Abstract
Malignant peripheral nerve sheath tumor (MPNST) is a rare tumor that can develop on the lining of nerves and within the network of nerve fibers in different organs, and it is commonly found in the head and neck, limbs, and trunk. These tumors can occur in patients of any age. They most commonly occur in adults aged 20 to 50 years; however, fewer cases of this tumor in children have been reported. To date, no neonatal case of MPNST in the nasal cavity has been reported. Here, we report the case of a 4-day-old female newborn who presented with a nasal mass that re-enlarged after surgery and was diagnosed as MPNST of the nasal cavity on the basis of pathological results. This is the first report of MPNST in the nasal cavity of a neonate. Differential diagnosis and treatment of nasal masses have been proposed in the related literature.
Introduction
Malignant peripheral nerve sheath tumor (MPNST) is a very rare disease, especially in the pediatric population. The total incidence of MPNST is approximately 1/100 000 in the overall population. 1 Malignant peripheral nerve sheath tumor develops on the lining of nerves and within the network of nerve fibers in different organs, and it commonly occurs in the head and neck, limbs, and trunk. 2 The majority of the nasal masses are diagnosed as nasal dermoid cyst, hemangioma, nasal glioma, and encephalocele. 3 We searched the PubMed database using the search term “(malignant peripheral nerve sheath tumor/MPNST) AND (newborn/neonatal) AND (nose/nasal cavity)” and found that no neonatal case of MPNST in the nasal cavity has been reported to date. Here, we report for the first time the case of a female newborn with MPNST in the nasal cavity.
Case Presentation
A female newborn weighing 2500 g was spontaneously delivered by a 27-year-old healthy mother without any family history of nasal mass. On the fourth day after birth, the baby had a slight nasal congestion and running nose. A prominent mass in the left nasal cavity appeared on the seventh day after birth. The symptoms of nasal congestion and running nose gradually deteriorated in the next few days, and the mass of the left nasal cavity gradually enlarged. Bloody pus started to exude on the 11th day after birth. The baby had moderate fever on the 14th day after birth. Her 1-year-old sister did not show similar symptoms. Physical examination of the neonate revealed the following: severe nasal congestion, mild mouth breathing, nose swelling, a prominent red mass in the left nasal cavity, and bloody purulent discharge from the left nasal cavity (Figure 1).
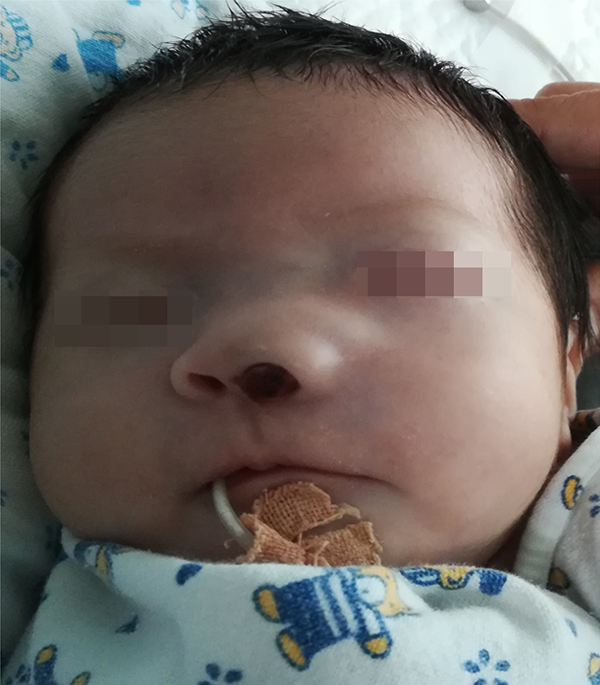
Gross observation of the left nasal mass.
Computed tomography (CT) scan showed an irregular lesion of 2.2 cm × 2.1 cm × 3.0 cm in the left nasal cavity, with an unclear margin and involving the left maxillary sinus, ethmoid sinus, and sphenoid sinus. The CT-determined density of the mass was approximately 19 HU. Multiple plaque calcifications were observed in the center of the lesion, without any enhancement. Adjacent bones, the right sinus, and the nasal cavity were compressed. The left ethmoid sinus, the medial wall of the orbit, and the nasal septum were destroyed. The hard palate on the left side had become thinner, and the left turbinate was destroyed. Magnetic resonance imaging showed an irregular mass occupying the left nasal cavity, with a clear boundary. T1 signal was hypointense and T2 signal was heterogeneous hyperintense. The enhanced scan showed obvious heterogeneous enhancement. The lesion showed a flaky nonenhanced area and mainly involved the left maxillary sinus and the left turbinate. The nasal septum was biased to the right, and the left eye was slightly stressed. No abnormal signals were noted in the brain (Figure 2). On the basis of clinical and radiographic findings, the patient underwent biopsy and the mass was resected. However, the tumor recurred after operation and increased rapidly in size, compressing the left eyeball. Histopathological analyses of the nasal mass showed that the surface of the tumor was covered with nasopharyngeal epithelium, subepithelial tissue, and submucous stroma. The tumor grew aggressively under the mucosa, with local tumor necrosis and abundant blood vessels. Tumor cells were distributed in nests or strands. The tumor cells mainly comprised epithelioid cells, rare spindle cells, and multinucleated giant cells. The size of the tumor cells was relatively consistent, with obvious cell atypia, cytoplasmic pink staining, coarse chromatin, obvious irregular or kidney-shaped nucleoli, and eccentric nuclei. Mucoid degeneration occurred in the interstitium of the tumor (Figure 3). Immunohistochemical staining showed that the tumor cells were positive for cytokeratin (partially), vimentin, S100, SOX10, TLE1, INI1, Ki-67 (60%+), and P53 (15%) but were negative for CD56, MyoD1, CD34, Syn, CD99, WT-1, NSE, CD138, CD38, CD68, LCA, Melan A, HMB45, p40, EMA, GFAP, D2-40, brachyury, and myogenin (Figure 4). The patient was tested with the dual-color EWSR1 probe. The EWSR1 translocation negative criterion was that >90% of nuclei should exhibit fusion signals (2 pairs of red and green signals are very close or overlapping to form yellow signal). The EWSR1 translocation positive criterion was that >80% of nuclei show translocation signal (one pair of red and green signals are very close or overlap to form a yellow signal, while the other pair of signals show a large separation at 1 signal). There were no separate signals of the EWS gene in this patient. We tested 180 cancer-associated NDA genes on tissue wax blocks and detected an FGFR2 exon10 c.1361C>T p.T454 M mutation in the patient. Tumor mutation burden (TMB) of the tumor was 3.6 mutations/Mb. The tumor exhibited microsatellite stability (MSS). The diagnosis of MPNST of the nasal cavity was confirmed by pathological analysis. Finally, the parents decided to abandon the treatment, and the newborn died.
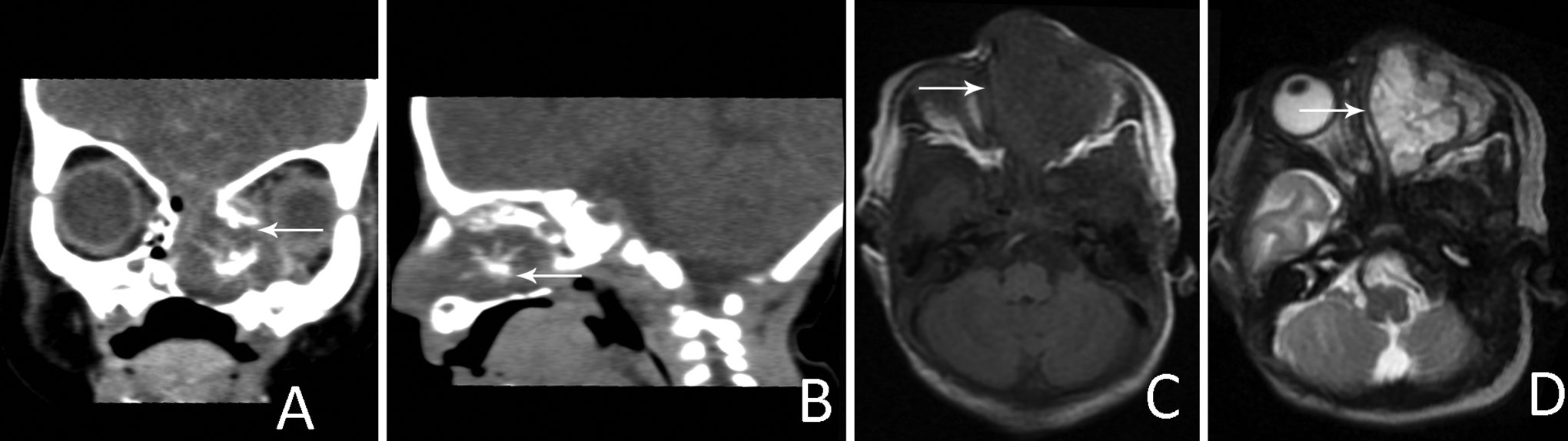
Imaging findings of the mass. A and B, CT scan images of the tumor; (C and D) T1-weighted and T2-weighted magnetic resonance images of the nasal mass. CT indicates computed tomography.
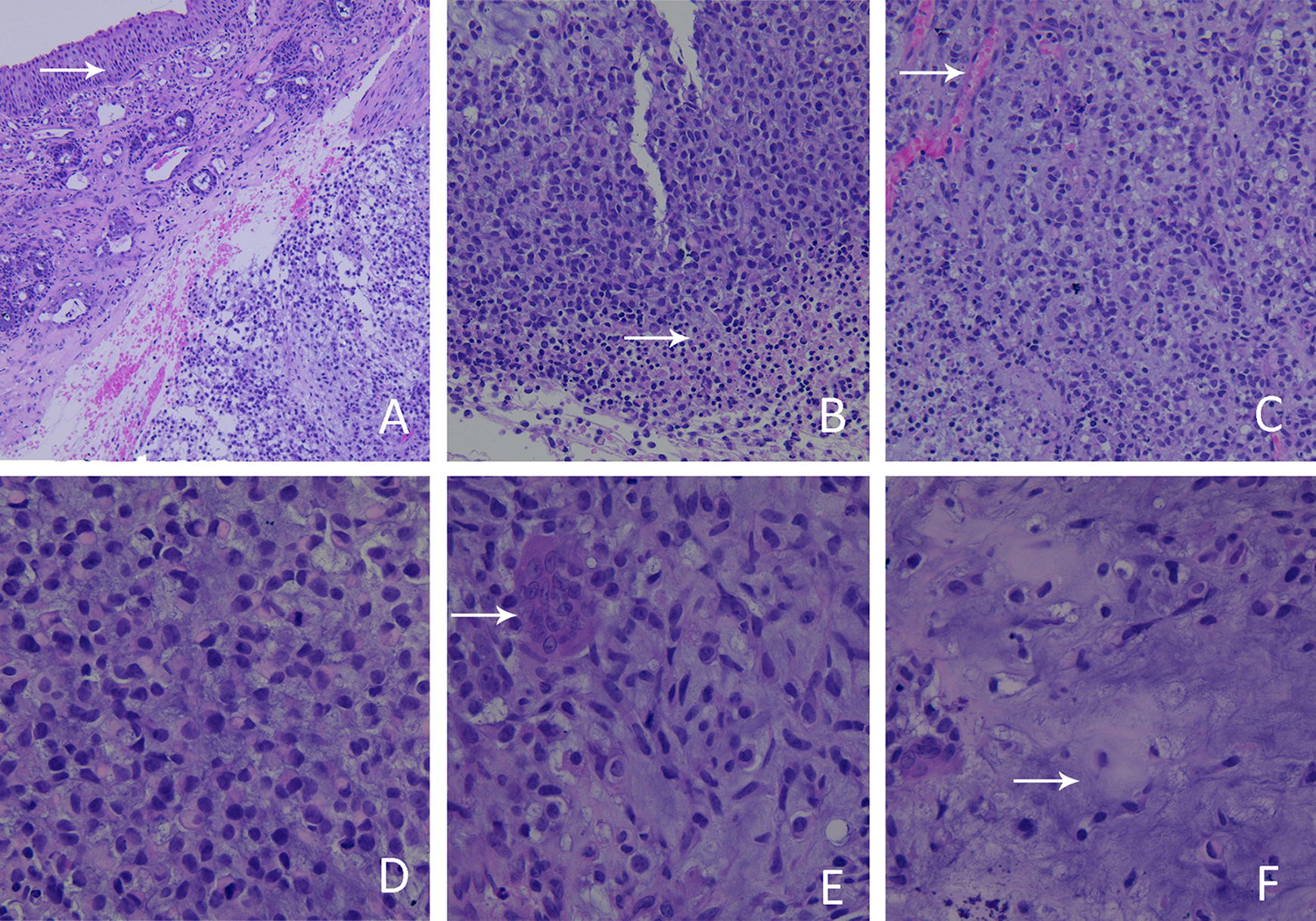
Histopathological analyses of the nasal mass (H&E staining). A, The surface of the tumor was covered with nasopharyngeal epithelium, subepithelial tissue, and submucous stroma (arrow). The tumor grew aggressively under the mucosa (×100). B, Local tumor necrosis (arrow, ×200). C, Abundant blood vessels were observed within the tumor (arrow, ×200). D and E, Tumor cells were distributed in nests or strands. Tumor cells mainly comprised epithelioid cells (D, ×400), rare spindle cells (E, ×400), and multinucleated giant cells (E, arrow). The size of the tumor cells was relatively consistent, with obvious cell atypia, cytoplasmic pink staining, coarse chromatin, obvious irregular or kidney-shaped nucleoli, and eccentric nuclei. F, Mucoid degeneration occurred in the interstitium of the tumor (arrow, ×400).
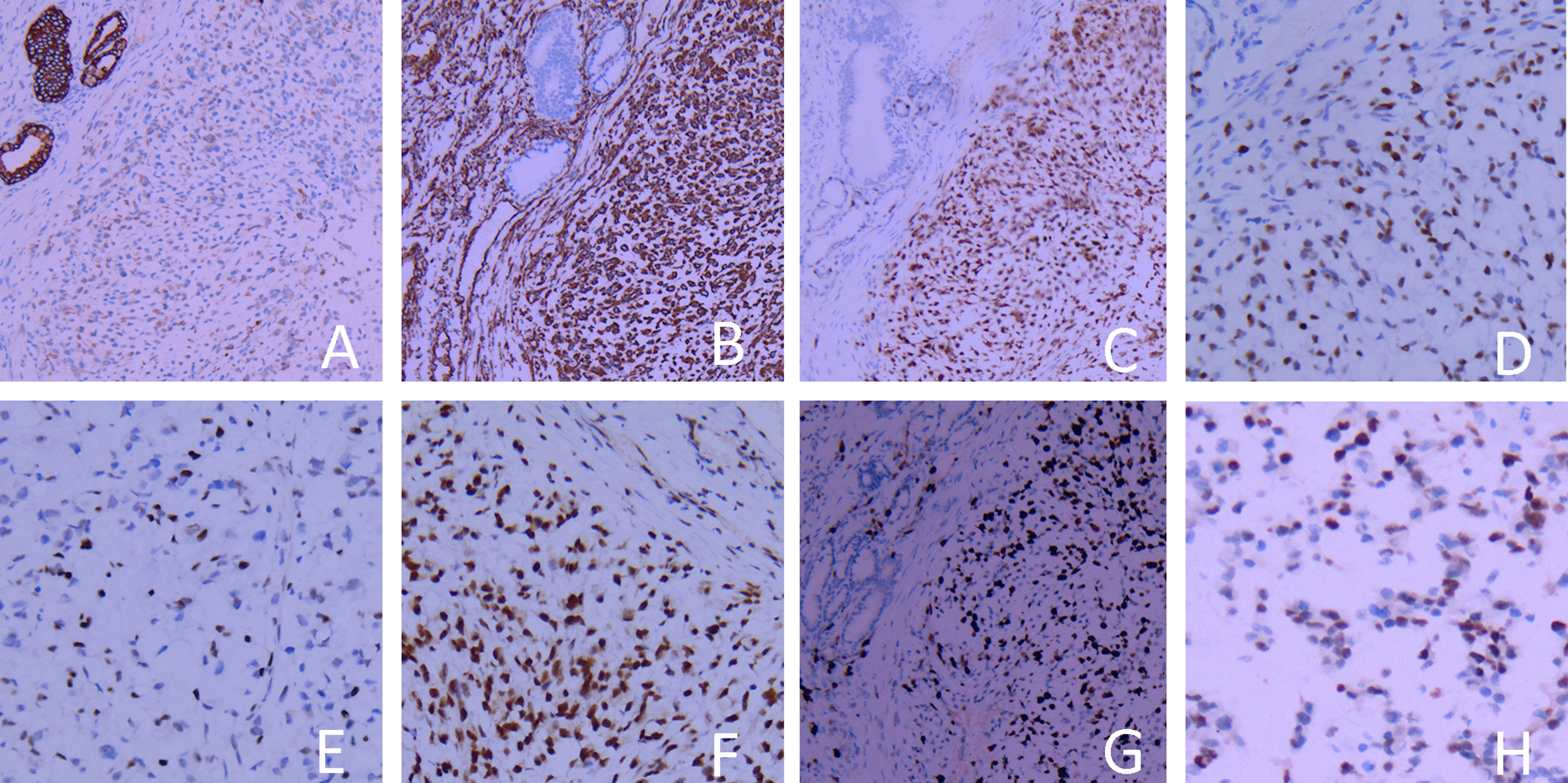
Immunohistochemical analyses of the nasal mass. A, Expression of cytokeratin in the nasal mass, ×200; (B) expression of vimentin in the nasal mass, ×200; (C) expression of S-100 in the nasal mass, ×200; (D) expression of SOX10 in the nasal mass, ×200; (E) expression of TLE1 in the nasal mass, ×200; (F) expression of INI1 in the nasal mass, ×200; (G) expression of Ki-67 in the nasal mass, ×200; (H) expression of P53 in the nasal mass, ×200.
Discussion
In the pediatric population, the incidence of MPNST is 0.56 per million person-years and is higher among postpubertal children aged 10 to 19 years. 1 Malignant peripheral nerve sheath tumor most commonly occurs on the extremities, especially at the proximal end, followed by the trunk and head and neck. 2 Malignant peripheral nerve sheath tumor usually manifests as a progressive enlargement of the tumor mass. There is no pain when the mass is small or at the early onset of tumor development. If the mass continues to grow, nerve compression symptoms appear. When the tumor invades the surrounding tissue, the patient may experience pain, which often involves nerves. The related area may also show neurological dysfunctions such as numbness and decreased muscle strength. To date, there are no reports of nasal MPNST in neonates. The 4 most common types of congenital diseases occurring as neonatal nasal masses are nasal dermoid cyst, hemangioma, nasal glioma, and encephalocele. 3 Table 1 shows the characteristics for differentiating MPNST from these diseases.
Differential Diagnosis of Nasal Masses.
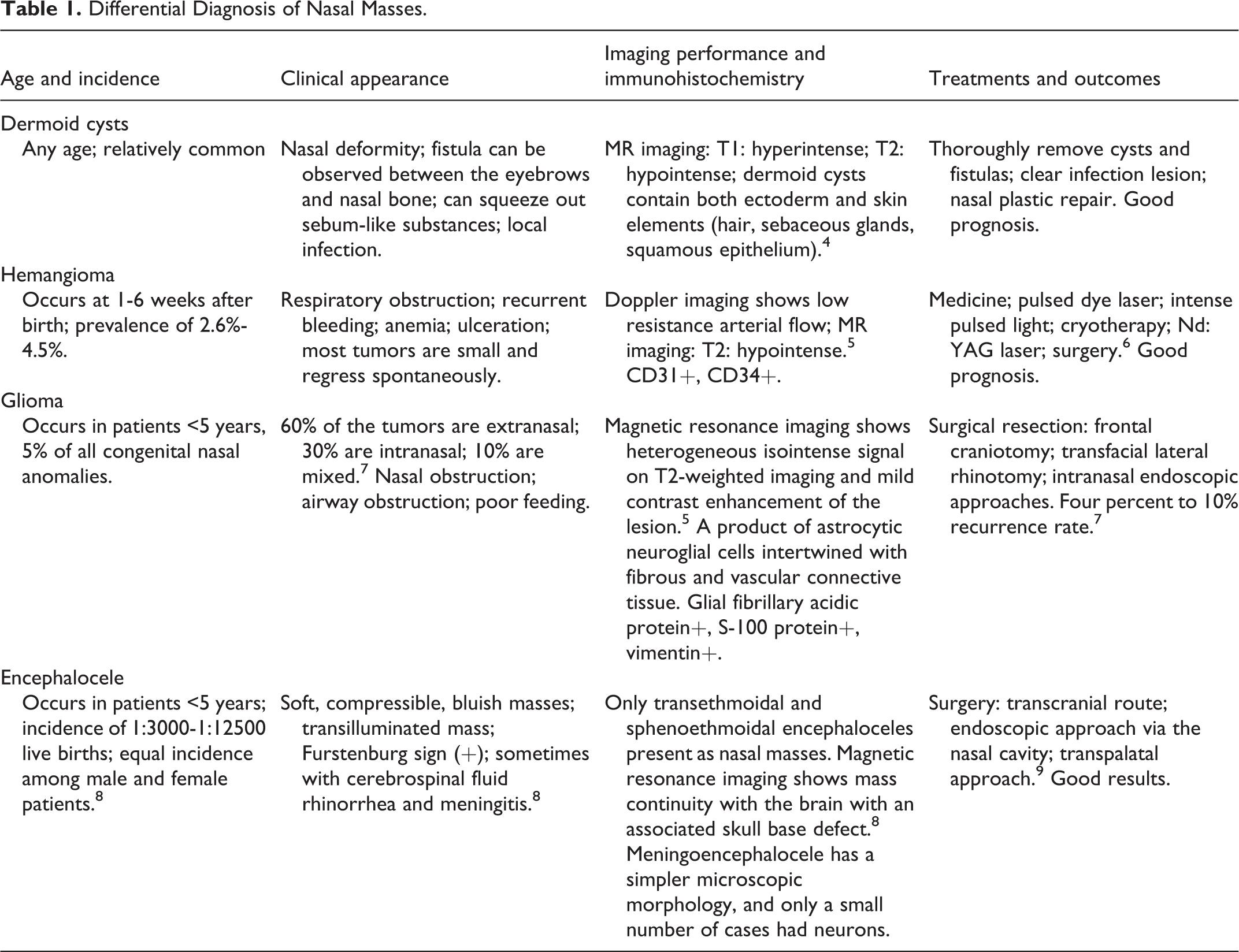
By performing MR examination, we excluded intracranial-related diseases such as meningocele. The diagnosis of MPNST is based on histopathology and immunohistochemistry. The tumor cells of MPNST are morphologically variable and may exhibit short fascicles, sweeping fascicles, whorls, hemangiopericytoma-like vascular pattern, and herringbone pattern. Malignant peripheral nerve sheath tumor can have hypocellular and hypercellular areas. Myxoid change, geographic necrosis, and subendothelial herniation by the tumor cells was noted in many cases. However, very few cases show heterogeneous elements of chondroid differentiation. Most MPNSTs are positive for S100, p16, p27, and p53; may be focally positive for SYN and vimentin; and are mostly negative for GFAP, neurofibromatosis (NF), and EMA. 10-11 A previous study showed that the sensitivity of SOX10 for the detection of MPNST was 27%. Hence, SOX10 combined with S100 expression can provide strong support for the diagnosis of MPNST. 12 Another study showed that approximately 30% of MPNST cases show positivity for TLE1. 13 In our present case, the tumor was positive for SOX10, vimentin, S100, TLE1, Ki-67, and P53 but was negative for EMA and GFAP. On the basis of the clinical, microscopical, immunohistochemical, and FISH data, this patient was diagnosed to have MPNST.
The histological morphology of MPNST is diverse, and it very rarely occurs in the nasal cavity of a neonate; hence, the diagnosis is difficult, and it is necessary to distinguish it from other tumors by combining clinical findings with immunohistochemical staining. The most common cancer type was lymphomas in children with head and neck tumors, followed by nasopharyngeal carcinoma and rhabdomyosarcoma (RMS). 14 Hence, the following diseases need to be considered for differential diagnosis.
(1) Lymphoma: It can occur in the nasal cavity and sinuses. The doctor mostly checks for nasal congestion, nasal pain, nasal spontaneous bleeding, and nasal and facial swelling, with fever and fatigue. Nasopharyngoscopy revealed diffuse swelling of the nasal mucosa or a new excrescence of soft tissue. Typical R-S cells or LCA+ is helpful for diagnosing lymphoma, but myoepithelial-derived expression is helpful to eliminate lymphoma. (2) Nasopharyngeal carcinoma: It is prevalent in Guangdong province in China. The prevalence age of onset is 10 to 19 years and 50 to 59 years. Clinical manifestations often include cervical lymphadenopathy, headache, nasal congestion, and nose bleeding. There is a certain correlation of nasopharyngeal carcinoma with Ebola virus infection. The expression of p63 and p40 proteins is related to the differentiation of nasopharyngeal carcinoma. The higher the expression of p63 and p40, the lower is the differentiation of nasopharyngeal carcinoma. Negative expression of p63 and p40 is helpful for the exclusion of nasopharyngeal carcinoma. (3) Rhabdomyosarcoma: It is the most common soft tissue sarcoma in children. The most common sites are head and neck, nasal cavity, sinuses, and orbits. Rhabdomyosarcoma lacks specific clinical manifestations. Some cases are asymptomatic, although painful swelling is the most common clinical manifestation in the head and neck region. Microscopically, RMS can be classified into different histologic subtypes. The most common is the embryonal subtype, characterized by undifferentiated, small, round, and hyperchromatic cells with a variable number of strap or tadpole-shaped, eosinophilic rhabdomyoblasts. The Desmin/Myogenin/Myo-D1 immunohistochemical positivity is useful to confirm the diagnosis. 15 (4) Olfactory neuroblastoma: NSE and CgA are often positive. Negative HMB-45 expression is useful to rule out the possibility of malignant melanoma. (5) Ewing sarcoma/primitive neuroectodermal tumor: CD99 is the most sensitive marker in the immunophenotype, and the expression of neural markers such as vimentin and NSE is also common. (6) Synovial sarcoma: TLE1 is an important marker for the diagnosis of synovial sarcoma, but it is not a 100% specific marker. 13 The lack of INl1 expression supports the diagnosis of synovial sarcoma. 16 The expression of the SYT/SSX fusion gene is helpful for the diagnosis of synovial sarcoma. 17 (7) Extraskeletal myxoid chondrosarcoma (EMC): Histomorphologically, the nodules are composed of tumor cells and a mucus-like matrix. The tumor cells are polygonal, spindle-shaped, or oval-shaped; with eosinophilic cytoplasm; arranged in a lace-like or strand-like manner; have small nuclei; stain intensely; and have rare mitotic images, which are characteristics of chondrocytes. The EMC immunophenotype includes diffuse positivity for vimentin and focal positivity for synaptophysin, NSE, and S-100 protein, while chromogranin is expressed in a minority of cases. Epithelial markers such as cytokeratins and EMA are usually negative. (8) Chordoma: Most tumors are located near the midline. The tumors are typically droplet-shaped, with strong positive expression of CK and S-100. Brachyury is considered to have high specificity and sensitivity in chordoma. Negative expression of brachyury is useful to exclude chordoma. (9) Central nervous system germ cell tumors: Podoplanin is a sensitive marker of germinoma and thus may be useful for diagnosis. Negative result for antibody D2-40, which specifically recognizes human podoplanin, is useful to exclude central nervous system germ cell tumors. 18
Malignant peripheral nerve sheath tumor occurs in 3 forms: approximately half of the cases are sporadic cases that originate in the large nerve trunk; 25% to 50% of the cases are malignant transformation of neurofibromatosis type 1 (NF1); and only a very few cases occur after radiotherapy. The newborn in our case had no clinical manifestations of NF1 and no history of radiotherapy; hence, it is considered that the tumor originated from the differentiation of potential stem cells into nerve cells. According to the morphological characteristics of MPNSTs, it can be divided into the following subtypes: conventional MPNSTs; epithelioid MPSNTs; MPSNTs with glands; malignant triton tumor; and plexiform MPNST of infancy and childhood. The common type of MPNST in children is plexiform MPNST of infancy and childhood, which mostly occurs in the limbs. However, in the present case, no plexiform structure was found, and the tumor did not occur in the limb. Given that the tumor expressed the epithelial cell marker CK, this case could be considered as epithelioid MPNSTs.
Malignant peripheral nerve sheath tumor is a highly malignant soft tissue sarcoma, which is prone to local recurrence and distant metastasis. Because of its aggressiveness and metastasis, it shows rapid progression and has a poor prognosis. In children, the overall 5-year survival rate was 43% to 59%, while the 5-year progression-free survival rate was 29% to 45%. 1 Neurofibromatosis type 1 is associated with worse survival. 19 In our present case, the Ki-67 labeling index was 60%, which indicated poor prognosis. This finding is consistent with the progressive enlargement of the tumor after surgery. High TMB and microsatellite instability usually predict treatment benefit. Although no radiotherapy and chemotherapy were performed in our case, low TMB and MSS indicate that the treatment may be poor. As MPNST is insensitive to chemotherapy and radiotherapy, surgery remains the main treatment option, and the role of adjuvant therapy is unclear. Studies have also found that responses to primary chemotherapeutic agents suggest that it may be effective in patients with tumor considered unresectable at diagnosis. 2 Ifosfamide combined with doxorubicin was found to be more effective than doxorubicin-based therapy. 20 Therefore, we can use radiotherapy and chemotherapy for children who cannot be operated or in whom the tumor cannot be completely removed by surgery. In the future, it may be possible to find effective alternative treatments by defining the characteristics of the disease at the genetic and molecular levels. We hope to develop molecular targets specifically for tumor cells. Studies have shown that MPNST has a higher expression of platelet-derived growth factor receptor α (PDGFRa) than benign schwannomas. 21 The PDGF system may contribute to the malignant progression of schwannomas. In our present case, a mutation of the PDGFRa gene was found; this finding suggests that it is related to the occurrence of MPNST and that this gene is a possible site for targeted molecular therapy.
Conclusions
The present case report is the first report of neonatal MPNST in the nasal cavity. Neonatal nasal masses are often benign, but the nasal mass in the present case was an MPNST. This case report will help to broaden the diagnosis and differential diagnosis of neonatal nasal masses.
Footnotes
Authors’ Note
All data generated or analyzed during this study are included in this article. Written informed consent was obtained from the legal guardian for publication of this case report and any accompanying images. Name of the ethics committee: Guangdong Women and Children Hospital Ethics committee.
Acknowledgments
The authors are grateful to otolaryngologists and radiologists for their efforts in treatment and for their assistance in writing the paper.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work was funded by the Guandong Natural Science Foundation (Nos. 2018A030313605 and 2018A030310598), the Guangzhou Science and Technology Project (No. 201804010090), and the National Natural Science Foundation of China (Nos. 81741083 and 81801492).
