Abstract

A 70-year-old male with no significant past medical history presented with a large fluctuating right neck mass present for the previous 4 months. The mass initially improved with a 10-day course of Augmentin and Bactrim but began to grow again slowly over the following number of months. The patient reported 7/10 neck pain, difficulty breathing while supine, dysphagia, and voice changes.
Contrast enhanced computed tomography (CT) of the neck revealed a 7.6-cm rim-enhancing area of cystic necrosis within a lesion of the right thyroid bed. This demonstrated extracapsular extension involving the infrahyoid strap muscles but not the overlying skin (Figure 1A). An additional hypodense nodule was also noted in the left thyroid lobe. The laryngeal skeleton and trachea remained patent with possible cricoid invasion, although there was mass effect on the trachea. No central or lateral compartment lymph node disease was identified. Positron emission tomography-CT demonstrated a hypermetabolic right thyroid mass with an standardized uptake value (SUV) max of 13.0 (Figure 1B). Numerous bilateral hypermetabolic cervical lymph nodes were seen in level IIb and right level IIa. There were also multiple hypermetabolic left internal mammary chain, bilateral axilla, mediastinal, and bilateral hilar lymph nodes concerning for metastases. Chest X-ray did not demonstrate concerning features (Figure 1C), but CT of the chest noted a number of pulmonary nodules, some with tree bud morphology, but several solid pulmonary nodules as well, suggestive of pulmonary metastasis. Flexible laryngoscopy did not demonstrate any abnormalities, revealing a patent airway with bilaterally mobile and symmetric vocal cords.
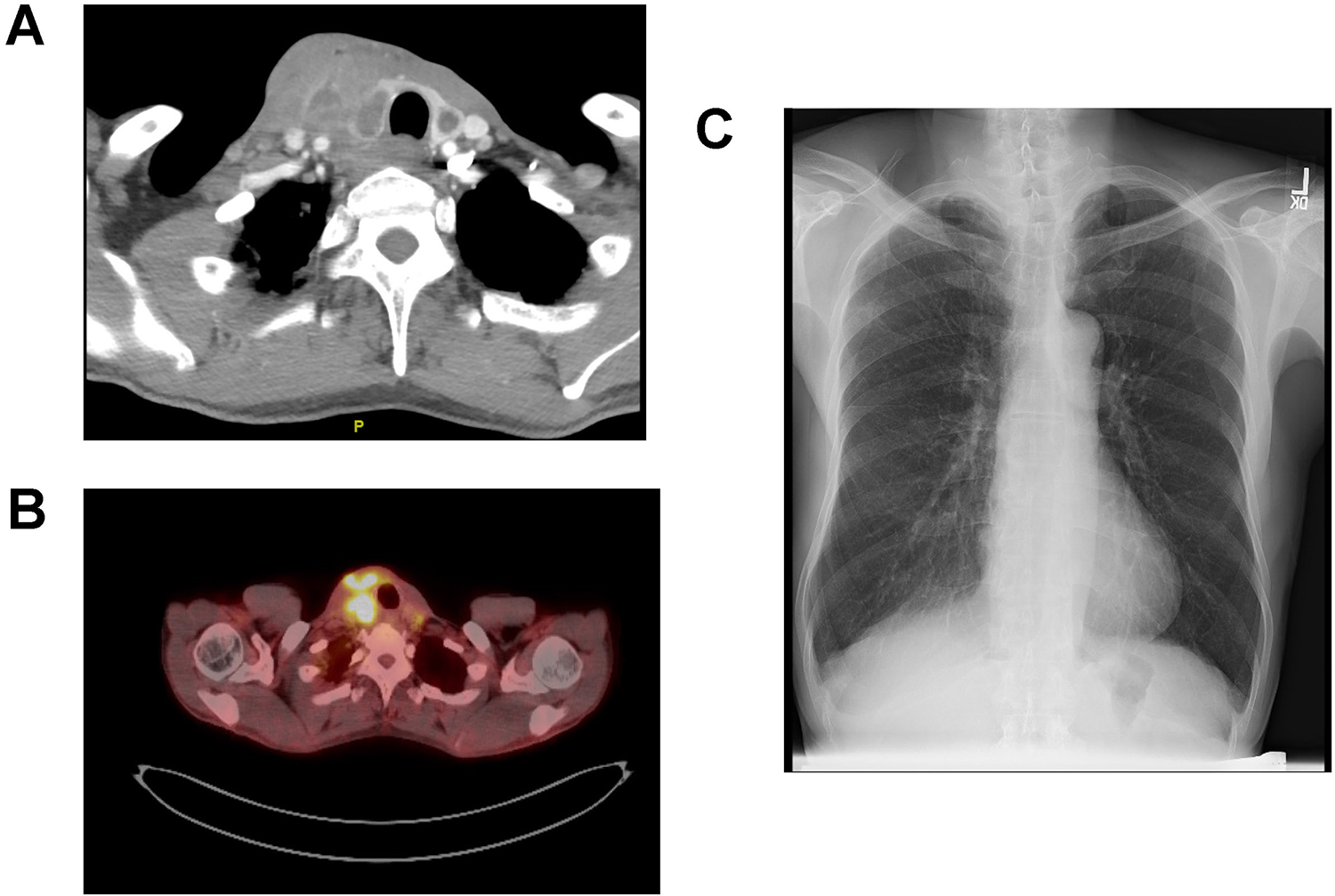
A, CT of the neck with contrast demonstrating the thyroid lesion with areas of necrosis in the right thyroid lobe in addition to areas of hypodensity in the left thyroid lobe. B, PET-CT showing hypermetabolic activity in the right thyroid lobe. C, Chest X-ray demonstrating no abnormalities or signs of active pulmonary tuberculosis. PET-CT indicates positron emission tomography computed tomography.
In the weeks prior to presentation, the mass began draining from a fine needle aspiration site. The biopsy itself was nondiagnostic (Bethesda I) showing only a fibrotic thyroid mass. It was challenging to obtain cells for definitive cell block/flow cytometry. Given the ambiguity of diagnosis, the patient was taken to the operating room for open biopsy.
It was unclear from our initial examination whether this patient had an aggressive thyroid malignancy, such as an anaplastic carcinoma, or another etiology which had been superinfected. The mass’s size fluctuation, drainage, and initial improvement with antibiotics favored the latter.
Intraoperatively, extreme fibrosis was noted just beneath the level of the platysma. The right sternohyoid and sternothyroid muscles were incised revealing further fibrosis as well as the thyroid mass. Sharp dissection was used to excise a generous amount of tissue for both permanent section and fresh for flow cytometry which revealed a necrotizing granuloma staining positive for acid fast bacilli (AFB) (Figure 2). Given the lack of pulmonary tuberculosis (TB) lesions on chest X-ray (Figure 1C), these results indicated primary TB in the thyroid gland. He was subsequently treated with standard RIPE therapy (rifampin, isoniazid, pyrazinamide, ethambutol) and is doing well.
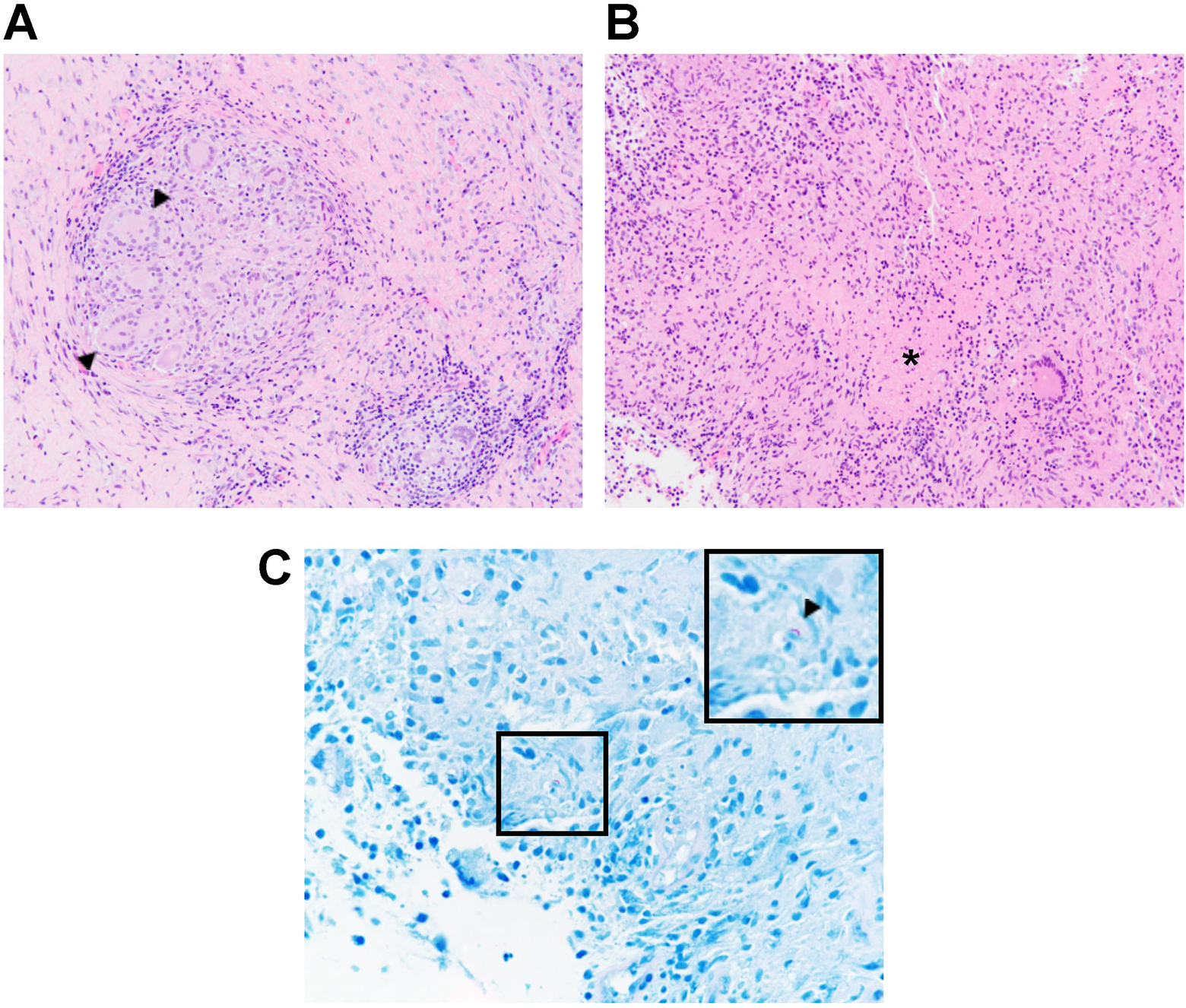
A, Thyroid tuberculosis featuring confluent epithelioid granulomas with numerous giant cells (arrowheads) (hematoxylin–eosin, 200×). B, Necrosis can be seen in the middle of granulomatous nodules which were marked with an asterisk (hematoxylin–eosin, 200×). C, Photomicrograph showing acid fast bacillus (arrowhead) magnified in inset in upper right corner (ZN stain, 600×).
Tuberculosis primarily affects the lungs but can be extrapulmonary in 15% to 20% of cases. 1 The thyroid gland is relatively resistant to TB infection; however, the underlying mechanisms are unknown.2,3 The presence of high iodine levels, a thyroid capsule, the bactericidal action of the colloid, or the gland’s rich lymphatic and vascular supply have been suggested to play a role in the gland’s relative immunity. 4
In the thyroid, TB can present as 1 of 2 forms: more commonly as miliary spread from pulmonary or cervical lymph node TB, or occasionally as the primary site of involvement. 3 Primary TB of the thyroid gland is extremely rare and an unusual presentation both of thyroid masses and of TB infections more generally, 5 occurring at an estimated rate of 0.1% to 0.4% of TB cases. Such rarity often causes a delay in diagnosis. The highest incidence of TB is in Asia and Africa, with over one-third of the world’s cases occurring in India and China alone. 6 Indeed, our patient was born in China and immigrated in 1997, traveling back twice per year.
The presentation of primary thyroid TB is variable. There can be diffuse or nodular swelling, discrete or cystic masses, or an acute abscess. 7 Patients with primary TB of the thyroid gland are typically euthyroid but can occasionally present with thyrotoxicosis2,8 or hypothyroidism. 9 The presentation of thyroid TB is usually subacute, mimicking granulomatous thyroiditis, but if presenting acutely can also mimic acute suppurative thyroiditis. The presence of pain often suggests an infectious etiology as in this case; in its absence, this may suggest malignancy. Radiologic features of thyroid TB are nonspecific and can mimic malignancy; however, CT with contrast has shown caseous necrotic lesions in multiple studies.10,11 This is in line with the radiologic findings we found in our patient.
While rare, our case suggests that TB of the thyroid gland should be included on the differential, especially when radiologic examination shows areas of central necrosis, when patients have a history of TB, or are from countries where TB prevalence is high.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
