Abstract
Objectives:
Head and neck squamous cell carcinoma (HNSCC) is rare in patients younger than 40 years. Many practitioners suspect HNSCC is a more aggressive disease in this age group, and perhaps increasing in incidence; however, there are scant and conflicting data to support this assertion. We sought to compare outcomes for young patients with non-human papillomavirus (HPV)–related HNSCC to those of older patients.
Methods:
A retrospective chart review of patients with HNSCC treated from 2004 to 2016 at 2 tertiary referral centers. Patients aged 18 to 40 with p16-negative HNSCC were included in the young patient cohort (n = 59). A randomly selected stage- and subsite-matched cohort aged 55 to 65 was analyzed for comparison (n = 114).
Results:
When considering all patients with HNSCC, patients younger than 40 were more likely to have oral tongue cancer (62.7%) compared to patients age 55 to 65 (16.9%). When an older patient cohort was stage- and subsite-matched to the young patient cohort, there were more never smokers (49.2% vs 17.5% of older patients, P < .01) and females (40.7% vs 24.6% of older patients, P = .028) in the young patient group. The young patient cohort had better average overall survival than the older group (14.4 vs 8.1 years, respectively, P = .02), but similar average disease-free survival (6.2 years vs 6.6 years, respectively, P = .67); 50.9% of young patients had tumors with adverse histologic features versus 42.0% of older patients (P = .28). The young patients demonstrated a superior average conditional survival after recurrence (9.8 years vs 3.2 years for older patients, P < .01).
Conclusions:
Despite the limitations of study design, these data suggest that young patients who develop non-HPV-related HNSCC tend to have similarly aggressive disease, but longer overall survival and better survival after recurrence. These findings may be attributable to better overall health as evidenced by fewer comorbidities.
Keywords
Introduction
Head and neck squamous cell carcinoma (HNSCC) has historically been considered a disease of older patients with a history of tobacco and alcohol use. 1 Since the 1970s, the incidence of HNSCC in the United States has been increasing in younger patients. This uptrend has been attributed largely to human papillomavirus (HPV)–related cancers; however, a subgroup of these patients has been shown to have non-HPV-related HNSCC. 2 In general, young patients with HNSCC have been thought to fare poorly compared to older patients, perhaps due to an inherently more aggressive form of the disease. This notion has been challenged more recently by the study of HPV-related cancers, a subgroup known to respond favorably to therapy. 3
Retrospective studies published in 1999 and 2005 compared outcomes of a cohort of patients with HNSCC younger than 40 years to an older cohort. Both studies reported similar disease-specific survival (DSS) between groups but better overall survival (OS) in the younger cohort.4,5 Friedlander et al. conducted a similar retrospective study focused on patients with oral tongue squamous cell carcinoma and found no significant difference in OS or DSS; this study noted a higher rate of locoregional recurrence in the young patient group. 6 None of these studies, or any others to our knowledge, separated patients based upon HPV status. Given that the young patient groups likely contained a higher proportion of HPV-related cancers, which are known to have improved outcomes, 7 the findings of these studies may have been impacted.
There remains a distinct and poorly studied group of young patients with non-HPV-associated HNSCC. In this study, we aim to compare this group to older patients with non-HPV-associated HNSCC and evaluate relative patient and disease characteristics and treatment outcomes.
Methods
This study was approved by the institutional review boards at the University of Virginia (UVA) and the University of Alabama at Birmingham (UAB). Patients treated for upper aerodigestive HNSCC from 2004 through 2016 were identified using the Head and Neck Cancer Research Database (UVA) or clinical billing database (UAB). Patients with p16-positive tumors, as well as patients with oropharyngeal tumors not stained for p16, were excluded in order to limit the study to non-HPV-related HNSCC. Ages were determined at the time of initial tumor board evaluation. Initially, all older patients aged 55 to 65 were identified (n = 421 at UVA). Only site, subsite, and stage data were collected on these patients. Next, all patients aged 18 to 40 were identified (n = 59, 37 from UVA and 22 from UAB). All data points were collected on these patients (see below). Relative subsite distribution of young versus all older patients was assessed by comparing these 2 groups. Finally, a control subset of older patients was created (n = 114, 72 from UVA and 42 from UAB) to evaluate relative outcomes of young versus older patients. This control subset of older patients (aged 55 to 65) was matched in a roughly 1:2 ratio by site and stage to the young patient group; when more than 2 appropriate matches were identified, older patients were selected randomly for inclusion prior to collection of clinical data. After selection of all matched patients, clinical data points were collected on this control subset, and outcome comparisons were made to the young group.
Data collection included age at time of diagnosis, gender, tobacco use, cancer stage, site/subsite, adverse pathological features (>3 positive nodes, extranodal extension, perineural invasion, lymphovascular invasion, positive margins), length of follow-up, recurrences, and deaths. Kaplan-Meier survival curves and associated statistical analyses were performed using SPSS Statistics Version 25.0 (IBM). Length of follow-up was calculated from the end-of-treatment date to the date of last evaluation for HNSCC (at otolaryngology, radiation oncology, or medical oncology clinics), whereas survival was calculated using any means of contact with the patient (other clinic visits, telephone encounters, etc). The significance of differences in survival was evaluated using a log-rank test. Other variables were compared using 2-tailed t test, Fisher exact test, or χ2 test as appropriate.
Results
As shown in Table 1, a different anatomic distribution of p16-negative upper aerodigestive HNSCC was observed in young versus older patients. The initial survey of all UVA patients aged 55 to 65 (n = 421, excluding p16+ tumors) demonstrated the most common subsite to be the supraglottis (24.9%). Comparatively, the most common subsite in the young patient group (n = 59, 37 from UVA and 22 from UAB) was oral tongue (62.7%). Also, it is notable that there was no p16− oropharynx or unknown primary tumors in the young patient group. The overall site distribution was significantly different in young patients (P < .001), as was the subsite distribution in the oral cavity (P < .001).
Sites and Subsites of HNSCC in All Older Patients Versus Young Patients.a
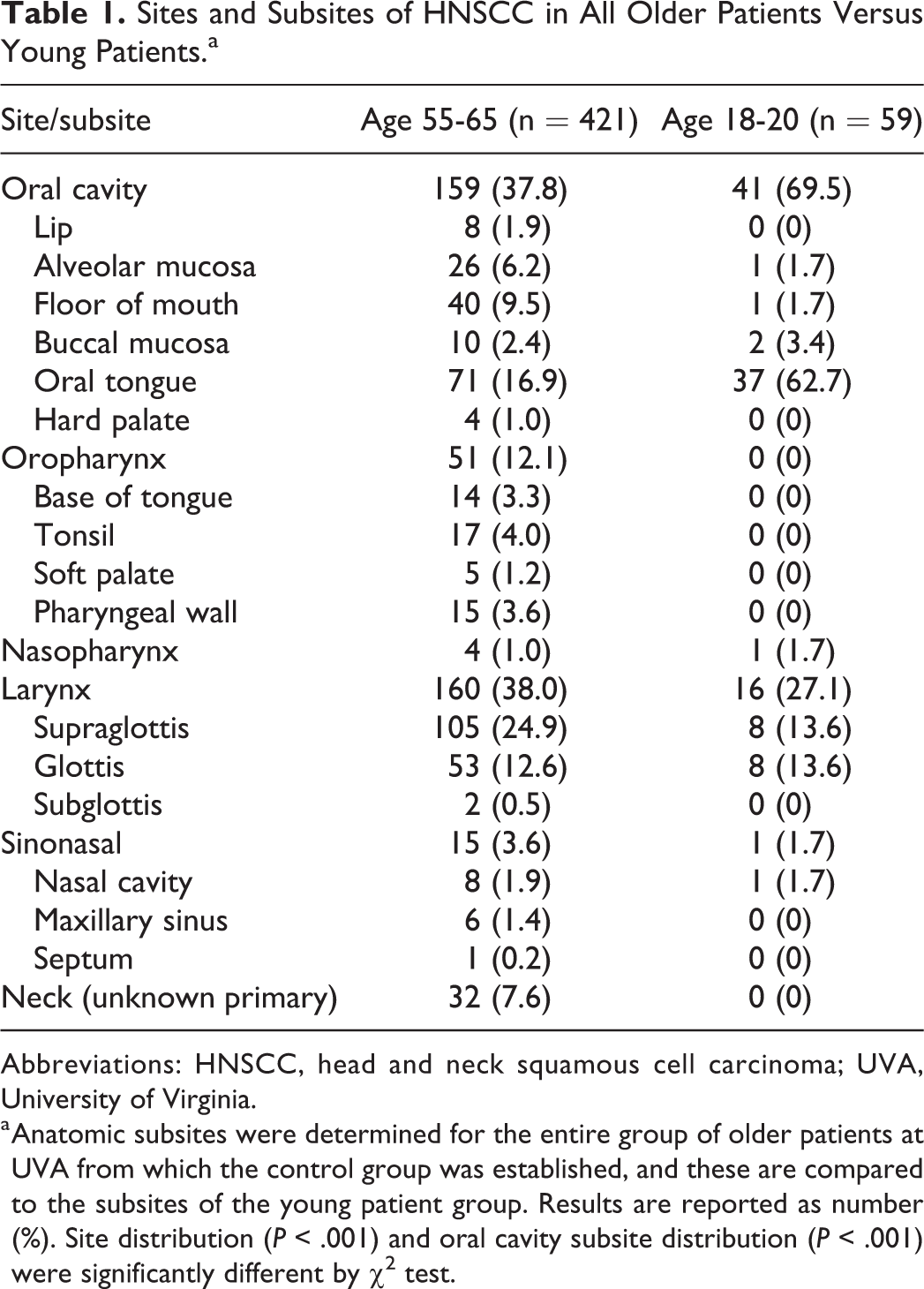
Abbreviations: HNSCC, head and neck squamous cell carcinoma; UVA, University of Virginia.
a Anatomic subsites were determined for the entire group of older patients at UVA from which the control group was established, and these are compared to the subsites of the young patient group. Results are reported as number (%). Site distribution (P < .001) and oral cavity subsite distribution (P < .001) were significantly different by χ2 test.
The young patient group was then compared to the site- and stage-matched older patient control group (n = 114, 72 from UVA and 42 from UAB); clinical characteristics of the 2 groups are shown in Table 2. Average age was 31.5 in the young patient group (median 32, range 18-40) and 60.2 in the control group (median 60, range 55-65). The young patient group was 40.7% female versus 24.6% in the control group (P = .028). There were significantly more never-smokers in the young patient group than the control group (49.2% vs 17.5%, P < .01). The average length of follow-up was 3.6 years (median 2.5, range 0-16.4) in the young patient group and 2.4 years (median 1.4, range 0-10.3) in the control group (P = .02). Initial treatment strategies including surgery (84.7% in young patients vs 82.5% in control group, P = .70) and radiation (13.6% in young patients vs 17.5% in control group, P = .50) were not significantly different between the 2 groups. Histological analysis of tumors revealed a larger subset of tumors with adverse pathological features in the young patient group (50.9% vs 40.2%); however, this did not achieve statistical significance (P = .28).
Clinical Characteristics of Young Patient Group Versus Control Group.a
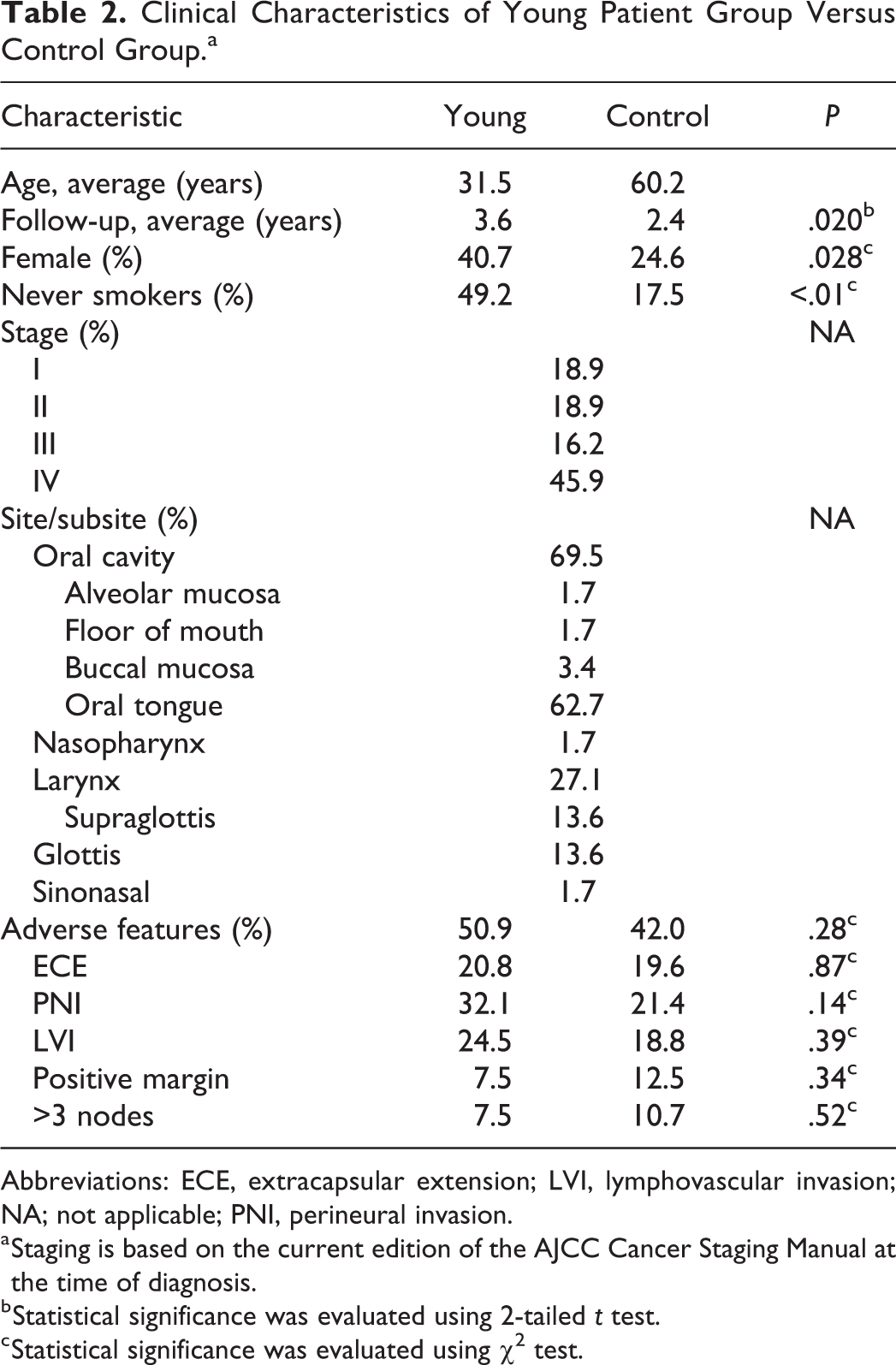
Abbreviations: ECE, extracapsular extension; LVI, lymphovascular invasion; NA; not applicable; PNI, perineural invasion.
a Staging is based on the current edition of the AJCC Cancer Staging Manual at the time of diagnosis.
b Statistical significance was evaluated using 2-tailed t test.
c Statistical significance was evaluated using χ2 test.
The average OS (Figure 1A) for the young patient group was 14.4 years compared to 8.1 years in the control group (P = .02). Disease-free survival (DFS) (Figure 1B) was similar between the 2 groups with average time to recurrence or death of 6.2 years in the young patient group versus 6.6 years in the control group (P = .18). Conditional survival (Figure 2), calculated as OS from the time of recurrence, was significantly better in the young patient group compared to the control group (9.8 years vs 3.2 years, P < .01). When oral tongue patients are analyzed separately, OS and disease-free survival curves were similar to those for the entire cohort; however, no significant differences were observed with this smaller subset (Figure 3).
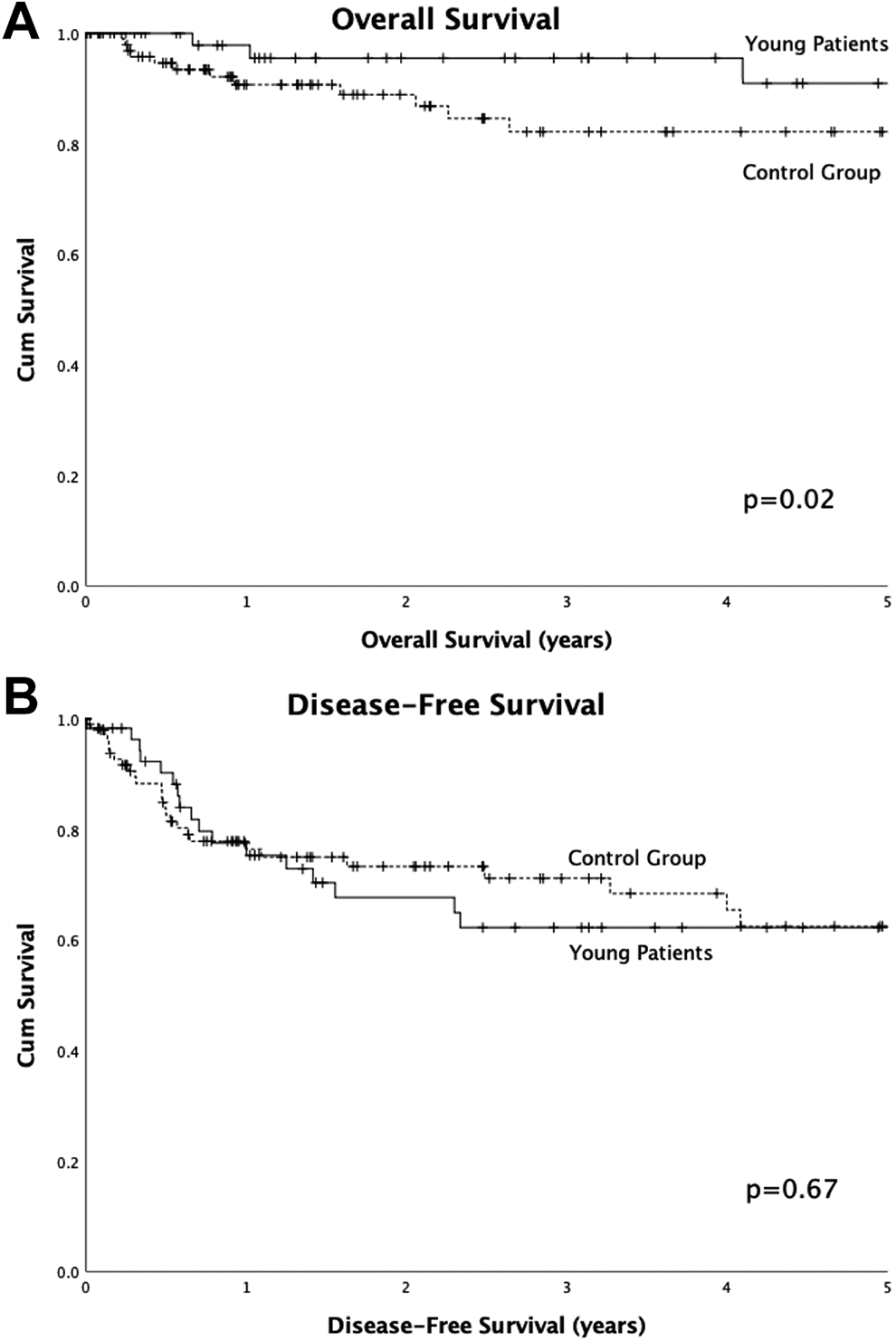
Survival of young patient cohort versus control cohort. A, Overall survival. Patients from University of Virginia (UVA) and University of Alabama at Birmingham (UAB) with no known death were censored at the date of last contact. Average survival was 14.4 years in the young patient group and 8.1 years in the control group (P = .02). B, Disease-free survival. Patients from UVA and UAB with no known recurrence/death were censored at the date of last contact. Average survival was 6.2 years in the young patient group and 6.6 years in the control group (P = .67).
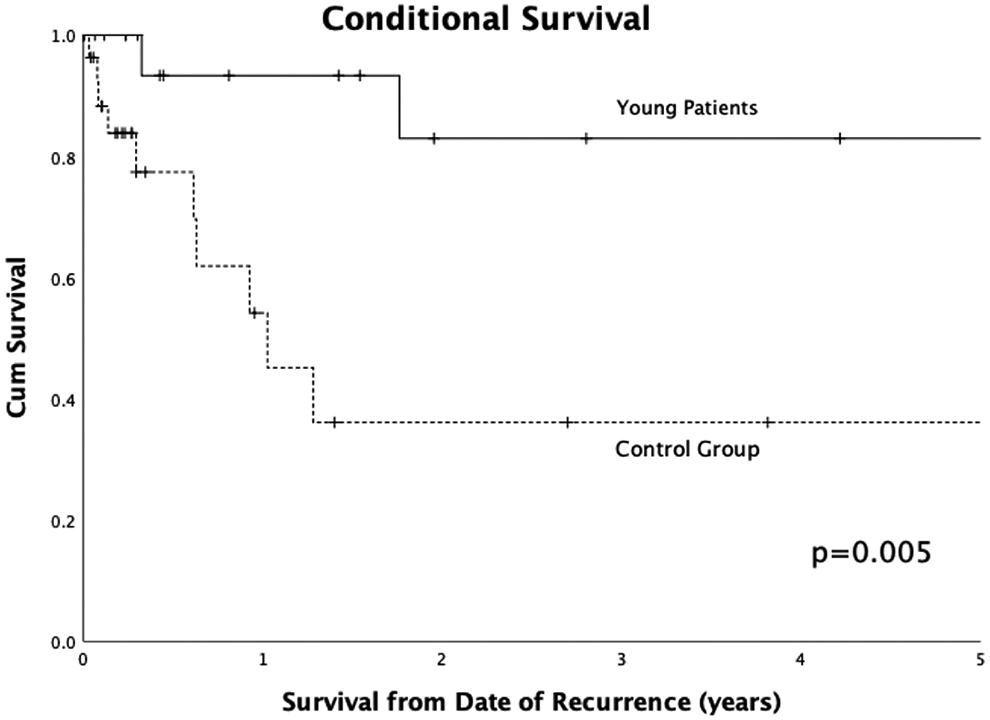
Conditional survival after recurrence of young patients versus control cohort. Patients from University of Virginia (UVA) and University of Alabama at Birmingham (UAB) with no known death were censored at the date of last contact. Average survival was 9.8 years in the young patient group and 3.3 years in the control group (P = .005).
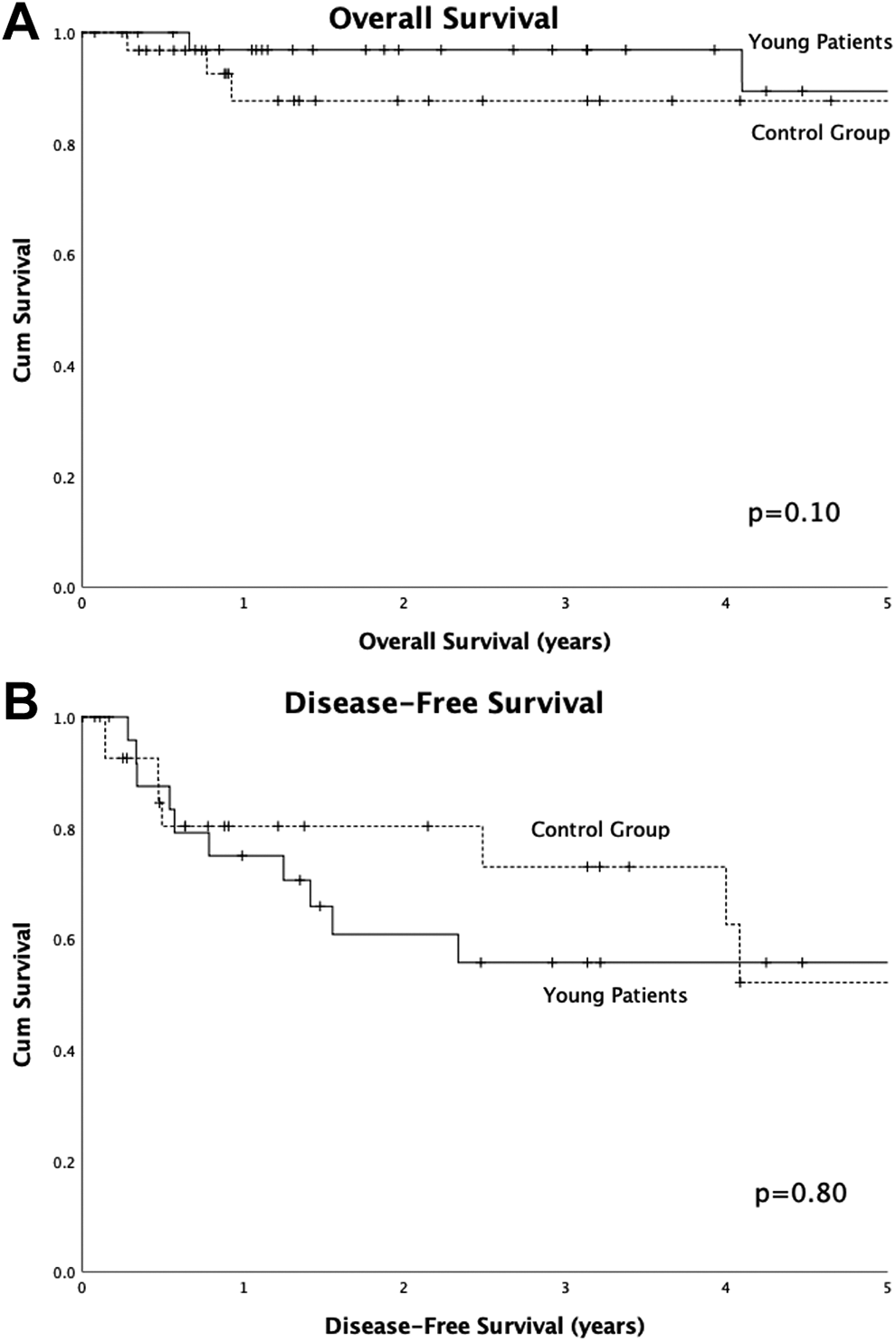
Overall survival of patients with HNSCC of the oral tongue. A, Overall survival. Patients from University of Virginia (UVA) and University of Alabama at Birmingham (UAB) with no known death were censored at the date of last contact. Average survival was 14.2 years in the young patient group and 8.1 years in the control group (P = .10). B, Disease-free survival. Patients from UVA and UAB with no known recurrence/death were censored at the date of last contact. Average survival was 5.2 years in the young patient group and 5.3 years in the control group (P = .80).
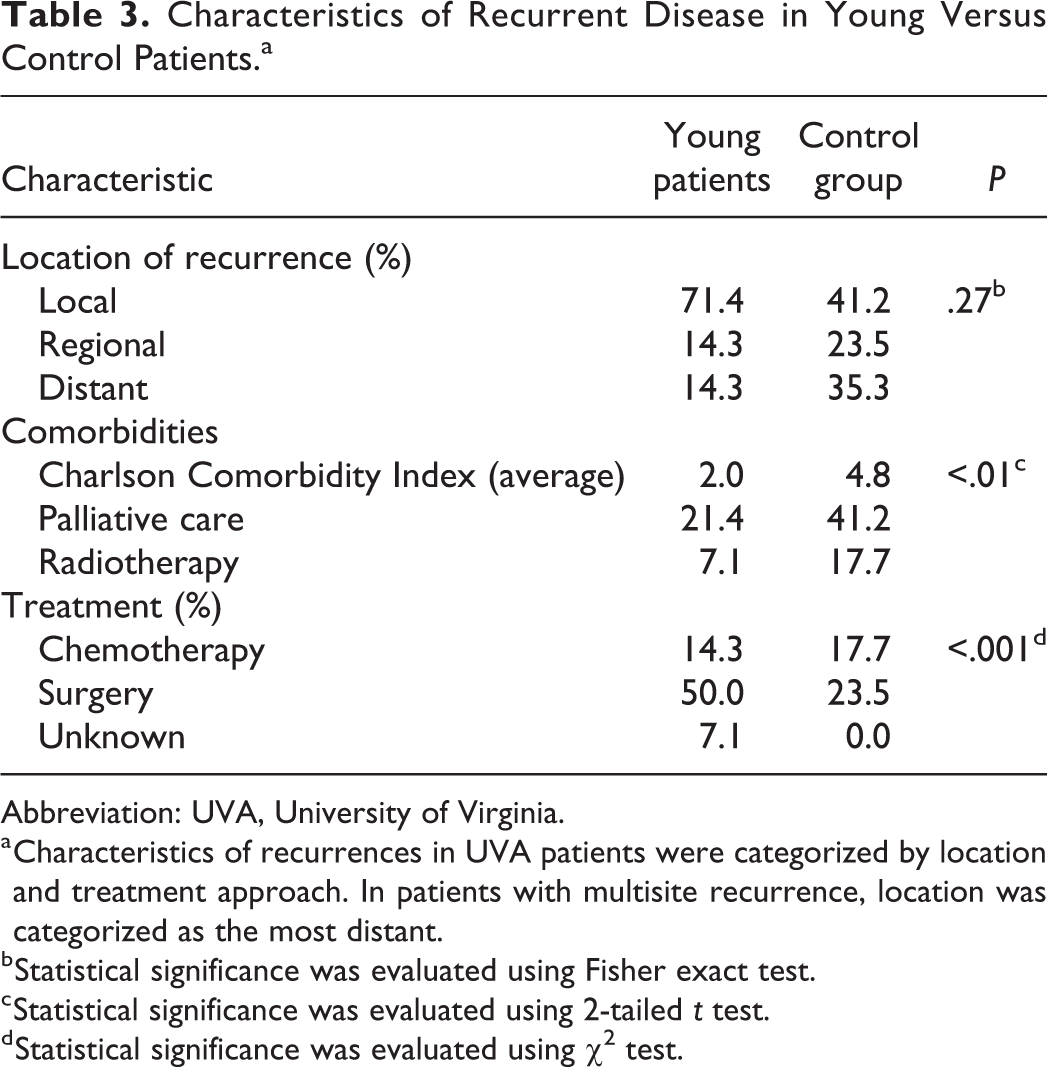
UVA patients who recurred (14 young patients; 17 control patients) were studied in greater detail (Table 3). Young patients had a higher rate of local recurrence than the control group (71.4% vs 41.2%, respectively), while control patients had a higher rate of distant metastasis (35.3% vs 14.3% in the young patient group). However, the overall distribution of recurrences was not statistically different between the 2 groups (P = .27, Fisher exact). The choice of treatment modality used to treat the first recurrences was significantly different between the groups (P < .001). Young patients were more likely to undergo surgery (50.0% vs 23.5% of control patients). Conversely, older patients were more likely to undergo palliation (41.2% vs 21.4% of young patients). Young patients with recurrences had an average Charlson Comorbidity Index (CCI) of 2.0 compared to 4.8 (median 5) for patients in the control group who recurred (P < .01), indicating significantly better overall health. This may have influenced treatment selection and/or outcome.
Characteristics of Recurrent Disease in Young Versus Control Patients.a
Abbreviation: UVA, University of Virginia.
a Characteristics of recurrences in UVA patients were categorized by location and treatment approach. In patients with multisite recurrence, location was categorized as the most distant.
b Statistical significance was evaluated using Fisher exact test.
c Statistical significance was evaluated using 2-tailed t test.
d Statistical significance was evaluated using χ2 test.
Discussion
The data from this 2-institution retrospective study highlight a unique group of patients with HNSCC. Although survival in young patients with HNSCC has been studied previously, no prior studies have isolated survival analysis to non-HPV-related cancers. In survival analysis, the young cohort had no significant difference in DFS with similar average time to recurrence compared to the site/stage-matched older control group. Despite this, younger patients had better OS which could be attributable to better overall health and fewer comorbidities. This pattern remained when only oral tongue cancers were studied. It was notable, however, that young patients have substantially better survival after the first recurrence (conditional survival) compared to the older cohort. Thus, although young patients with non-HPV-related HNSCC are at similar risk for recurrence, they fare far better after recurrence and have longer OS. The reason for this may be due to the decreased likelihood of distant metastasis in the young patient group, although this difference did not reach significance likely due to the small number of recurrences. Additionally, among patients with recurrences, the young patients had a significantly lower CCI and were more likely to undergo surgery while older patients had more comorbidities and were more likely to be managed with palliation.
Based on these outcome data alone, it is difficult to discern whether non-HPV-related HNSCC in young patients represents a distinct disease process at the molecular level, or alternatively if it is the same disease in a physiologically different host. Analysis of the 2 groups reveals several differences. The young patients in this study had a significantly lower incidence of tobacco exposure, a more even gender distribution, and possibly a higher number of adverse pathological features than those in the older cohort (this difference did not reach statistical significance). Additionally, young patients had a preponderance of oral tongue tumors (62.7%) compared to all older patients with HNSCC prior to matching for subsite and stage (11.5%). These data lead us to believe that the HNSCC we observe in young patients may have a unique molecular character compared to HNSCC in older patients.
Others have previously suggested a unique subset of HNSCC in young white females with limited exposure to traditional risk factors with superior survival.8,9 However, the present study is the first to exclude HPV-related cancers on the basis of p16 immunohistochemical staining. Positive p16 staining has been independently associated with HPV infection and improved outcomes in young patients.10,11 The molecular basis of HNSCC in young patients has been investigated, although deliberate exclusion of HPV-related cancers has been rare. TP53 mutations are common in all cancers including HNSCC, with a higher incidence correlated with tobacco and alcohol use. 12 A study of oral tongue cancers by Li et al. found fewer TP53 mutations in young nonsmokers. 13 In contradistinction to this, De Paula et al. reported higher rates of TP53 expression in tumors of young patients with few risk factors. 14 Other genetic alterations noted at higher rates in young patients include loss of heterozygosity and altered methylation of histone H3. 12 Further studies of molecular changes in non-HPV-related HNSCC in young patients are needed to clarify the extent of these molecular differences and to determine whether such differences constitute a fundamentally different form of HNSCC compared to that in older patients exposed to traditional risk factors.
This study is limited by its retrospective nature, by the rarity of non-HPV-related HNSCC in patients younger than 40 years and by the limited number of recurrence and death events in this already small population. For some analyses, data were only available for UVA patients, introducing the possibility of institution-dependent bias, although such bias is commonly present in single-institution studies. Additionally, average follow-up times for both groups were relatively short, which may have impacted OS estimates.
Conclusion
Compared to older patients, young patients who develop non-HPV-related HNSCC tend to be more gender-balanced and are more likely to be never smokers. They have similarly aggressive disease with comparable DFS. However, they have superior OS and conditional survival after recurrence, which may be due to fewer comorbidities and thus better overall health.
Footnotes
Authors’ Note
This paper was presented at the 2018 American Head and Neck Society Annual Meeting and the 2018 Virginia Society of Otolaryngology Annual Meeting.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
