Abstract
Introduction
Vestibular migraine (VM) causes repeated dizziness in people with a history of migraines. VM is one of the most common causes of recurrent dizziness in the general population. Diagnostic criteria for VM have been established by the International Headache Society and the International Bárány Society in a consensus document.1,2 According to these criteria, VM is characterized by experiencing a minimum of 5 episodes of vestibular symptoms, a current or previous history of migraine with or without aura, and 1 or more migraine features present in at least half of vestibular episodes. And these symptoms cannot be explained by a different vestibular diagnosis or a diagnosis according to the International Classification of Headache Disorders.
Vertigo is a stressful experience that may trigger fear, phobia, and other psychiatric disorders. There have been reports that there are high rates of comorbidity, ranging from 30% to 50%, between vestibular and psychiatric syndromes.3,4 VM is known to accompany many psychiatric disorders, including anxiety and depression. 5 Patients with vestibular symptoms also had three-fold increased odds of depression and anxiety compared to other U.S. adults. 6 Anxiety and depression were more frequent in patients with VM than in those with other types of peripheral vertigo, such as vestibular neuritis (VN) and benign paroxysmal positional vertigo (BPPV). 7 In another investigation, VM patients also showed significantly higher dizziness handicap and depressive symptoms contrasted to other vestibulopathies, including BPPV and VN, which is in concordance with the aforementioned study results. 8
An association between vestibular dysfunction and VM has been proposed and actively investigated although the association between VM and vestibular function remains uncertain.9,10 The caloric test is a useful clinical tool to identify and quantify peripheral vestibular dysfunction. One study revealed abnormal caloric test results in 22% of VM patients, 11 while another revealed in 42% of VM patients. 12 Abnormal vestibular function for low-frequency stimuli tested by caloric testing was frequent in VM, suggesting a substantially decreasing low-frequency canal function in VM. Although caloric testing has been considered the gold standard method for demonstrating vestibular hypofunction, it has some limitations. Caloric irrigation produces unilateral, low-frequency stimulation and provides a nonphysiological stimulation of the peripheral vestibular organ. Additionally, it can only assess the functioning of the horizontal semicircular canal and of the superior vestibular nerve. On the other hand, the video head impulse test (vHIT) evaluates the vestibular ocular reflex in response to high-frequency head movements. Moreover, it can measure the functioning of all 6 semicircular canals. When compared to caloric testing, it has been shown that vHIT has high specificity (90%-100%) but low sensitivity (34%-56%) in several studies with different vestibular disorders.13,14 The combination of caloric test and vHIT can more systematically evaluate vestibular function.
Since there are no pathognomonic physical examinations or laboratory assessments available, the diagnosis of VM relies on the clinical history and the exclusion of another vestibular diagnosis to better account for the presentation. Migraine features, including photophobia, phonophobia, and visual aura during vestibular episodes, are characteristic clinical symptoms of VM patients. Each of these migraine features is reported to be accompanied by varying frequencies, ranging from 25% to 75%, in VM patients.15,16 Additionally, transient otologic symptoms, including hearing loss, tinnitus, and ear fullness, may occur in VM, but these symptoms do not progress. 17 VM patients may present with various migraine features and otologic manifestations accompanied by headache and vestibular symptoms. Therefore, to increase the understanding of psychiatric disorders that may accompany VM, it is necessary to analyze whether they are related to these various symptoms.
Anxiety and depression are the most relevant psychiatric comorbidities associated with vertigo and migraine, greatly influencing the degree of disease, prognosis, and clinical outcomes. In the present study, we estimated the prevalence of anxiety and depression symptoms, as assessed by the Hospital Anxiety and Depression Scale (HADS), in patients with VM. We have also tried to identify the contributing factors, including the association of vestibular dysfunction with anxiety and depression in patients with VM.
Materials and Methods
We conducted a retrospective chart review of patients aged ≥18 years who visited the vertigo clinic in the Kangwon National University Hospital for the first time from June 2018 to July 2020. All patients underwent a standard clinical oto-neurological examination and audio-vestibular testing, including oto-endoscopy, pure-tone audiometry, tympanometry, finger-to-nose test, rapid alternating movements tests, the examination of spontaneous nystagmus, Dix–Hallpike maneuver, supine-roll test, vHIT, and caloric testing on the day of the visit. The charts were reviewed for the following information: sex, age, vestibular and migraine features of VM attack, brain magnetic resonance imaging (MRI) or computed tomography, clinical oto-neurological examination, and audio-vestibular testing.
The inclusion criteria were the following: (1) patients with a diagnosis of VM according to the criteria of the Bárány Society and International Headache Society1,2 and (2) patients who were measured using the Korean versions of the Dizziness Handicap Inventory (DHI) and HADS at the first visit. The Korean version of these scales is a translation of the original English sentences into Korean to allow patients to understand the questions. The Korean version of these scales is a valid and reliable instrument.18,19
The exclusion criteria were the following: (1) patients with another diagnosis related to vestibular symptoms, for example, VN, BPPV, superior semicircular canal dehiscence, vestibular schwannoma, and Menière’s disease, (2) patients with external or middle ear diseases including conductive hearing loss (CHL) and previous ear surgery, (3) intake of sedating drugs including benzodiazepines and barbiturates, and (4) a history of psychiatric disorders or psychological treatments.
Hospital Anxiety and Depression Scale
The HADS is a self-assessment tool consisting of 14 items that screen for depression and anxiety disorders in nonpsychiatric hospital outpatients. 20 The HADS is a valuable screening test for general psychiatric distress in anxiety and depression in patients experiencing dizziness. 21 The HADS comprises two 7-item subscales, the HADS subscales for Anxiety (HADS-A) and Depression (HADS-D), which are rated on a 4-point Likert-type (from 0 to 3) scale. The scores in each subscale are computed by summing the corresponding items, with maximum scores of 21 for each subscale. A score of 8 or higher on both subscales was found to have an optimal balance between sensitivity and specificity.22,23 A score of 0 to 7 is considered normal, and 8 or above as having clinically relevant anxiety or depression.
Intensity of Vestibular Symptoms
The Korean versions of DHI was used to measure the intensity of vestibular symptoms. The DHI is a 25-item self-assessment questionnaire used to evaluate self-perceived handicaps in patients with dizziness. 24 Each statement is scored using a point system of “yes” (4 points), “sometimes” (2 points), or “no” (0 points). This test is brief, easy to score, and yields quantitative data. The DHI has demonstrated good validity and high test-retest reliability and correlates well. Patients were categorized based on their total score, with a score of 0 to 30 indicating a mild handicap, a score of 31 to 60 indicating a moderate handicap, and a score greater than 60 indicating a severe handicap.25,26 The intensity of vestibular symptoms is rated “moderate” when they have a moderate handicap, and “severe” if patients have a severe handicap.
Spontaneous Nystagmus
Spontaneous nystagmus was assessed using a videonystagmography (VNG) system (SLMED, Seoul, Republic of Korea) in the sitting position. The examination was performed with and without fixation, while the subjects attempted to maintain their gaze straight ahead. The average of 10 representative slow phases was used to quantify the slow phase velocity (SPV) of nystagmus. Nystagmus was considered insignificant if the SPV was less than 3°/s.
Video Head Impulse Test
We assessed the horizontal semicircular canal functions using a video-oculography system (SLMED, Seoul, Republic of Korea). The patients were positioned 1 m away from the target at the level of their eyes and seated in an adjustable chair to optimize examination. The examination was conducted by an experienced examiner and manually performed more than 20 times (head rotation 15-20°, duration 150-200 ms, peak velocity >150°/s) on both sides of each plane. Normal horizontal vHIT was defined as having a gain of ≤2 standard deviation (SD) of the age-matched normal gain reference range and no fixation catch-up saccades.27,28
Caloric Test
The binocular video-based system (SLVNG, SLMed, Seoul, Republic of Korea) was used to conduct a bithermal caloric test while the patient was lying in the supine position with 30° of head flexion. Water at temperatures of 30°C and 44°C was administered separately to each ear canal for 30 seconds, with a 5-minute pause between irrigations. The caloric result was defined as the sum of the SPV of nystagmus induced by both warm and cold irrigations. Canal paresis was defined as ≥25% asymmetry based on Jongkees’ formula. 29 Bilateral canal hypofunction was defined based on caloric responses of <5 degrees/second in both ears. 30
Peripheral Vestibular Dysfunction
The peripheral vestibular dysfunction was diagnosed when at least one of these criteria met: (1) direction-fixed, horizontal spontaneous nystagmus and >50% decrease of SPV of spontaneous nystagmus with fixation, (2) caloric canal paresis, and (3) an abnormal horizontal vHIT result.31,32
Pure-Tone Audiometry
The MADSEN Astera audiometer running OTOsuite software (GN Otometrics, Taastrup, Denmark) was used to assess the pure-tone air (0.25-8 kHz) and bone (0.25-4 kHz) conduction thresholds in a double-walled soundproof room. Low- to medium-frequency sensorineural hearing loss (SNHL) is defined as a bone conduction threshold increase of at least 30 dB HL (Hearing level) at each of two contiguous frequencies below 2000 Hz, which are worse in the affected ear compared to the contralateral ear. For cases with bilateral low- to medium-frequency SNHL, the bone conduction thresholds must be 35 dB HL or higher at each of 2 contiguous frequencies below 2000 Hz. 33 If vestibular episodes are accompanied by low-to-medium-frequency SNHL, the possibility of Menière’s disease should be considered.
Statistical Analysis
All statistical analyses were conducted using SPSS 21.0 (SPSS Inc., Chicago, IL, USA). Means with SD and frequencies with percentages are presented. Statistical analysis was conducted using a chi-squared test for categorical variables and an unpaired t-test for continuous variables. The normality test used the Shapiro–Wilk test. Univariate logistic regression analyses were conducted to identify the association of each independent variable with anxiety and depression. Variables with a P-value less than 0.1 by univariate logistic regression were included in the multivariate analysis to determine the effect of each independent variable on anxiety and depression. We performed a backward stepwise method when conducting the multivariate analysis. The fit of the multivariate logistic regression model in this study was verified by the Hosmer–Lemeshow test. A P-value <0.05 was considered statistically significant.
Ethical Approval
The study protocol was approved by the Institutional Review Board of Kangwon National University Hospital (IRB A-2020-08-012-001). Informed consent was waived due to the retrospective nature of this study.
Results
Demographic and Clinical Characteristics of VM Patients
Table 1 shows the demographic and clinical characteristics of VM patients. A total of 74 patients with VM were enrolled in this study (12 males, 62 females; mean age: 45.12 years, SD: 11.75, range: 23-65 years). The mean HADS score was 13.08 (SD: 5.77; range: 1-24). The mean HADS-A was 7.12 (SD: 3.29; range: 0-14), and the mean HADS-D was 5.96 (SD: 3.33; range: 0-14). The mean duration of vestibular symptoms was 10.69 hours. The average number of vestibular episodes was 11.57. Twenty-two patients had severe intensity vestibular symptoms, and 52 had moderate intensity symptoms. During VM attacks, the majority experienced headaches (79.7%). Photophobia accompanied VM episodes in 55.4% of patients, 40.5% reported phonophobia, and 21% reported visual aura. The most frequent aural symptom was ear fullness (16.2%), followed by tinnitus (13.5%) and hearing loss (8.1%). None of the patients presented with low-mid-frequency SNHL, CHL, or mixed hearing loss, except for 3 patients with both-sided high-frequency SNHL. Two patients had direction-fixed, horizontal spontaneous nystagmus to the left, and 9 patients showed abnormal vHIT results. Twenty-three patients had unilateral canal paresis, and 16 of these patients have canal paresis on the left side. No patients showed bilateral canal hypofunction. Overall, 25 patients were diagnosed with peripheral vestibular dysfunction. Twenty-two patients underwent brain MRI, there were no abnormal MRI findings in those patients.
Demographic and clinical characteristics of patients with vestibular migraine.

Values are presented as the mean ± standard deviation and frequencies (percentages).
Abbreviations: HADS, Hospital Anxiety and Depression Scale; HADS-A, anxiety subscale of HADS; HADS-D, depression subscale of HADS; No., number; vHIT, video head impulse test.
Comparison of Demographic and Clinical Characteristics of Psychiatric Distress Experienced by VM Patients
Table 2 compares the demographic and clinical characteristics of the psychiatric distress of VM patients. Thirty-six (48.6%) patients demonstrated clinically relevant anxiety levels, and 24 (32.4%) demonstrated depression. There were no statistical differences in demographic and clinical characteristics, including sex ratio, mean age, duration of vestibular symptoms, and migraine features during VM attacks, between the anxiety and non-anxiety groups. There were significantly more VM patients with vestibular dysfunction, frequent VM episodes, and severe intensity vestibular symptoms in the anxiety group than in the non-anxiety group (P < 0.05). The depression group showed no statistical differences in demographic and clinical characteristics, including sex ratio, mean age, duration of vestibular symptoms, migraine features during VM attacks, and episode frequencies of VM. There were significantly more VM patients with vestibular dysfunction, and severe intensity vestibular symptoms in the depression group than in the non-depression group (P < 0.05).
Comparison of demographic and clinical characteristics of the psychiatric distress experienced by patients with vestibular migraine.
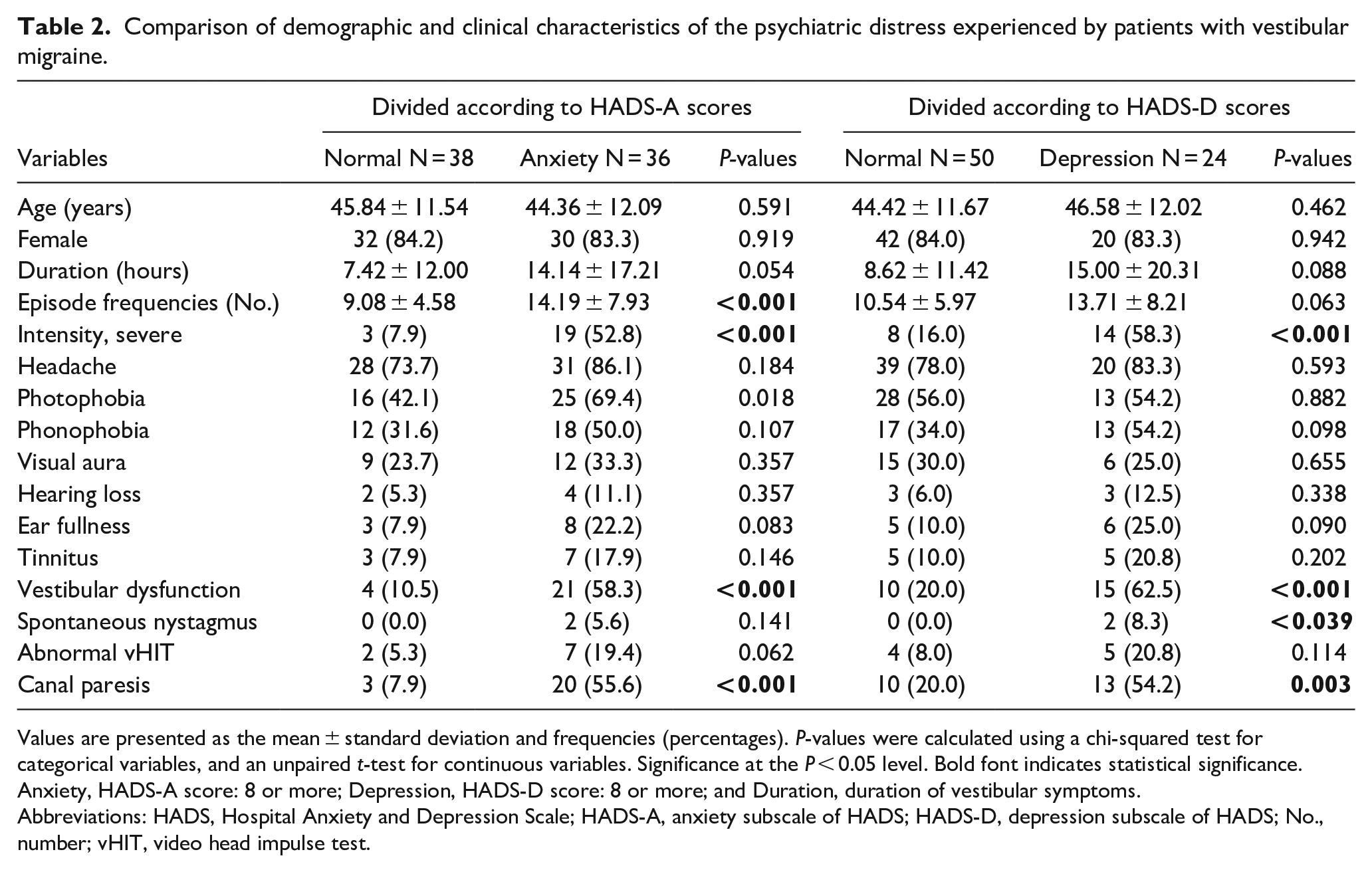
Values are presented as the mean ± standard deviation and frequencies (percentages). P-values were calculated using a chi-squared test for categorical variables, and an unpaired t-test for continuous variables. Significance at the P < 0.05 level. Bold font indicates statistical significance. Anxiety, HADS-A score: 8 or more; Depression, HADS-D score: 8 or more; and Duration, duration of vestibular symptoms.
Abbreviations: HADS, Hospital Anxiety and Depression Scale; HADS-A, anxiety subscale of HADS; HADS-D, depression subscale of HADS; No., number; vHIT, video head impulse test.
Contributing Factors Associated with Anxiety and Depression in VM Patients
This study used univariate and multivariate logistic regression analyses to determine the factors that contribute to anxiety and depression in VM patients. In univariate logistic regression analyses, duration of vestibular symptoms, episode frequencies, severe intensity vestibular symptoms, photophobia, ear fullness, and vestibular dysfunction were associated with more anxiety at a significance level of P < 0.1. In the multivariate model (Table 3), vestibular dysfunction [odd’s ratio (OR) = 6.33, 95% confidence interval (CI) = 1.45-27.56], severe intensity vestibular features (OR = 6.62, 95% CI = 1.36-32.31), and frequencies of VM attack (OR = 1.13, 95% CI = 1.01-1.27) were significantly associated with anxiety. Table 4 shows the findings from logistic regression analysis on the determinants of depression. In univariate logistic regression analyses, episode frequencies, severe intensity vestibular symptoms, ear fullness, and vestibular dysfunction were associated with more depression at a significance level of P < 0.1. In the multivariate model, severe intensity vestibular features (OR = 4.31, 95% CI = 1.28-14.54) and vestibular dysfunction (OR = 3.87, 95% CI = 1.18-12.71) were significantly associated with depression.
Logistic regression analysis of determinants of anxiety in patients with vestibular migraine.
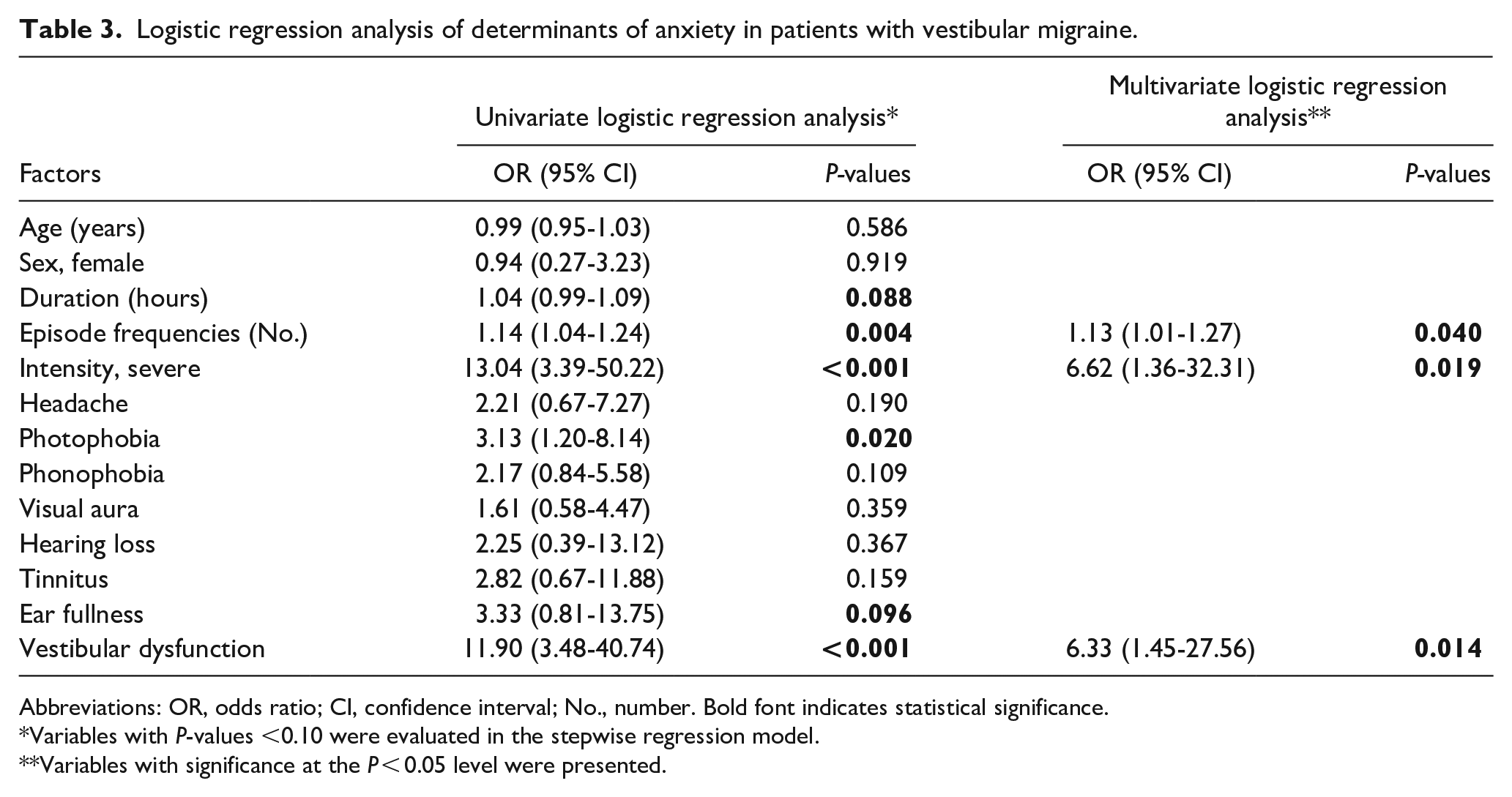
Abbreviations: OR, odds ratio; CI, confidence interval; No., number. Bold font indicates statistical significance.
Variables with P-values <0.10 were evaluated in the stepwise regression model.
Variables with significance at the P < 0.05 level were presented.
Logistic regression analysis of determinants of depression in patients with vestibular migraine.
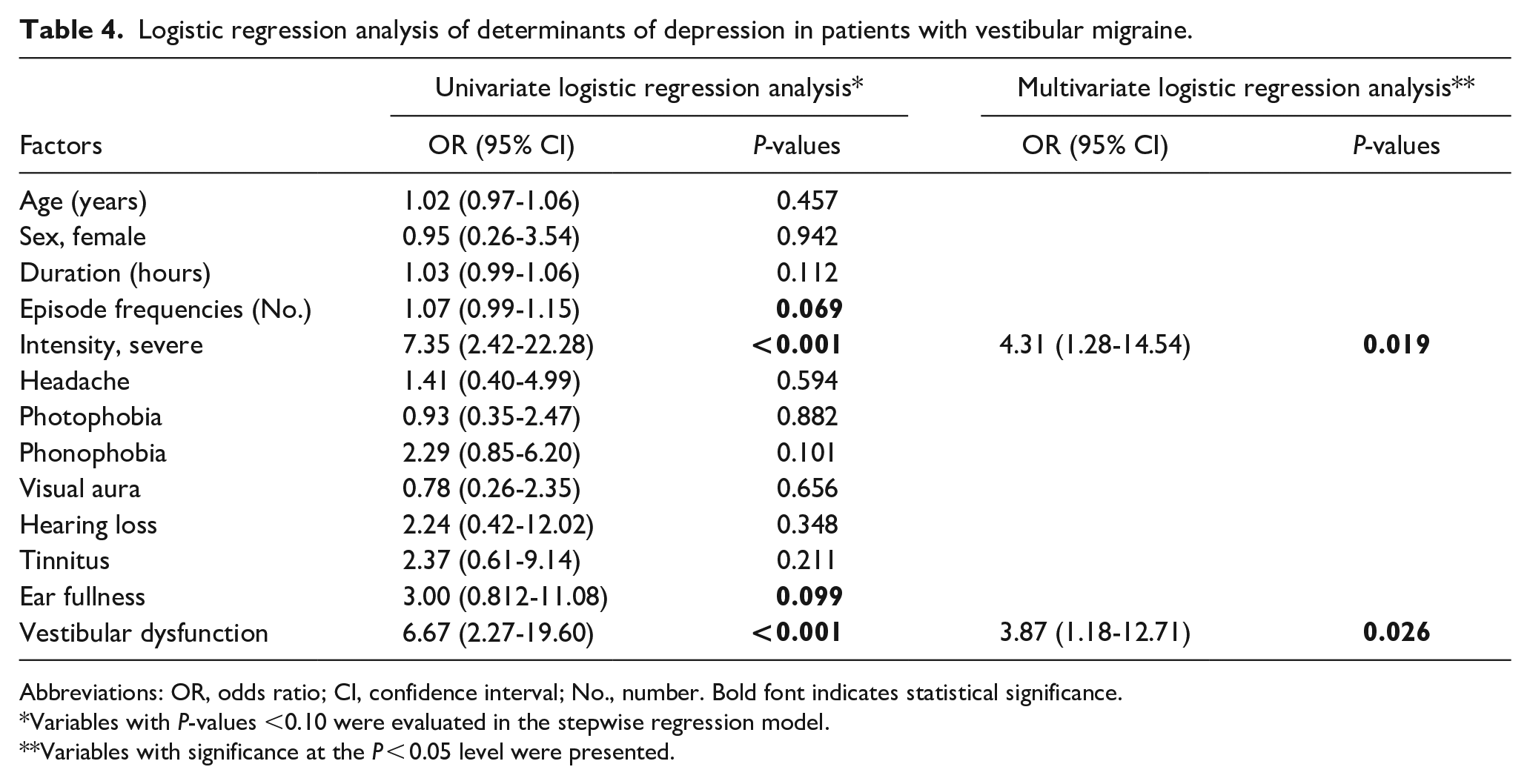
Abbreviations: OR, odds ratio; CI, confidence interval; No., number. Bold font indicates statistical significance.
Variables with P-values <0.10 were evaluated in the stepwise regression model.
Variables with significance at the P < 0.05 level were presented.
Discussion
In this study, we analyzed 74 patients with VM and found that peripheral vestibular dysfunction was a contributing factor influencing anxiety and depression. The main results are as follows: (1) The prevalence of anxiety in patients with VM is considerably higher than in the depression. (2) There were more VM patients with vestibular dysfunction and severe intensity vestibular symptoms in the group with greater anxiety and depression scores. (3) Multivariate analyses by logistic regression model show that peripheral vestibular dysfunction and severe intensity vestibular symptoms contribute to anxiety and depression in VM patients.
The HADS is commonly used to assess emotional distress in nonpsychiatric clinics. Several studies have found that the HADS score is higher in patients with vestibular disorders.22,34,35 There is a high correlation between the dizziness handicap and HADS scores in patients with dizziness. 34 In the present study, the prevalence of anxiety in patients with VM is considerably higher than in the depression. In previous studies, vestibular disorders were mainly related to anxiety rather than depression.3,5,7,34 Yuan et al 7 found that the prevalence of anxiety (45.9%) was significantly higher than that of depression (27%) in VM patients. Depression was not significantly different between VM and migraine patients, but VM patients were more anxious. 5 Therefore, the relationship between vertigo and anxiety might be more pronounced in patients with VM than in those with depression.
Caloric testing is one of the most commonly used methods to identify peripheral vestibular dysfunction by assessing the function of the horizontal semicircular canal and the superior vestibular nerve. Peripheral vestibular dysfunction and abnormal caloric results in VM patients increased in the anxiety and depression groups. Kang et al 36 previously showed that 19% of patients with VM exhibited abnormal caloric responses and showed that the abnormal caloric group was more likely to require continued medical treatment at 6 months than patients with normal caloric results. However, they did not comment on any specific reasons for symptom control. Here, we found a significant difference in patient-reported anxiety and depression between patients with normal and abnormal caloric results.
While the exact pathophysiology of VM remains unclear, electrophysiological and neuroimaging studies have shown that multisensory integration occurs at the vestibular thalamus in VM. 37 The convergence of sensory modalities at the cortical level, combined with sensitization at the thalamic level, may contribute to the perceptual hypersensitivity commonly observed in VM patients. 38 Thus, VM can be described as central hypersensitization and aberrant sensory integration in terms of vestibulo-thalamic-cortical processing. Anxiety and depression play a central role in dizziness symptoms in patients with VM. 39 The vestibular system has reciprocal connections with various nodes of the multi-local psychiatric system and can affect cognition and emotional regulation in animals and humans. 40 This intricate vestibular-psychiatric circuitry involves thalamocortical and cerebellar networks, and interaction within the circuitry is mediated by intrinsic neurotransmitters of the thalamocortical and limbic pathways. 41 Anxiety and depression also exacerbate autonomic symptoms, such as fear and avoidance of activities caused by vestibular dysfunction. Increased autonomic symptoms interfere with compensation and prolong dizziness symptoms. 35 Although the pathophysiology related to VM has not yet been elucidated, it seems likely that peripheral vestibular dysfunction exacerbates anxiety and depression in VM patients.
Our study has several limitations. First, anxiety and depression were assessed through self-reported questionnaires rather than structured interviews based on appropriate diagnostic processes. More comprehensive psychiatric interviews based on the Diagnostic and Statistical Manual of Mental Disorders (DSM-5) and the International Statistical Classification of Diseases and Related Health Problems (ICD-10) criteria should be added to future studies. Second, this study was designed using a cross-sectional approach, and follow-up studies of patients with VM are required to verify these results. Follow-up studies after medication would yield insights into whether psychological tendencies, such as anxiety and depression, remained after the recovery of peripheral vestibular function. In follow-up studies, increasing the number of enrolled patients and analyzing gender differences would be necessary. Lastly, due to its retrospective nature, the functioning of the vertical semicircular canal through vHIT and otolith organs through vestibular evoked myogenic potentials (VEMP) could not be analyzed. In addition to assessing the horizontal semicircular canal, evaluating the function of the vertical semicircular canal through vHIT provides relevant information on vestibular function. VEMP is an effective tool specifically used for assessing the otolith organs (saccule and utricle). Future prospective research is needed to evaluate the associations between psychiatric problems and peripheral vestibular functions, including vHIT of the vertical semicircular canal and VEMP, in VM patients.
Conclusion
Our study showed that peripheral vestibular dysfunction and severe intensity vestibular symptoms contributed to anxiety and depression in VM patients. These results show that peripheral vestibular dysfunction may affect the vestibular-psychiatric circuitry in patients with VM. The vestibular-psychiatric circuitry plays a substantial role in patients’ emotional state and dizziness status with VM. VM patients with peripheral vestibular dysfunction are even more susceptible to anxiety and depression. Therefore, screening for peripheral vestibular dysfunction and psychiatric disorder in VM patients should be considered in a timely manner.
Footnotes
Acknowledgements
Data Availability
The data that support the findings are available from the corresponding author, upon reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a 2019 Research Grant from Kangwon National University and a National Research Foundation of Korea (NRF) grant funded by the Korean government (MSIT) (No.2017R1C1B5076065). The funders had no role in the study design, data collection or analysis, decision to publish, or article preparation.
