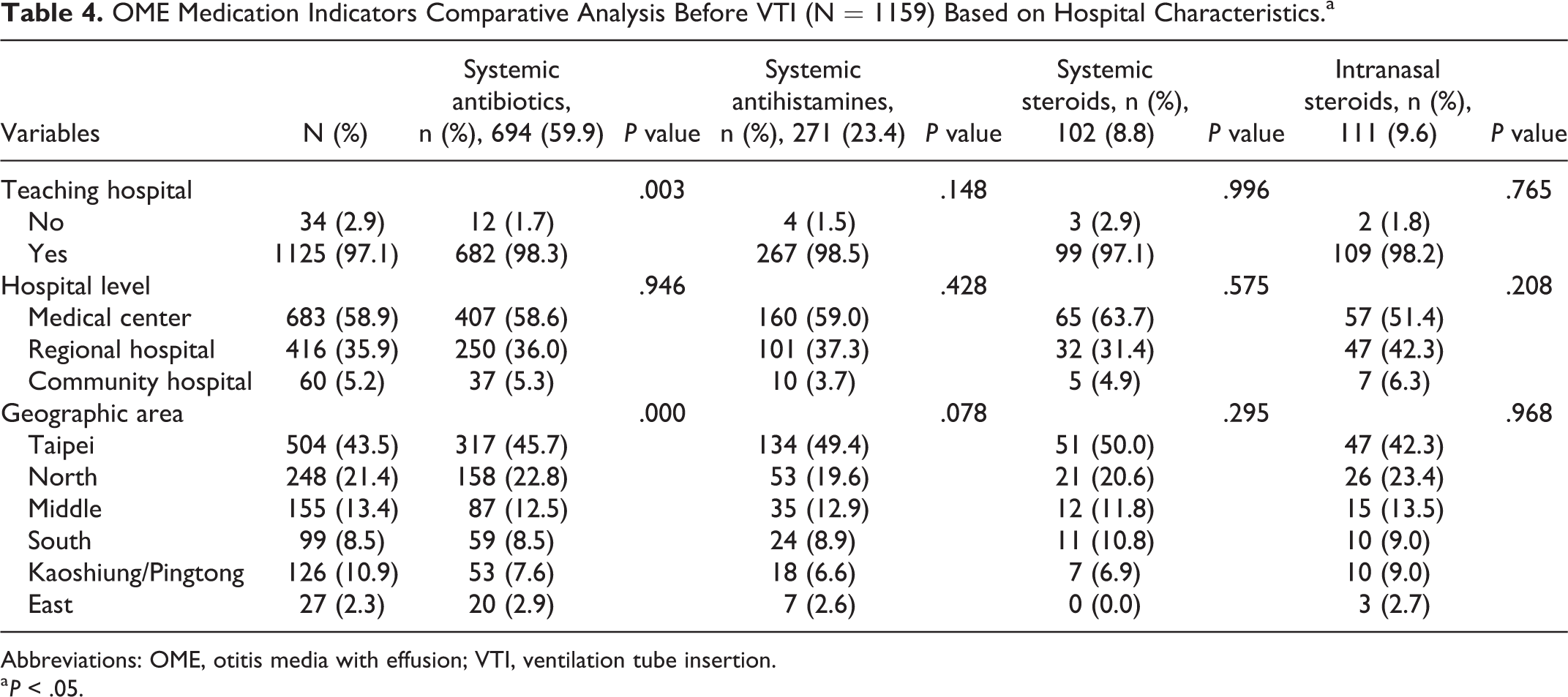
Abbreviations: OME, otitis media with effusion; VTI, ventilation tube insertion.
a P < .05.
We also used a multivariate logistic regression analysis to investigate the extent to which factors such as male/female sex, age, watchful waiting, comorbidity, catastrophic illness, teaching hospital, hospital level, and geographical area affected the use of drugs in OME patients, and the results are presented in Table 5. Four factors might increase the use of systemic antibiotics, namely age (age > 2 years), comorbidity, teaching hospital, and community hospital; 2 factors might reduce the use of systemic antibiotics, namely catastrophic illness and watchful waiting; and only watchful waiting appeared to influence the decreased use of systemic antihistamines.
Multivariate Logistic Regression Analysis of OME Medication Indicators Before VTI.a
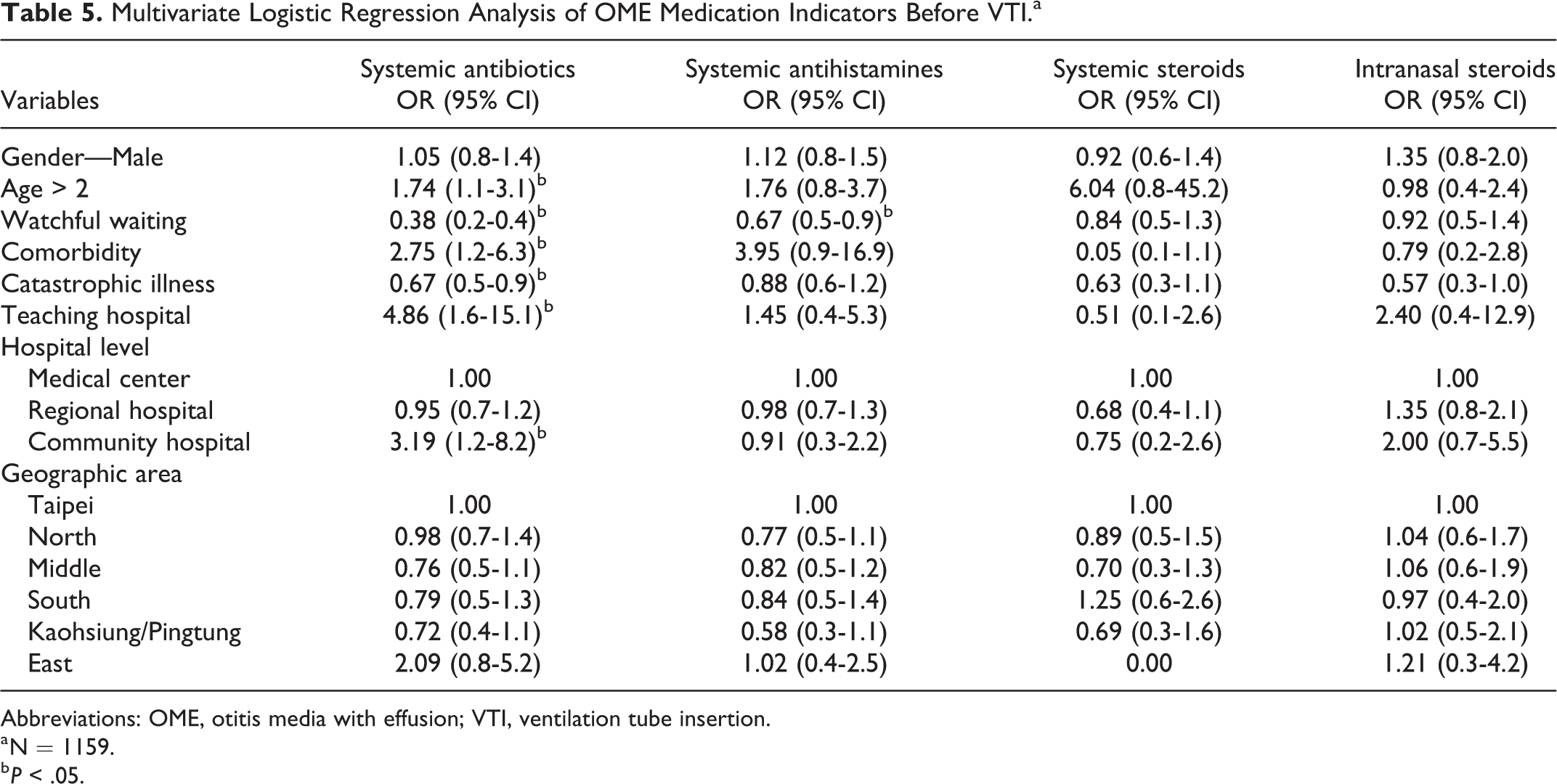
Abbreviations: OME, otitis media with effusion; VTI, ventilation tube insertion.
a N = 1159.
b P < .05.
Among the factors that significantly affected drug use in OME patients, comorbidity was the most relevant; 98.7%, 99.3%, 100%, and 97.3% of patients who used systemic antibiotics, systemic antihistamines, systemic steroids, and intranasal steroids, respectively, also had comorbidities. However, the effect was nonsignificant for patients using intranasal steroids. These results clearly demonstrated that among patients who received treatment that was not in accordance with the clinical practice guidelines and who had been prescribed the aforementioned 4 classes of drugs, the percentage of comorbidities was highly similar; the 3 most common diseases (nasal allergy, APS, and acute bronchitis) occurred at least 3-fold more frequently than the remaining 5 comorbidities.
The results revealed that patients enrolled in the NHIRD had a higher rate of receiving systemic antibiotics (59.9%), systemic antihistamines (23.4%), systemic steroids (8.8%), and intranasal steroids (9.6%). In addition, the study demonstrated a positive correlation between drug use and influencing factors—the higher the number of factors that related to the patients’ drug use pattern, the higher the rate of drug use. Systemic antibiotics had the highest rate of use (59.9%), and 12 influencing factors might have associated with its use; among them, 10 factors increased the drug use rate, namely age (age > 2 years), comorbidities (APS, CPS, acute bronchitis, pneumonia, bronchopneumonia, nasal allergy, and asthma), teaching hospital, and community hospital; and 2 factors reduced the drug use rate, namely catastrophic illness and watchful waiting. The rate of use of systemic antihistamines was 23.4%; 6 factors influenced its use, of which watchful waiting reduced its use. Systemic steroids had the lowest rate (8.8%) of use, and 5 factors influenced its use. The rate of use of intranasal steroids was also relatively low (9.6%), and 2 factors influenced its use.
Discussion
This study found that the drug use pattern recorded in the NHIRD was significantly different from the recommendations of clinical practice guidelines. 1,5,6,9,10 Pharmacological treatments may have short-term symptomatic effectiveness, the absence of long-term effectiveness, the associated adverse events, and the cost mean that they cannot be recommend in the treatment of OME. 11 Moreover, the methods of relevant studies published outside Taiwan 12 -15 mainly involved collecting statistics of the clinical rate of medication use. The rate of use of systemic antibiotics was 32% (US), 64.1% (Turkey_pediatricians), and 60.5% (Turkey_otorhinolaryngologists) that of systemic antihistamines was 9.5% (US), 26.2% (Turkey_pediatricians), and 14.0% (Turkey_otorhinolaryngologists) and that of intranasal steroids was 10.0% (US), 21.5% (Turkey_pediatriclans), and 57.0% (Turkey_otorhinolaryngologists). The rate of medication uses in Turkey literature published in 2018 was similar to Taiwan’s NHIRD in 2011 to 2012. They have a higher frequency of medication use than US.
Recurrence AOM
The 2016 guideline 1 recommended that clinicians should manage OME in children who are not at risk through watchful waiting for 3 months from the date of effusion onset. The proportion of completion for sufficient watchful waiting was 30.1% in Taiwan. In the current study, all patients received VTI; therefore, their clinical conditions are speculated to be more complicated. Such patients are most likely to have AOM recurrence. The annual incidence rate of AOM was estimated to be 64.5 cases per 1000 children, and the overall 1-year cumulative incidence rate of recurrence was 33.1% in Taiwan’s pediatric population. 16 In such patients with AOM recurrence, doctors cannot confidently persuade parents against the use of any drugs; consequently, drugs belonging to the studied 4 classes are prescribed against the clinical practice guidelines.
Comorbidity Among Patients
The current study highlights that higher the number of comorbidities, higher the rate of drug use. Among the influencing factors, comorbidity has the most relevant effect on rate of drug use. The comorbidities included in the current study were the most common diagnoses coded by ENT physicians in Taiwan. Among the comorbidities included in this study, nasal allergy, APS, and acute bronchitis had the most relevant effect on rate of drug use. These 3 conditions present concomitant with OME. When a patient presents certain symptoms that cannot be differentiated using a specific diagnosis at that moment, the patient might receive a diagnosis of all concomitant conditions in order to manage symptoms as soon as possible. This process is called “upcoding.” Further, up to 59.9% of OME patients received systemic antibiotics, which might be associated with the remarkably high rate (98.7%) of comorbidity coded with OME at the same time. A total of 23.4% of OME patients had received treatment with systemic antihistamines, and among these patients, 99.3% also had comorbidities. The rate of use of systemic steroids and intranasal steroids was 8.8% and 9.6%, respectively, and the corresponding comorbidity rate was 100% and 97.3%. Among the comorbidities, nasal allergy, APS, and acute bronchitis were vital factors that associated with the use of the 4 classes of drugs in this study.
Catastrophic Illness
Catastrophic illness significantly reduced the rate of drug use. A total of 26.3% of patients with catastrophic illness received treatment with drugs belonging to the 4 drug classes studied.
Hospital Characteristics
In terms of the characteristics of health care institutions patients visited (teaching hospital, administrative level of hospital, and location of hospital), the high nonadherence rates may be attributed to the following: (1) Health system–related factors: patients who visit teaching hospitals tend to have more complicated diseases and therefore receive treatment with drugs more frequently; (2) economic and social factors: patients with parents with a high social-economic status are prone to request the type of treatment they want to receive; (3) health care provider–related factors (physician inertia): physicians who practice at community hospitals may less confidently persuade parents against the use of drugs in treating OME; therefore, the rate of drug use tends to be higher; and (4) patient-related factors: there may be increased concerns regarding not using any drug among patients and parents; consequently, physicians have no choice but to prescribe drugs, thus increasing the rate of drug use.
Geographical Area
The lowest rate of drug use in the Kaohsiung/Pingtung area might be associated with the weather. The weather in the south of Taiwan is drier and warmer, which may render nasal allergy and acute infection less prevalent. After treatment, physicians may not know the effectiveness of treatment if patients did not attend the follow-up visit. Therefore, the analysis of data from the Taiwan NHIRD in this study might elucidate the overall drug use among OME patients in Taiwan, which might serve as a reference for physicians for modifying their prescription patterns in the future.
There was no clinical information from the claim database, the association between guideline compliance in medication use and the otitis media treatment outcomes cannot be established. The exploration of this study is limited. In the future, studies should adopt methods such as questionnaire survey and medical chart review to determine the reason for the noncompliance by physicians to clinical practice guidelines in treating OME patients.
Conclusion
The rate of drug use among OME patients aged ≤12 years before VTI receipt, who were enrolled in the Taiwan NHIRD, differed from the rates recommended by commonly used clinical practice guidelines. We found that the higher the number of associated factors of the patients’ drug use pattern, the higher the rate of drug use. The rate of use of the 4 classes of medications included in this study (systemic antibiotics, systemic antihistamines, systemic steroids, and intranasal steroids) was influenced by factors namely age, watchful waiting, comorbidities (nasal allergy, APS, and acute bronchitis), catastrophic illness, and hospital characteristics. Among the 4 classes of drugs, systemic antibiotics had the highest rate of use, and the relevant factors that might have increased the rate of use are age (age > 2 years), comorbidities, teaching hospital, and hospital level (community hospital). We suggest that the study results should be used as reference for drafting treatment guidelines for OME patients in accordance with current clinical practices in Taiwan in the future. Further, the auditing measures should be undertaken and reinforced for the use of drugs in the treatment of OME patients at all levels of hospitals in Taiwan.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
