Abstract
Objectives:
Obstructive sleep apnea (OSA) is a common sleep disorder, which is associated with recurrent oxygen desaturation during sleep. It has already been shown that nocturnal hypoxia may lead to cochlear dysfunction in patients with OSA. Less is known whether hypoxia during sleep also impacts vestibular function in those patients. Thus, the aim of the presented study was to assess a potential vestibulotoxic effect of nightly desaturations with hypoxia in patients with OSA by investigating a possible correlation between respiratory parameters and vestibular function tests.
Methods:
A total of 56 patients were included in the study and underwent a fully attended cardiorespiratory polysomnography (PSG). Vestibular function was assessed using video head impulse test to evaluate horizontal semicircular canal function and cervical vestibular evoked myogenic potentials (cVEMPs) and ocular vestibular evoked myogenic potentials (oVEMPs) to measure otolith function. Descriptive data analysis was conducted and correlation analysis between selected PSG parameters and the results of vestibular testing was performed using Kendall τ coefficient.
Results:
A significant correlation between vestibular function and respiratory polysomnographic parameters could not be demonstrated in the study (P > .05) but cVEMP and oVEMP results showed a trend toward a correlation with oxygen desaturation indices and apnea–hypopnea index. Additionally, otolith hypofunction was more prevalent in patients with hypertension as well as OSA.
Conclusion:
The results of our study show that there is no significant correlation between vestibular function and sleep apnea parameters, although otolith dysfunction might be more prevalent in patients with OSA and hypertension.
Introduction
Obstructive sleep apnea (OSA) is a common sleep disorder with a prevalence of 6% to 13% in adults.1,2 It is characterized by obstruction of the upper respiratory tract during sleep. The nocturnal apnea and hypopnea phases are characterized by a reduction in air flow and/or oxygen saturation mostly in slow wave sleep and rapid eye movement sleep. These events cause repetitive arousals. As a result, sleep is no longer considered restorative and restful, potentially resulting in daytime sleepiness, reduced daytime performance, and reduced sleep-related quality of life. 3 Since the mid-1980s, OSA has been recognized as a risk factor for cardiovascular, cerebrovascular, endocrine, and even psychiatric diseases such as hypertension, stroke, diabetes, and schizophrenia.4-6 The pathogenesis of some OSA-associated diseases likely involves nocturnal repetitive hypoxemia (oxygen desaturation and reoxygenation) resulting from respiratory distress, which consecutively causes activation of the inflammatory cascade, oxidative stress, vascular endothelial dysfunction, and activation of the autonomic nervous system.7,8 Additionally, the vasa nervorum can be damaged by insufficient oxygenation, resulting in neuronal loss in the central and peripheral nervous systems, which can affect various physiological reflex circuits. 9 Initially, acute hypoxemia can be compensated for by various mechanisms, persistent hypoxemia however can trigger a neuropathy. 10 With this regard, it seems possible that nocturnal hypoxia may also affect other sensory systems such as the cochlea or the vestibular system. A possible link between daytime sleepiness, sleep apnea, and dizziness was reported in a recent study, 11 and the effects of oxygen desaturation on the cochlea have already been studied in patients with OSA. For instance, Casale et al confirmed higher thresholds in pure tone audiograms, lower otoacoustic emissions, and prolonged latencies in brainstem audiometry in patients with severe OSA. 12 Less is known about how nightly hypoxia might affect vestibular function, although all 5 vestibular receptors can now be objectively tested using video head impulse test (vHIT) and vestibular evoked myogenic potentials (VEMPs).13,14 Mutlu et al demonstrated that cervical VEMPs (cVEMPs) revealed reduced otolith function in patients with severe OSA compared to healthy volunteers. 15 In addition, patients with moderate to severe OSA scored significantly higher in the Dizziness Handicap Inventory questionnaire, which quantifies the impact of dizziness on quotidian activity by measuring self-perceived handicap, compared to patients with mild OSA.16,17 Nevertheless, a prospective study which performs a receptor-specific testing of all vestibular organs using video head pulse test and VEMP examination in patients with OSA is still missing. Therefore, we performed a prospective study to evaluate vestibular function in patients with OSA.
Methods
This prospective study was conducted at a tertiary referral center and approved by the local ethics committee (2015-626N-MA). In total, 56 patients were recruited from the sleep laboratory. All of the patients presented to the hospital to undergo diagnostic polysomnography (PSG) for suspected sleep-disordered breathing and underwent a fully attended overnight PSG and vestibular function tests. Polysomnography was conducted with Grael and Somté PSG headboxes and Profusion software version 3.0 (Compumedics, Australia) and was evaluated according to the 2012 update of the 2007 procedures and definitions from the American Academy of Sleep Medicine. 18
Polysomnography analysis included the apnea–hypopnea index (AHI), minimal oxygen saturation (minO2), average oxygen saturation (ØO2), 3% and 4% oxygen desaturation indices (ODI3/4%), and sleep time with an oxygen saturation <90% (t90). According to the International Classification of Sleep Disorders, OSA is defined as an AHI ≥5/h in combination with symptoms or comorbidities or an AHI ≥15/h. 19 In general, mild OSA is defined as an AHI of 5 to 14/h; intermediate OSA is defined as an AHI of 15 to 29/h; and severe OSA is defined as an AHI ≥ 30/h. These definitions were also used in the present study.
Participants were excluded if they were blind or had poor range of motion in their neck, which would preclude them from performing the vestibular function tests. Additionally, patients with known vestibular disorders and profound hearing loss were excluded from the study. The vestibular function tests included the horizontal vHIT as well as the cVEMP and ocular VEMPs (oVEMP). For the vHIT, a portable video-oculography goggle system (EyeSeeCam, Munich, Germany) was used. A high-speed infrared camera and a built-in accelerometer were used to measure head and eye movements at a sampling rate of 250 Hz. The vestibular-ocular reflex (VOR) was quantitatively assessed during horizontal head impulses. The target head velocity was 100° to 200°/s with amplitudes between 5° and 15° from the center to the lateral position. The impulses were unpredictable in both directions, and at least 15 impulses were performed for each side. The VOR gain was calculated as the ratio of the eye to head velocity at 40, 60, and 80 ms. Furthermore, the presence of refixation saccades was analyzed.
For cVEMP testing, which was recorded with the patient in a supine position and the head elevated 30° from the horizontal, the stimulus was a 500 Hz, 100 dB SPL tone burst at a repetition rate of 4/s delivered monaurally. Electromyography findings for the oVEMPs from the ipsilateral sternocleidomastoid muscle and from the contralateral inferior oblique muscle were recorded with an EMG system and noninvasive surface electrodes.
Additionally, the demographic data of all patients, including comorbidities, age, gender, weight, and body mass index (BMI), were collected and analyzed. Subsequently, polysomnographic parameters were analyzed and correlated with vestibular function test results using Kendall τ. As vestibular function test results are age dependent,20,21 they were analyzed with regard to existing age-appropriate normative data. Statistical analyses and plotting were performed using R, an open source environment for statistical computing and graphics. 22
Results
Demographic Parameters
The patients’ age ranged from 24 to 80 years (mean age: 56 ± 13 years and median age: 58 years; Figure 1). The age and BMI of patients with OSA did not significantly differ from those of patients without OSA. Most patients with OSA were male (78.6%), which represents a male predominance in the gender distribution of this disease.
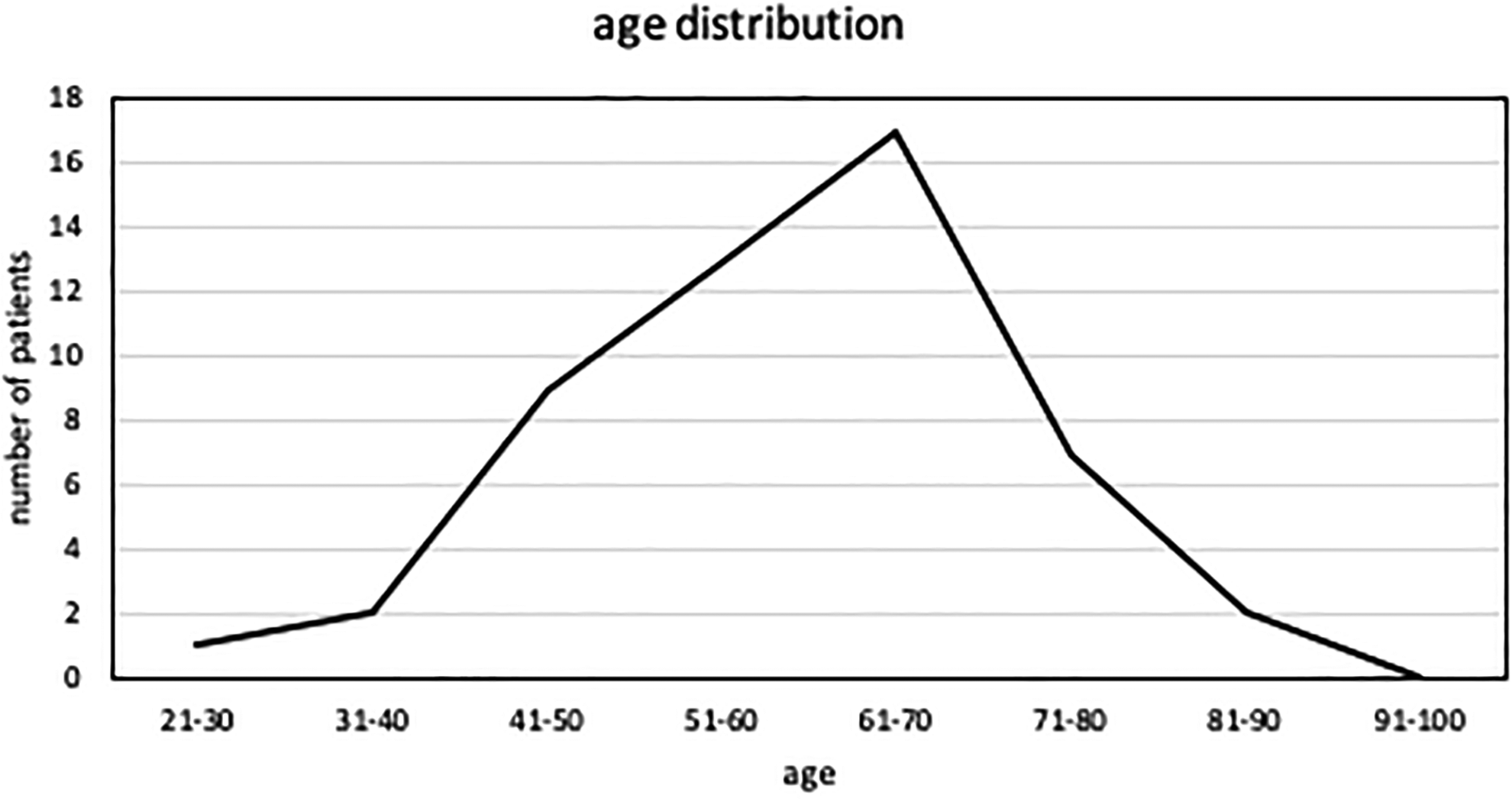
Age distribution of the included patients (age in years).
Age, BMI, and gender are shown in Table 1. There was a higher incidence of diabetes and hypertension in patients with an AHI ≥15 (Table 2). All included patients (especially those with an AHI <15/h) had OSA-defining symptoms or comorbidities according to the definition mentioned earlier. The incidence of diabetes and hypertension in the patients with OSA is displayed in Table 2.
Apnea–Hypopnea Index Displayed in Relation to Gender, Age, and BMI.a
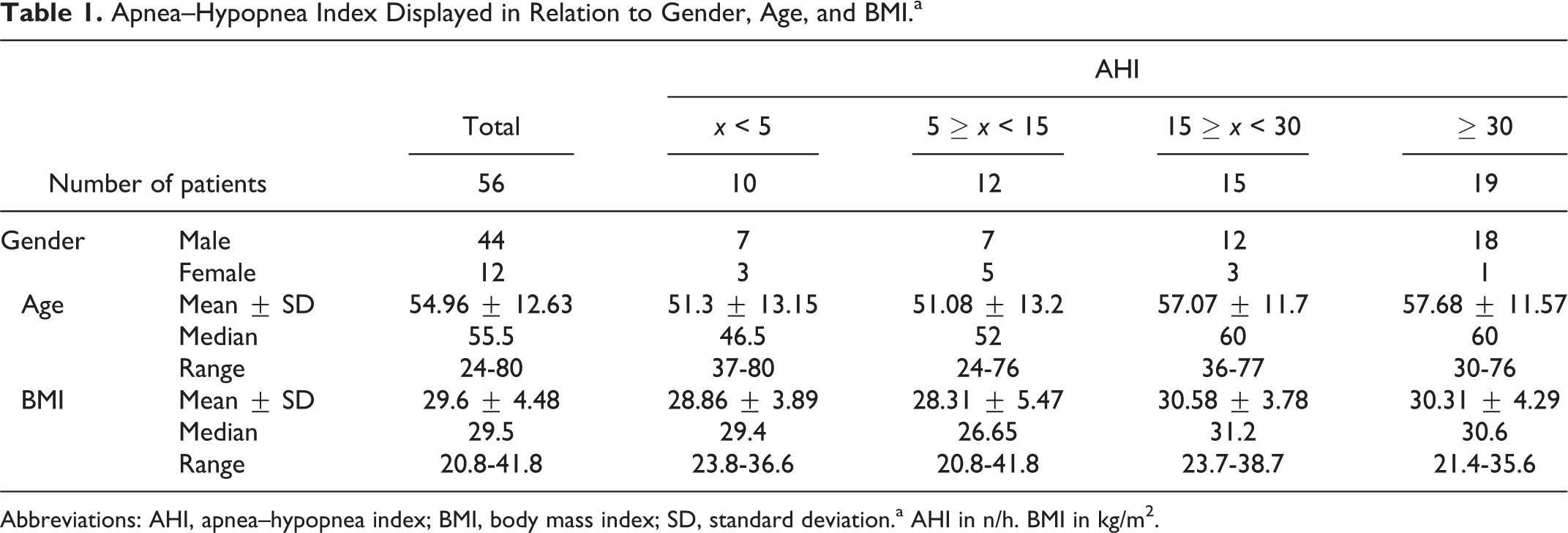
Abbreviations: AHI, apnea–hypopnea index; BMI, body mass index; SD, standard deviation.a AHI in n/h. BMI in kg/m2.
Incidence of Hypertension and Diabetes Mellitus.

Abbreviation: AHI, apnea–hypopnea index.
Polysomnographic Data
Ten patients showed no evidence of OSA (AHI < 5). Obstructive sleep apnea was detected in 46 patients (mild: 12, moderate: 15, and severe: 19). The AHI data are displayed in Table 1.
Video Head Impulse Test Results
The vHIT was performed in 50 of the 56 recruited patients. Six patients (2 with AHI < 5 and 4 with moderate to severe OSA) could not be included due to technical reasons. The results were subsequently assessed as pathological or physiological. A total of 5 cases showed a pathological result (reduced gain) on one side. One of these patients was female, and 4 were male. In 3 patients, bilateral pathological gains were detected. In 2 patients, normal vHIT results were observed when impulses were delivered to the left side, while pathological vHIT results were only observed when impulses were delivered to the right side. Two patients with pathologic vHIT results had an AHI > 30, and one of the patients also had arterial hypertension. None of the patients had diabetes. Further details are displayed in Table 3.
Results of vHIT Testing in the Cohort.a
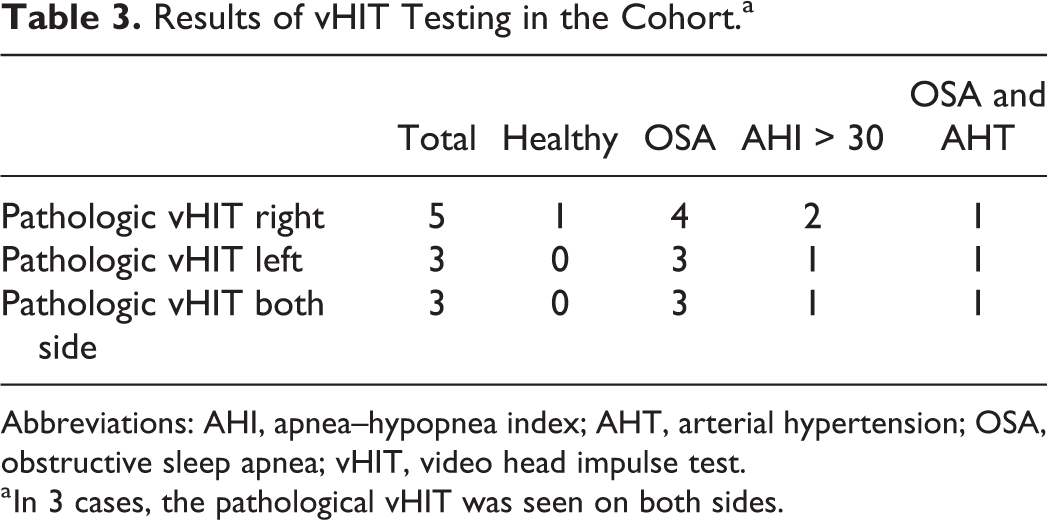
Abbreviations: AHI, apnea–hypopnea index; AHT, arterial hypertension; OSA, obstructive sleep apnea; vHIT, video head impulse test.
a In 3 cases, the pathological vHIT was seen on both sides.
Vestibular Evoked Myogenic Potential Results
The VEMP measurements were possible in 47 patients. A total of 40 of those 47 patients had OSA. Nine patients (3 with an AHI < 5 and 6 with moderate to severe OSA) could not be included due to technical reasons. Reliable cVEMP results were observed in approximately half of the patients, while reliable oVEMP measurements could only be elicited in 10 patients. In 4 patients, both cVEMP and oVEMP results could be consistently assessed. In 17 patients, neither cVEMP nor oVEMP responses could be detected. Details and descriptive data for patients with OSA and comorbidities are shown in Table 4.
Results of VEMP Testing in the Cohort.a
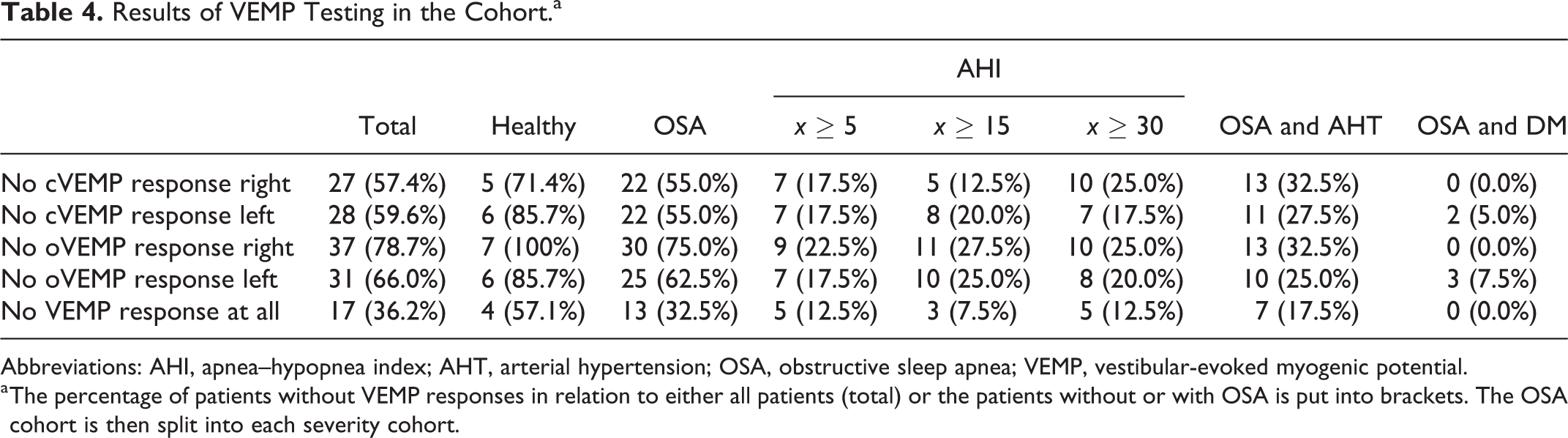
Abbreviations: AHI, apnea–hypopnea index; AHT, arterial hypertension; OSA, obstructive sleep apnea; VEMP, vestibular-evoked myogenic potential.
a The percentage of patients without VEMP responses in relation to either all patients (total) or the patients without or with OSA is put into brackets. The OSA cohort is then split into each severity cohort.
Overall, there was no significant difference between VEMP results in patients with or without OSA. However, with regard to the descriptive indices, there were more pathological VEMP results in patients with OSA and hypertension (Table 4). Table 5 presents further details for patients with pathologic VEMP results. In this group, no significant differences could be observed between patients having different severities of OSA.
Distribution of AHI in the OSA Cohort With Pathologic VEMP Responses.
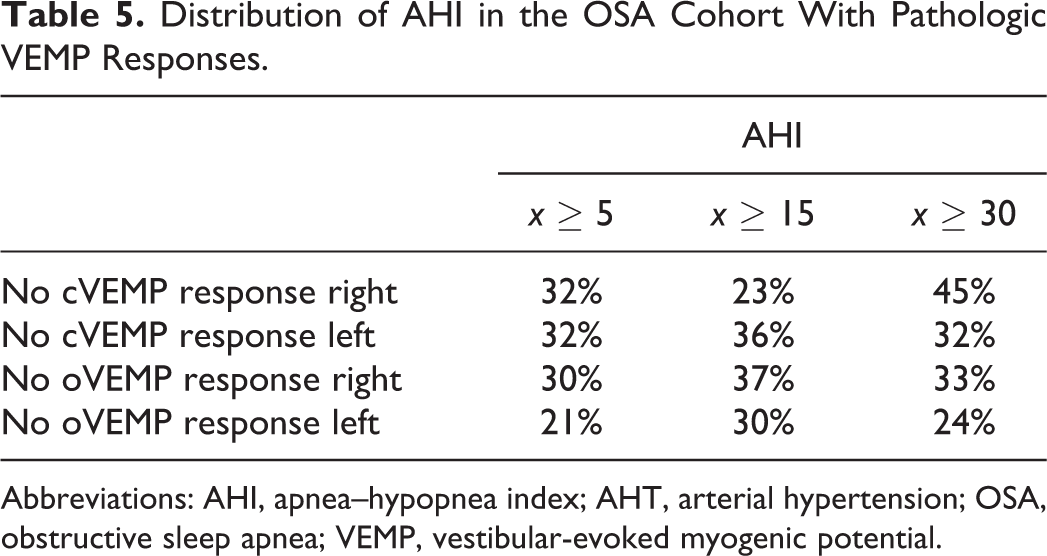
Abbreviations: AHI, apnea–hypopnea index; AHT, arterial hypertension; OSA, obstructive sleep apnea; VEMP, vestibular-evoked myogenic potential.
Kendall τ
Lastly, the correlation analysis between the PSG parameters and the vestibular function tests was performed using Kendall τ. In addition to the OSA-defining AHI, correlation analysis could be performed for minO2, ODI3/4%, t90, and ØO2. Overall, no statistically significant correlation could be determined for the above-mentioned parameters. A tendency toward a statistically significant correlation was observed between cVEMP (left) and AHI (P = .13) and ODI 4% (P = .17) and between oVEMP (right) and AHI (P = .14). The P values for the correlation tests are shown in Table 6.
Correlation Analysis Between PSG Parameters and Vestibular Testing.a
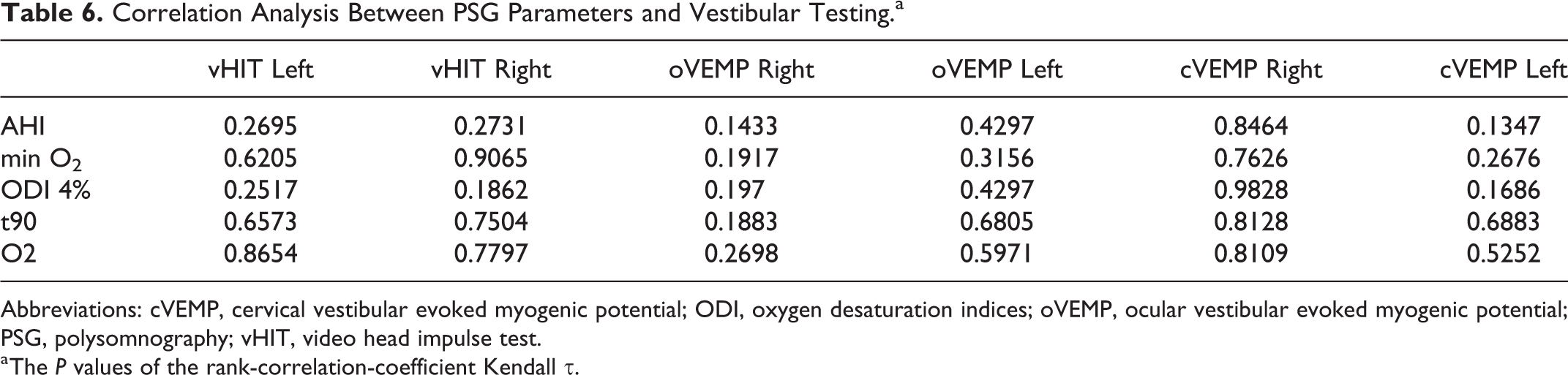
Abbreviations: cVEMP, cervical vestibular evoked myogenic potential; ODI, oxygen desaturation indices; oVEMP, ocular vestibular evoked myogenic potential; PSG, polysomnography; vHIT, video head impulse test.
a The P values of the rank-correlation-coefficient Kendall τ.
Discussion
The results of our study show that there is no statistically significant correlation between vestibular function and respiratory parameters in patients with OSA compared to controls. Nevertheless, our results indicate that there might be a higher prevalence of impaired otolith function in patients with OSA compared to healthy individuals, especially when the patient with OSA is also having arterial hypertension. We were able to identify the potential pathological structure more precisely, as vHIT as well as VEMPs were performed in the individuals. Horizontal semicircular canals did not show any reduced function in patients with OSA, whereas otolith end organs might be affected by nightly oxygen desaturation.
Our findings might have several reasons: nocturnal hypoxia in patients with OSA may cause damage in the following structures: the peripheral vestibular end organs or the peripheral and central vestibular pathways. Looking at the level of the peripheral vestibular end organs, VEMP stimuli are primarily thought to be transduced by type 1 vestibular hair cells. 23 Ototoxic medication, such as gentamycin, or metabolic disturbances, such as hyperglycemia in patients with diabetes mellitus, 24 also seem to have stronger toxic effects in vestibular hair cells type I. 25 Additionally, it is also known that those cells are more likely to suffer from age-related degeneration. 26 In total, vestibular hair cells type I seem to be more vulnerable to toxic effects, including metabolic disturbances and vestibulotoxic medication. Therefore, it seems conceivable that nocturnal oxygen deprivation in patients with OSA in combination with elevated blood pressure also may lead to altered vestibular hair cell function.
Another potential explanation for our findings might be that neurovascular damage to the vestibular nerve might cause delayed or absent VEMP responses. Untreated nightly oxygen desaturations favor the development of neuropathies, 10 which has been described for several sensory neuronal systems. In the human vision system, for example, patients with OSA have a compromised oxygenation of the optic nerve, which may lead to glaucomatous optic neuropathy and changes in retinal microstructures. Moreover, it has recently been shown by Lin et al that successful surgical treatment improves these ophthalmologic findings in patients with OSA. 27 As a result, the vestibular nerves themselves, as locations for a potential vestibular dysfunction in patients with OSA, should be also taken into consideration. Interestingly, the utricle and the superior and horizontal semicircular canals share the superior divisions of the vestibular nerve trunk and therefore a common vascular supply, whereas the posterior semicircular canal as well as the saccule share a different supply and the inferior part of the vestibular nerve. 28 This might explain why our results show normal horizontal semicircular canal function but altered otolith function, since the saccule and the horizontal semicircular canal differ in their vascular supply and the superior and inferior divisions of the vestibular nerve may have different risks of ischemia or small vessel disease. As hypertension is an independent risk factor for cerebral small vessel disease,29,30 it might be conceivable that especially the combination of comprised oxygenation in patients with OSA and hypertension causes neurovascular damage to the vestibular nerve.
As noted above, nightly hypoxia and hypertension might lead to otolith hypofunction due to several factors: one potential site of damage might be the peripheral vestibular end organs themselves or the vestibular nerves. Yet a potential site of damage in the brain stem is also imaginable. Vestibular evoked myogenic potentials have been increasingly described as a sensitive and noninvasive method to evaluate brain stem function.
31
It has been asserted, for example, that patients with multiple sclerosis might present a prolongation of latencies or reduced wave amplitudes and that VEMP testing can be a sensitive tool for the evaluation of brain stem involvement in multiple sclerosis.32-34 Therefore, altered VEMP results in patients with OSA might be due to neuronal degeneration in the brain, as it has already been described that apnea produces neuronal degeneration in the pons and medulla of guinea pigs.
9
In turn, these results from the animal model might be transferable to humans and ground an explanation for our VEMP findings. Interestingly, Degache et al could demonstrate that daytime postural stability is influenced by nocturnal breathing disorders.
35
The authors could, inter alia, prove an association between mean nocturnal Sp
In sum, though we did not find any statistically significant correlation between vestibular function and sleep apnea parameters, otolith dysfunction seems to be more prevalent in patients with OSA and arterial hypertension. This might be caused by above-mentioned mechanism, nevertheless the role of arterial hypertension still needs to be further clarified. There is still limited data of how arterial hypertension affects vestibular function36,37 mainly because of the reason that many patients are not only having hypertension alone but also having other cardiovascular risk factors (eg, diabetes, hyperlipidemia) as well and therefore the risk factors and their impact of vestibular function cannot be easily evaluated separately. To further investigate our findings, larger sample sizes are needed to verify our results. Ideally, patients with severe OSA but no other cardiovascular comorbidities would be compared to patients with severe OSA and hypertension and to patients with only hypertension but not OSA to also further clarify the role of hypertension.
Our study has limitations as among other things our sample size was limited to 57 patients. One reason is that diagnostic PSGs are performed less frequent in our sleep laboratory and in sleep laboratories in common, as the role of portable polygraphy monitoring for confirming OSA is recognized more widely. Future studies with larger sample sizes are needed to verify our results and further localize which structure of the vestibular system is affected most by nocturnal hypoxia and which other cardiovascular risk factor might also be involved in causing otolith dysfunction. For this future studies, portable polygraphy monitoring systems might be used to further increase the number of patients tested. In addition, our study has a strong selection bias as we do not have included an appropriate aged-matched control of healthy volunteers. All patients recruited for the presented study came to our sleeping laboratory with a complaint of disruptive sleep and/or snoring. An age-matched control of healthy volunteers would have minimized potential selection bias and would have possibly provided further inside and stronger results.
Conclusion
The results of our study show that there is no significant correlation between vestibular function and sleep apnea parameters, though otolith dysfunction might be more prevalent in patients with OSA and hypertension.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
