Abstract
Introduction:
While the cochleotoxicity of cisplatin has been well investigated, less is known about the effects of platinum-based chemotherapy on the vestibular system. In particular, there is a lack of prospective studies using modern laboratory vestibular testing that examine the effects of cisplatin on the semicircular canals and on the otolith organs. The aim of the present study was, therefore, to investigate the vestibulotoxic effect of cisplatin in patients with head and neck tumors who are undergoing chemoradiation.
Methods:
Forty-five patients undergoing cisplatin-based chemoradiation for head and neck cancer received a vestibular assessment consisting of anamnesis, a horizontal video head impulse test (vHIT), ocular and cervical vestibular evoked myogenic potential testing, as well as pure tone audiometry. This assessment was performed before therapy, 6 weeks after therapy, and 3 months after therapy.
Results:
Video head impulse test showed a significantly reduced median gain 6 weeks after chemoradiation. In addition, significantly more refixational saccades could be detected after therapy. Vestibular evoked myogenic potential testing results also revealed significant changes, whereas pure tone audiometry did not. None of the patients mentioned “dizziness” during the follow-up examinations.
Conclusion:
We demonstrated a vestibulotoxic effect of cisplatin-based chemoradiation in patients with head and neck cancer. Future studies are needed to better understand cisplatin-induced vestibulotoxicity and to identify possible vestibuloprotective substances. Still, before and after chemoradiation, patients should undergo not only auditory testing but also vestibular testing in order to detect potential vestibular loss as soon as possible and to quickly initiate vestibular physiotherapy.
Introduction
Ototoxicity—which is often synonymously used to describe cochleotoxicity—is a common side effect of a number of cytostatics; damage to the cochlea and associated cochlear symptoms such as hearing loss and the appearance of tinnitus have now been well studied and are well understood.1,2 In contrast, vestibulotoxicity, the undesired side effect of various medications on the balance organs, has so far been less studied. It may also caused by a number of chemotherapeutic agents, in particular platinum-containing cytostatics and aminoglycoside antibiotics, especially gentamicin.2-4 The potentially toxic effect of various drugs on the balance organs has been shown in numerous clinical studies and in vitro experiments.5-10 The consequences of a vestibulotoxic disorder may include dizziness, gait insecurity (especially in the dark), oscillopsia, and possibly an increased tendency to fall.11-13
While the cochleotoxicity of cisplatin has now been well-investigated, knowledge about the effects of platinum-containing cytostatics on the balance organs is much less extensive, as Prayuenyong et al highlight in their review on this topic. 14 A differentiated, receptor-specific diagnosis has only been possible since the introduction of the video head impulse test (vHIT) and the vestibular evoked myogenic potential (VEMP).15,16 Only through these vestibular tests can the distinctive effects on the vestibular organs be detected, understood, and documented.
To the best of our knowledge, there are currently no prospective clinical studies that investigate the effects of platinum-containing drugs on the horizontal semicircular canal as well as on the otolith organs using modern neurotological diagnostics. Existing studies are only partially retrospective and/or only assess the vestibulo–ocular reflex (VOR) using bithermal testing 3 and/or rotary testing 17 and do not consider the otolith or semicircular function.
Early detection and localization of vestibulotoxicity under platinum-containing cytostatic therapy is of considerable relevance since chronic balance and dizziness complaints can lead to a significantly reduced quality of life (QoL) for the patient. 18 Therefore, treatment should be initiated as early as possible using vestibular physiotherapy. 19 In addition, it is quite conceivable that the symptoms caused by a bilateral toxic-induced loss of function are often not noticed early or are related to other side effects of cytostatic therapy (eg, nausea, vomiting).
The aim of the present prospective study was therefore to determine the possible toxic effect of cisplatin-containing chemoradiation therapy on the function of both the otolith organs and the semicircular canal.
Methods
Forty-five patients who underwent radiochemotherapy (RCT) for head and neck tumors between November 2014 and October 2017 were admitted after receiving ethics approval (2014-567N-MA) by the medical ethics committee II of the Medical Faculty Mannheim, Ruprecht-Karls-Universität Heidelberg, and were included in the present prospective, monocentric study. Included were patients who were able to give consent and who underwent platinum-containing adjuvant or primary chemoradiation therapy for the treatment of a head and neck tumor. Exclusion criteria were previously known peripheral vestibular disease (eg, hydropic inner ear disease) or vestibulotoxic chemotherapy that had already taken place. Before the start of RCT, after the end of therapy (approx. 6 weeks) and 3 months after the start of therapy, all patients received a vestibular assessment as part of the first post-therapeutic tumor follow-up. The assessment consisted of an anamnesis survey, a horizontal vHIT and ocular and vestibular evoked myogenic potentials (VEMPs). In addition, a pure tone audiogram was performed all 3 times to assess cochlear function and to detect possible cochleotoxicity. The patients received simultaneous RCT with cisplatin and 5-fluorouracil. Cisplatin was administered as part of the scheme at weeks 1 and 4 with a dosage of 80mg/m2.
Video Head Impulse Test
A portable video oculography system (EyeSeeCam) was used for the vHIT examination. A high-speed infrared camera and built-in accelerometer were used to measure head and eye movements at a sampling rate of 250 Hz. The VOR was assessed quantitatively during horizontal head impulses. The target head speed was 100 to 200/s with amplitudes between 5° and 15° from the center to the lateral position. At least 15 pulses per page were carried out in the horizontal plane. The target values were the mean gain at 40 ms, 60 ms, and 80 ms as well as the occurrence of catch-up saccades.
Cervical VEMP Testing
For the cervical VEMP test, a tone burst stimulus (500 Hz; 125 dB SPL; rise/fall time each 1 millisecond, plateau 2 milliseconds) with a repetition rate of 5/s was used, which was presented monaurally. Electromyographic changes in the sternocleidomastoid muscle were derived. Responses were averaged over 100 stimuli. The first positive and negative peaks recorded between 13 ms and 23 ms were referred to as p13 and n23 responses. Cervical VEMPs were considered absent if no reproducible p13 and n23 responses could be determined.
Ocular VEMP Test
The ocular VEMPs (oVEMPs) were recorded with eyes open and a maximal gaze upward. For recording of oVEMP, tone bursts (4/s, rise/fall time each 1 millisecond, plateau 2 milliseconds) at 500 Hz were applied with 100 dB nHL. All EMG signals were amplified and filtered (band-pass filtering with high and low pass filtering, software filtering 5-800 Hz, hardware filtering 10 Hz/6 oct). The VEMP recordings were made with a reproducibility of greater than 90% after 100 to 200 stimuli. The responses were averaged over 50 stimuli. The first negative and positive peaks of the ocular VEMP response after the stimulus began were designated n1 and p1. Ocular VEMPs were also considered to be absent if no defined n1 response could be identified. For the purpose of this study, mean c and oVEMP latencies were analyzed as prolonged latencies have been proven to be a better indicator for retrolabyrinthine lesions as amplitudes. 20
Pure Tone Audiogram
Before performing pure tone audiometry, otoscopy was performed to exclude a sound conduction component or other pathology. The air conduction hearing threshold was then standardized at 250 Hz, 500 Hz, 1000 Hz, 2000 Hz, 4000 Hz, and 8000 Hz. Then, the average hearing threshold (pure tone average [PTA]) was determined from the tested frequencies 500 Hz, 1000 Hz, 2000 Hz, and 4000 Hz calculated on each side. Additionally, changes in hearing thresholds for 8000 Hz were analyzed. All tests were carried out in a soundproof cabin with regularly calibrated devices (ANSI S3.6-1996).
Statistical analysis was done using SPSS Statistics 17.0 (SPSS Inc). Before the study started, a power analysis was carried out to determine that a sample size of 40 participants would provide a power of 0.80 at a level of .05 to detect clinically significant differences. The nonparametric Wilcoxon rank-sum test was used for all non-normally distributed results of the vestibular tests; normally distributed results were tested using the t test. In addition, parameters such as age, sex, tumor location, and stage of the included patients were evaluated descriptively. A P value less than .05 was considered to be statistically relevant.
Results
A total of 45 patients (31 men, 14 women) aged 45 to 76 years (average 61.1 years) were included in the study. The individual entities and TNM stages as well as further descriptive statistics and radiation dose can be found in Table 1.
Further Descriptive Data Is Shown for All 45 Patients Enrolled in this Study.
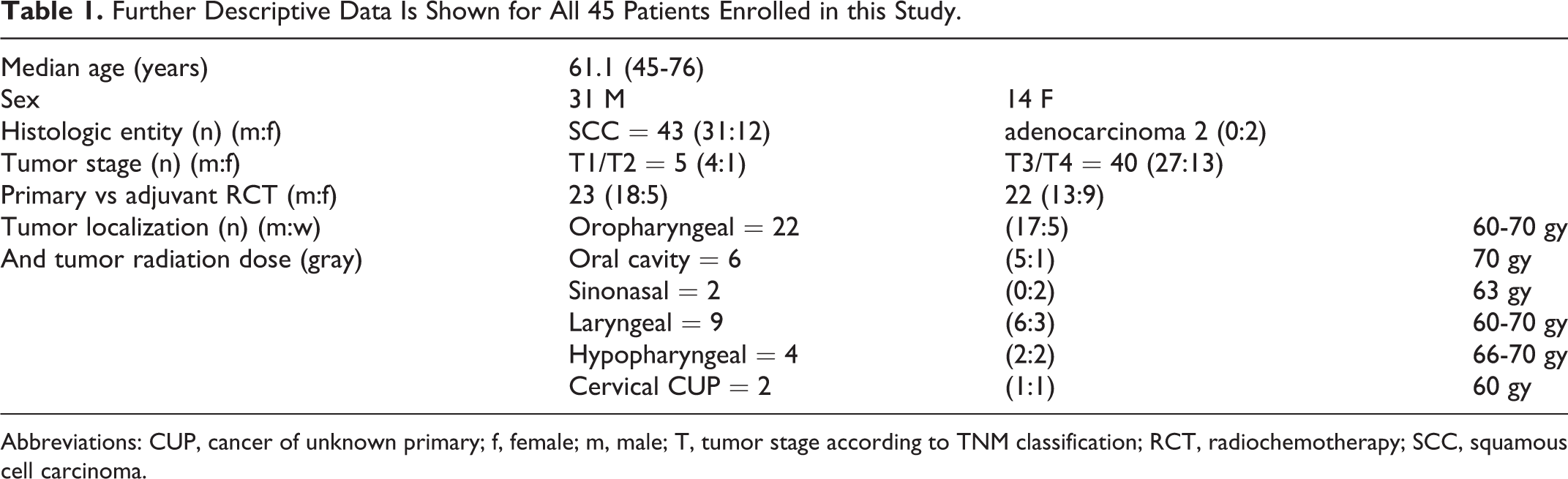
Abbreviations: CUP, cancer of unknown primary; f, female; m, male; T, tumor stage according to TNM classification; RCT, radiochemotherapy; SCC, squamous cell carcinoma.
In 41 of the 45 included patients, all of the above tests were carried out at each of the prior and 6 respectively 3 months after therapy. One patient (n = 1, 2.2%) died before the last checkup, which was to take place 3 months after the end of therapy. In 2 other patients (n = 2, 4.4%), the second cycle of RCT was performed with carboplatin instead of cisplatin due to a pronounced nephrotoxicity. Another patient in post-therapeutic checkups refused any electrode placement as part of the VEMP test due to a pronounced radiogenic dermatitis. A total of 41 (27 men, 14 women) of the 45 patients (91.1%) could therefore carry out all planned tests. Of all included patients, 23 (51.1%) patients received adjuvant RCT and 22 (48.9%) of the patients received primary RCT. In any of the tests performed, there was no significant gender difference. Moreover, there was no significant difference between the results of patients who received adjuvant or primary RCT.
History of Dizziness
As part of the targeted dizziness anamnesis on the days of the tests, no patients expressed an “occurrence of spinning or swaying vertigo.”
Video Head Impulse Test
Median gain before RCT was 1.01 (n = 41) when tested horizontally to the right and 1.03 when tested to the left. Six weeks after RCT, the gain was significantly reduced on both sides (P < .05). Even 3 months after the completion of therapy, it continued to be reduced but was no longer statistically significant (see Table 2).
vHIT Results Prior to RCT, As Well As 6 Weeks and 3 Months After Treatment.a
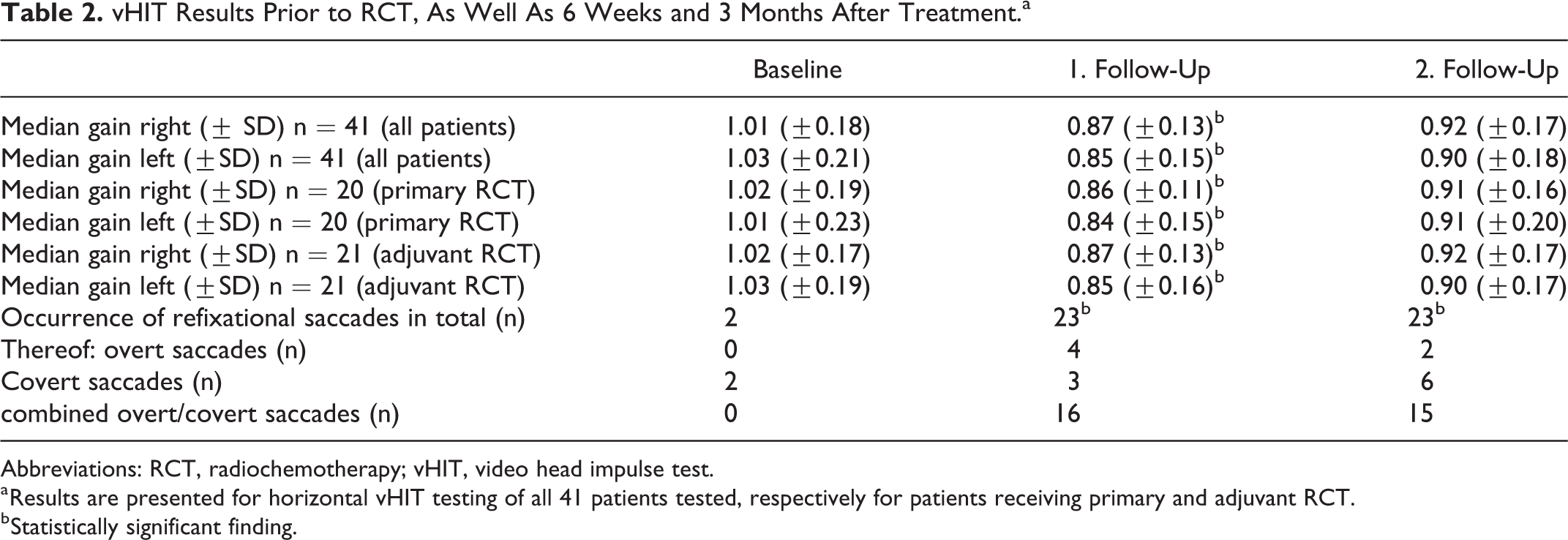
Abbreviations: RCT, radiochemotherapy; vHIT, video head impulse test.
a Results are presented for horizontal vHIT testing of all 41 patients tested, respectively for patients receiving primary and adjuvant RCT.
b Statistically significant finding.
Catchup saccades occurred in 23 of the 41 patients 6 weeks after RCT (n = 23, 56.1%). Combined catchup overt and covert saccades were present in 16 patients (n = 16, 39.0%). The remaining 7 patients either had isolated overt saccades (n = 4) or covert saccades (n = 3) (see Table 2). The saccades persisted for 3 months after RCT.
There was no significant difference between the results of patients who received adjuvant or primary RCT either for the evaluation of the gain or for the occurrence of catchup saccades (P = .12 or P = .34; further details see Table 2).
Vestibular Evoked Myogenic Potential
The cervical VEMPs (cVEMPs) were reproducible in all patients before the start of therapy. The mean latency was 14.6 ms for p13 and 22.5 ms for n23 on the right side and 14.3 ms and 22.3 ms on the left side. Three months after RCT, reproducible responses could not be carried out in 12 patients (n = 12, 29.3%), which corresponds to a statistically significant increase in the cVEMP results rated as “missing” (P < .05). In the other patients for whom cVEMP showed reproducible results (n = 29, 70.7%), latencies were prolonged, but this was not statistically significant (P = .19).
The oVEMPs could also be derived reproducibly in all patients before RCT. The mean latency for n1 was 10.4 ms and 15.6 ms for p1 when measuring the right side and 10.2 ms and 15.2 ms for the left side. In the oVEMPs, too, no reproducible potential was measured in about a third of the patients, a statistically significant proportion, both 6 weeks and 3 months after RCT (n = 14, 34.2%, P < .05). Latencies were extended 3 months after RCT to 11.4 ms for n1 and 16.1 ms for p1 (P = .21) on the right and 11.0 ms and 15.9 ms on the left but were not statistically significant. Table 3 provides an overview of the VEMP results.
Results of VEMP Testing at Baseline, 6 Weeks, and 3 Months After Treatment.a
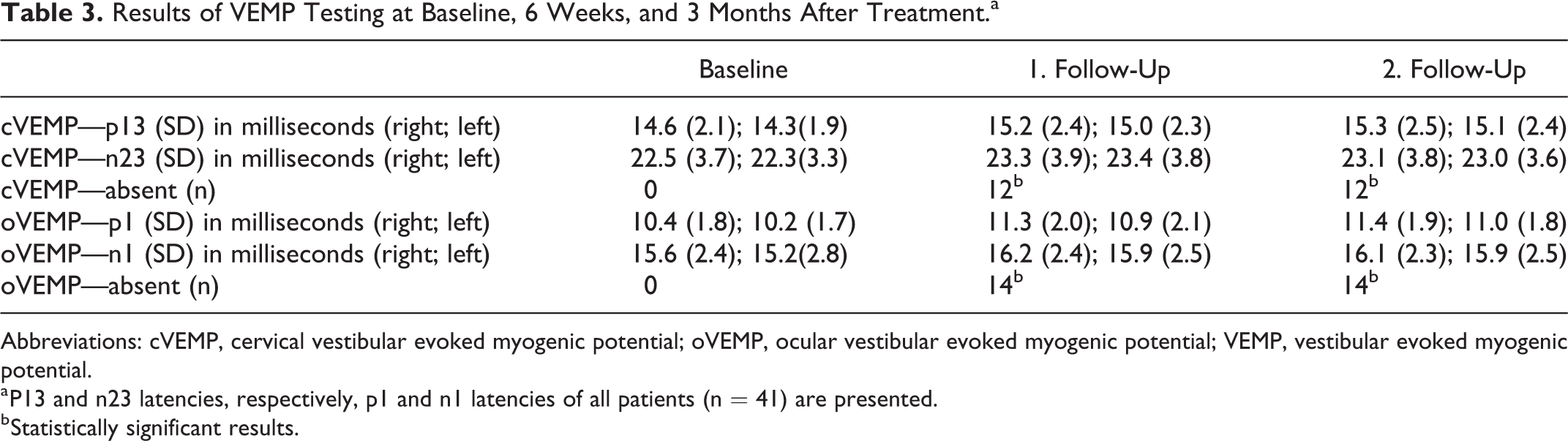
Abbreviations: cVEMP, cervical vestibular evoked myogenic potential; oVEMP, ocular vestibular evoked myogenic potential; VEMP, vestibular evoked myogenic potential.
a P13 and n23 latencies, respectively, p1 and n1 latencies of all patients (n = 41) are presented.
b Statistically significant results.
Finally, in the results of the VEMP test, no statistically significant difference was determined between patients with primary or adjuvant RCT (P = .45).
Pure Tone Audiogram
The mean PTA of all (n = 41) patients was 22.3 dB before the start of the RCT. It was 24.1 dB 6 weeks after RCT and 23.7 dB 3 months after RCT. These differences were not statistically significant (P = .37 and .41). Also hearing thresholds for 8 kHz did not show significant changes (P = .32). Mean hearing threshold for 8 kHz was 39.3 Hz prior to RCT and 41.2 dB and 40.9 during follow-up tests. Furthermore, there was no statistically significant difference between the results of the patients who received primary or adjuvant RCT (P = .13).
Discussion
Within the scope of this monocentric prospective clinical study, the vestibulotoxic effect of cisplatin-containing combined radio-chemotherapy on the otolith organs as well as on the horizontal semicircular canals was demonstrated objectively and clearly. After RCT, significantly less reproducible oVEMP and cVEMP responses could be performed. In addition, the vHIT measurement showed a significantly reduced gain 6 weeks after RCT and an increased occurrence of saccades.
The tests demonstrated vestibular loss of function after cisplatin-containing RCT, but the exact location and target points of toxicity are still unclear. One possible location is the vestibular end organs themselves. For example, Breglio et al demonstrate that cisplatin is detectable in the human cochlea months or years after ingestion and that the stria vascularis is the site of the highest exposure. 21 Although there is no analogous structure to the vascular stria in the vestibular labyrinth, damage to both locations (the cochlea and the vestibular organs) appears to be possible due to the common blood supply of the auditory and vestibular systems in the inner ear. The outer hair cells and the spiral ganglion cells are also considered damage sites.22,23
Lo et al were also able to demonstrate reduced ATPase activity and increased oxidative stress in the otolith organs in the murine model after cisplatin administration, which also led to changes in oVEMP results. 24 Overall, the inner ear therefore appears likely to be the location of the damage. What is striking here, however, is that the patients included in the present study did not experience a significant drop in average hearing loss. There are several different reasons for this. For example, cisplatin primarily leads to hearing loss in the higher frequencies. This has been demonstrated in particular by long-term results in patients with testicular cancer.25,26 The determination of the PTA and hearing thresholds at 8 kHz as done in the present study may therefore be the wrong parameter for evaluation and represents a limitation of the present study. Higher frequencies should have been evaluated as well, as high frequencies are initially altered. In addition, other objective test methods, such as otoacoustic emissions or combinations of objective and subjective testing make more sense 27 and should have been performed evaluate cochleotoxic effects more sensitively. However, the nonsignificant reduction in hearing in the present study may have other causes. There are indications that cisplatin-induced cochleotoxicity can also be progressive years after taking medication.28,29 Accordingly, the follow-up examinations carried out in this study 6 weeks or 3 months after taking the medication may have not allowed sufficient time to be able to make a definitive statement about the occurrence of possible cochleotoxicity and associated hearing loss. In addition, the anatomical differences between vestibular and cochlear hair cells should be considered. 2 As has already been demonstrated for aminoglycoside antibiotics, these lead to different levels of toxicity. 30 Even if there are morphological indications that cisplatin in the murine model seems to induce a greater cochlear loss than vestibular hair cell loss, 8 further basic research is needed to better understand how the ototoxic mechanisms of cisplatin in the auditory and vestibular systems can be distinguished.
In addition to the peripheral end organs, other locations of the central vestibular system could also be targets of toxicity. Toxicity in the brainstem has already been discussed as a possible cause of vestibulopathy in patients with obstructive sleep apnea or diabetes mellitus.31,32 Toxicity in the brainstem could therefore also be a possible explanation for the results outlined above. On one hand, the vestibular cores could themselves be sensitive to the toxicity induced by cisplatin. On the other hand, damage to the vestibular cores as a result of radiation therapy is also possible. For example, radiogenically induced brainstem lesions or myelitis are known complications in the context of RCT in head and neck tumors, and cognitive and neuropsychological changes also occur.33,34 In order to be able to distinguish whether the damage is primarily caused in the peripheral end organs or in the central vestibular structures such as the vestibular cores, this study would need to be expanded to a patient population that received cisplatin-based chemotherapy but no radiotherapy in the area of the head.
It also seems to be important to determine whether the existence of vestibulotoxicity is influenced by age or gender. In our cohort, we did not identify any statistically significant differences between men and women. In our cohort, we tried to identify if the individual patients—independent of age—show a decline in vestibular function during the 3-month observational period. In order to further analyze the role of age and whether older patients are potentially more susceptible to vestibulo- and cochleotoxic damage by cisplatin, much larger cohorts would be needed in order to identify possible statistically significant differences between certain age groups.
What is striking about our results is that none of the included patients subjectively had dizziness or balance problems in the course of the follow-up examinations. This may have different causes. For example, there was a symmetrical reduction in vestibular function, which is most likely to develop gradually. A gradual, symmetrical loss of function, unlike a 1-sided hypofunction, causes more subtle symptoms such as postural imbalance and imbalance and only with the progress of oscillopsia and dizziness. This symmetrical loss of function can also be obscured by central compensation or compensation by the visual or proprioceptive systems. Initially uncharacteristic symptoms such as imbalance or malaise could also affect other side effects of RCT such as anemia of chronic disease, dehydration, nausea, or vomiting. A limitation of our study in this regard is the fact that dizziness was only assessed once per visit but no standardized questionnaire was completed. A questionnaire such as the Dizziness Handicap Inventory 35 would have helped determine further restrictions on the affected patients.
Finally, our results were able to show that vestibular dysfunction can be caused by RCT-containing cisplatin—both in the adjuvant and in the primary therapy regimen—in patients with head and neck tumors. Therefore, especially with this vulnerable population, not only auditory but also vestibular testing should be carried out for medicolegal reasons.
The new neurotological test methods such as VEMPs and vHIT are suitable for this, as they already enable objective detection of a subclinical hypofunction. However, it is a limitation to our study that we did not include rotatory chair testing, as this would have given us further detailed information about central vestibular compensation mechanisms. Central compensation might explain why we saw a gain reduction in vHIT during first follow-up, which was not reproducible during second follow-up. Another limitation of our study is that we only included vHIT for the lateral semicircular canal and did not also test the anterior and posterior canals. Including Left anterior Right posterior (LARP) and Right anterior Left posterior (RALP) testing would have given us further detailed and precise information about the platinum induced toxicity.
Nevertheless, detection done as early as possible allows for the initiation of vestibular physiotherapy as early as possible. For example, in the context of subsequent follow-up treatments, often administered to inpatients after RCT, bilateral vestibular dysfunctions lead to a considerable reduction in QoL.36,37 In addition, screening before and after RCT also makes sense because falls and mobility restrictions are more common among cancer survivors compared to healthy people in the same age-group.38,39 This applies not only to patients with head and neck tumors but also to other patients such as the pediatric oncological population, to whom special attention should be paid.
Further studies are needed to better understand cisplatin-induced vestibulotoxicity and to identify vestibuloprotective substances that can be given alongside RCT, if necessary. Sodium thiosulfate appears promising here; the cochleoprotective potential of cisplatin has already been shown in children. 40 Until vestibuloprotective therapy has become established, however, there remains early detection and the rapid initiation of structured balance training.
Conclusions
The data presented show that there is a significant change in vestibular function under primary or adjuvant RCT containing cisplatin. The exact location of the damage (central vs peripheral) is still unclear. Future studies are needed to better understand cisplatin-induced vestibulotoxicity and to identify possible vestibuloprotective substances. Nevertheless, before and after cisplatin-containing RCT, patients should not only undergo auditory testing but also undergo objective vestibular testing in order to detect functional losses early and to initiate vestibular physiotherapy as quickly as possible.
Footnotes
Acknowledgements
The authors would like to thank Dr Lena Zaubitzer for assistance in data collection.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
