Abstract
Objectives:
Ear reconstruction is a challenging surgery for the complicated conditions in patients with microtia. The tissue expansion techniques were necessary and relatively safe for patients with insufficient soft tissue. However, complications such as necrosis of expanded flap and exposure of tissue expander limited the popularization of this method. This study described the use of modified Brent method to handle the exposure of the postauricular tissue expander.
Methods:
From January 2013 to December 2019, 27 ear reconstruction patients with trauma or necrosis on an expanded skin flap and subsequent exposure of tissue expander were treated with modified Brent method, which consisted of 3 stages: removal of the expander, tension-free closure of wound, and framework fabrication; elevation of reconstructed ear; lobule rotation; and minor modification.
Results:
Fifty-six percent of exposures occurred in the lower pole of the tissue expander. Exposure usually occurred 54.5 days after implantation. The majority of reconstructed ears had a satisfactory appearance and showed relatively stable outcomes. Only one case of cartilage exposure required revision surgery and was repaired by the temporoparietal fascia.
Conclusion:
With reasonable distribution of expanded flap, prolonged interval, and sutures under tension-free conditions, complications like the occurrence of trauma or necrosis-induced exposure of tissue expander can be repaired efficiently by a staging modified Brent method.
Keywords
Introduction
Microtia is an uncommon congenital deformity that ranges in severity from maldevelopment of the auricle and/or middle ear to a total absence of the ear. Currently, ear reconstruction with costal cartilage remains the mainstream method worldwide. 1 On the basis of Tanzer modern ear reconstruction method, Brent improved this technique to a 4-stage method. 2 -4 Nagata utilized the incision of lobule rotation and tragus to implant the ear framework and reduced the stages into 2, which is the most popular method for ear reconstruction currently. 5 -7
Neumann was the first to apply a tissue expander in postauricular flap expansion to overcome skin inadequacy. 8 Park continued to develop this method and described an expanded 2-flap method. 9 Pan, Jiang, and Zhang popularized the expanded method in China and obtained satisfactory results in thousands of patients. 10 -12 Considering that the postauricular skin of hemifacial microsomia (HFM) is often tight and taut, resulting in insufficient soft tissue to cover the costal cartilage framework, Qian et al explored the practice of expanders in HFM patients. 13 Furthermore, a single expanded flap method was derived to avoid free skin grafting, as reported by Xing et al in 2018. 14
With the increase in the number of applications, complications have increased. Complications such as hematoma, infection, and leakage can be managed by reoperation, replacement, or removal of the expander. 12,15 However, the handling of complications like trauma and necrosis caused by poor blood supply are more troublesome. The most prominent question is whether to continue the expansion of the postauricular flap. Skin flap necrosis eventually leads to expander exposure. The contractibility of the expanded flap increases with expander exposure and peri-expander infection and creates a force larger than that of wound contraction, causing enlargement of the wound. 16 Patients and surgeons would face great risks of failure if expansion continues. The removal of the expander is the last thing that patients and surgeons would consider.
In order to close the wound and remove the necrotic area of the skin flap without tension, we applied the modified Brent method to reduce extensive tension of the expanded skin flap. In the first stage, after removal of the tissue expander, a fabricated base framework of the auricular was implanted in the skin envelope, and the wound or necrosis was managed by debridement and tension-free closure. Ear elevation was performed at least 6 months later in the second stage. The third stage mainly consisted of lobule transposition, tragus construction, and trimming of the remnant ear. Brent technique consists of 4 stages: implantation of ear framework, transposition of the lobule, construction of the tragus, and construction of the cranio-auricular sulcus. 3,4 The third stage of our strategy corresponds to the second and third stages of Brent technique.
We applied this treatment strategy in 27 patients with expanded flap necrosis or exposure from 2013 to 2019. Except for one case of cartilage exposure, which was covered by temporoparietal fascial flap and full-thickness free skin graft, the majority of patients obtained good appearances of reconstructed ear. The results showed that the strategy of applying the modified Brent method to repair undesired expanded flaps is safe and effective.
Patients and Methods
Patients Information
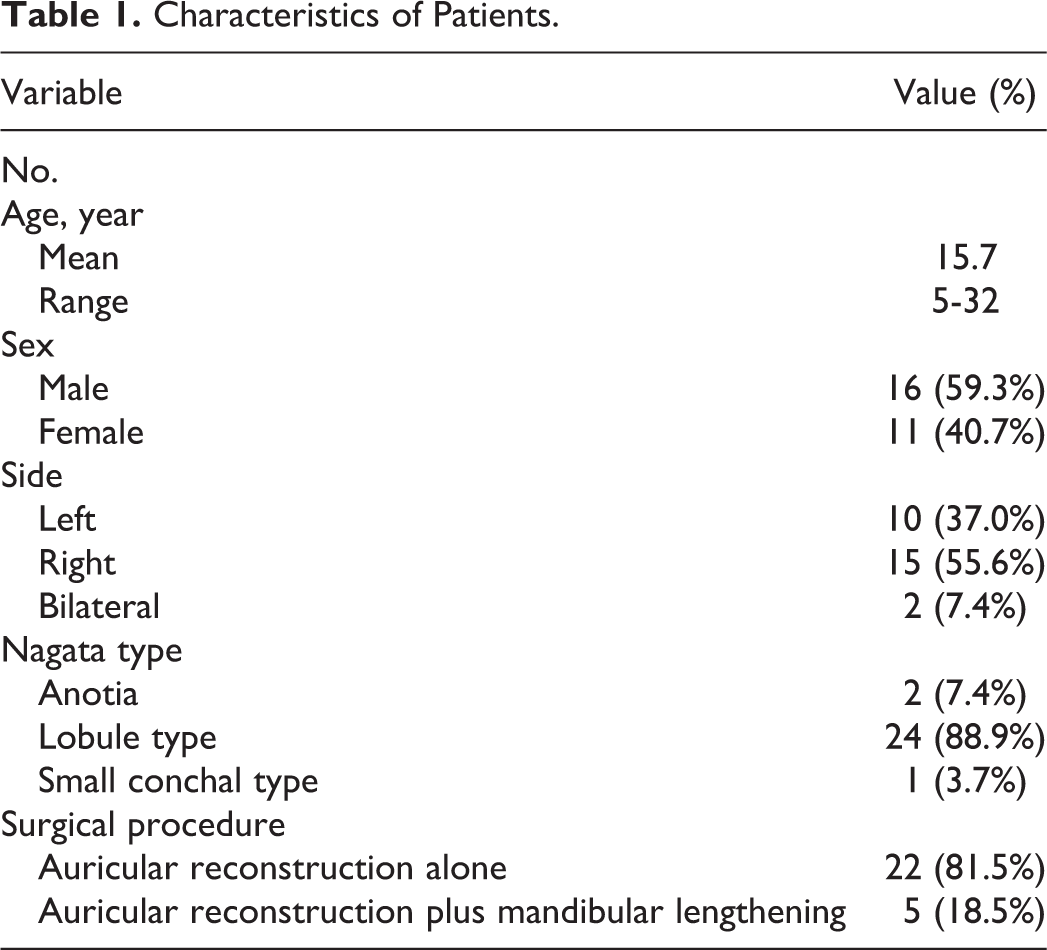
From January 2013 to December 2019, our center performed modified Brent method on 27 patients. (See details in Table 1) Twenty-five (92.6%) patients had HFM. All patients were initially planned to undergo the tissue expander method, but unfortunately, they had to suspend it for various reasons. Flap necrosis caused by thrombosis or ischemia was found in 21 (77.8%) patients; it was caused by trauma in 5 (18.5%) patients, and 1 (3.7%) patient had a canaloplasty scar. Twenty-one (77.8%) patients received tissue expander implantation in our center, while 6 (22.2%) patients got theirs from other hospitals. It needs to be noted that the tissue expander was implanted between subcutaneous layers and fascial layer. In addition, tissue expansion period consists of injection period and inactivity period, when the expansion maintains after the injection of final volume.
Procedures of Operation
First stage: Removal of the expander and framework fabrication
The operation area was sterilized twice with iodine and alcohol before deployment of the drapes. The expander was removed from the initial incision. Then, the skin envelope was irrigated with normal saline, hydrogen peroxide, and diluted iodophor twice. The axial half of the sixth, seventh, and sometimes eighth costal cartilages were harvested from the contralateral or bilateral chest. With reference to an X-ray film template, the sixth and seventh costal cartilages were sculptured and assembled into a base frame (Figure 1). The eighth costal cartilage was carved for the helix. In adults, the seventh cartilage was wide enough and its outer edge could be split and form a helix. Since the pedicle of the expanded flap mainly stemmed from the remnant ear, the residual ear was reserved and the tragus was not fabricated. After assembling all these components, a framework was established and placed in the skin pocket. With negative pressure from drainage, the skin flap was plastered down to the framework. The wound area or necrosis was completely excised and sutured close. By adjusting the distribution of the skin flap, the wound achieved tension-free closure. A crescent-shaped pad was fabricated and implanted in the subcutaneous layer of the cartilage-harvesting incision.
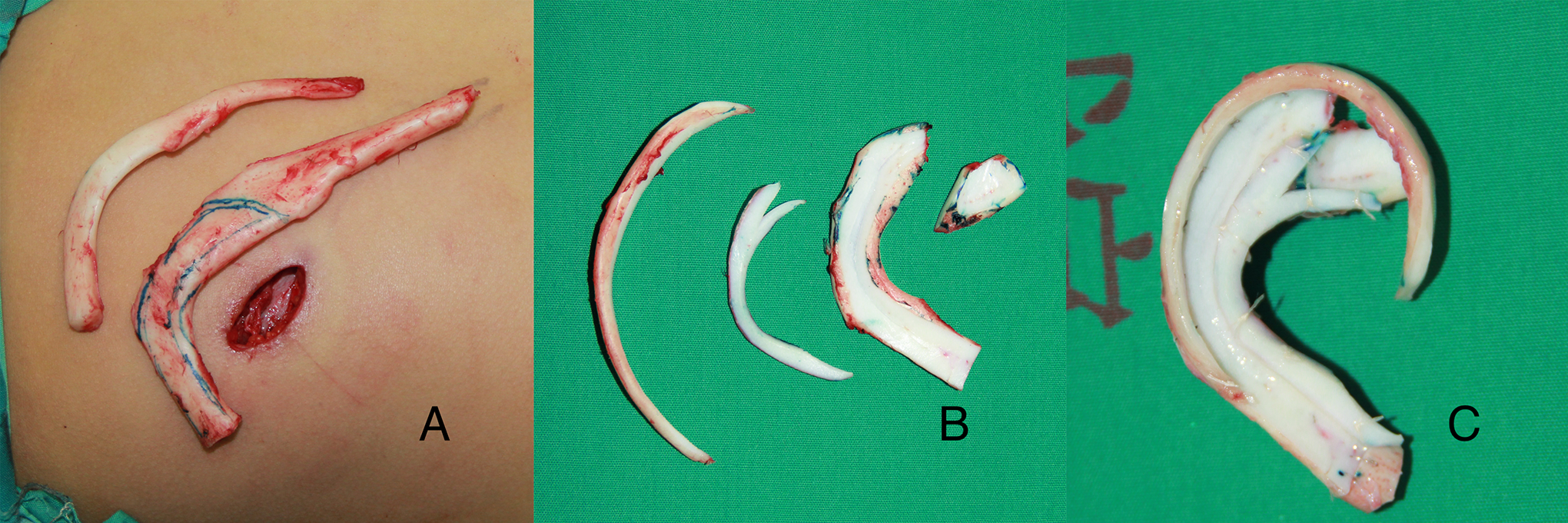
A, A 3-cm-long incision to harvest costal cartilage. B, Costal cartilage was carved into helix, antihelix, and components of base frame. C, Ear framework.
Second stage: elevation of the reconstructed ear
At least 6 months later, an elevation of the reconstructed ear was performed. This stage can be performed under local anesthesia for adult patients and some adolescents. The crescent-shaped pad was removed and trimmed into a suitable shape to enhance its projection and form the wall of concha. A postauricular fascial flap with a pedicle on the remnant ear was raised and turned over to the helix to cover the posterior surface of the cartilage framework. A full-thickness free skin graft was obtained from the cartilage-harvesting incision or groin to cover the fascial surface.
Third stage: the modification of the remnant ear
This stage can be performed under local anesthesia. The modification of the remnant ear was performed at least 6 months after ear elevation. Lobule transposition is usually performed by Z-plasty to cover the distal helix. The skin of remnant ear was separated into 3 flaps: the lobule flap, anterior, and posterior flap (Figure 2). The remnant auricular cartilage was removed and reshaped into the tragus, covered by the anterior and posterior flaps (Figure 3). With the removal of remnant cartilage and redistribution of the mastoid skin flap, the conchal cavity deepened.
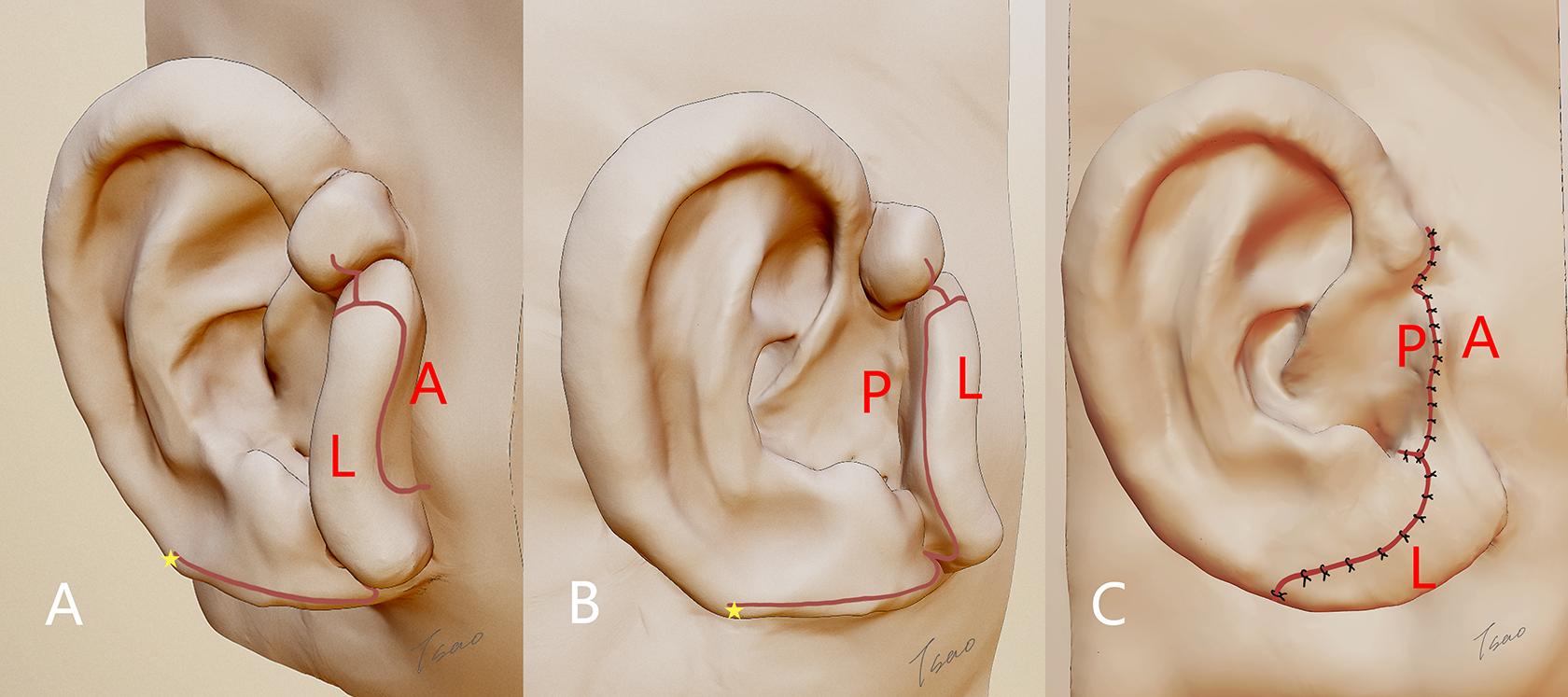
A-B, From the incision (red line), the skin of remnant ear was separated into 3 skin flaps: lobule flap (L), anterior flap (A in red text), and posterior flap (P). The distal end of lobule should be placed 2 to 5 mm exceeding the ending of helix cartilage (yellow star). C, The final contour of the reconstructed ear.
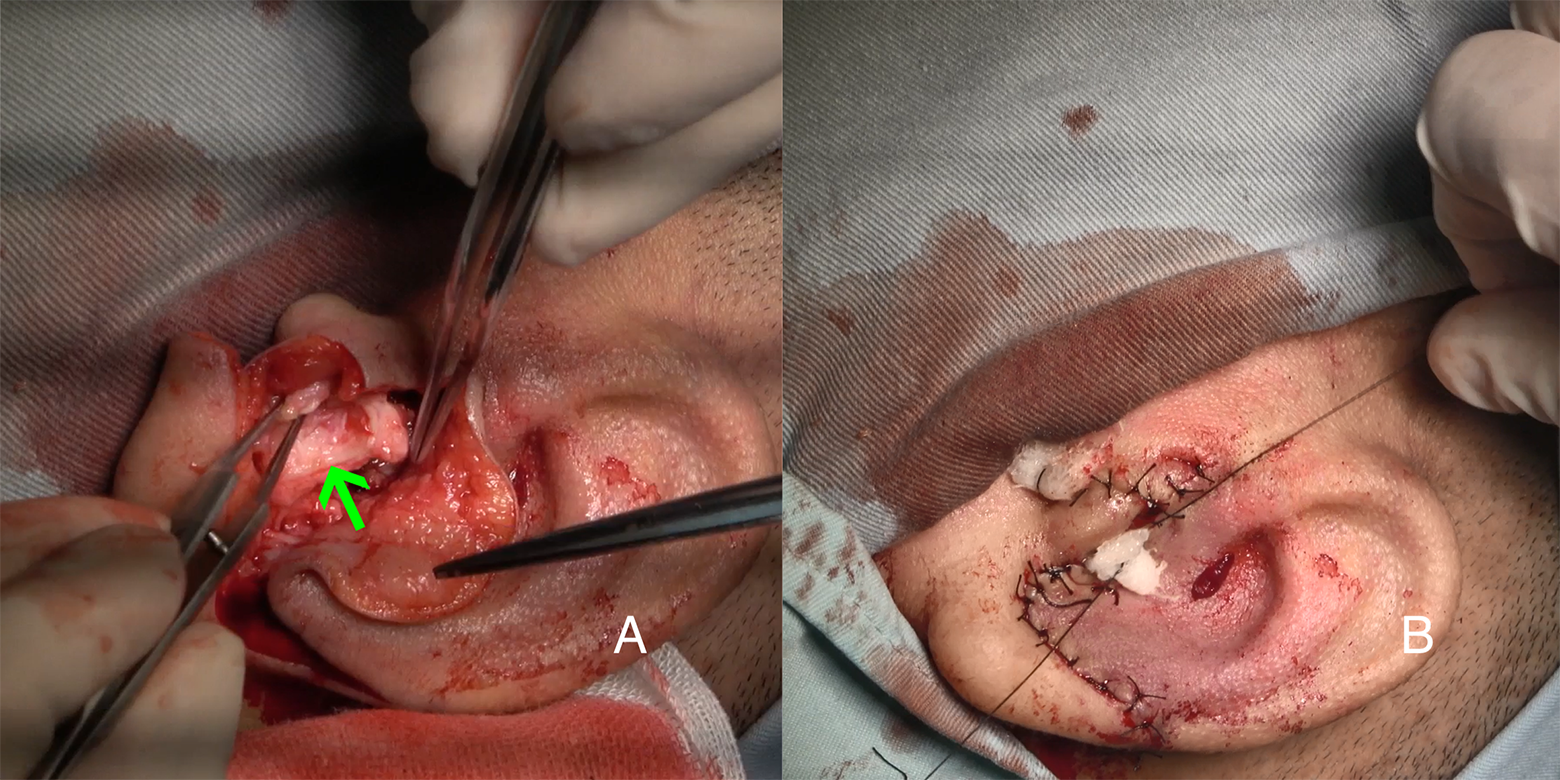
A, The cartilage vestige of remnant ear was trimmed into tragus (green arrow). B, The tragus was covered by anterior and posterior flap.
Evaluation and Follow-Up
Social applications, phone calls, and clinical appointments were used to conduct follow-up. A modified evaluation system of the reconstructed ear was proposed (Table 2), which was derived from Qian and Chen’s work. 9,13
Characteristics of Patients.
Evaluation System of Reconstructed Ear.
Results
From January 2013 to December 2019, a total of 953 patients received the expanded 2-flap method for ear reconstruction by one of the authors (Q.Z.), while 31 (3.3%) patients showed tissue expander exposure. Of those, 21 received modified Brent method to salvage the exposure; 7 patients’ tissue expander was removed and considered another operation at least 6 months later; 3 continued the tissue expansion technique.
Exposure of the tissue expander occurred during the injection and inactivity periods in 14 (51.9%) and 13 (48.2%) patients, respectively. With an average of 54.5 (range, 32-122 days) days and a volume of 57.8 mL saline (range from 27 to 78), 23 (85.2%) patients showed necrosis of the expanded flap in the late injection period and early inactivity period. The most frequent location of tissue expander complications was the lower part (15, 55.6%), with the middle area of the lower part as the most problematic area (Table 3).
Location of Tissue Expander Complications.
Complications occurred in all 3 stages of surgery, but only one required remedy operation. In the first stage, delayed healing of the exposure wound was found in one (3.7%) patient. The wound gradually closed after disinfection and dressing changes every day and finally showed no obvious scar. Exposure of the helix cartilage occurred in 1 (3.7%) patient with a diameter of 5 mm after the second stage, which enlarged over the following days. Temporoparietal fascia flap with a free skin graft was used to cover the cartilage. After detumescence, the reconstructed ear showed a good appearance. After the second stage, mild seroma under the concha was found in 1 (3.7%) patient and was soon absorbed. Two (7.4%) patients showed delayed healing of the incision after the third stage, all of which were located on the tragus.
The follow-up duration of these cases ranged from 1 to 7 (average, 4.5 years) years. During follow-up, 96.3% of patients and their families showed high satisfaction with their reconstructed ear. The mean score after the third stage surgery and at the follow-up was 11.1 and 10.3, respectively.
Case Reports
Case 1
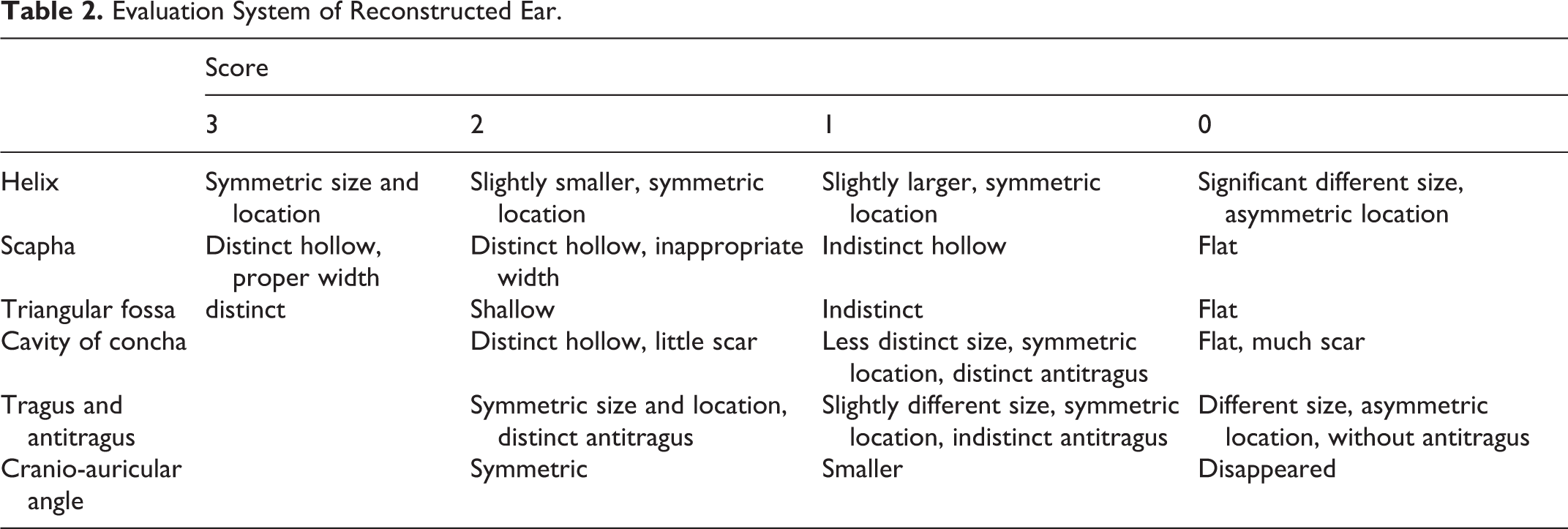
This was a 22-year-old female with right lobule-type microtia. One year before beginning ear reconstruction, she underwent canaloplasty in another hospital. The reconstructed auditory meatus was located on the top of the residual ear with obvious scar hyperplasia (Figure 4A). The periauricular skin was thick, scarring, and excessively taut (Figure 4B). Given the obstacle of the canaloplasty scar, a single tissue expander could not reach all the areas of the front margin. Therefore, we designed a special strategy for tissue expansion. A 30-mL rectangular tissue expander was placed horizontally above the auditory meatus, and a 50-mL kidney-shaped tissue expander was implanted upright below the auditory meatus (Figure 4C). Unfortunately, the scar location showed poor blood supply after 39 days with 75 mL of normal saline injected in total. This area gradually became necrotic and exposed (Figure 4F). By day 48, the patient underwent debridement and the modified Brent method to salvage the wound area. Before the second stage operation, the wound was closed and the scar on the reconstructed ear diminished (Figure 4E). After a 5-year follow-up, reconstructed ear showed a quite distinct appearance and the patient was very satisfied (Figure 4G-H).
A, Side view before operation. B, Postauricular tension. C, Operation design of tissue expanders implantation. D, Posterior view before operation. E, Side view after the first stage operation. F, Exposure of tissue expander. G, Side view after 5 years. H, Posterior view after 5 years.
Case 2
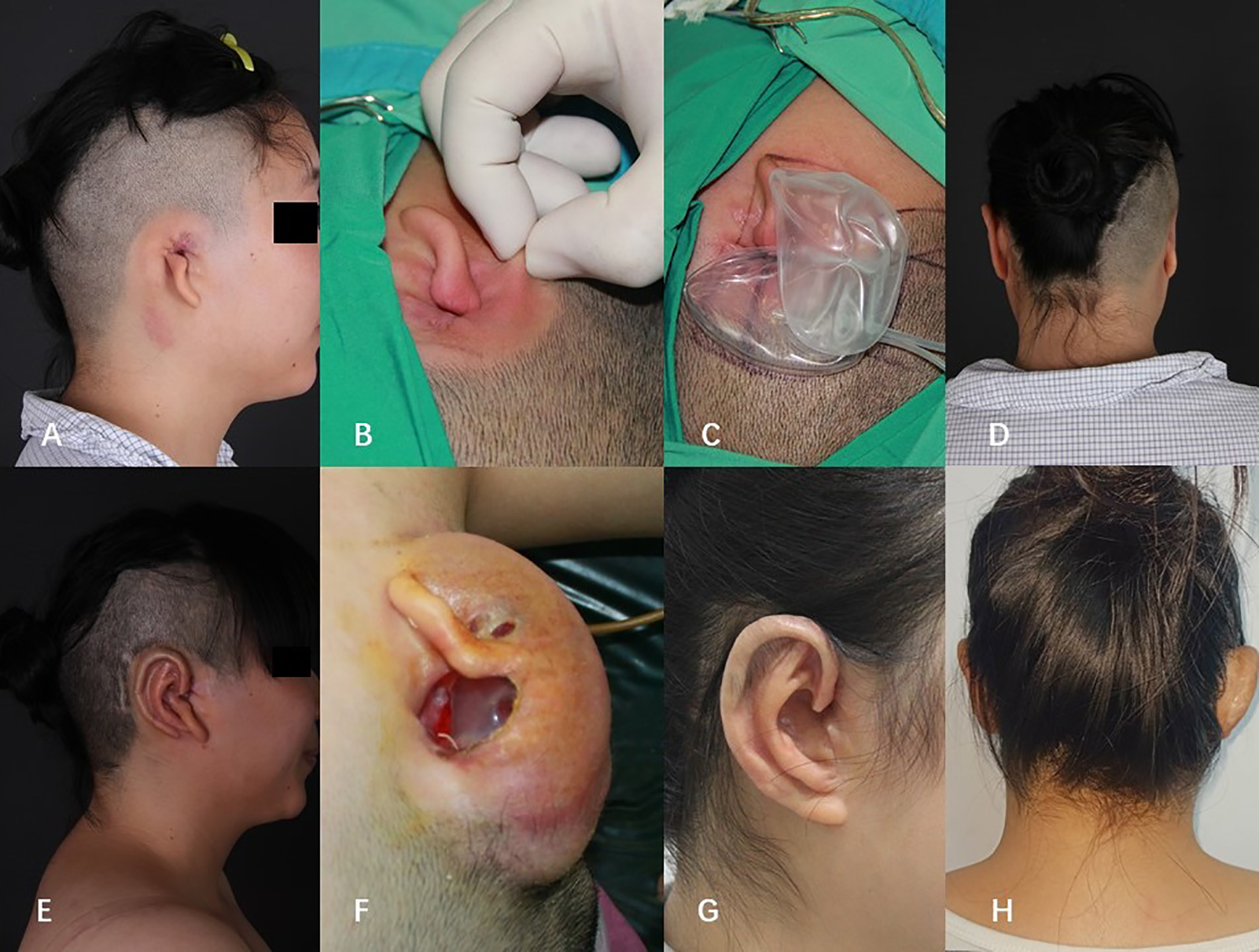
This was a 9-year-old boy with right lobule-type microtia and depressed temporal (Figure 5A). The residual auricular represented a relatively large lobule and low-set residual ear. This boy received a 50-mL kidney-shaped expander implantation and mandibular lengthening at the same stage. Thrombosis in the upper pole occurred on the 65th day with a volume of 70 mL normal saline and induced a 1.5 × 1.7 cm exposure of the tissue expander (Figure 5D). The wound was relatively clean. We used the modified Brent method for repair in 3 stages and obtained an 11-point ear according to the evaluation system (Figure 5B-C). After 3 times of laser hair removal, the hair on the scapha was visibly reduced. After a 6-year follow-up, the anti-helix cartilage showed little absorption (Figure 5E). The position of the reconstructed ear moved down, and the middle and lower part elongated with the lengthening of the mandible (Figure 5F). Although the final score of this reconstructed ear was decreased to 9 points, the parents of this patient expressed complete satisfaction and reported that the reconstructed ear showed quite good resistance to outer pressure.
A, Anterior view after tissue expander exposure. B-C, Side and posterior view of reconstructed ear before the third stage operation. D, Tissue expander exposure. E-F, Side and posterior view of reconstructed ear after 6-year follow-up.
Case 3
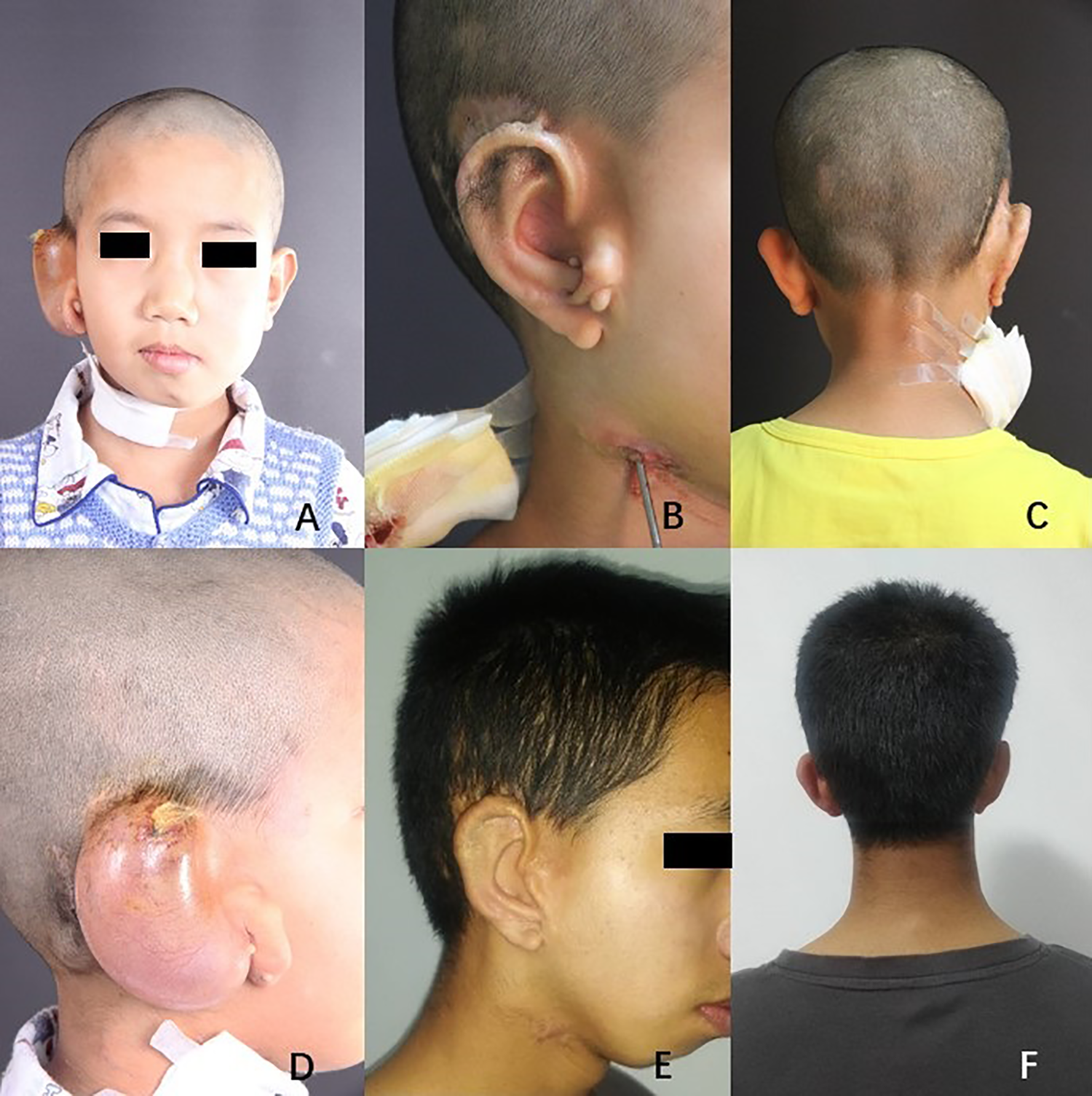
This patient was a 15-year-old girl with right lobule-type microtia. The residual ear represented a small lobule (Figure 6A). This girl was initially scheduled to receive the expanded 2-flap method, and a 50-mL kidney-shaped tissue expander was implanted. At the 35th day of injection period, necrosis appeared and showed no improvement after 5 days of observation (Figure 6B). With a volume of 52 mL saline, ear reconstruction was performed using a modified Brent method (Figure 6C). The 6-year follow-up showed a distinct contour, and the score of the reconstructed ear was 11 point (Figure 6D).
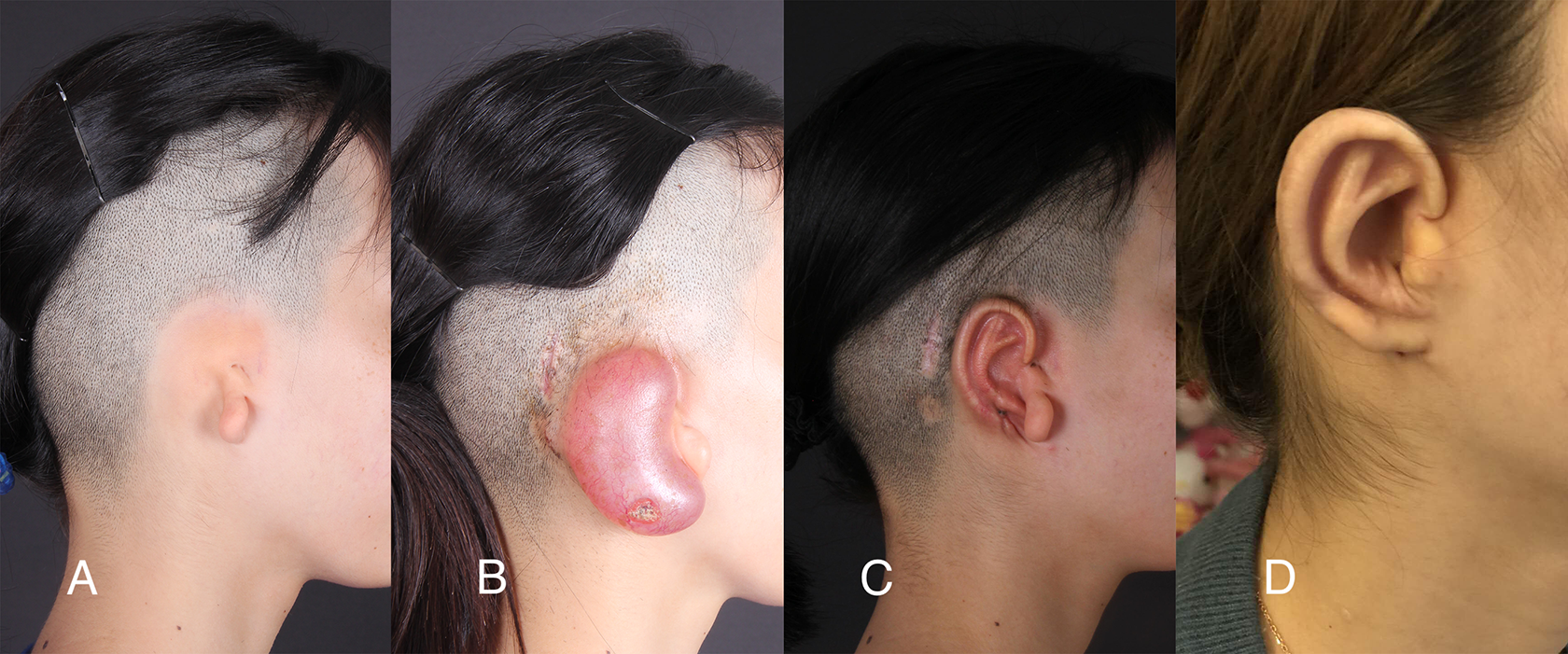
A, Right view of effected ear. B, Necrosis of expanded flap on the lower pole. C, Right view after the first stage operation. D, Reconstructed ear after 6 years.
Discussion
With worldwide efforts of surgeons, expanded skin flap method has been modified and is now widely used to overcome skin inefficiency. 9 -14,17 -23 Complications came after the increasing use of expansion technique. Chen reported 24 cases of partial necrosis of the expanding postauricular flap, with an incidence of 2.9% of necrosis, which is similar to the current study (3.3%). 15
Several factors contributed to necrosis and trauma of the expanded flap. The skin of the mastoid region is usually thin, taut, and have few subcutaneous tissues, especially in HFM patients. 13,24 In this study, the percentage of patients with HFM was 92.6%. As Qian et al described in 2017, the postauricular skin of HFM patients was overly taut and thin, which bears more risks in tissue expansion than others because of thinner skin with poor vascular supply as well as less adjacent tissue migration into the expanded field (case 2). 13 In addition, an expanded flap requires careful nursing to protect it from external violence in at least a 3-month-long expansion period. 15 Furthermore, for patients without an ear canal, many studies recommended that ear framework should be implanted in advance to canaloplasty because soft tissue removal could damage the vascular supply of the residual ear, and scar hyperplasia of canaloplasty is common, which makes ear reconstruction more difficult. 15,25 -27 In case 1, the scar on the upper residual ear showed little extensibility and poor vascular supply. With the dissection of the expander envelope, the vascular supply of scarring skin was affected and necrosis ensued.
Management of complications of tissue expander are usually found in the Results section of articles. Few studies have focused on summarizing experiences. When exposure of the tissue expander occurred, a decision was to be made between carrying through with the expansion method in a timely manner and removing the tissue expander only to consider other surgical method 6 months later. 15 The former option can be done only when the skin is expanded enough to cover the framework even after removal of necrosis and exposure area. The latter leaves wound healing and pocket closing in an absolutely tension-free condition, yet may result in scarring and thickening of subcutaneous layer, causing vulnerability and restriction for the next operation. Removal of tissue expander can be considered when uncontrolled infection occurs. Compared to the aforementioned 2 options, repairing the tissue expander exposure using modified Brent method requires minimal skin area and can reach a tension-free condition for wound healing as well as avoid adhesion of scar in internal soft tissue. Covering on the rough surface of framework and through debridement would make skin flap inadequate and thus induce excessive tension, which features color change and tightness of skin and is adverse to vascular perfusion and healing of incision. This strategy is safer and more convenient. From our experience, strategies for managing expander exposure are summarized in this flow chart (Figure 7). In this chart, the size of exposure and the amount of skin flap coverage were relative to the size of the contralateral ear and cranio-auricular sulcus, which were measured by the operator to judge the tension preoperatively.
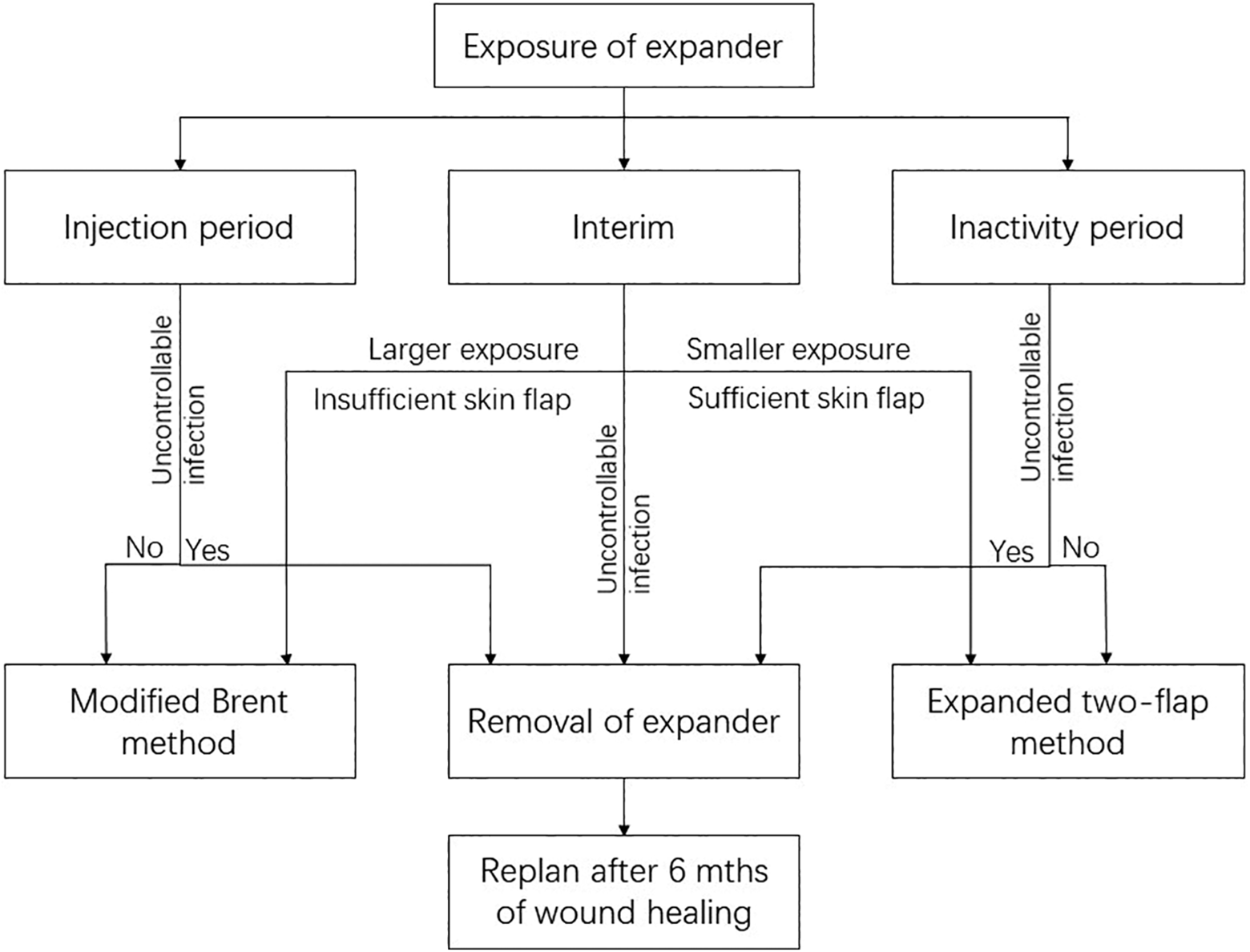
Flow chart of recommended management.
Some Technical Points Should Be Noted in This Strategy
The dissection for the implantation of tissue expander damaged perforator vessels from postauricular fascia to skin. The blood supply of expanded flap came from the margin of expander envelope, especially the remnant ear. Necrosis of expanded flap indicated poor blood perfusion in that region. The pedicle of remnant ear was necessary to be maintained to protect the expanded flap, so the implantation of ear framework in the first stage was through the initial incision.
In addition, the dissection of tissue expander also damaged the subcutaneous pedicle in concha region, which was proposed by Nagata in order to provide vascular supply for the mastoid region and the posterior flap of remnant ear. The efficacy of this pedicle was controversial first, 28,29 but it was proved by laser fluorescence and clinical trials in previous studies. 30,31 Furthermore, the attachment of skin to fascia can form deeper concave concha and eliminate the cavity between them to prevent potential seroma in ear elevation stage. For patients in this study, however, we ensured the blood supply of mastoid region and posterior skin by a prolonged interval and staging operation. The length of interval is empirical and we proposed that the duration between every stage should be no less than 6 months. Brent noted that the reduction of surgical stages was built around the premise of prevention of complications and repair. 32 Considering that patients in this study had already experienced undesired procedures, the security of surgery should be top priority.
Reasonable distribution of the skin flap over the cartilage framework was important for the recovery of the damaged area. An expanded flap with necrosis often presents with uneven thickness, with thinning of the central part of the expanded flap and the margins of broken area. After excision and closure of necrotic tissue, we distributed more skin in the thin and broken areas to create a tension-free condition for wound recovery as well as increase the blood supply. The drainage suction of the first stage should be adjusted to an appropriate pressure to improve vascular perfusion of the skin flap. In terms of framework fabrication, a distinct contour required more skin coverage and had higher demand of blood perfusion and was thus sometimes sacrificed to ensure skin flap survival.
The fabrication of ear framework in the first stage is similar to what Li et al described in previous study. 33 The base frame was spliced utilizing the sixth and seventh cartilage, which were fastened firmly by titanium suture. The eighth costal cartilage, which is a floating one, is trimmed to form the helix and fixed to the base frame. The tragus pocket was not dissected in the first stage in order to preserve the blood supply from the remnant ear, so the construction of tragus was arranged to the final stage. Remnant cartilage of microtia ear was removed in the final stage and trimmed into tragus, which reduced the amount of costal cartilage harvested in the first stage. For concha-type microtia, it is no need to reconstruct tragus.
Management of the capsule of the tissue expander was also important. The thick capsule led to an indistinct outline of the framework. Nevertheless, total capsulectomy usually results in hematoma and insufficient blood supply. 1,25 Particularly, for patients with tissue expander exposure, sufficient thickness of the skin flap was mandatory for operational security. Hence, we recommend that unless the capsule is excessively thick and its supply area shows no sign of poor blood irrigation, the capsule can be dissected properly.
Conclusion
Microtia can be treated effectively and relatively safely with the expanded 2-flap method. The occurrence of tissue expander exposure, which is caused by trauma and necrosis, can be repaired efficiently by a staging modified Brent method. With prolonged interval, reasonable distribution of the skin flap, and cautious capsulectomy, the damaged area was closed in a tension-free condition. In the long follow-up period, reconstructed ears showed a relatively stable appearance. It is meaningful for operators to continue exploring this strategy in excessive thin and taut skin conditions.
Supplemental Material
Supplemental Material, sj-pdf-1-ear-10.1177_01455613211007967 - Repair Exposure of the Postauricular Tissue Expander Using the Modified Brent Method: A 7-Year Experience
Supplemental Material, sj-pdf-1-ear-10.1177_01455613211007967 for Repair Exposure of the Postauricular Tissue Expander Using the Modified Brent Method: A 7-Year Experience by Tongyu Cao and Qingguo Zhang in Ear, Nose & Throat Journal
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grants from the National Natural Science Foundation of China (81571924 to Q.Z.).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
