Abstract
Objective:
The purpose of this scholarly review is to present an update of the efficacy, safety, and distribution of intranasal corticosteroids (INCS) in the context of treatment for chronic rhinosinusitis (CRS).
Materials and Methods:
A literature review from 1999 to 2020 of MEDLINE, PubMed, and EMBASE databases was performed, using a comprehensive search strategy. Studies reporting on efficacy, safety, and distribution of all INCS formulations, both Food and Drug Administration (FDA) and non-FDA approved, were reviewed.
Results and Conclusions:
High-level evidence publications and position papers support the role of INCS in medical treatment for CRS. Significant improvement in disease-specific and general quality of life measures is observed with all formulations of INCS. Overall, the use of both FDA and published non-FDA INCS appears to be safe. Several novel distribution devices might improve penetration to specific areas within the sinuses.
Introduction
Chronic rhinosinusitis (CRS) is a multifactorial condition with unclear etiology. Although the underlying causes and inciting events remain debated, the resultant inflammation has been well characterized. Irrespective of its phenotype, the inflammation in CRS remains extremely responsive to corticosteroid medications in the vast majority of patients. Unlike oral corticosteroids, which have a significant side effect profile, topical intranasal corticosteroids (INCS) remain well tolerated with an excellent long-term safety profile. It is for this reason that they are considered first-line therapy for CRS. 1,2
The mode of action of INCS is complex. Although it remains unknown whether INCS penetrate the nasal mucosa, their lack of systemic absorption supports a local action on the nasal mucosa. Intranasal corticosteroids are known to influence epithelial cells through their direct binding to glucocorticoid receptors within the cells 3 and are believed to directly affect mast cells, Langerhans Cells, macrophages, and fibroblasts and also reduce the influx of inflammatory cells. In this way, they are thought to have direct effects on both the early-phase response as well as the secretion of inflammatory mediators (interleukin-1, -2, -4, -6, -8, tumor necrosis factor-α, Granulocyte-macrophage colony-stimulating factor), released during the delayed inflammatory response. 4,5
Numerous well-performed high-level randomized controlled trials (RCTs) have been published supporting the effectiveness of INCS in the management of CRS with and without nasal polyposis. A recently published Cochrane review, including 18 randomized placebo-controlled trials, demonstrated improvements in symptom scores and endoscopic disease severity scores in both chronic rhinosinusitis without nasal polyps (CRSsNP) and chronic rhinosinusitis with nasal polyps (CRSwNP) patients. In the CRSwNP cohort, reductions in polyp size and improvements in quality of life scores and olfaction scores were also observed. 6 This high level of evidence has resulted in position papers recommending the routine use of INCS in the management of CRS with and without nasal polyposis with a level A-1 recommendation. 1,2
This review aims to present an update of the current INCS available for the treatment of CRS. Formulations and delivery devices will be reviewed with a particular focus on available evidence for their efficacy and safety. Factors affecting distribution, as well as safety in the pediatric population and during pregnancy, will also be discussed.
Material and Methods
We performed a search in PubMed using the Medical Subject Headings (MeSH) terms “administration, intranasal” “steroids” “chronic rhinosinusitis” and “efficacy [subheading]” “distribution [subheading]” “complications [subheading]”. A comprehensive review of the English-language literature was performed using the following databases: Ovid MEDLINE (1999 to July 2020), the National Library of Medicine’s version of PubMed and EMBASE (1999 to July 2020).
The following MeSH terms were used in the literature review: steroids; corticosteroids; intranasal corticosteroids; distribution; efficacy; complications; safety; chronic rhinosinusitis; nasal polyposis; administration, topical; nasal sprays; nasal lavage; toxicity and delivery.
Formulations
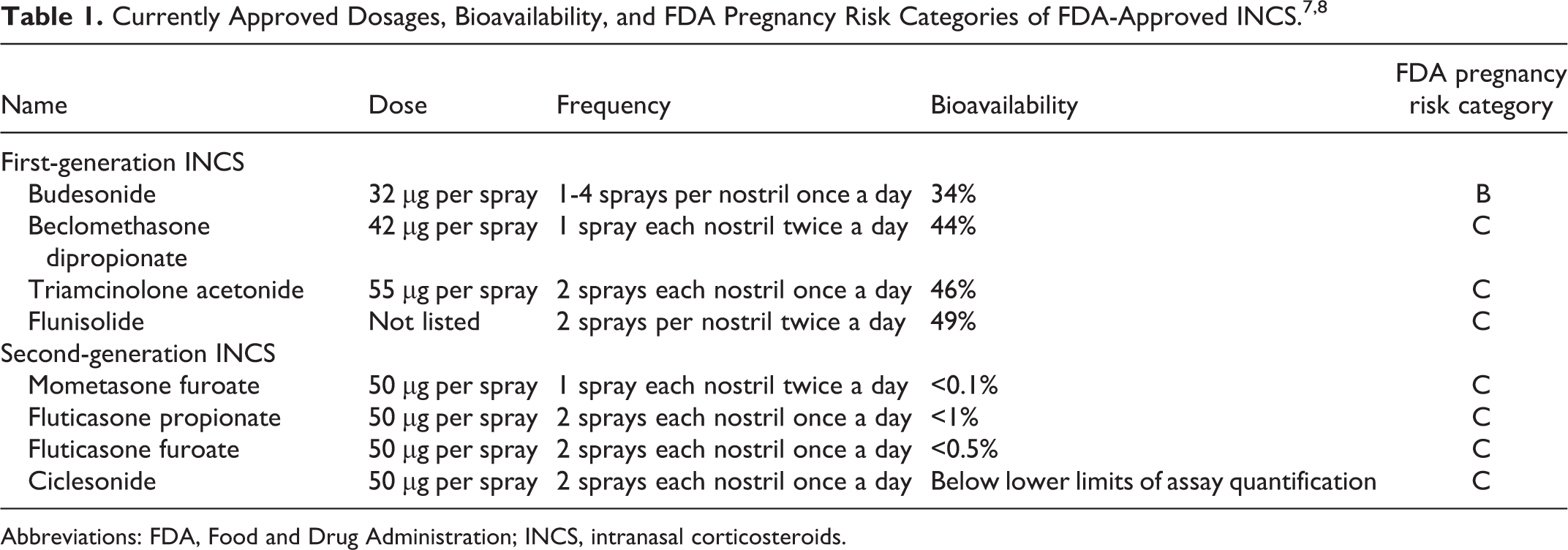
Intranasal corticosteroids are classified into first- and second-generation formulations (Table 1). The first-generation INCS include budesonide, triamcinolone acetonide, beclomethasone dipropionate, and flunisolide. Second-generation preparations include mometasone furoate, fluticasone furoate, fluticasone propionate, and ciclesonide. Although initial concerns existed regarding possible systemic absorption from daily use, studies of newer generation INCS have shown these agents to be well tolerated with a low side effect profile and no significant impact on the hypothalamus–pituitary–adrenal axis (HPAA). 7
First-generation INCS have higher systemic bioavailability compared to newer generation INCS, ranging from 10% to 49%. Second-generation INCS have a significantly lower rate of systemic absorption, being less than 0.1% for both ciclesonide and mometasone furoate 8 (Table 1). Although different compounds have different absorption rates in the nasal mucosa and gut, systemic bioavailability depends primarily on the degree of first-pass hepatic metabolism. 9 Newer generation INCS like ciclesonide and fluticasone propionate owe their low bioavailability to extensive hepatic metabolism and low gastrointestinal absorption. Differences in lipid solubility between generations have also been described with higher and faster uptake by the nasal mucosa in newer generation INCS because of their lipophilic substances. It is thought that this may improve their efficacy and onset of action. 10
Abbreviations: FDA, Food and Drug Administration; INCS, intranasal corticosteroids.
Corticosteroids can be delivered topically via sprays, in high-volume irrigation devices, or via nebulization. All 8 INCS are currently approved by the Food and Drug Administration (FDA) to treat perennial and seasonal allergic rhinitis when delivered as a spray. Mometasone carries an additional indication for seasonal allergic rhinitis prophylaxis and has been clinically approved to be used in the pediatric population from the age of 3 years. 11 At present, there is no FDA approval for the use of INCS for CRS in irrigations or nebulized form.
Delivery Devices
Intranasal corticosteroids sprays
Intranasal corticosteroids were initially delivered via Freon propelled aerosols; however, these devices yielded poor intranasal drug distribution. 12 Metered-dose pump sprays and aqueous pump sprays have since been shown to deliver a more significant percentage of INCS dose to the patient with less retention in the device. Aqueous pump sprays remain the most commonly used delivery device for INCS, given their more preferably distribution pattern. Unlike metered-dose aerosols and pump sprays, which result in drug deposition primarily within the anterior, non-ciliated region of the nose, aqueous pump sprays appear to also deposit drugs to the ciliated mucous membrane regions of the nasal mucosa. 13 With this said, intranasal distribution from an aqueous pump spray can be affected by various factors, including the volume of nasal spray and the spray cone angle.
The clinical efficacy seems to be similar among most forms of INCS sprays. In 2016, a Cochrane Review by Chong et al evaluated the effects of different types of INCS in all published RCTs with a minimum follow-up of 3 months to determine whether a difference existed between the different formulations. Nine RCTs, with a total of 911 participants, were included. The authors found insufficient evidence to suggest that one type of INCS spray was more effective than another in patients with CRS. 14
In general, INCS sprays have proven to be safe. Minor local adverse effects such as bleeding, nasal dryness, throat dryness, and burning have been reported. There is moderate quality evidence to suggest that epistaxis is associated with higher doses of INCS (risk ratio 2.06, 95% confidence interval 1.2 to 3.54) 7,14,15 as well as incorrect use. Severe complications such as nasal mucosa atrophy and septal perforation are infrequent and almost always prevented by instructing the patient on appropriate technique with the spray aimed away from the nasal septum. Studies that have examined the effect of INCS on the HPAA have shown an overall minimal impact on cortisol metabolism. 16 Although 2 studies have shown a decreased output of urinary cortisol during treatment with either fluticasone propionate or beclomethasone dipropionate in adults, most studies of these agents have shown no effect on the HPAA. 17 Mometasone furoate has been extensively studied in more than 20 trials of adults and children. No effects on the HPAA were detected in either children or adults. 18 No clinical relevant impact of INCS was found on intraocular pressure (IOP), glaucoma, lens opacity, nor cataract formation in recently published systematic reviews. 19,20 A meta-analysis and systematic review by Donaldson et al of 60 studies concluded that INCS are safe in the adult population. 21
High-volume irrigations
Given the well-documented increase in sinus penetration with high-volume irrigations, particularly in postoperative cases, the off-label use of budesonide irrigations has become increasingly popular in CRS treatment. 22 Budesonide repulses (0.5 mg-1 mg) diluted in high-volume, low-pressure devices (200-240 mL) have been shown to confer a greater symptom reduction and a more significant improvement in endoscopic appearance in unoperated patients when compared with saline irrigations. 23 Table 2 summarizes the most recent high-level studies published regarding high-volume budesonide irrigations for CRS treatment. 23 -26 Budesonide irrigations have also demonstrated a reduction in oral steroid rescue treatment in postoperative functional endoscopic sinus surgery patients with asthma. 27
Summary of High-level Studies Evaluating Budesonide Irrigations in Patients With CRS.
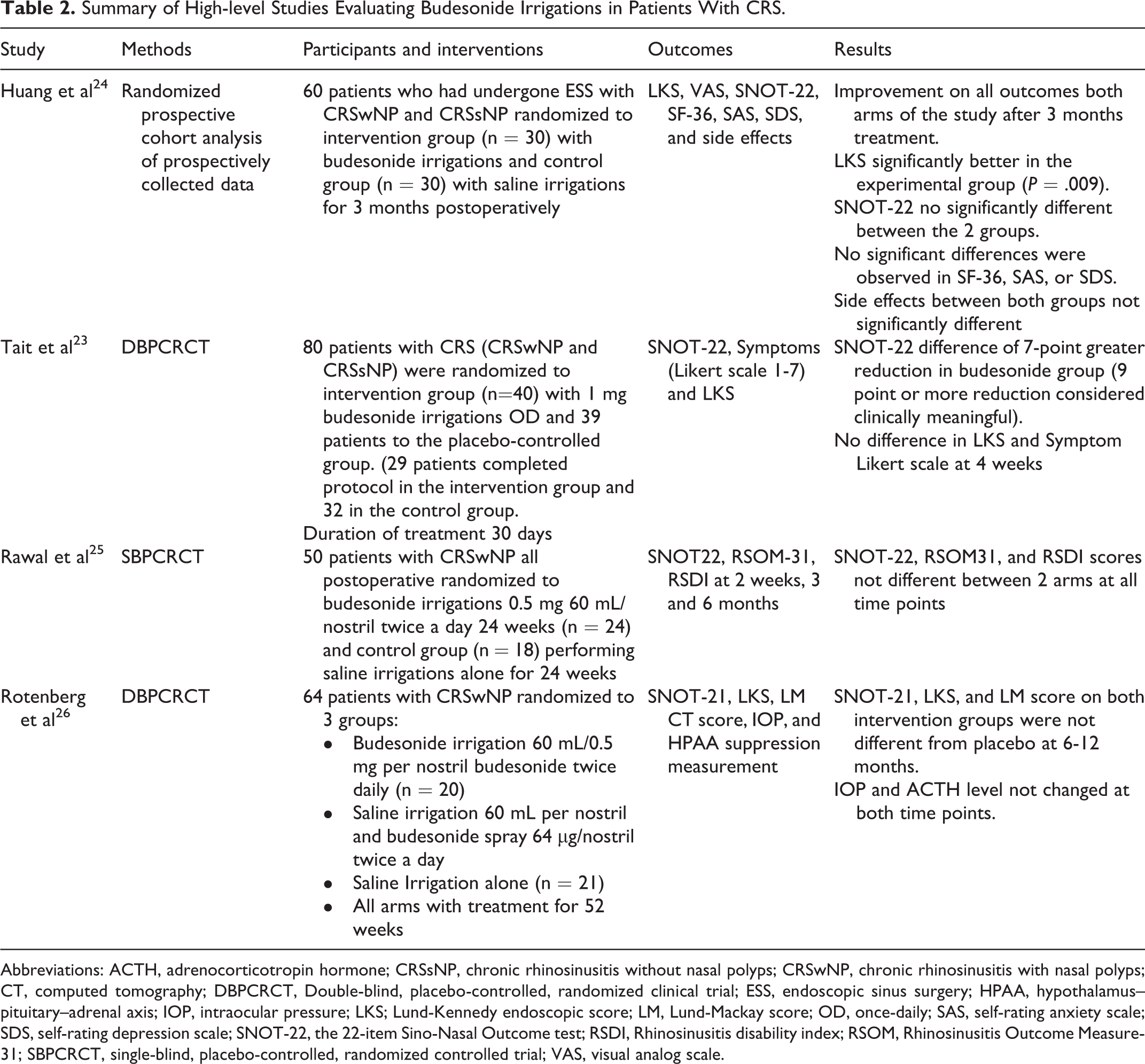
Abbreviations: ACTH, adrenocorticotropin hormone; CRSsNP, chronic rhinosinusitis without nasal polyps; CRSwNP, chronic rhinosinusitis with nasal polyps; CT, computed tomography; DBPCRCT, Double-blind, placebo-controlled, randomized clinical trial; ESS, endoscopic sinus surgery; HPAA, hypothalamus–pituitary–adrenal axis; IOP, intraocular pressure; LKS; Lund-Kennedy endoscopic score; LM, Lund-Mackay score; OD, once-daily; SAS, self-rating anxiety scale; SDS, self-rating depression scale; SNOT-22, the 22-item Sino-Nasal Outcome test; RSDI, Rhinosinusitis disability index; RSOM, Rhinosinusitis Outcome Measure-31; SBPCRCT, single-blind, placebo-controlled, randomized controlled trial; VAS, visual analog scale.
High-volume irrigations with other INCS have also been studied in patients with CRS. Harvey et al published a placebo-controlled randomized clinical trial comparing mometasone irrigations with mometasone nasal spray. 28 High-volume intranasal irrigation with mometasone (2 mg in 240 mL saline) showed a greater improvement in total visual analog scale scores, endoscopy scores, and CT scores at 12 months compared to mometasone sprays.
With the increased off-label use of steroid irrigations, concerns have been raised about their potential toxicity. Based on the study by Harvey et al, <3% of sinus irrigation fluid remains in the sinuses post high-volume irrigation. 29 As a result, the total dose of steroids retained in the sinonasal cavity is significantly lower than that after INCS spray use. Given the long-term safety profile of INCS, the safety of budesonide irrigations had been assumed. This has now been confirmed with well-performed long-term studies. Smith et al identified no evidence of HPAA suppression or elevation in IOP with daily use of budesonide irrigations for 12 months. 30 Although a study by Soudry et al showed similar results, an asymptomatic HPAA suppression in a small subset of patients concurrently using nasal steroid sprays or pulmonary steroid inhalers was observed. 31 Reassuringly, this HPAA suppression was found to be reversible when the budesonide irrigations were ceased. Caution is, therefore, recommended in patients using multiple forms of topically delivered steroid therapy.
Atomizers and nebulizers
In an attempt to improve the distribution of INCS, alternate methods of application and distribution have been explored in recent years. Thamboo et al 32 published a study in which they used budesonide via a mucosal atomization device (MAD; Wolfe-Tory Medical). This device atomizes medication into particles ranging between 30 and 100 µm in size. Their research suggests that the application of budesonide with the MAD results in a non-diluted low-volume, high-concentration distribution of the drug intranasally, which in theory should translate to increased anti-inflammatory activity. In terms of budesonide’s long-term safety with this device, 3% of patients developed a diminished cortisol response test, and 6% had an elevated IOP after a mean duration of treatment of 23 months. 33
Another delivery system designed to improve penetration and availability of INCS, particularly to the posterior and superior sinonasal spaces, is the exhalation delivery system with fluticasone (EDS-FLU) Xhance by OptiNose (OptiNose Inc). The EDS-FLU consists of a mouthpiece and a sealing nosepiece with an optimized frustoconical shape that expands the nasal valve area. When exhaling into the mouthpiece against the device’s resistance, the soft palate is elevated by positive oropharyngeal pressure, isolating the nasal cavity from the rest of the respiratory system. 34 Gamma deposition studies have demonstrated that with the EDS-FLU, there is less drug deposition in the non-ciliated nasal vestibule and significantly greater distribution to the upper posterior regions when compared to conventional delivery with a spray pump. 35 The EDS-FLU has been proven to decrease off-target drop deposition and drug loss by swallowing and drip out. 36,37
In a 3-month placebo-controlled study in 109 patients with CRSwNP, EDS-FLU was reported to be well tolerated. Combined symptom scores, nasal blockage, and endoscopic polyp scores were improved at 12 weeks. 38 In large, open-label cohorts, the degree of symptom improvement from baseline in both CRSwNP and CRSsNP populations was comparable. 39,40
The NAVIGATE I and II trials evaluated the efficacy and safety of the EDS-FLU versus placebo in patients with CRSwNP in double-blind RCTs. All EDS-FLU doses produced statistically significant improvement in the SNOT-22 total score and all 4 cardinal symptoms of nasal polyposis (congestion/obstruction, facial pain/pressure, rhinorrhea/postnasal drip, hyposmia/anosmia). 41,42
Factors Affecting Distribution
Various factors have been shown to affect the topical delivery of medications to the sinuses. These include anatomical variations, the delivery device itself, head-positioning, and surgical status. Delivery devices include low volume devices such as metered dose sprays, droppers, atomizers, nebulizers, and high-volume devices such as irrigation bottles and neti pots. Studies involving commercially available INCS sprays have demonstrated that most atomized drugs are deposited in the anterior part of the nasal cavity and do not reach the sinus mucosa on the initial actuation of the device. 43,44 Similarly, poor sinus distribution has also been demonstrated for the other low-volume devices irrespective of the extent of surgery. While sinus penetration is also minimal for high-volume devices in the unoperated state, 45,46 both surgery and the extent of surgery have a significant favorable influence on sinus penetration for these devices.
Cadaveric studies have shown that surgery increases irrigation fluid distribution to all sinuses, particularly the sphenoid and frontal sinuses. 29,47 -51 Several studies have focused on the different distribution rates of high-volume irrigations to the frontal sinus with graded surgical approaches. Barham et al found greatest distribution scores in cadaver heads after performing a Draf III procedure (90.7% distribution) when compared with Draf IIb and Draf IIa (81.3% and 50.1%, respectively). 49,50 Middle turbinate resection appears to positively impact distribution to all sinuses in cadaveric studies. 51 However, no in vivo studies have been performed to date, and the risk of developing empty nose syndrome should be considered. 52
Computational fluid dynamic (CFD) simulation is a novel technique for evaluating irrigant penetration of the individual sinus cavities. Computational fluid dynamic technique has been widely used in the field of rhinology to simulate multiple variables such as aerosol particle deposition 53 and nasal airflow. 54 Computational fluid dynamic studies have also demonstrated that extended surgical approaches to the frontal sinus (ie, Draf III procedure) significantly increase penetration of high-volume irrigations. 55 Head positioning has been studied using CFD simulations, finding that the nose-to-ceiling head position is superior to the nose-to-floor position in delivering irrigation to the sphenoid sinus. 56
Based on the available evidence, a recently published evidence-based review recommended for surgery and the use of high-volume devices after conventional sinus surgery (aggregate quality evidence C) and against low-volume devices (drops, sprays, nebulizers) based on their poor sinus distribution. 57 In patients who could not perform or tolerate high-volume irrigations, they recommended the use of low-volume devices in the supine head back or lateral head position to improve nasal distribution to olfactory cleft and middle meatus, respectively. A Cochrane review by Snidvongs et al has also independently concluded that topical steroid delivery for patients with CRSsNP had more responders and increased beneficial effects in symptoms when delivered directly to the sinus cavity with high-volume irrigations when compared with nasal sprays. 58
Safety in Pediatric and Pregnant Patients
There are currently only 4 INCS approved by the FDA for use in children less than 6 years of age. These include mometasone furoate, triamcinolone acetonide, fluticasone furoate, and fluticasone propionate. A recent meta-analysis evaluated 33 studies and concluded that INCS in FDA-approved administration routes appear to be safe in the pediatric population. 59 No persistent abnormalities in cortisol levels or IOP changes were found. Growth velocity changes were not significant in most evaluated RCTs; a few studies noted a temporary reduction in short-term growth velocity.
All INCS were previously labeled as category C based on the FDA’s pregnancy risk classification system. Budesonide was upgraded to category B based on 3 studies using a Swedish birth registry from 1995 to 2001 for mothers using budesonide during pregnancy (Table 1). 60,61 Literature about the safety and efficacy of INCS in pregnancy is still lacking, and concrete recommendations cannot be made. Previously published expert panel recommendations consider all modern INCS safe to use for CRS maintenance during pregnancy, and value should be placed on maternal sinonasal inflammation control during pregnancy. 62 Based on a review by Alhussein et al, the use of fluticasone furoate, mometasone, and budesonide is considered safe if used at the recommended therapeutic dosages. 63
Summary
Intranasal corticosteroids play a significant role in the medical treatment of CRS, particularly in improving disease-specific and general quality of life measures.
Intranasal corticosteroid efficacy in CRS is supported by high-level evidence and endorsed by the European Position Paper on Rhinosinusitis and Nasal Polyps 2020 and International Consensus Statement on Allergy and Rhinology: Rhinosinusitis documents.
Intranasal corticosteroids appear to be generally safe in all routes of administration; however, long-term use of INCS irrigations with concomitant corticosteroid pulmonary inhalers may warrant active screening for subclinical adrenal suppression.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article. Alkis J. Psaltis is a consultant for ENT technologies, Medtronic, Tissium and FuseTech and is on the speakers bureau for Baxter.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.