Abstract
Objective
This investigation aims to review the known genetic mutations associated with oral cavity squamous cell carcinoma (OCSCC) in young adults with limited environmental risk factors (YLERs).
Data Sources
A comprehensive search strategy was designed to identify studies in MEDLINE (Ovid), Embase (Ovid), and Scopus from database inception to May 2017 that included adults ≤50 years of age with OCSCC and minimal tobacco use history (≤10 pack-years) who had their tumors genetically sequenced or mutational profiles analyzed.
Review Methods
Identified articles were screened by 2 reviewers. Quality of evidence was graded by the MINORS criteria for case-control studies; other studies were graded by assigning a level of evidence for gene mutation literature.
Results
Thirteen studies met our inclusion criteria, and 130 patients met our criteria for age and tobacco history.
Conclusions
Keywords
Traditionally, oral cavity squamous cell carcinoma (OCSCC) has been observed in older men with a history of environmental risk factors, such as tobacco and alcohol use. While the incidence of oropharyngeal cancer has been increasing due to carcinogenic strains of human papillomavirus (HPV), the overall incidence of squamous cell carcinoma seen in the oral cavity has decreased.1,2 It is hypothesized that this is due to the declining rates of tobacco use in younger patient populations. 3 While the overall incidence of OCSCC is decreasing, the frequency of oral tongue squamous cell carcinoma (OTSCC) has been observed to be increasing, particularly in this younger cohort of patients.4,5 Specifically, a higher rate has been reported among young White individuals (<50 years) without traditional risk factors and predominantly among women.6-8 Tobacco use in this young cohort with oral tongue cancer has been reported to range between 10% and 50%.7,9 Furthermore, this increase in OTSCC has been observed among the same birth cohorts in White men and women, particularly those born after the 1940s. 10 Strikingly, this same increase in incidence has not been observed among other subsites of the oral cavity, such as the buccal mucosa, upper and lower alveolus and gingiva, hard palate, or floor of mouth. 11
To date, there has been no definitive etiology proposed for the increasing incidence of OTSCC, despite the suggestion that a combination of genetic predisposition and environmental exposures may portend an elevated risk. 12 As the application of next-generation sequencing technologies continue to rapidly develop, identifying genetic mutations present in this young OCSCC population with low environmental risk may provide insight into causality and/or potential targeted therapies. This article aims to review the known mutational landscape of OCSCC, particularly of the oral tongue subsite, in young adults with low environmental risk (YLERs).
Methods
This systematic review was conducted in accordance with the recommendations of the PRISMA statement (Preferred Reporting Items for Systematic Reviews and Meta-analyses). 13
Search Strategy
A comprehensive search strategy was designed and executed by R.P. in MEDLINE (Ovid), Embase (Ovid), and Scopus for results published from database inception through May 17, 2017. The search strategy utilized all relevant controlled vocabulary and keyword terms in an effort to yield all results with data pertaining to the mutational landscape of OCSCC in adults with a tobacco use history ≤10 pack-years.
Eligibility Criteria and Study Selection
Studies in the English literature were included that pertained to adults ≤50 years old (“young”) who had OCSCC, a smoking history ≤10 pack-years (“nonsmoking”), and their tumors genetically sequenced or mutational profiles analyzed. Ten pack-years was chosen as the cutoff for smoking history due to a previous analysis showing significant escalation in the risk of OCSCC development above this amount for adults of all ages. 4 Studies without information pertaining to genetic mutations were excluded. While OTSCC was the primary focus of our review, given the low number of relevant studies, all oral cavity subsites were included in the eligibility criteria to capture the maximum number of cases. For the same reason, records of alcohol and smokeless tobacco habits were not factored into the eligibility criteria. Study selection was conducted independently by 2 reviews (K.O.S. and R.R.B.) in 2 successive rounds. Records that appeared to meet eligibility criteria or could not be definitively excluded were advanced to the second round, wherein they were screened on the basis of full text.
The articles describing gene mutations discovered through our search strategy were reviewed and assigned a level of evidence for gene mutation literature, as adapted to our study from a previous study of genetic mutations in pulmonary artery hypertension. 14 Case-control studies were graded according to the MINORS criteria (Methodological Index for Non-randomized Studies; Table 1 ). 15
Summary of the Included Articles and Mutations.
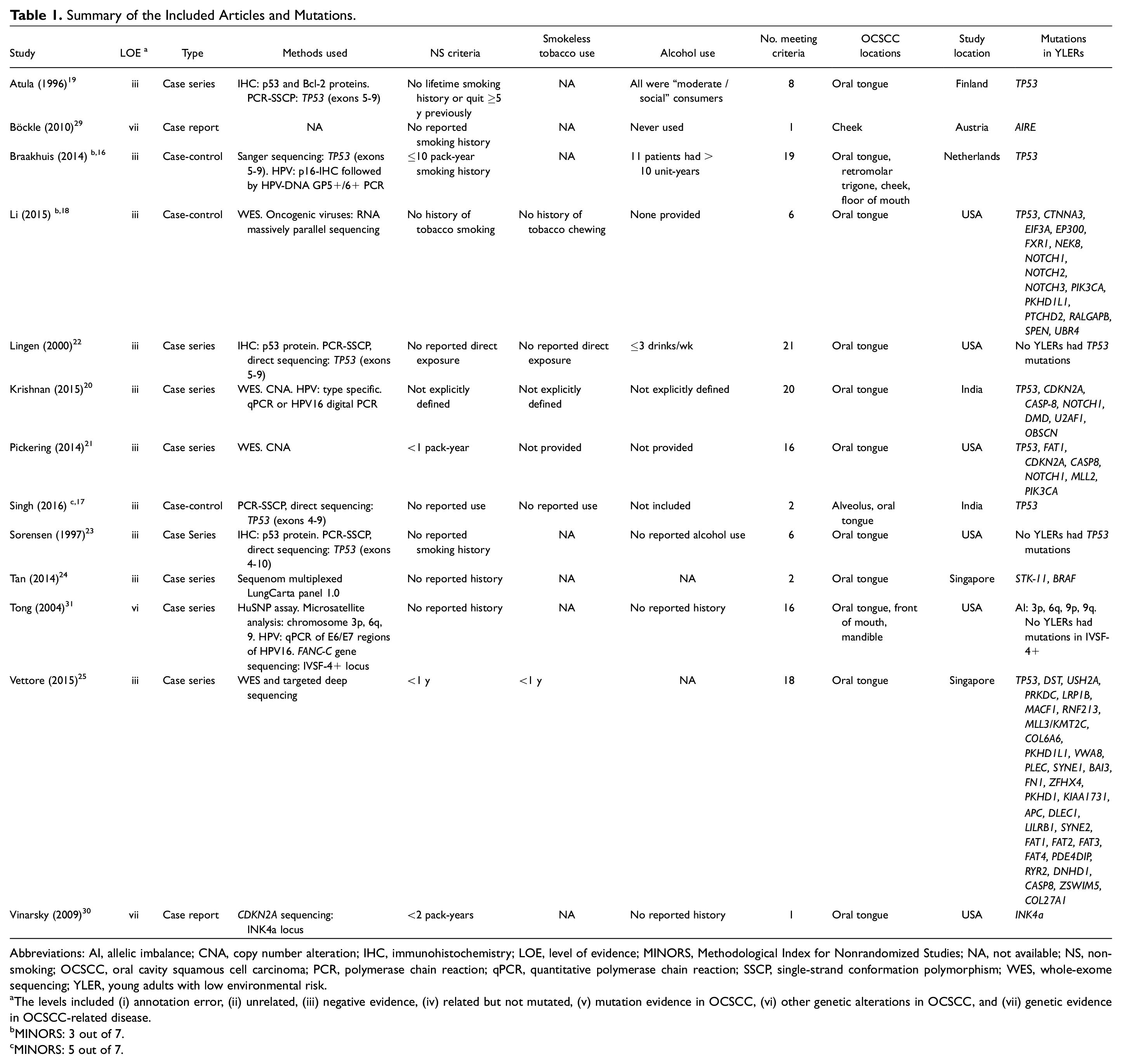
Abbreviations: AI, allelic imbalance; CNA, copy number alteration; IHC, immunohistochemistry; LOE, level of evidence; MINORS, Methodological Index for Nonrandomized Studies; NA, not available; NS, nonsmoking; OCSCC, oral cavity squamous cell carcinoma; PCR, polymerase chain reaction; qPCR, quantitative polymerase chain reaction; SSCP, single-strand conformation polymorphism; WES, whole-exome sequencing; YLER, young adults with low environmental risk.
The levels included (i) annotation error, (ii) unrelated, (iii) negative evidence, (iv) related but not mutated, (v) mutation evidence in OCSCC, (vi) other genetic alterations in OCSCC, and (vii) genetic evidence in OCSCC-related disease.
MINORS: 3 out of 7.
MINORS: 5 out of 7.
Results
Literature Overview and Study Selection
Our search yielded a total of 187 records across all databases. After deduplication, 181 unique records remained. The PRISMA flow diagram in Figure 1 summarizes the results of the study selection process. Thirteen articles met the inclusion criteria: 8 case series, 3 case-control studies, and 2 case reports.
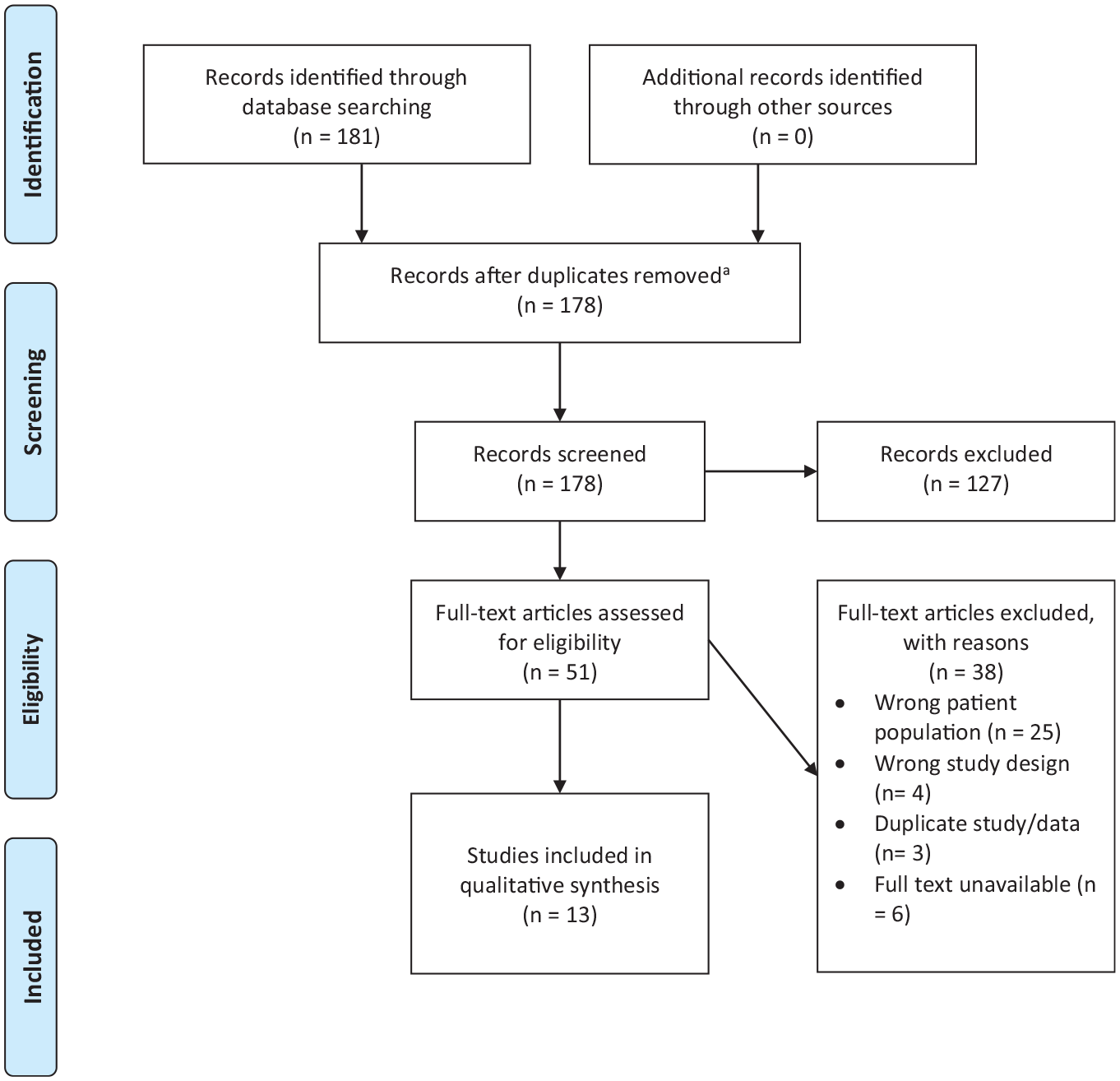
PRISMA flow diagram illustrating the selection process of the included articles.
Risk-of-Bias Assessment
Ten studies showed negative evidence for increased
Patterns of TP53 Mutation and Expression
One study revealed that nonsmokers had significantly fewer
Other Mutational Differences
Other mutations unique to the population of YLERs were
Discussion
While traditional environmental risk considerations regarding the development of OTSCC have largely implicated tobacco use, OTSCC in young nonsmokers is becoming increasingly prevalent despite declining smoking rates worldwide. 5 Interestingly, the rate is decreasing among non-Whites and non-Hispanics, as well as in those >45 years of age across all ethnicities. 6 Though OTSCC in YLERs has emerged as a distinct clinical entity, studies to date have failed to identify a clear etiology. Our review attempts to synthesize our existing knowledge of the genetic changes underlying carcinogenesis in this unique cohort.
TP53 Mutations
The rate of
Truncation events may lead to lack of expression or inability to detect the truncated protein through available methods, thus making it challenging to define the affiliation between mutation and expression. Importantly, these mutational differences may have implications in terms of prognosis and effectiveness of potential therapies in this population. Overall, it appears that
Other Potentially Implicated Genes
The pathogenesis of OTSCC in YLERs is likely to be multifactorial, and
Differing Mutational Landscapes and Genetic Contexts
The genome-wide mutational landscape may be different in YLERs with OCSCC. For instance, one study in our review found elevated rates of allelic imbalance in chromosomes 6q and 9q in nonsmokers.
31
A study not included in our review found increased rates of LOH in distal regions of chromosome 17p, home to
It should be noted that race and sex may affect the genetic changes underlying tumorigenesis in particular populations, and it has been demonstrated that certain genes are differentially mutated in OTSCC across ethnicities. For instance, Asian patients with OTSCC were found to have mutations in genes such as
Finally, while OTSCC in YLERs exists as a distinct clinical and epidemiologic entity, studies to date have yet to identify any genomic or etiologic factors unique to this subpopulation. Furthermore, often contrasting observations related to
Emerging research suggests that the primary difference in OCSCC between smokers and nonsmokers lies in the immune microenvironment, particularly in the PD-1 and PDL-1 pathways.46,47 Despite having similar or higher levels of inflammatory infiltrates as compared with older smokers, the immune response in YLERs may be attenuated, particularly with regard to immune activation and immune exhaustion. 47 These findings elicit promising therapeutic considerations, particularly with regard to immunotherapies, warranting further genetic analysis to potentially direct future management strategies.
Limitations
Overall, few studies have been conducted on genetic mutations in the young nonsmoking population with OCSCC. Furthermore, most are restricted by small sample sizes and are case reports, case studies, or case-control studies, ultimately limiting the applicability of these results. Moreover, inadequate sample sizes prevent appropriate stratification by age, sex, and risk habits. Our analysis was also constrained by discrepancies in the annotation of environmental risk factors. To capture all relevant studies, the use of smokeless (chewing) tobacco was not included in our selection criteria due to its inconsistent incorporation among studies. However, it is an important consideration in this context given that it may reflect tobacco habits across different populations and is a known risk factor for the development of OCSCC. Alcohol use was also not consistently reported, though it is a well-documented risk factor for the development of OCSCC. 48 In addition, the definition of nonsmoking varies considerably among studies, and while all studies met our criteria of a ≤10–pack-year history, specific criteria among individual studies differed. The variations in age and tobacco criteria used among studies hinder the interpretation of individual findings and limit our ability to aggregate data. Last, comprehensive mutational analysis requires significant resources, which often limits the scope of such studies. Significant variation in the methods used to perform mutational analysis complicates the comparison of findings among studies. The relative rarity of OTSCC in our targeted cohort makes it difficult to appropriately power these studies and draw accurate conclusions. While it is apparent that further study is needed to gain meaningful insight into this unique disease process, this review serves as a first step toward appreciating our existing knowledge base and identifying what remains to be elucidated.
Conclusions
The rise in OTSCC among young patients without traditional risk factors represents a unique and puzzling clinical entity. Though a discrete epidemiologic cohort, distinctive underlying etiologic and genomic factors have yet to be identified. Our review suggests that
Author Contributions
Disclosures
Footnotes
This research was presented at the American Academy of Otolaryngology–Head and Neck Surgery Annual Meeting & OTO Experience; September 15-18, 2019; New Orleans, Louisiana.
