Abstract
Background:
Children with cleft palate commonly experience otitis media with effusion (OME), leading to conductive hearing loss. The effectiveness of tympanostomy tube placement in improving hearing outcomes in this population remains limited.
Methods:
A retrospective cohort study included 44 non-syndromic children undergoing primary single-stage cleft palate repair at Maastricht University Medical Centre. Tympanostomy tubes were placed during primary surgery. The primary outcome was the change in hearing thresholds post-intervention. Secondary outcomes included OME prevalence, hearing loss type, frequency of tube placements, and tympanic membrane abnormalities.
Results:
OME was present in 88.6% of patients. Conductive hearing loss was found in 31.8% and sensorineural loss in 4.5%. A significant mean improvement of 14.13 dB (P < .001) was observed post-intervention. No significant difference was found between ears (P = .916). Random intercept variance at the child level was 49.07 (standard deviation = 7.00), indicating substantial interindividual variability. Tympanic membrane abnormalities were observed in patients with repeated tube placements, but did not cause long-term hearing deterioration.
Conclusion:
Tympanostomy tube placement improves hearing outcomes in children with cleft palate. When OME is present at the time of cleft surgery or there is a documented history of recurrent OME or acute otitis media, concurrent tube placement should be considered to alleviate conductive hearing loss. Prospective studies are warranted to confirm these findings and optimize care.
Keywords
Introduction
Orofacial clefts, among the most common congenital anomalies, have a global prevalence estimated at ~1 in every 1000 to 1500 newborns. 1 Cleft lip results from incomplete fusion of the maxillary prominences and medial nasal processes between the sixth and seventh weeks of gestation. In contrast, cleft palate arises from the failure of the palatal shelves to fuse between the 9th and 12th weeks. These anomalies can occur independently or together and may lead to feeding difficulties, speech impairment, and hearing loss. 2
In addition to well-documented feeding and speech difficulties, hearing loss is a major concern in children with a cleft palate. This is primarily caused by persistent otitis media with effusion (OME) due to Eustachian tube dysfunction. This dysfunction arises due to the abnormal anatomical positioning of the soft palate musculature in cleft palate, impairing the normal opening of the Eustachian tube. Poor function of the tensor veli palatini muscle, combined with velopharyngeal insufficiency, leads to fluid accumulation in the middle ear. 3
OME is a chronic condition, highly prevalent in children with cleft palate. It often recurs despite treatment, and it can persist into later childhood, leading to prolonged hearing deficits.3 -5 Compared to the general pediatric population, in which ~60% experience OME before the age of 2, the incidence is significantly higher among children with cleft palate, with rates reaching up to 97%. 6 Moreover, recurrence and persistence are more common in this group: while OME recurrence occurs in about 30% to 40% of children without a cleft, it affects up to 50% of those with cleft palate. Persistence beyond 1 year is observed in only 5% to 10% of non-cleft children, but in as many as 90% to 96% of cleft palate cases. 6 Tympanostomy tubes are frequently placed to address this issue by ventilating the middle ear, reducing fluid retention, and improving hearing outcomes. While conductive hearing loss is most common, less frequent cases of sensorineural or mixed hearing loss may also occur,3 -5 particularly in the context of underlying syndromic conditions such as 22q11.2 deletion syndrome 7 or Stickler syndrome. 8
Given that untreated hearing loss during critical periods of development can result in permanent speech delays and educational challenges, early diagnosis and intervention are essential to ensure optimal outcomes. In cases where cleft palate is accompanied by OME and hearing loss, simultaneous tympanostomy tube insertion at the time of palatoplasty is recommended, as palatoplasty alone may not sufficiently improve hearing in all patients. 9
While the efficacy of tympanostomy tubes in children without cleft palate is well established, evidence regarding their effectiveness in cleft palate-affected populations remains limited. This study contributes to the current body of evidence on this topic. In addition, there is currently no national consensus among cleft centers in the Netherlands regarding the routine placement of tympanostomy tubes during cleft palate repair. 10
Despite their clinical benefits, tympanostomy tubes are not without risk. Potential adverse effects include otorrhea, tympanosclerosis, focal atrophy, retraction pockets, persistent tympanic membrane perforation, and complications related to general anesthesia. 4 Some studies report positive outcomes of simultaneous tympanostomy tube placement during palatoplasty, suggesting that this approach may reduce OME recurrence and minimize the need for repeated anesthesia episodes, 11 while others are more cautious due to potential adverse effects. 12
Therefore, this study aims to assess the prevalence of OME and hearing loss, examine the frequency of tympanostomy tube placement, and evaluate hearing outcomes by comparing audiometric data before and after intervention.
Materials and Methods
Study Design
This study is a retrospective cohort study conducted at the Department of Otorhinolaryngology and Department of Plastic and Reconstructive Surgery of the Maastricht University Medical Centre (MUMC), MosaKids Children’s Hospital. Data was collected to evaluate hearing outcomes before and after tympanostomy tube placement. Placement was performed simultaneously with primary cleft palate repair. In addition, the prevalence and impact of OME and hearing loss in individuals with cleft-related anomalies were analyzed.
Study Population
We included children who underwent cleft palate repair at the MUMC+, involving a surgical approach defined as single-stage closure of both the hard and soft palate. Furthermore, tympanostomy tube placement and subsequent follow-up were also required to have been performed at MUMC+, with enrollment in the cleft team within 6 months postoperatively. Syndromic patients were excluded to ensure a homogenous study population. The primary surgical techniques used for cleft palate repair were the 2-flap palatoplasty and the Furlow double opposing Z-plasty.
All cleft palate repairs were performed by 2 experienced surgeons specialized in cleft surgery, ensuring consistency and reducing operator-related variability.
Tympanostomy tubes were placed simultaneously with cleft palate repair, with an average age at the time of surgery and tube placement of 9.9 months. Tympanostomy tubes were placed in all patients with hearing loss, and in selected cases without measurable loss as a preventive measure (eg, history of OME or recurrent acute otitis media).
Data Collection
Data was extracted from medical records (between June 19, 2012 and April 25, 2024), incorporating several patient characteristics, including demographic information (gender and age at the time of surgery) and cleft type (eg, soft palate only [Veau I], soft and hard palate [Veau II], unilateral cleft lip and palate [Veau III], and bilateral cleft lip and palate [Veau IV]). Hearing-related parameters included the presence of hearing loss, type of hearing loss (ie, sensorineural, conductive, and mixed), occurrence of OME, the number of tympanostomy tube placements (analyzed separately for each ear), and any tympanic membrane anomalies. Hearing was assessed before and after tympanostomy tube placement, exclusively using pure tone audiometry to determine air conduction thresholds. Changes in hearing thresholds refer to differences in air conduction thresholds, not the air-bone gap. Hearing loss was defined as a high Fletcher index (average pure tone threshold of 1, 2, and 4 kHz) >30 dB hearing level. In this study, hearing loss severity was categorized as follows: normal (up to 30 dB), mild (30-40 dB), moderate (41-55 dB), moderately severe (56-70 dB), severe (71-90 dB), and profound (>90 dB). 13 Patients not meeting the 30 dB threshold were classified as not having hearing loss for the purpose of this study, although minor or subclinical hearing impairments cannot be excluded.
Outcome Measures
The primary outcome measure was the evaluation of hearing loss, evaluated through changes in audiometric thresholds before and after tympanostomy tube placement.
Secondary outcome measures included the prevalence of OME, the type of hearing loss (conductive or sensorineural), the frequency of tympanostomy tube placements, and the presence of eardrum abnormalities.
The prevalence of OME was assessed based on previously documented diagnoses in the patients’ medical records up to the time of study assessment, which were established using a combination of micro-otoscopy and tympanometry. A type B tympanogram with a flat curve and normal canal volume was considered diagnostic of OME. Hearing loss was characterized using the most recent audiometric examination available for each patient, acknowledging that these assessments were conducted at varying ages. Postoperative audiometric evaluations were performed between 1 and 12 months after tympanostomy tube placement, with a median interval of 84 days (interquartile range: 55-105 days), generally as part of routine follow-up appointments. Similarly, the frequency of tympanostomy tube placements was summarized cumulatively across each patient’s lifetime. Eardrum abnormalities were identified during tympanostomy tube placement procedures, which were performed at various time points throughout the patients’ treatment histories. It should be noted that these abnormalities may not necessarily reflect the current condition of the tympanic membrane.
Ethical Considerations
Ethical approval for this study was granted by the Institutional Review Board of the MUMC, Maastricht, The Netherlands (METC 2024-0524).
Data Analysis
Baseline characteristics were summarized as mean and standard deviation (SD) or as count and percentage. Normality of the distribution of continuous outcome data was assessed through visual inspection of Q-Q plots. Given that the data were normally distributed, a linear mixed-effects model was employed to compare pre- and post-intervention levels of hearing loss, accounting for repeated observations within patients by including a random intercept for each individual.
Secondary outcome measures, including the prevalence of OME and hearing loss, and the frequency of tympanostomy tube insertions, were analyzed descriptively as count and percentage or as median and first and third quartiles, where appropriate.
All statistical analyses were performed in RStudio (version 2024.12.0 + 467), with a threshold of significance set at P < .05. No a priori sample size calculation was conducted, as we intended to include all available records for this exploratory study.
Results
Following an initial prescreening of the local cleft database, 48 children with cleft palate or cleft lip and palate, born between January 2011 and November 2018, were assessed for eligibility. Of those, 4 were excluded because their tympanostomy tube placement occurred at an external hospital, precluding consistent follow-up. The remaining 44 patients were included in the descriptive analyses. Of these 44, 14 were excluded from the audiometric analyses: 12 due to the absence of a postoperative audiogram conducted at our institution, and 2 who had not undergone tympanostomy tube placement. Consequently, the final analysis included 30 patients for the primary audiometric outcomes, while 44 patients (22 female, 22 male) were included in the descriptive analyses (secondary outcomes). This study population had a mean age of 9.9 years (± 2.1 SD) at the time of assessment. Details on the distribution of cleft types are presented in Table 1.
Patient Characteristics.
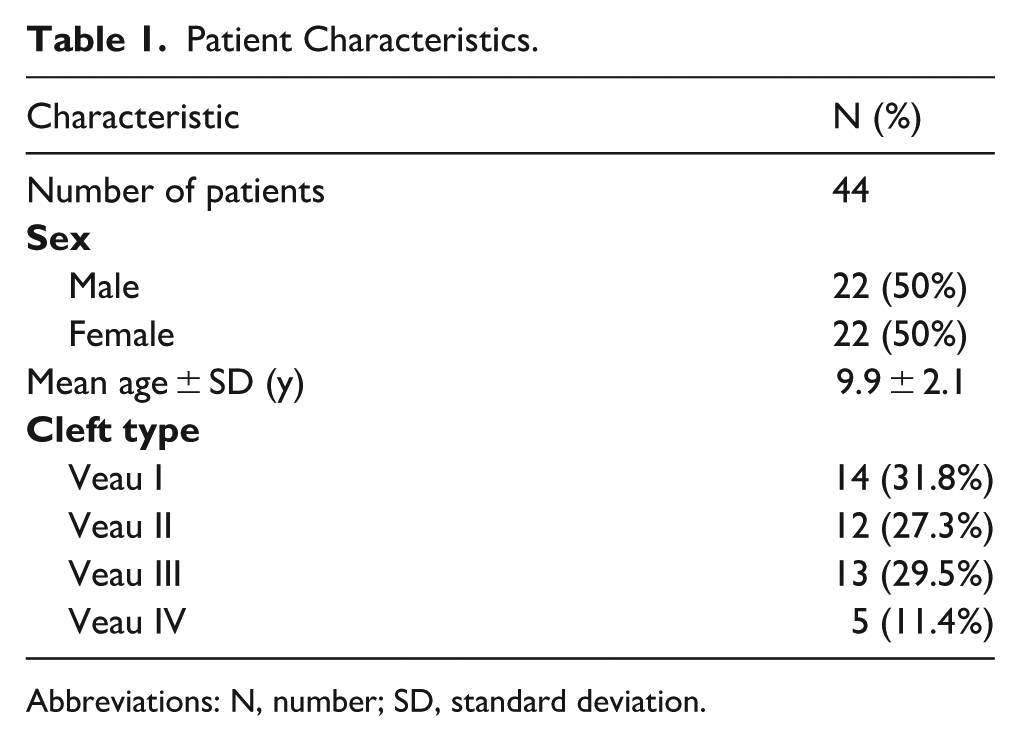
Abbreviations: N, number; SD, standard deviation.
Primary Outcome
The mean difference in hearing loss between pre- and post-tympanostomy was 14.13 dB (95% CI: 11.11-17.15; P < .001), indicating a statistically significant improvement in hearing following tympanostomy tube placement, with 86.7% of patients showing improvement. No statistically significant difference was observed between the left and right ears (mean difference = −0.16, P = .916). The random intercept variance at the child level was 49.07 (SD = 7.00), suggesting substantial interindividual differences in baseline hearing loss. Figure 1 presents boxplots illustrating the distribution of hearing loss before and after the intervention, stratified by ear (left vs right).
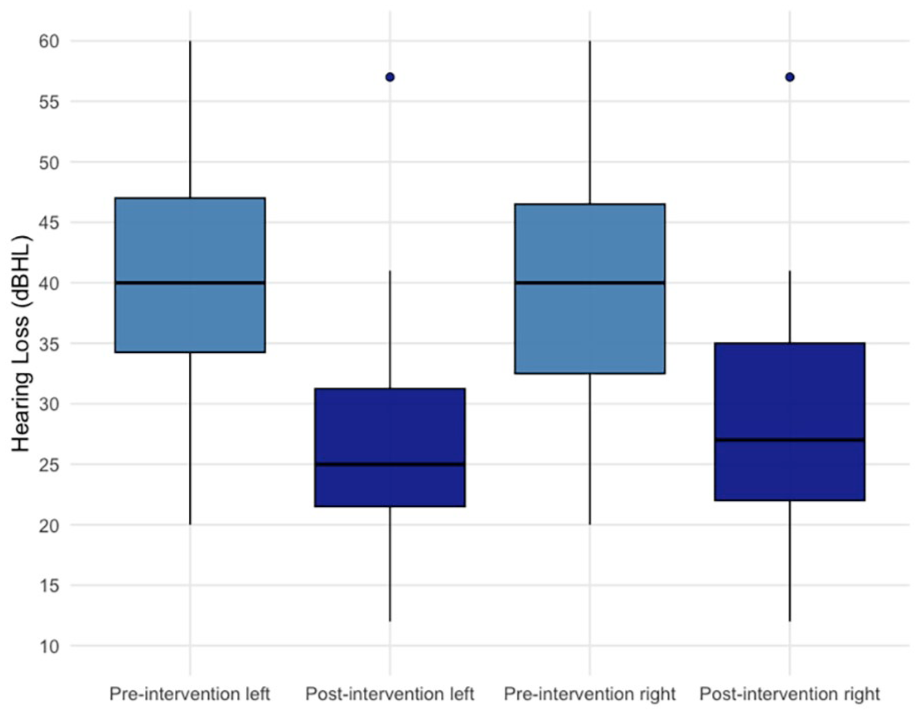
Boxplots of hearing loss before and after tympanostomy tube placement, separated by ear (left and right). Postoperative measurements correspond to the first audiogram performed after the intervention. An outlier is present and reflects a patient with persistently elevated hearing thresholds post-intervention, likely due to ongoing or recurrent OME. dB HL, decibel hearing level; OME, otitis media with effusion.
Secondary Outcomes
Among the 44 patients included in the study, 39 (88.6%) had a documented history of OME. Conductive hearing loss was observed in 17 patients (38.6%) of the total cohort (n = 44), involving 14 left ears and 14 right ears. Among the affected ears, 22 showed mild and 5 moderate conductive hearing loss. Sensorineural hearing loss was less common, occurring bilaterally in 2 patients (4.5%).
In total, 2 ears had moderate, 1 ear had moderately severe, and 1 ear had profound sensorineural hearing loss. One of these patients (2.3%) also had a conductive component, indicating mixed hearing loss.
The median number of tympanostomy tubes placed was 1 (first and third quartile: 1 and 3) for both the left and right ears. Cumulatively, a total of 90 tubes were placed in the left ear and 84 in the right ear.
Eardrum anomalies were observed in a subset of patients. Tympanic membrane perforations were reported in 2 patients, and atelectasis was noted in 4 patients, including 1 with concurrent bony ear canal stenosis at the level of the eardrum. In addition, bilateral myringosclerosis was observed in 1 patient, and 1 patient exhibited atelectasis with an atrophic eardrum.
A detailed overview of these secondary outcome measures is provided in Table 2.
Secondary Outcomes.
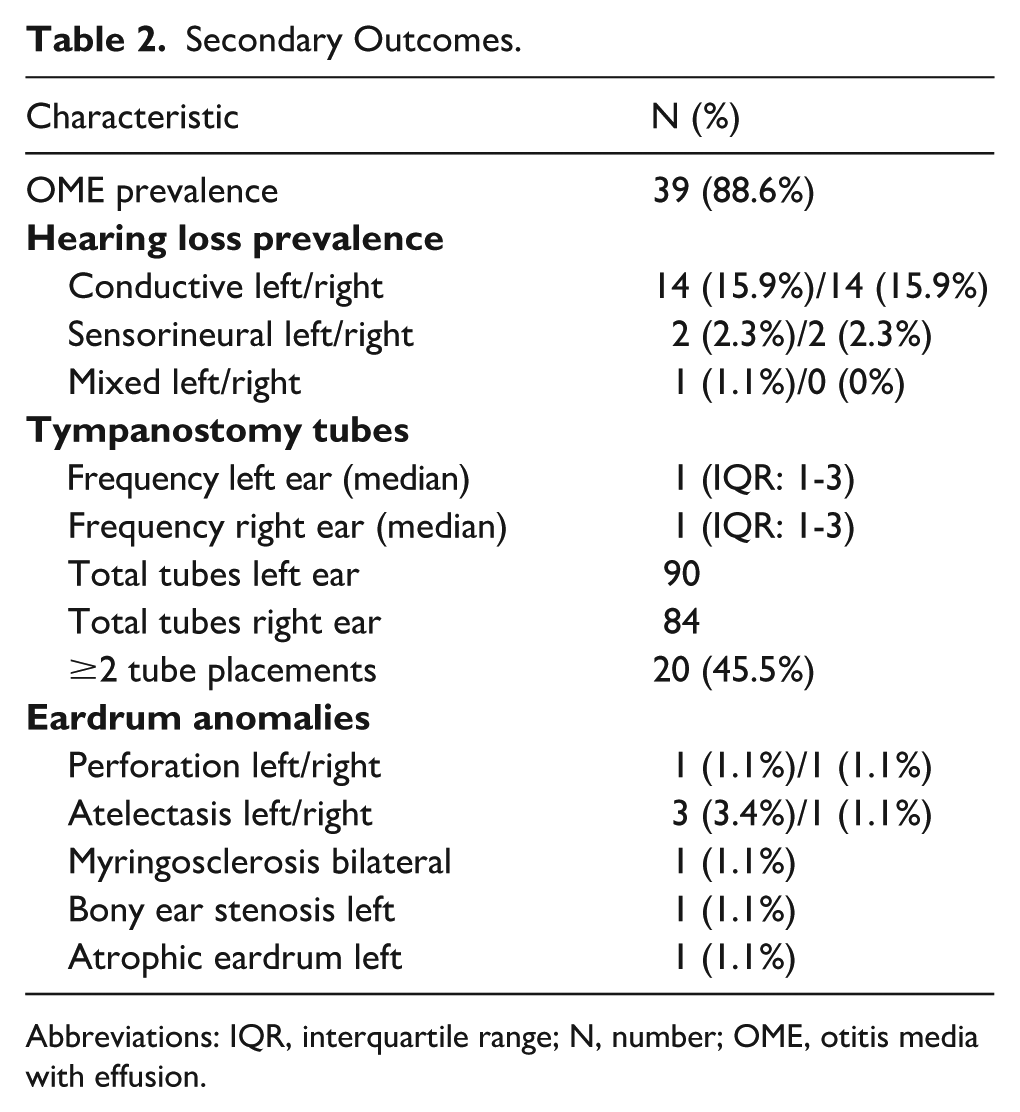
Abbreviations: IQR, interquartile range; N, number; OME, otitis media with effusion.
Discussion
This study demonstrates both a statistically and clinically significant improvement in hearing outcomes following tympanostomy tube placement in children with cleft palate or cleft lip and palate. The primary outcome measure showed a mean hearing improvement of ~14 dB following intervention. Given that audiological guidelines define a hearing improvement >10 dB as clinically relevant, the observed effect size further supports the clinical relevance of tympanostomy tube placement in this population and aligns with previously documented efficacy in alleviating OME and mitigating conductive hearing loss.3,5,14 Of all included patients, 86.7% demonstrated a measurable improvement in hearing thresholds following tympanostomy tube placement. In the remaining cases, no hearing improvement was observed. Only one of these patients had documented conductive hearing loss at the time of surgery; the others had normal hearing thresholds preoperatively. A possible explanation for the lack of improvement is the relatively long interval between tube placement and the postoperative audiogram in some cases, potentially diminishing the measurable effect. In addition, tympanostomy tubes were occasionally placed prophylactically based on a history of recurrent OME or acute otitis media, even in the absence of active middle ear effusion or hearing loss at the time of surgery. In such cases, further audiological improvement was not expected. No statistically significant difference was observed between the left and right ears, indicating that the intervention was equally effective bilaterally. Moreover, the random intercept variance at the child level was substantial, suggesting considerable interindividual differences in baseline hearing thresholds. This variability is not unexpected in a cleft-affected population and likely reflects the heterogeneous nature of middle ear pathology and cleft-related anatomical differences, potentially influenced by factors such as cleft type, age at the time of surgery, and the frequency or chronicity of OME episodes. 2
Consistent with previous reports,2,3,5 the high prevalence of OME in this cohort—88.6%—further highlights the central role of Eustachian tube dysfunction in cleft palate-affected children. In addition, eardrum anomalies such as atelectasis, tympanic membrane perforations, and myringosclerosis were identified in a subset of patients with repeated tube placements. As mentioned in the introduction, tympanostomy tube placement may be associated with adverse effects, such as otorrhea and persistent tympanic membrane perforation. These complications can be particularly problematic in children with sensorineural hearing loss, as they may hinder both the wearing and effective use of hearing aids. 4 Therefore, rather than placing tubes in all children with cleft palate, we advocate for a more selective approach. Tympanostomy tube placement should be considered in children who have active OME at the time of surgery, a documented history of recurrent OME, or several episodes of acute otitis media. In children without such indications, the potential risks may outweigh the immediate benefits, and a watchful waiting strategy may be more appropriate, allowing for tube placement at a later stage if clinically indicated. Notably, a longitudinal study in a cleft palate population has shown that OME may resolve spontaneously in a significant proportion of cases. Alper et al reported that by the age of 6, ~60% of children no longer exhibited persistent OME, suggesting that spontaneous resolution is not uncommon even in this high-risk group. 12 Similar findings were reported in a recent Portuguese study, in which 56.5% of children with cleft lip and palate did not require tympanostomy tube placement during follow-up after surgical repair. The authors emphasized the importance of pre- and postoperative otolaryngological evaluation to guide individualized decision-making and avoid unnecessary procedures. 15 These results align with our recommendation for selective tube placement, rather than routine prophylactic insertion. At the same time, other studies have demonstrated that tympanostomy tubes can be highly effective when OME and hearing loss are present. Tellioğlu et al found that children with mild to moderate preoperative conductive hearing loss were significantly more likely to benefit from tube placement, emphasizing the importance of a detailed audiological assessment prior to intervention. 16 Similarly, Shaffer et al observed a substantial reduction in the prevalence of hearing loss after tube insertion (from 84.0% to 26.5%), providing further support for intervention in selected cases. 17
These findings are supported by several prior studies. For instance, Azman et al reported a high prevalence of middle ear effusion (88.8%) in cleft palate-affected children, with most cases showing conductive hearing loss and improved outcomes following tube placement. 18 Similarly, Kwan et al noted that 87% of non-syndromic cleft palate patients required tube placement due to OME, with notable audiometric improvements post-intervention. 19 Shaffer et al further demonstrated a marked reduction (84.0% prior vs 26.5% afterward) in hearing loss prevalence after tympanostomy tube placement. 20
While hearing outcomes can be influenced by factors such as recurrent infections, chronic OME, and anatomical variations, the overall trend toward improvement after intervention appeared consistent. Johnston et al reported that although children with frequent insertions had more tympanic membrane abnormalities over time, overall hearing thresholds remained within normal limits at later follow-up. 21 In line with this, Szabo et al found that whereas some children exhibited tympanic membrane changes such as myringosclerosis or residual perforations, these did not invariably lead to further hearing deterioration. 22
Further supporting these findings, Kobayashi et al have noted that tympanostomy tubes are beneficial when OME is present, leading to a demonstrable hearing improvement. 23 Although studies differ in terms of re-insertion rates and the criteria used to define “clinically significant” hearing improvement, several reports suggest that tympanostomy tubes can contribute to improved auditory outcomes in children with cleft palate, particularly in those with OME.
This study has limitations that should be addressed. First, the relatively small sample size limits the precision of our findings. Second, the lack of a nonintervention control group precludes direct comparison with the natural course of OME. Third, the variation in follow-up intervals among patients, which ranged from 1 to 12 months, could affect the comparability of audiometric outcomes and may partly explain why no hearing improvement was observed in 13.3% of the patients following tympanostomy tube placement. Furthermore, while the exclusion of syndromic patients ensures a more homogeneous study population, it limits the applicability of the findings to this subgroup. In addition, potential associations between cleft type and hearing outcomes, as well as between the number of OME recurrences and hearing outcomes, were not systematically analyzed due to inconsistent documentation and the limited sample size. Lastly, different surgical techniques were employed based on cleft severity and surgeon preference, with the majority of patients undergoing either a Furlow double opposing Z-plasty or a 2-flap palatoplasty. Due to the limited number of patients within each surgical subgroup, the potential influence of surgical technique on hearing outcomes could not be reliably analyzed and is therefore acknowledged as a limitation of the study. Future research should address these limitations by incorporating larger, prospective cohorts with standardized follow-up intervals and inclusion of diverse patient populations.
In summary, this study highlights 3 main points:
OME is highly prevalent in children with a cleft palate, commonly causing conductive hearing loss;
tympanostomy tube placement is effective at reducing this hearing loss;
and while tympanic membrane anomalies do occur—particularly in cases with repeated tube placements—they are often not associated with severe or progressive hearing decline.
Conclusion
Our findings demonstrate the efficacy of tympanostomy tube placement in improving hearing outcomes in cleft palate-affected children, reinforcing the evidence from prior studies. Although OME and its sequelae remain common, when OME is present at the time of cleft surgery or there is a documented history of recurrent OME or acute otitis media, concurrent tube placement should be considered to alleviate conductive hearing loss.
Footnotes
Ethical Considerations
This retrospective cohort study was approved by the Institutional Review Board of the Maastricht University Medical Centre, The Netherlands (METC 2024-0524).
Consent to Participate
Informed consent was waived due to the retrospective nature of the study and the use of anonymized data.
Author Contributions
Nina Wijnants: conceptualization, methodology, formal analysis, investigation, data curation, writing—original draft, visualization, project administration. Josine C.C. Widdershoven: conceptualization, writing—review and editing. Stan C.J. van Boxel: writing—review and editing. Sander M.J. van Kuijk: methodology, formal analysis, writing—Review and editing. Jan W. Brunings: writing—review and editing. Darren I. Booi: writing—review and editing. René R.W.J. van der Hulst: writing—review and editing. Rutger M. Schols: conceptualization, writing—review and editing, supervision.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets generated and/or analyzed during the current study are not publicly available due to privacy restrictions but are available from the corresponding author on reasonable request.
