Abstract
The neuroendocrine carcinomas (NECs) of the head and neck are rare. The purpose of this article is to explore the diagnosis and treatment of NECs in the ear and larynx. We report a case of a patient with NECs found in the ear and throat simultaneously, and the relevant literatures are reviewed. It is difficult to identify which is the original site. There is no specific clinical manifestation of NECs in the ear and throat, and carcinoid syndrome is a rare situation. Surgery is still the preferred treatment for this disease. For patients with metastasis, radiotherapy and/or chemotherapy are required.
Introduction
Neuroendocrine carcinomas (NECs) originate from the neuroendocrine cells found in various organs and systems throughout the human body. As neuroendocrine cells are widespread in the digestive and respiratory systems, NECs are most commonly seen in the gastrointestinal tract, pancreas, and lungs. Rarely are there reports of NECs that are primarily in the head and neck; simultaneous occurrence in the ear and throat is even rarer. Here, we report a patient with NECs in both the ear and throat, and we review the relevant literature.
Case Report
An 82-year-old male patient presented with left ear pain and left facial dyskinesia, which he had begun suffering 3 months before. At onset of symptoms, he started to have left ear pain and hearing loss with occasional bloody discharge for no known reason, as well as left side facial palsy. His House-Brackmann facial nerve functional assessment was grade V. He received anti-inflammatory and nutritional neurotherapy, but he did not improve. He reported that 15 years ago he had been diagnosed with right nasopharyngeal squamous cell carcinoma with ipsilateral cervical lymph node metastasis. The pathological examination showed poorly differentiated squamous cell carcinoma. He received radiotherapy and had no metastasis or recurrence afterward. During the physical examination on this hospital admission, a new light pink, irregular growth was found completely blocking the external auditory canal of the left ear. The tympanic membrane was not visible (Figure 1A). Laryngeal examination revealed a new dark red, smooth growth in the epiglottis blocking the laryngeal cavity (Figure 1B). The patient denied any difficulty breathing, but he said he had recently experienced pharyngeal foreign body sensation while swallowing. A computed tomography (CT) scan was performed on the ear; images showed pneumatization of the left mastoid. A high-density shadow was seen in the left tympanic cavity and mastoid, with the ossicular chain embedded in the shadow, but no bony destruction. There was also a high-density shadow in the left external auditory canal (Figure 2A). Laryngeal CT showed an ovoid, well-defined, and isodensity shadow in the epiglottis (Figure 2B). Nasopharyngeal CT showed no tumor recurrence at the primary site. A hearing test suggested severe mixed-type hearing loss in the left ear. Pathological examination of ear biopsy tissue under a light microscope showed nests of tumor cells with large, dark-stained nuclei. Immunohistochemical (IHC) staining revealed that these cells were positive for chromogranin A (CgA), synapsin (Syn), cytokeratin (CK), and cluster of differentiation 56 (CD56); 80% positive for Ki-67; scantly/weakly positive for epithelial membrane antigen (EMA); and negative for vimentin (VIM), P63, and P40 (Figure 3), which was consistent with ear NECs. Light microscopy showed that tumor cells biopsied from the larynx were relatively uniform in size, distributed in nests. The IHC staining revealed that these cells were CgA+, Syn+, CK+, VIM−, EMA+ (focal), CD56+, 90% Ki-67+, P63−, and CK5/6− (Figure 4), which was consistent with laryngeal NECs. No metastasis was found during the whole-body examination. The patient and his family refused further treatments, and he was signed out of the hospital against medical advice.
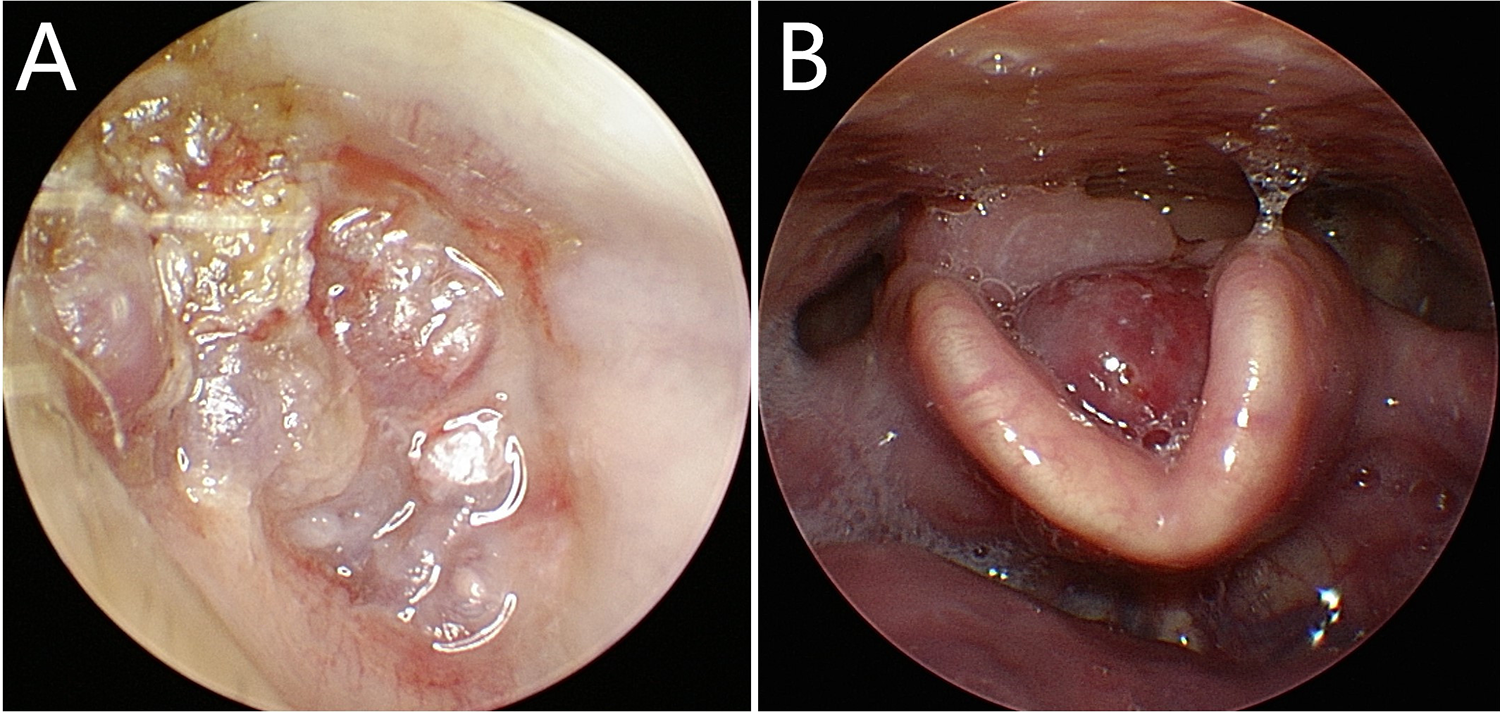
A, Picture of the patient’s otoscope of the left ear. B, Picture of the patient’s laryngoscopy.
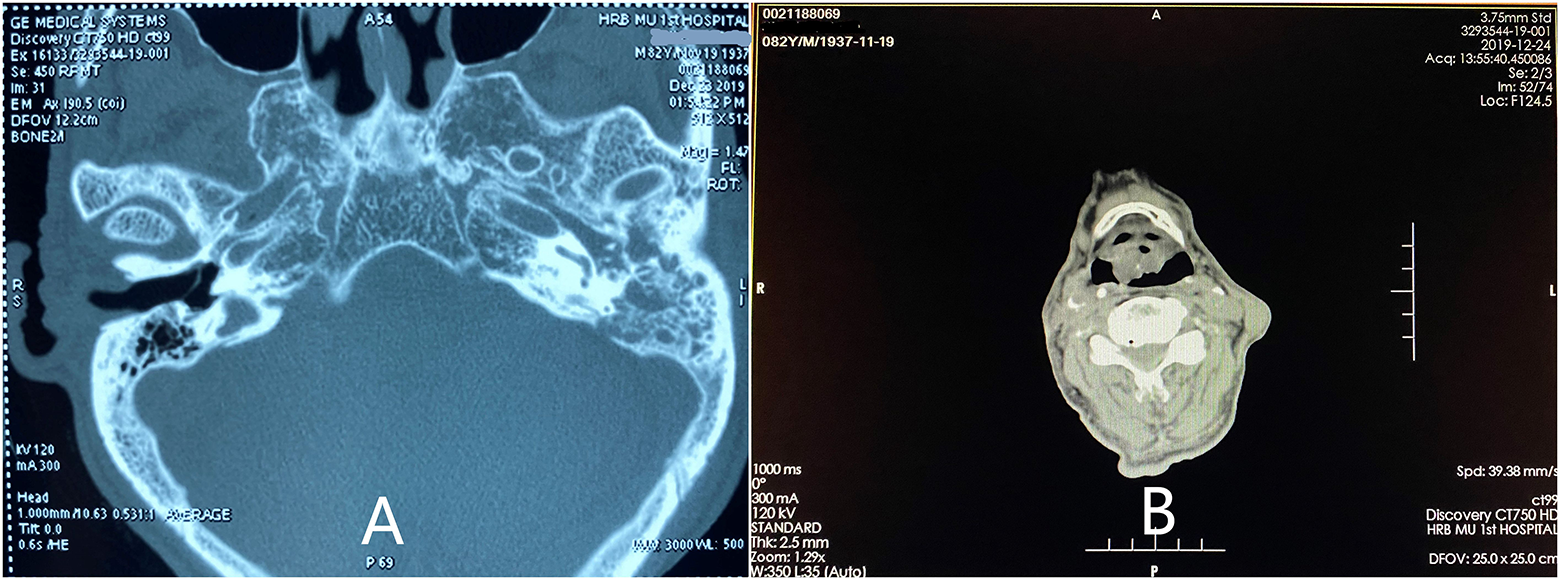
A, High-resolution computed tomography (CT) of the temporal bone. B, Laryngeal CT.
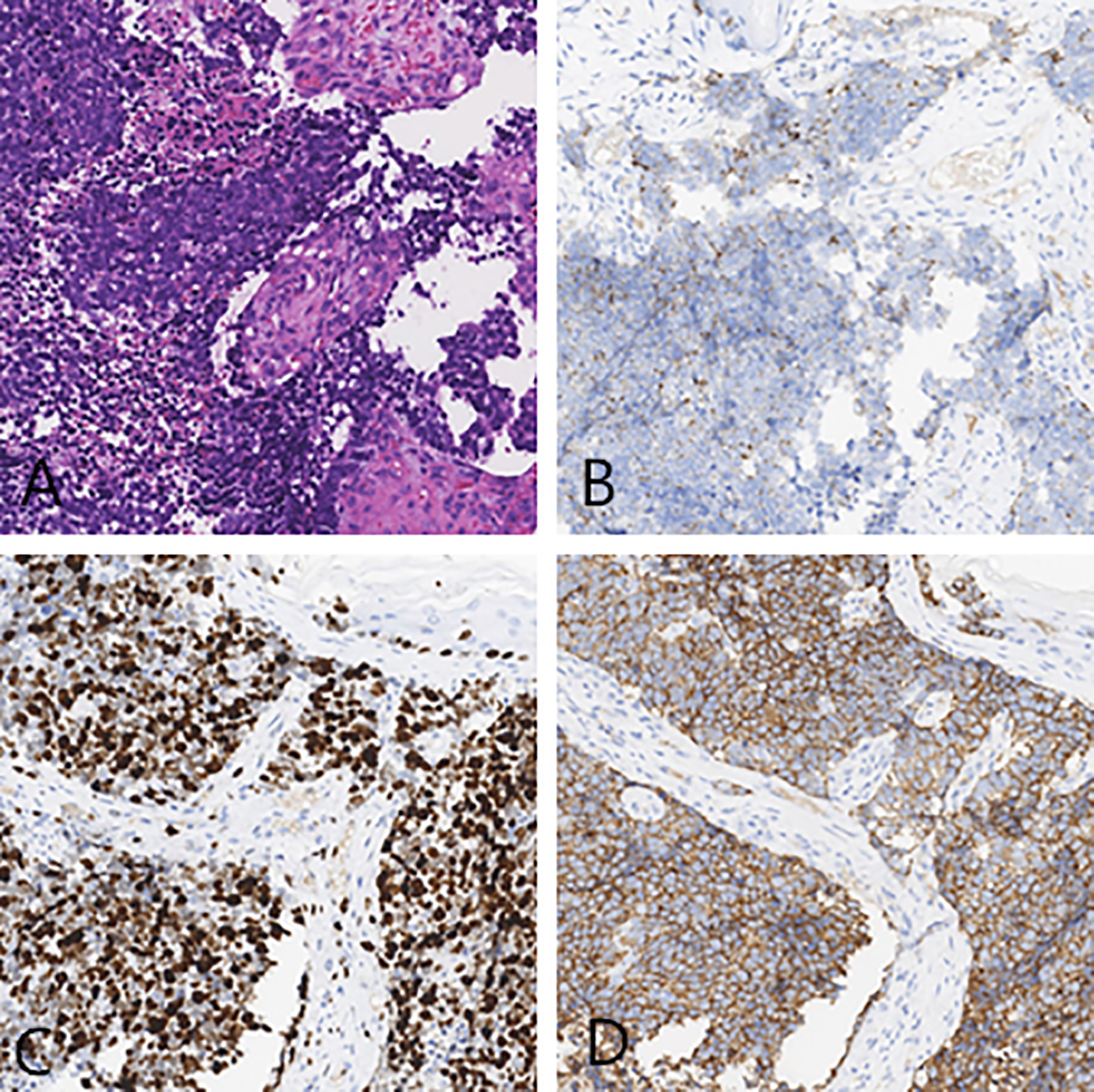
Pathological and immunohistochemical images of the left ear. A, H&E staining. B, chromogranin A (CgA+). C, Ki-67 (80%+). D, synapsin (Syn+).
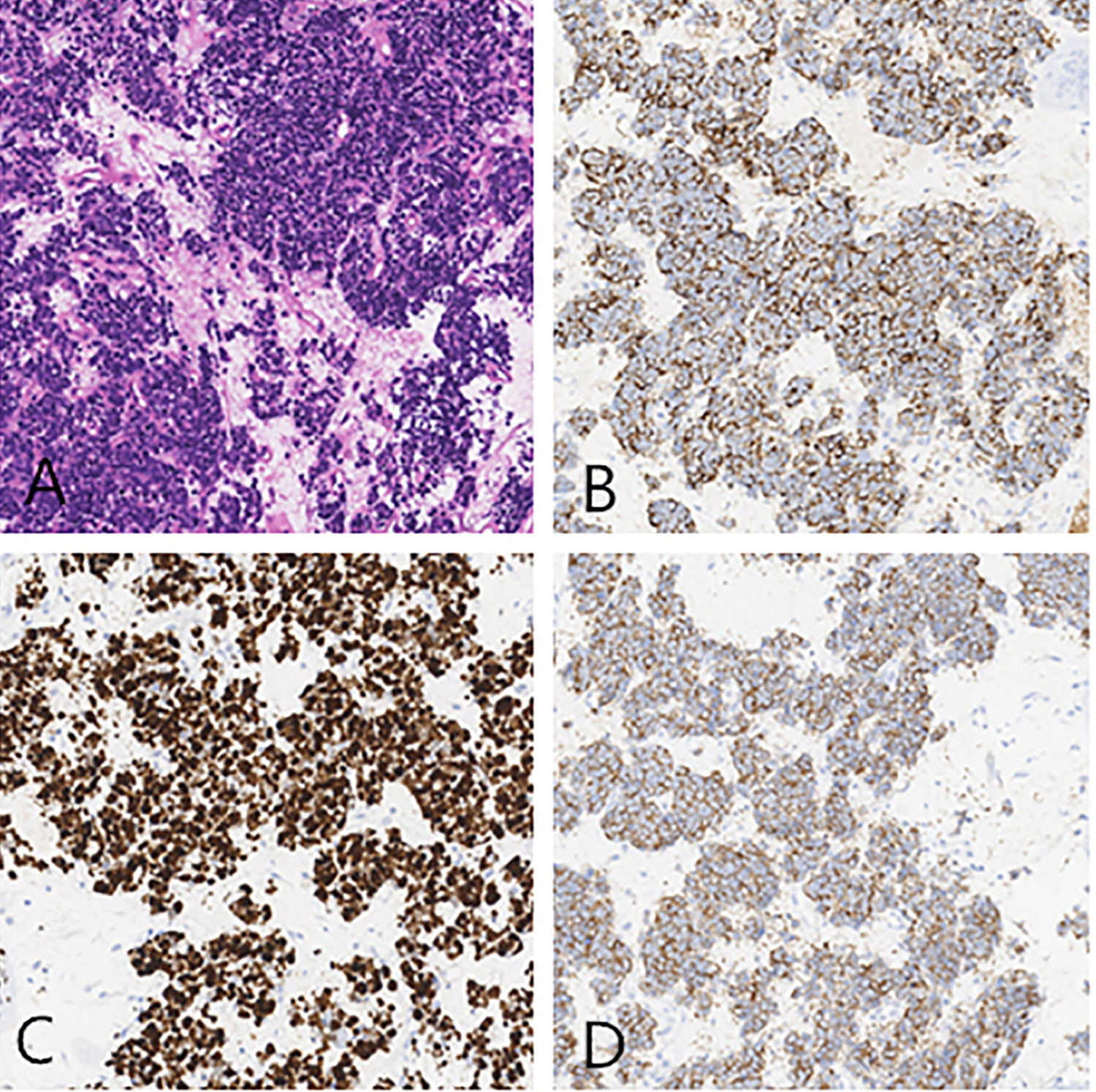
Pathological and immunohistochemical images of the larynx. A, H&E staining. B, chromogranin A (CgA+). C, Ki-67 (90%+). D, synapsin (Syn+).
Discussion
Neuroendocrine carcinomas are rare tumors and especially so in the head and neck. Occurrence of NECS in this region of the body usually involves the larynx; NECs account for 1% of malignant tumors therein.1,2 The ear is the second most common head and neck location for NECs. 3 Currently, about 1000 cases of NEC have been reported in the larynx, and fewer than 100 cases in the ear. But we couldn’t find 2 simultaneous tumors until now.
Neuroendocrine carcinoma in the larynx usually happens in patients aged 60 to 70 years. It commonly arises in the supraglottic region, most commonly in the arytenoid cartilage and epiglottis and second most commonly in the epiphyseal wrinkles. 4 Patients with NECs can present clinically with nonspecific symptoms, including hoarseness, cough, blood in sputum, and dyspnea, all of which can be seen in other laryngeal cancers. Neuroendocrine carcinomas more commonly occur in men than women, with an incidence ratio of 3:1. Smoking increases the risk for NECs. It is also reported that human papillomavirus infection is associated with the occurrence of NECs. 5 In rare situations, abnormal hormones secreted by NECs can cause carcinoid syndrome, including diarrhea, flushing of the skin, and bronchoconstriction. The patient in this case study had only abnormal pharyngeal foreign body sensations during swallowing with no other chief complaints, which caused the delay in his diagnosis.
The presence of neuroendocrine cells in the middle ear has not been definitively confirmed, and the exact source of carcinoids therein has not been clarified. 3 One possible explanation is that undifferentiated pluripotent endoderm stem cells might still exist on the surface of the middle ear mucosa and could be the origin of NECs. 6 Patients with ear NECs commonly have conductive hearing loss, accompanied by clogged ears, blockage, tinnitus, otalgia, and external auditory canal discharge (often bloody). Facial palsy can occur if the tumor invades the facial nerve. Ramsey et al analyzed 46 patients with NECs and found that the incidence ratio was 1.4:1 men to women. Percentages of patients with hearing loss, ear fullness, tinnitus, ear discharge, and otalgia were 87%, 33%, 24%, 17%, and 9%, respectively. A few patients also reported transient facial palsy. Carcinoid syndrome was not common in ear NECs. 7
Recent studies have found that laryngeal NECs are more likely to metastasize. Van der Laan et al analyzed 416 patients with laryngeal NECs and found that the distant metastasis rate in highly differentiated cancer was very low, whereas in poorly and moderately differentiated cancer, it could be as high as 90%. 8 Ear NECs have a low risk of distant metastases; most of them are in situ recurrences with local metastases.7,9 Another study found that metastases from ear NECs usually occur 8 months to 43 years after treatment. 10 Our patient’s NECs had high Ki-67 values and were highly proliferative tumors with high degrees of malignancy, rapid growth rates, and a high risk of metastasis. Since he had had ear discomfort with peripheral facial palsy for 3 months, which was longer than he experienced the mild and insignificant symptom of pharyngeal foreign body sensation during swallowing, we suspected that the ear NEC was the primary tumor and had metastasized to the larynx. However, elderly patients commonly have delayed pharyngeal reflexes and can have tumors for a while before they might present with any complaints in the larynx. Therefore, the location of the primary NEC in this patient was not clear.
Neuroendocrine carcinomas can secrete a variety of neuroendocrine hormones, such as serotonin, growth hormone, insulin, gastrin, glucagon, and CgA; this enables the NECs to be detected by serological and IHC methods. Of these hormones, serum CgA is the most commonly tested for. Its serum level correlates with the tumor’s volume, differentiation, and secretory activity, and so serum CgA tests can facilitate assessment of treatment response and prognosis. 11 Imaging examinations often show no specific presentations. Computed tomography examinations can show soft tissue density shadows, with or without bony destruction. Magnetic resonance imaging scans can show T1 isointensity signals and T2 signals similar to gray matter. 12 A preoperative pathological examination should be performed if possible, since its results will play a critical role in determining diagnosis and treatment strategy, as well as in evaluating prognosis. One study also reports that fine needle aspiration cytology (FNAC) could be used in diagnosis. 13 The authors of that study believe that FNAC is safe for biopsy, posing few complications, especially for subcutaneous masses. Incisional biopsy, by contrast, could cause greater tissue damage. For masses in the external auditory canal and middle ear, cytology can supplement or even be substituted for biopsy, but only if immunocytochemical examination is available to the clinician.
Pathological examination is the gold standard for the diagnosis of NEC. Well-differentiated NECs consist of round- or spindle-shaped cells, which are arranged in nests, trabeculae, or large pieces. The cytoplasm is pink, with punctate or dense chromatin visible in the nucleus. Nucleoli and mitosis are rarely observed. There is no necrosis or pleomorphism. The nuclei of moderately differentiated NECs are vesicular and have visible nucleoli, mitosis, necrosis, and cell pleomorphism, with frequent vascular lymphatic invasions. Poorly differentiated NECs consist of nested, lamellar, or banded cells with scant cytoplasm. The nucleus is round, oval, or spindle shaped; it has polymorphism, prominent nucleoli, and mitosis. The center of the nests frequently has necrosis and apoptosis. In diagnostic IHC evaluation of NECs, tests for CgA and Syn are essential, as they can demonstrate the neuroendocrine properties of these cancer cells. Somatostatin, glucagon, and neuron-specific enolase can also be tested for. 14 Ki-67 is usually a required test for evaluating the patient’s prognosis. Patients can be classified by the tumor’s Ki-67 labeling index: if Ki-67 is ≤2%, 3% to 20%, or >20%, tumors are respectively grade 1 (low grade), 2 (intermediate grade), or 3 (high grade). A higher grade indicates greater malignancy. 15 In our patient, biopsy specimens from both the larynx and ear showed visible nucleoli and mitosis, which were consistent with poorly differentiated cancer. Positive IHC tests for CgA and Syn indicated that the cancer cells had neuroendocrine properties. Both his larynx and ear cancers were determined to be grade 3 NECs, as Ki-67 index exceeded 20% in both locations (80% and 90%, respectively).
Surgical resection is the preferred method of treatment for ear NECs. Surgical methods include complete tumor resection, radical mastoidectomy, subtotal temporal bone resection, and lymph node dissection in the mastoid region. When the ossicular chain is embedded in the tumor, the long-term recurrence rate in patients who undergo ossicular chain resection is 18% to 22%, which is better than that of patients with unresected ossicular bone. When the tympanic membrane needs to be reconstructed, it is better to choose fascia instead of cartilage graft to facilitate the postoperative middle ear examination. 9 The efficacy of postoperative radiotherapy, chemotherapy, and other adjuvant treatments is not known at present, since the number of patients who have been studied is too small to draw convincing conclusions from. It is proposed that ear NECs might originate from carcinogenesis caused by chemotherapy on the surrounding lesions. 16 Most physicians suggest that patients with poorly differentiated NECs receive radiotherapy and chemotherapy after surgery, which can improve their disease-free survival rates.
The main treatment method for well- and moderately differentiated laryngeal NECs is also surgical resection. The surgical method is selected based on the property, location, and area of the tumor. 3 Well-differentiated NECs progress slowly with a low risk of metastasis and can be removed by local mass resection. Due to the high risk of metastasis, moderately differentiated NECs should be treated with partial or total laryngectomy combined with selective bilateral cervical lymph node dissection. Local radiotherapy is recommended after the procedure. Poorly differentiated NECs are highly malignant with high rates of metastasis and recurrence and have poor surgical outcomes. Therefore, chemoradiotherapy is the preferred choice. However, some physicians still choose to operate on these NECs based on tumor stage. Zhang et al analyzed the treatment of 25 patients with laryngeal NECs and found no significant differences in the 5-year survival rate between those with early-stage (T1, T2) laryngeal NECs who received CO2 laser therapy under a supportive laryngoscope and those who received open surgery. 17 Patients with stage T3 and T4 NECs should receive partial or total laryngectomy with neck lymphadenectomy, followed by postoperative radiotherapy or chemotherapy. They should then be followed up and receive repeat, close postsurgical examinations. Routine imaging examinations should be performed to determine any recurrence.
Footnotes
Authors’ Note
Tianhong Zhang is the corresponding author, and she contributed to the conception of the study. Tianyu Ma and Chunrui Wang are the co-first authors who contribute to writing this manuscript. Xiaowei Qin contributes to collecting all images. All authors read and approved the final manuscript. Tianyu Ma and Chunrui Wang contributed equally to this work.
Acknowledgments
This work is supported by Qi Liu from the Pathology Department of the First Affiliated Hospital of Harbin Medical University who provides the pathological images.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The project was funded by the Harbin Science and Technology Bureau (NO.2016RAXYJ070).
