Abstract
Primary heterotopic meningiomas are lesions that are not associated with a cranial nerve foramen, vertebral canal, or intracranial structure. The most common histopathological subtype is meningeal epithelioma. In clinical practice, primary heterotopic nasal meningioma occurs relatively rarely, and its most common pathological type is psammomatous meningioma, whereas nasal fibrous meningioma is infrequent. In our case, a 31-year-old male patient was admitted to the hospital with “progressive nasal obstruction on the right side for half a year.” Computed tomography examination of the paranasal sinuses revealed a polyploid mass in the right nasal cavity. The patient underwent surgical resection of the mass under nasal endoscopy. Histopathological examination confirmed that the mass was an ectopic meningioma of the nasal cavity. The patient was regularly followed up for 2 years without recurrence of the tumor. Primary heterotopic fibrous meningioma of the nasal cavity is clinically rare but should be considered as a differential diagnosis for hemangioma of the nasal cavity, inverted papilloma, and nasal polyp. The final diagnosis is based on pathology and immunohistochemistry analysis results.
Background
Meningiomas are extra-axial tumors of arachnoid cap cells. In 2016, the World Health Organization Classification of the Central Nervous System Tumors divided these tumors into WHO types I, II, and III, and 15 subtypes. 1 The biological characteristics of different pathological types and grades of meningiomas are dissimilar. 2 For example, ectopic meningiomas are tumors with meningioma morphology that occur in tissues and organs without meningioma cover, which, based on the connection with the central nervous system, can be divided into primary and secondary types. Among them, primary heterotopic meningioma refers to lesions without associations between the tumor tissue and the cranial nerve foramen or vertebral canal and intracranial structure. These tumors can occur in the middle ear, mouth, nose, parotid gland, neck, and other parts of the body. We herein report a case of fibrous meningioma originating from the nasal cavity.
Case Presentation and Literature Review
The patient, 31-year-old male, was admitted for “progressive nasal obstruction on the right side for half a year.” The patient claimed that he had neither major disease, such as infectious or family hereditary diseases, nor surgical history. Physical examination revealed no local rupture or uplift of the nasal skin. A dark red mass with a smooth surface was visible in the posterior part of the right nasal cavity. As can be seen in the computed tomography (CT) examination, presented in Figure 1, a soft tissue mass with expansive growth was detected in the right nasal cavity. Its maximum diameter was approximately 26 × 45 mm2. Partial bone absorption that was slightly shifted to the left was observed in the bony nasal septum. That soft tissue mass was moderately enhanced in the enhanced scan image. The right turbinate was not clearly visible due to soft tissue mass compression. Preoperative CT diagnoses of right nasal space occupation, hemangioma, or varus papilloma were considered.
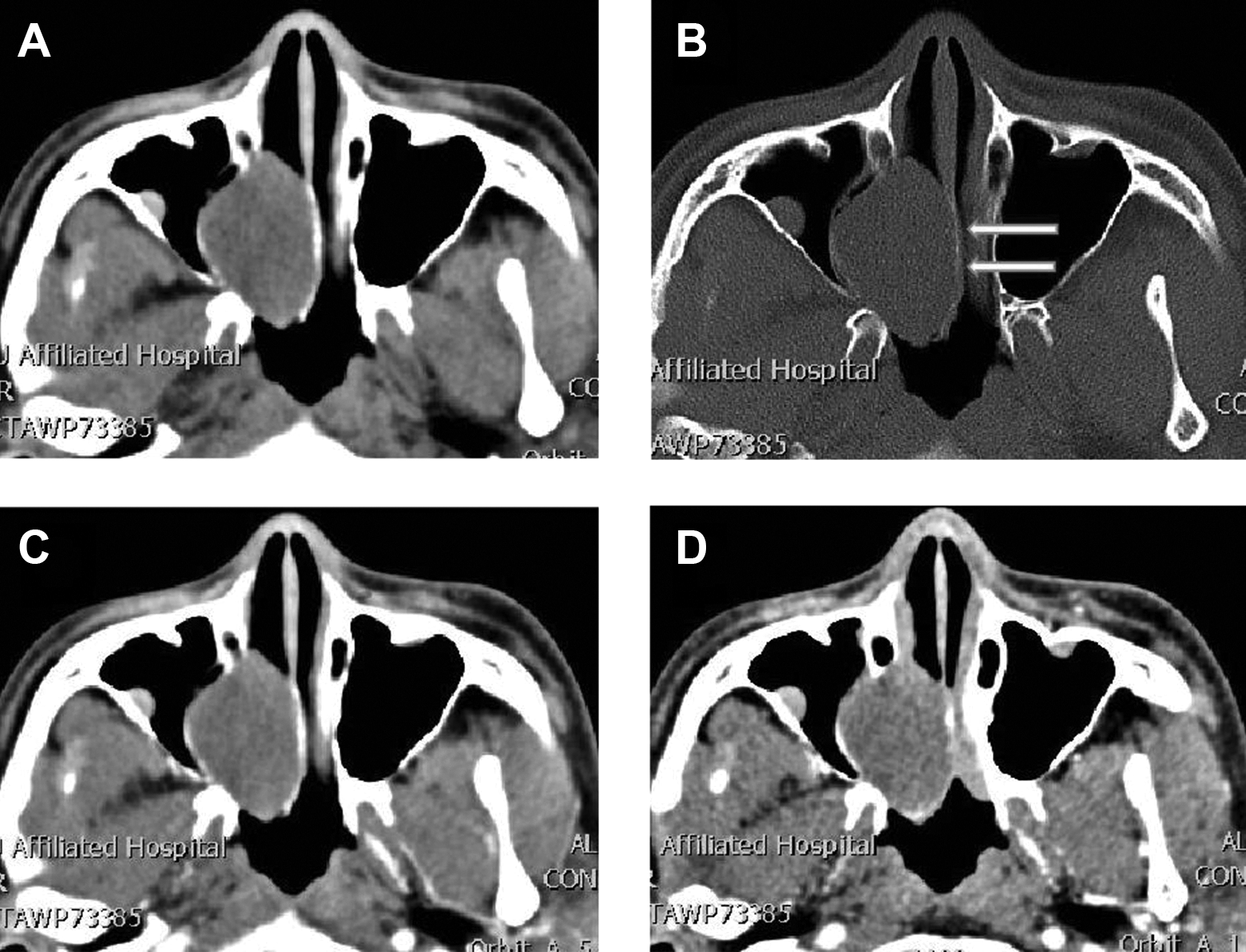
A, Axial CT soft tissue window shows the density shadow of nasal soft tissue measured on the right, with a clear boundary. B, Bone window shows nasal septal compression slightly to the left, nasal septal compression shifting to the left, and local bone resorption thinning (arrow). C, Arterial phase of contrast-enhanced CT showed mild uniform enhancement. D, Contrast-enhanced CT venous phase, tumor continuous uniform enhancement. CT indicates computed tomography.
The following intraoperative findings were observed: A dark red neoplasm in the right nasal cavity, protruding to the posterior nostril, with tumor tissue originating from the nasal mucosa. The tumor was resected under a nasal endoscope, then the nasal cavity was irrigated with Taeshu, and collagen and gelatin sponges were stuffed into the surgical cavity to stop bleeding. After the surgery, the tumor tissue removed was sent for pathological examination. The histopathological examination results, depicted in Figure 2, were as follows: Tumor cell vimentin, S100, EMA, and Ki67 (approximately 1%) were positively expressed, whereas SMA and HMB45 were not expressed. Observation under a light microscope revealed abundant cytoplasm in the tumor cells, small round nuclei, little heteromorphism, and few nucleoli. Final diagnosis of the tumor as nasal meningioma (fiber type, WHO I) was made on the basis of pathology findings in combination with immunohistochemical analysis results. The patient was followed up for 2 years postoperatively, and no definite tumor recurrence was found.
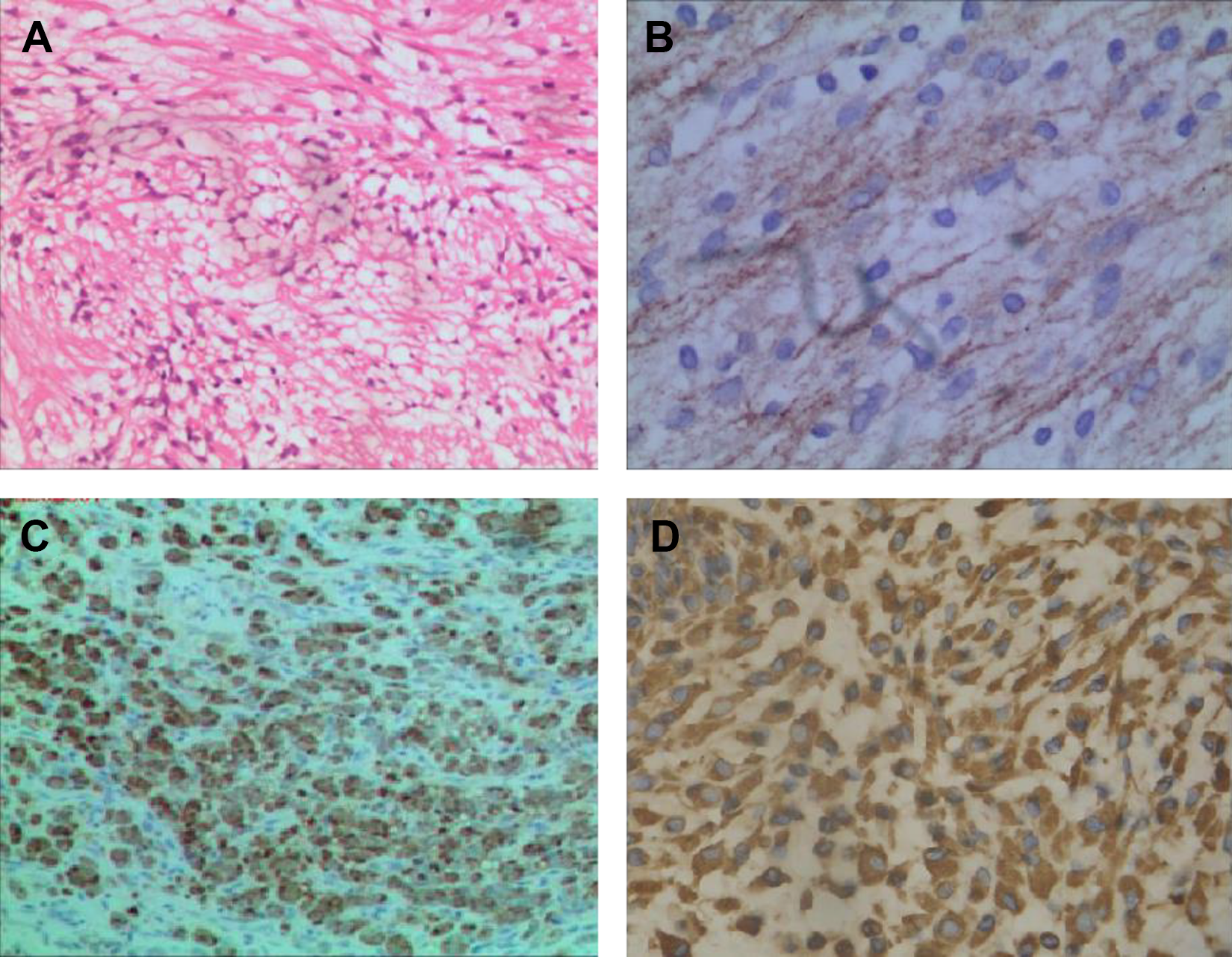
A, Pathological examination: (H&E, ×100) tumor cells have abundant cytoplasm, small round nuclei, and no obvious atypia. B, EMA positive expression in tumor cells. C, S100 positive expression in tumor cells. D, Immunohistochemical staining showed a strong positive for Vimentin. H&E indicates hematoxylin and eosin.
We searched and retrieved English-language case reports and case series on primary heterotopic nasal meningiomas published in the PubMed, Embase (including Medline), and Cochrane Library databases as of September 01, 2020. The following key words were used: (Nasal or Nose) and (Meningiomas OR Meningioma OR Meningoma OR Meningothelioma). The literature screening flow chart is presented in Figure 1. A total number of 8 full-text publications of studies on 8 patients were included in our analysis. The following data were recorded of each case: the first author, year of publication, and nationality as well as the patients’ age, gender, mass size, the onset of side, imaging features, clinical history, and follow-up time (Shown in Table 1). The scatterplot of gender, affected side, and age distribution of 9 patients (including our case) with nasal ectopic meningioma is illustrated in Figure 3. The 9 cases of nasal ectopic meningioma consisted of 5 females and 4 males, with no significant difference in the incidence between males and females. The age of onset ranged from 31 to 63 years, with a median age of 60 years. Except for one case, in which the appearance of the tumor was not documented in the literature, all cases presented as polyploid masses. Previous publications reported of meningothelial and transitional pathological types of ectopic meningioma of the nasal cavity, whereas, here, we report for the first time a case of fibrous meningioma of the nasal cavity. The clinical manifestations of nasal ectopic meningiomas reported in the literature are nonspecific, most predominant of which are nasal obstruction or/and nasal bleeding, whose histopathological nature tend to be benign. In the 9 patients studied, only 1 with atypical meningioma had recurrence and tumor transformation into a malignant type (grade III) 2 years after surgical resection. The imaging findings of nasal ectopic fibrous meningiomas were soft tissue masses with uniform density or signal, with rare calcification (only 1 in 9 patients). Therefore, the reported nasal ectopic meningiomas have been often misdiagnosed as nasal polyps or inverted papillomas.
Information on Heterotopic Meningiomas of the Nasal Cavity in the Literature.
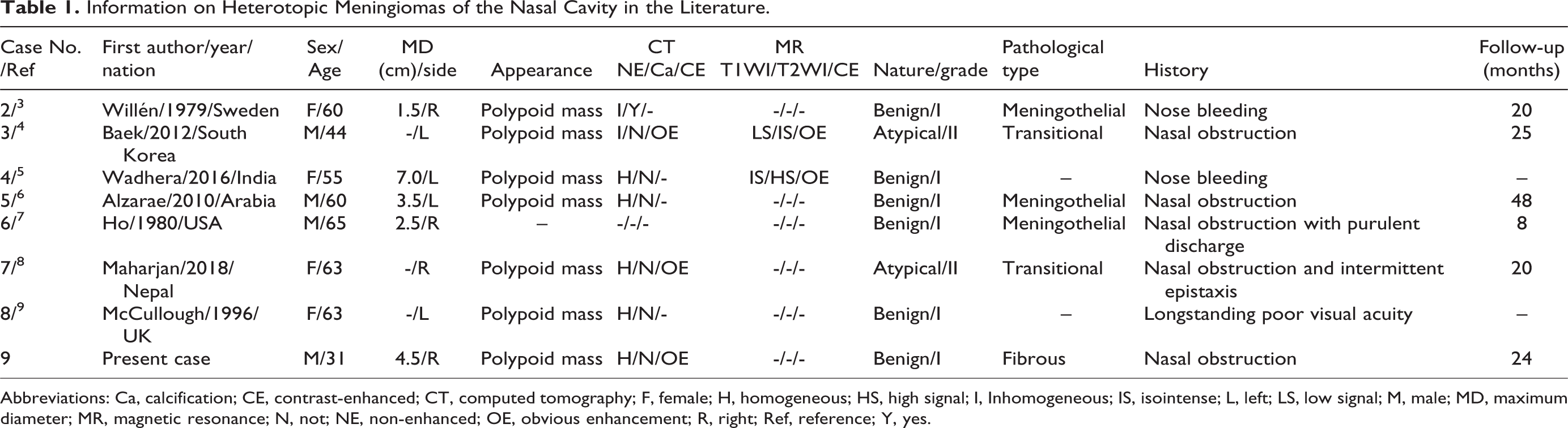
Abbreviations: Ca, calcification; CE, contrast-enhanced; CT, computed tomography; F, female; H, homogeneous; HS, high signal; I, Inhomogeneous; IS, isointense; L, left; LS, low signal; M, male; MD, maximum diameter; MR, magnetic resonance; N, not; NE, non-enhanced; OE, obvious enhancement; R, right; Ref, reference; Y, yes.
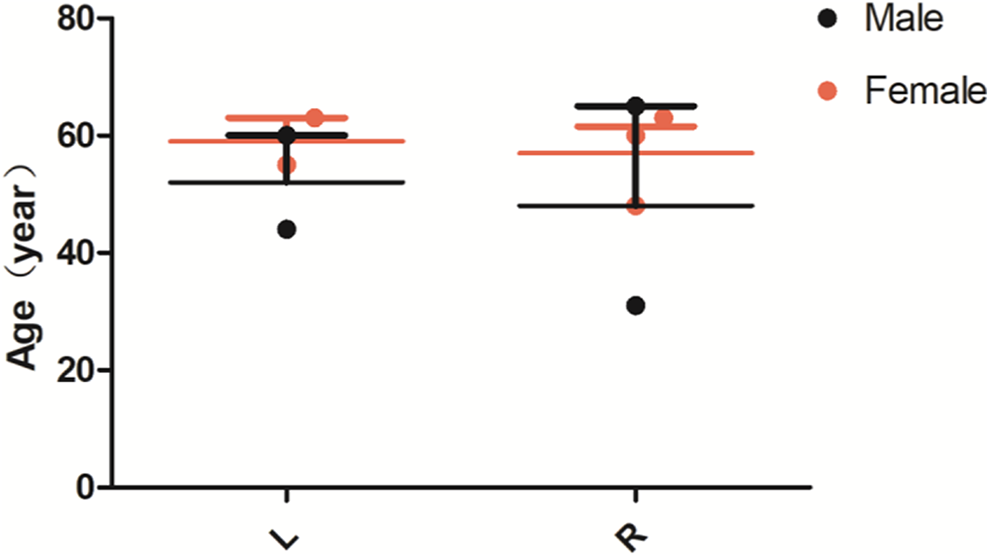
The scattered distribution of gender, diseased side and age of meningioma of the nasal cavity in 9 cases. There was no significant difference in the incidence of nasal meningioma between males and females and between the left and right sides. The age distribution was concentrated, and the median age of onset was 60 years old.
Discussion
Meningioma is a common tumor of the central nervous system, accounting for approximately 1/4 of intracranial tumors. It can occur at all ages and is more common in women. Ectopic meningiomas are rare, accounting for less than 1% of non-epithelial tumors. 10 A previous study of 163 primary extracranial meningiomas 11 showed that the most common histopathological subtypes were meningioma epithelioma, accounting for 53%, followed by the transitional type and psammomatous type, accounting for 12% respectively, while fibrous type accounted for less than 7%. Moreover, another investigation 12 confirmed that most of the ectopic meningiomas originating in the nasal cavity were of the psammomatous type, whereas fibrous type was rare. The clinical symptoms of nasal ectopic meningiomas are nonspecific and usually include nasal congestion, nosebleed, headache, and facial deformities. A longer history is associated with more serious symptoms and signs. Most of the nasal meningiomas are benign; local infiltration and distant metastasis may appear in the malignant types. Importantly, benign meningiomas are often misdiagnosed as nasal polyps due to their polyp appearance. Currently, CT examination is not able to accurately identify and distinguish the nature of a nasal cavity tumor, but it is essential in determining the location, size, adjacent bone structures, and the relationship between the tumor and the surrounding tissues and organs. Fibrous meningioma of the nasal cavity may present as a soft tissue mass with equal density and clear boundaries in CT, with a shape similar to that of nasal polyp. Compression deformation and hyperplasia can be observed in adjacent bones. Calcification is rare in the tumor, but no specificity is present. Therefore, it is difficult to differentiate from some benign nasal tumors such as inverted papilloma, hemangioma, nasal polyps, and isolated fibroma. In our case, CT examination showed a soft-tissue, polyploid mass in the right nasal cavity with clear boundaries. The adjacent bone structure (septum nasi osseum) had partial bone absorption, slight compression, and a shift to the left, which was consistent with the findings of the aforementioned study. The diagnosis of nasal meningioma is based on histopathological examination. The histopathological morphology is consistent with that of intracranial meningioma, and the positive expression of both immunohistochemical markers vimentin and EMA facilitates diagnosis. 13 In our case, the immunohistochemical results of the patient were in agreement with these signs. Surgical resection is the preferred method for the treatment of nasal meningioma, but sometimes resection may be difficult to complete, and the recurrence rate can reach more than 25%. In case of incomplete resection or malignant transformation, postoperative radiotherapy or anti-progesterone therapy should be considered.14,15 The case we report here did not receive radiotherapy or chemotherapy after surgical resection of tumor tissue. He was followed up for 2 years, with CT of paranasal sinus showing no obvious signs of recurrence.
According to the clinical characteristics and CT findings of the nasal cavity tumor, the primary heterotopic fibrous meningioma needs to be differentiated from the following tumors:
(1) Nasal hemangioma: in 2/3 of the patients, it occurred in the nasal septum or the middle inferior turbinate. Computed tomography plain scan reveals a soft-tissue, polyploid mass with clear edges, low-density necrosis in the center of the lesion, and partial calcification. Most of the enhanced scan show obvious uneven enhancement, which is not consistent with the enhanced characteristics of meningioma; (2) varus papilloma is the most common benign tumor in the nasal cavity and paranasal sinus, accounting for up to 5% of nasal tumors. It tends to occur in male patients over 50 years old. Varus papilloma usually originates from the lateral nasal wall. The CT manifestations of varus papilloma are soft-tissue masses connected with the nasal cavity or paranasal sinus. The edge is usually less smooth than the edge of meningioma, high-density calcification, or residual bone can be seen inside. The enhanced scan is with uniform or nonuniform enhancement; the adjacent bone may show compression changes and local hyperplasia sclerosis 16 ; (3) nasal polyps, usually occurring bilaterally in the nasal cavity, are difficult to distinguish from the aforementioned benign nasal tumors when they are located unilaterally in the nasal cavity. Computed tomography manifestations of unilateral nasal polyps are uniform or nonuniform soft-tissue masses in the nasal cavity, most of which with clear boundaries, leading to partial or complete obstruction of the nasal cavity. The bone of the adjacent sinus wall and nasal septum can shift to the healthy side due to tumor compression, which can be accompanied by local bone absorption and destruction; (4) solitary fibroma rarely occurs in the nasal cavity. It originates from CD34(+) dendritic mesenchymal cells and can differentiate into fibroblasts, myofibroblasts, vascular endothelium, or epidermis, with a peak age of onset ranging from 40 to 60 years. The CT appearance of solitary fibroma is isolated, round, or irregular mass of soft tissue with clear boundaries. It has swelling potential, and its growth can fill the whole nasal cavity, involving the sinuses and pushing away the surrounding tissues. This causes oppression and absorption of the adjacent bone. Furthermore, it is difficult to distinguish the lesion from a nasal meningioma in cases it presents as a well-defined soft tissue mass. In addition, nasal ectopic fibrous meningiomas still need to be differentiated from some early malignant neoplastic lesions of the nasal cavity, such as malignant melanoma, lymphoma, and adenocarcinoma.
Conclusion
Briefly, the primary occurrence of ectopic fibrous meningioma is relatively rare, but it should be considered as one of the differential diagnoses of inverted papilloma, nasal polyp, and hemangioma in clinical work. In the absence of specific clinical features of nasal ectopic meningiomas, CT examination shows uniform density in the nasal cavity, smooth edges, and the presence of a soft-tissue mass, as well as enhanced scanning homogeneity, which all have prognostic implications for that disease. The final diagnosis of this disease is based on pathological and immunohistochemical examination findings, and transnasal endoscopic resection of the tumor tissue is an effective treatment option.
Supplemental Material
Supplemental Material, sj-doc-1-ear-10.1177_0145561320974863 - Primary Heterotopic Meningioma of Nasal Cavity: Case Report and Literature Review
Supplemental Material, sj-doc-1-ear-10.1177_0145561320974863 for Primary Heterotopic Meningioma of Nasal Cavity: Case Report and Literature Review by Xianwen Hu, Maoyan Jiang, Zelong Feng, Juan Wang, Pan Wang and Jiong Cai in Ear, Nose & Throat Journal
Footnotes
Authors’ Note
Hu X conceived the case statement and drafted the manuscript. Wang P collected the patient’s data and performed the follow-up. Feng Z and Hu X wrote the manuscript together. Jiang M and Wang J wrote the commentaries below pathological pictures. Cai J and Wang P revised and finalized the manuscript for publication. Written informed consent was obtained from the patient for publication of this case report and any accompanying images. A copy of the written consent is available for review by the editor of this journal. All the authors read the article and agreed to publish it. All procedures carried out in the study met the ethical standards of the National Research Council, as well as the Declaration of Helsinki and its subsequent amendments or similar ethical standards. The patient’s oral and written informed consent was obtained for this case report and related pictures.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the National Natural Science Foundation of the Peoples Republic of China, NSFC (grant numbers: 81571712). The sponsor, Jiong Cai, was responsible for designing the study and the final review.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
