Abstract
Importance
The inclusion of depth of invasion (DOI) in the American Joint Committee on Cancer’s staging system for oral tongue squamous cell carcinoma (OTSCC) has major clinical implications. Few studies have evaluated the accuracy of preoperative biopsy to predict DOI.
Objective
To evaluate the reliability of preoperative punch biopsy for measuring DOI in early OTSCC and compare it to evaluation by digital palpation. Secondarily, to assess the punch biopsy’s ability to differentiate between carcinoma in situ (Tis) and invasive carcinoma.
Design
A prospective single-center cohort study.
Setting
Center Hospitalier de l’Université de Montréal, a tertiary center in Canada.
Participants
Patients with suspected early stage OTSCC.
Intervention
Punch biopsy was used to sample the deepest part of tumors to measure biopsy-derived DOI (bDOI). In addition, DOI was estimated via digital palpation: clinical DOI (cDOI) by surgeons.
Main Outcome Measures
Pathologic DOI (pDOI) from final histopathology reports was the gold standard. Spearman’s correlations were calculated between cDOI, bDOI, and pDOI. Diagnostic performance metrics were calculated for the ability to distinguish pDOI of ≥2 mm, and to differentiate Tis from invasive carcinoma.
Results
Among 27 patients, correlation coefficients between bDOI and pDOI, and cDOI and pDOI were 0.603 (95% CI: 0.202-0.884) and 0.894 (95% CI: 0.749-0.955), respectively. Punch biopsy sensitivity and specificity were 0.88 (95% CI: 0.62-0.98) and 0.91 (95% CI: 0.59-0.99) to detect pDOI ≥ 2 mm, and 0.89 (95% CI: 0.65-0.99) and 0.86 (95% CI: 0.42-1.00) for distinguishing Tis from invasive carcinoma. Digital palpation sensitivity and specificity for pDOI ≥ 2 mm were 0.86 (95% CI: 0.57-0.98) and 1.00 (95% CI: 0.63-1.00).
Conclusions
Punch biopsy and clinical palpation demonstrate high diagnostic yield for identifying lesions with pDOI ≥ 2 mm. Punch biopsy appears to be reliable to distinguish Tis from invasive carcinoma.
Relevance
Larger studies are needed to corroborate these findings and assess the role of punch biopsy in guiding elective neck dissection decisions.

Keywords
Key Message
• Depth of invasion is a prognostic factor in oral tongue squamous cell carcinoma.
• Depth of invasion measured on punch biopsy is able to distinguish final pathologic depth of invasion ≥2 mm from <2mm.
• In carcinoma in situ, punch biopsy can reliably exclude invasion.
Background
In contemporary literature, pathologic DOI (pDOI) has proven to be an important prognostic factor in early oral cavity squamous cell carcinoma (OTSCC). pDOI is defined as the distance from the level of the epithelial basement membrane (BM) to the tumor-invasive front. 1 This measure is distinct from tumor thickness (TT), which is defined as the total thickness of the tumor, combining both the invasive depth and the exophytic portion above the BM. 2 Studies show that pDOI correlates with disease-specific survival, as well as occult lymph node metastasis.1,3,4 This has led to its incorporation in the 8th edition of the American Joint Committee on Cancer tumor TNM staging system (AJCC8) (Table 1). Although the optimal cutoff value above which an elective node dissection (END) is indicated has been debated in the literature,5-8 the most recent clinical guidelines from the National Comprehensive Cancer Network (NCCN) now mandate an END for any T1-2, N0 oral cavity SCC with a pDOI above 4 mm and recommend that END should be considered for a pDOI of 2 to 4 mm. 9 The NCCN guidelines state that pDOI should be preoperatively determined by palpation.
The American Joint Committee on Cancer Tumor Node Metastasis Staging System, Eighth Edition. 10

The adequate determination of pDOI in the preoperative setting is critical to plan proper surgical management. Resection margins depend on pDOI, as they differ between carcinoma in situ (Tis) (pDOI = 0) and invasive carcinoma. Moreover, accurate preoperative determination of pDOI can make the difference between performing concomitant glossectomy with END instead of waiting for pDOI to be measured on a gross tumor specimen before performing END in a second surgery. Many recent studies have correlated DOI measured on ultrasound (US),11,12 magnetic resonance imaging (MRI) 13 or computed tomography (CT),14-17 with pDOI. Only 2 studies have evaluated the reliability of DOI measured on biopsy (bDOI) as a predictor of pDOI.18,19 Thus far, no study has evaluated the reliability of digital palpation to estimate pDOI in the preoperative setting. The primary objective of this prospective pilot study was to evaluate the reliability of preoperative punch biopsy to measure preoperative pDOI in early (Tis-T1-T2, N0) OTSCC and to compare it to digital palpation. The secondary objective was to evaluate the reliability of digital palpation to measure DOI, as it is considered the standard evaluation modality in the NCCN guidelines. The tertiary objective was to evaluate punch biopsy’s reliability to distinguish Tis from invasive carcinoma.
Methods
This unicentric prospective diagnostic test accuracy study was conducted between March 2019 and March 2022 at the Center Hospitalier de l’Université de Montréal (CHUM), a tertiary reference center in Montreal, Canada. This study was approved by the local institutional review board (CRCHUM IRB#: 18.296; January 31, 2019). Patients were recruited either at the time of diagnosis or before surgery. Written informed consent was obtained from all patients. Eligibility criteria included patients aged 18 years or above with a clinically early stage (cTis-T1-T2, cN0) OTSCC that had not been previously operated. Other oral cavity subsites were excluded. Clinical DOI (cDOI) was assessed on palpation by fellowship-trained head and neck surgeons (all ≥10 years in practice). Surgeons provided an estimated number of mm of invasion, and any number of mm above zero was considered invasive. bDOI was measured on a 3 or 4 mm diameter punch biopsy specimen. The punches used were Integra Miltex biopsy punches that have a maximal biopsy depth of 12 mm (7 ± 5mm) (Integra LifeSciences, Princeton, USA).
For ulcerated or flat lesions, a biopsy was taken in the center of the lesion, where DOI was maximal (Figure 1). For exophytic lesions (Figure 1b), biopsy was taken at the site of maximal thickness. All biopsies were taken at a 90-degree angle with the tumor surface. Patients recruited before ever being biopsied were biopsied in the clinic under local anesthesia (less than 2 mL of 2% xylocaine in epinephrine 1:100 000). Patients previously biopsied in other centers were biopsied again at the time of glossectomy, under general anesthesia. pDOI retrieved from the final histopathology reports was used as the gold standard. All specimen samples were analyzed by 1 subspecialized head and neck pathologist (author OEG). DOI was calculated in millimeters.

Punch biopsy technique. Punch biopsies were taken in the thickest part of the tumor. This was generally the central portion of the tumor, whether it was (a) plane, (b) exophytic, or (c) ulcerative lesion. Care was taken to insert the punch to its full depth in the tongue to include the tumor-invasive front. In highly exophytic lesions, such as one with a stem-like base, sampling the tumor at the junction between the tumor stalk and the normal tongue surface may optimize DOI measurement, but no highly exophytic lesions were included in this study’s patient population.
Histopathologic analysis of the punch biopsy specimens was performed after formalin fixation, standard paraffin embedding, 4-micron sectioning, and hematotoxin and eosin staining. pDOI was measured by drawing a reference line at the BM and drawing a “plumb line” perpendicular to the reference line, down to the tumor-invasive front into the stroma. The same method was used on both biopsy specimens and final glossectomy specimens (Figure 2).

pDOI measurement technique. (a) A line is drawn at the junction between the basement membrane and the epithelium (dotted line), and a plumb line (solid line) is then drawn perpendicular to the basement membrane to measure pDOI. (b) The tumor specimen shows the defect created by two punch biopsies taken at 90 angles to the tumor surface. The first punch was taken at partial depth (dash arrow), while the second one was taken at full depth (solid arrow). This picture illustrates that sampling with a punch biopsy may not be done at the deepest point of the tumor. Here, the point of maximal tumor depth was not sampled and is located at the double line arrow.
Statistical analyses were carried out using SPSS statistical software (version 29.0; IBM Corporation, Armonk, NY, USA). Descriptive statistics are expressed as mean ± standard deviation for continuous variables and as frequency (percentage) for categorical variables. Spearman’s correlation coefficients were calculated to compare bDOI to pDOI, cDOI to pDOI, and cDOI to bDOI. Secondarily, we aimed to evaluate bDOI and cDOI ability to predict final specimen pDOI as a dichotomous outcome variable with a cutoff ≥ 2 mm. This cutoff was chosen because of its clinical relevance; as mentioned above, for T1-T2, N0 OTSCC, the NCCN guidelines suggest that an END should be considered starting at DOI ≥ 2 mm (9). For predicting pDOI ≥ 2 mm, we performed receiver operating characteristic (ROC) curve analysis for bDOI and cDOI. We used Youden’s index to find the optimal cutoff point for both modalities. Based on the optimal cutoff point we calculated sensitivity, specificity, positive predictive value (PPV), negative predictive value (NPV), and accuracy of each modality. Confidence intervals (95% CI) were calculated using the Clopper-Pearson method. Finally, we calculated the sensitivity, specificity, PPV, NPV, and accuracy of bDOI for distinguishing invasive carcinoma from Tis. Missing values were omitted from the statistical analysis.
Results
Demographics
Twenty-seven patients were included in the study. Relevant demographic data and tumor specifications are summarized in Table 2. Most participants were male (67%). Patients’ ages ranged from 25 to 97 years old at the time of biopsy (mean = 67.2). Most of the biopsies were done in the operating room (OR) at the time of glossectomy, while only 7 were done in the clinic. For patients whose biopsy was done in clinic (most of which were T1 or Tis), the mean delay between bDOI and pDOI measurements was 43.2 days (±17.7). The vast majority of lesions (82%) were located on the lateral surface of the tongue, whereas the ventral and dorsal surfaces comprised only 11% and 7%, respectively. The average greatest diameter of the included lesions was 16.8 mm (±9.5). Overall, there were 7 Tis, 10 T1 lesions, 8 T2 lesions, and 1 T3 OTSCC. Perineural invasion and lymphovascular invasion were identified in 8 and 2 specimens, respectively. Biopsy specimen sampling was adequate in all cases with a full depth of invasion (DOI) reported. Biopsy was well tolerated by all enrolled patients. No significant bleeding occurred.
Patient Demographics and Tumor Characteristics.

Abbreviations: END, elective neck dissection; LN, lymph node; LVI, lymphovascular invasion; PNI, perineural invasion; SD, standard deviation.
Correlations Between DOI Measures
Scatterplots showing correlations between cDOI and bDOI, bDOI and pDOI, and between cDOI and pDOI, and corresponding Spearman’s correlation coefficients are shown in Figure 3. It appears that the lowest correlation coefficients were between cDOI versus bDOI, and bDOI versus pDOI, with values of 0.743 (95% CI: 0.484-0.887) and 0.603 (95% CI: 0.202-0.884), respectively. In comparison, the correlation coefficient between cDOI and pDOI was higher, at 0.894 (95% CI: 0.749-0.955).

Scatterplots showing the correlations between cDOI, bDOI, and pDOI. Scatterplots showing correlations between (a) cDOI and bDOI (b) bDOI and pDOI, and (c) cDOI and pDOI, with corresponding Spearman’s correlation coefficient. cDOI and pDOI show a stronger correlation.
Predicting pDOI ≥ 2 mm
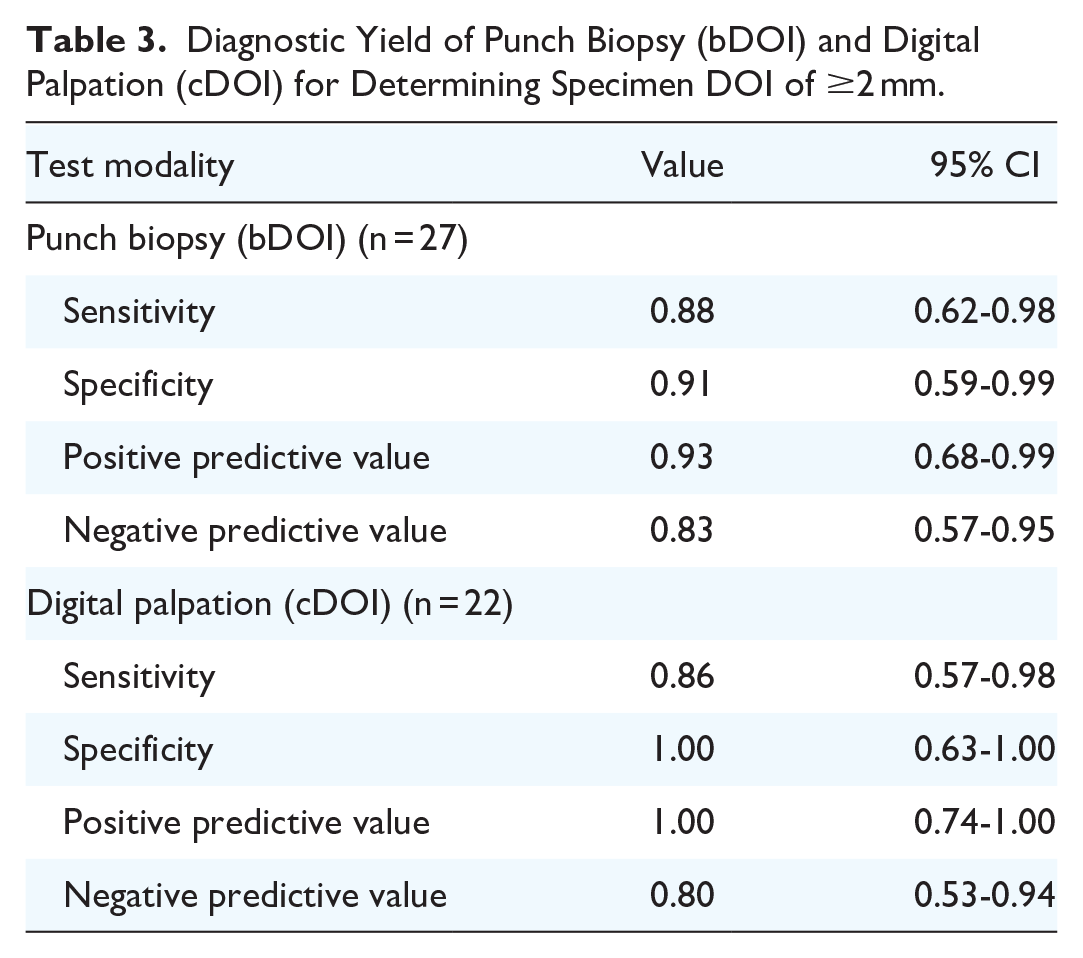
ROC curves for predicting pDOI ≥ 2 mm using both bDOI and cDOI are presented in supplement material (Figure S1). The areas under the ROC (AUROC) curve for bDOI and cDOI were 0.89 (95% CI: 0.72-1.06) and 0.93 (95% CI: 0.82-1.04), respectively. The point that maximized Youden’s index (0.78) for bDOI was at ≥1.5 mm, which corresponded to sensitivity, specificity, PPV, and NPV of 0.88 (95% CI: 0.62-0.98), 0.91 (95% CI: 0.59-0.99), 0.93 (95% CI: 0.68-0.99) and 0.83 (95% CI: 0.57-0.95), respectively. The point that maximized Youden’s index (0.86) for cDOI was at ≥3.5 mm, which corresponded to sensitivity, specificity, PPV, and NPV of 0.86 (95% CI: 0.57-0.98), 1.00 (95% CI: 0.63-1.00), 1.00 (95% CI: 0.74-1.00) and 0.80 (95% CI: 0.53-0.94), respectively. Diagnostic yields of both modalities are summarized in Table 3.
Diagnostic Yield of Punch Biopsy (bDOI) and Digital Palpation (cDOI) for Determining Specimen DOI of ≥2 mm.
Among 10 patients who underwent END, 2 (20%) had positive nodes. Those patients had final pDOI of 5 and 7 mm, respectively, and punch biopsy DOI was ≥ 2 mm for both (3 and 4 mm, respectively). Of the remaining 8 patients with negative nodes, 7 patients had pDOI ≥2 mm and were adequately classified as ≥2 mm based on bDOI, whereas 1 patient had pDOI <2 mm, and was also adequately classified as <2 mm based on bDOI.
Distinguishing Tis From Invasive Carcinoma
Table 4 presents the sensitivity, specificity, and predictive values of punch biopsy for distinguishing Tis from invasive carcinoma. As expected, the PPV of punch biopsy was high, at 0.94 (95% CI: 0.71-1.00), whereas NPV was 0.75 (95% CI: 0.35-0.97). The sensitivity and specificity were 0.89 (95% CI: 0.65-0.99) and 0.86 (95% CI: 0.42-1.00), respectively.
Diagnostic Yield of Punch Biopsy to Distinguish Tis From Invasive Carcinoma in Clinically Suspected Tis-T1-T2, N0 OTSCC (n = 25).

Discussion
Reliability of bDOI
To our knowledge, this is the first prospective proof of concept on the reliability of preoperative punch biopsy to predict pDOI in early OTSCC. The primary endpoint of the study was to assess the correlation between bDOI, cDOI, and pDOI. In light of our results, it appears that cDOI correlates better with pDOI than bDOI when these are considered continuous variables. However, when evaluating pDOI as a dichotomous outcome, with a threshold (here ≥2 mm) above which END should be considered, 9 both bDOI and cDOI appear to demonstrate promising diagnostic yield (AUROC 0.89 and 0.93, respectively). Interestingly, both patients with positive nodes had pDOI > 2 mm and were adequately classified as >2 mm based on bDOI. It should be noted, however, that the aim of this study was not to evaluate the association between bDOI and clinical prognostic outcomes, such as nodal positivity. Future studies on larger cohorts to explore these associations are warranted.
In the current literature, few studies have evaluated the reliability of bDOI, and most did not use a standardized punch biopsy technique. The 2 most important considerations when measuring bDOI are to take the sample in a representative area of the tumor and to include the tumor-invasive front in the biopsy sample. Dhanda et al. 20 suggested that a biopsy depth of 10 mm was required to include the tumor-invasive front in at least 79% of tumors. Their retrospective review of 139 biopsy samples from various oral cavity subsites showed that only 28% of punch biopsies had a biopsy depth greater than tumor depth. This may be partially attributed to anatomical constraints in other oral cavity subsites such as the floor of the mouth and alveolar crests. In another retrospective study by Moor et al., 18 bDOI could not be measured in 16 of 36 (47%) included biopsy specimens as tumors extend to the full depth of the specimen. The biopsy technique was not described and presumably not standardized. In our study, all samples were deep enough to measure bDOI, as they included the deep tumor-stroma junction. This was the result of the standardized technique used and the depth of the selected punches. Therefore, the inclusion of the tumor-invasive front could not have affected the results and cannot explain the relatively low correlation between bDOI and pDOI.
Potential injuries to Warthin’s duct, the floor of mouth structures and airway compromise may be concerns when using a deep punch. 20 Our study focused only on oral tongue tumors where deep punches are generally safe. Although this was not a primary objective of the present study, and the number of included patients is low, punch biopsy in the clinic was not harmful, even when taken at a maximal punch depth of 12 mm. No patient experienced significant bleeding, nor required electric cauterization. Some required silver nitrate and a suture.
Sampling the most invasive part of the lesion may also present a challenge. In our samples, the mean greater diameter of included tumors on final histopathology was 16.8 mm. A 3- or 4-mm punch diameter represents 18% to 24% of this tumor diameter. Therefore, the site of maximal DOI may not be adequately sampled by such a small punch (See Figure 2b). Overall, sampling of the tumor-invasive front outside of the site of maximal tumor depth may represent a major confounder. It may limit punch biopsy’s representativeness of pDOI and explain the low correlation. In the current study, no difference between 3- and 4-mm punch was noted.
Of note, the pathologist (author OEG) never had difficulty finding the level of the BM even if the peripheric normal tissue was not included in the biopsy sample and was always able to report the full DOI.
Another potential factor that can explain our results was the delay between bDOI and pDOI measurements for patients whose biopsy was not taken in the OR. Tumor growth during that time confounds the correlations. Taking biopsies in the clinic may reduce the reliability of bDOI, but it maximizes the external validity and clinical applicability of our findings.
In the clinic, infiltration with xylocaine and epinephrine may also have distorted the tumor anatomy and artificially increased bDOI. Furthermore, tumor shrinkage is a well-studied phenomenon that may arise during the process of formalin fixation and paraffin embedding. Studies suggest that shrinkage can reduce tumor dimensions between 10% and 24%.21,22 The impacts of xylocaine infiltration combined with tumor shrinkage have not been evaluated in the present study. Of note, cDOI was estimated by clinicians before xylocaine infiltration, which could explain why the correlation between cDOI and pDOI was greater than that of bDOI and pDOI. Because the included population was small, no significant subgroup analysis has been performed on patients who were not infiltrated before the punch biopsy.
Another potential confounder was that some biopsies were taken in tumors that had already been biopsied, where scarring and fibrosis might thicken tumoral tissue. Again, these tumors were not excluded in an effort to maximize the external validity of this study, as many tumors arise in areas of previous premalignant changes that may have been biopsied multiple times.
In recent years, systematic reviews and meta-analyses (SRMA) have been published on radiologic measurement of DOI (rDOI) or tumor thickness (rTT). In an SRMA conducted by our group on preoperative modalities for measuring DOI, it seemed MRI was the best imaging modality with the highest pooled correlation coefficient with pDOI (0.86 [95% CI: 0.82-0.88]). 23 US has mostly been used to measure rTT. In that same SRMA, it was found that there were only 2 retrospective studies evaluating the use of preoperative biopsy to measure DOI. Moor et al. 18 did not specify what technique was used for biopsy but reported a moderate correlation coefficient with pDOI of 0.51. Almangush et al. 19 looked at incisional biopsies. They did not report any correlation coefficient. However, like us they dichotomized pDOI, but at a threshold of ≥4 mm, whereas we dichotomized at ≥2 mm, and they reported sensitivity of 0.77 and specificity of 0.77 of bDOI (compared to 0.88 and 0.91, respectively, in our study). In another recent SRMA on the use of US to measure rDOI or rTT, Klein Nulent et al. 24 found a pooled Pearson correlation coefficient of 0.88 (P < .001) between US measures and histopathology measures. In another SRMA, Tarabichi et al. 25 found a higher pooled correlation coefficient of 0.95 (P < .0001). As for MRI, an SRMA by Li et al. 13 obtained an overall inter-class coefficient of 0.89 (95% CI: 0.84-0.90) between rTT or rDOI and pTT or pDOI. Overall, these correlation coefficients are considerably higher than the ones we obtained with punch biopsy (bDOI vs. pDOI coefficient of 0.603 (95% CI: 0.20-0.88)). Interestingly, they do compare with clinical palpation of the tumor, as reflected by our 0.894 (95% CI: 0.75-0.96) coefficient between cDOI and pDOI.
Distinguishing Tis From Invasive Carcinoma
A secondary aim of this study was to evaluate the reliability of punch biopsy to distinguish Tis from invasive carcinoma. To our knowledge, this subject has not been studied prospectively in the literature. Central sampling of the tumor with a deep punch biopsy may be a reliable tool to rule out the presence of invasion and prevent surgeons from resecting larger margins than necessary when proceeding with excision. Maximizing the sensitivity of punch biopsy is of the utmost importance to adequately rule out the presence of invasive carcinoma when the biopsy shows only Tis. This allows the surgeon to not only take narrow excision margins but also to potentially vaporize the lesion instead of proceeding with wide local excision. In the present study, the sensitivity of the punch biopsy was 0.89 (95% CI: 0.65-0.99). This suggests that a central, deep punch biopsy may be a good tool to rule out the presence of invasion, but a larger sample size would be necessary to reduce the confidence interval and validate this hypothesis. Of note, the punch biopsy in 1 patient suggested invasive carcinoma, while the final specimen showed only Tis. It is possible that the only area of invasive carcinoma was excised by the biopsy. This explains why the PPV is less than 100% in the present study.
We still advise that any lesion showing significant macroscopic changes, induration, ulceration, rapid growth, or any other clinical signs of invasion should be excised rather than vaporized. Final histopathologic analysis remains the gold standard to distinguish Tis from invasive lesions. In the future, larger studies could ascertain the role of punch biopsy to adequately rule out neoplasm invasiveness.
Finally, some studies that measured rDOI and rTT have previously focused on correlating radiologic measurements not with pDOI or pTT, but directly with other prognostic factors.26,27 Few studies on oral cavity punch biopsy’s reliability to predict nodal metastasis have been published. Seki et al. 28 correlated a bTT of more than 3 mm with lymph node metastases (OR: 47.5, 95% CI: 3.9-511.2), making it an interesting prognostic tool, irrespective of the correlation between bTT and pTT. Since then, DOI has surpassed TT as a prognostic factor.1,4 A direct correlation between bDOI and the number of positive lymph nodes on END would be the most clinically useful tool to implement the use of bDOI in clinical practice. In the present study, only 10 patients had an END, of which only 2 had a single positive lymph node on final histopathology. These patients had a bDOI of 3- and 4-mm and a pDOI of 5- and 7- mm, respectively. No significant subgroup analysis can be made yet to correlate bDOI to clinical prognostic factors.
Conclusions
To our knowledge, this study is the first prospective study on punch biopsy’s reliability to measure pDOI in early OTSCC. It is the first proof-of-concept using standardized deep central punches to measure bDOI. Even though a standardized biopsy technique allowed a deep enough tumor sampling, bDOI did not correlate strongly with pDOI. However, it demonstrated a high diagnostic yield for distinguishing lesions with a pDOI of ≥2 mm from those with less. Clinical palpation by experienced surgeons correlated better with pDOI and was comparable to the limited data on radiologic measurement of DOI. In the present study, xylocaine infiltration, tissue manipulation, delay between biopsy and final pDOI measurement, and sampling outside the deepest tumor site may explain the results. Interestingly, punch biopsy’s sensitivity to rule out the presence of invasiveness in Tis was 0.89. It should be noted that this was a pilot proof-of-concept study, and results should be considered as hypothesis-generating for future larger studies. Subsequent studies on this subject may also directly correlate bDOI with clinical outcome measures, as this could prove whether the use of punch biopsy could effectively alter preoperative treatment planning. Larger studies are needed before recommending its use to decide whether elective neck dissection is necessary in early OTSCC and whether it can reliably rule out focal invasiveness in Tis.
Supplemental Material
sj-docx-1-ohn-10.1177_19160216251321452 – Supplemental material for Punch Biopsy for Preoperative Depth of Invasion Assessment in Early Oral Tongue Squamous Cell Carcinoma: A Prospective Pilot Study
Supplemental material, sj-docx-1-ohn-10.1177_19160216251321452 for Punch Biopsy for Preoperative Depth of Invasion Assessment in Early Oral Tongue Squamous Cell Carcinoma: A Prospective Pilot Study by Béatrice Voizard, Gabriel S. Dayan, Olguta-Ecaterina Gologan, Tareck Ayad, Eric Bissada, Louis Guertin, Paul Tabet, Guillaume B. Cardin, Laurent Létourneau-Guillon and Apostolos Christopoulos in Journal of Otolaryngology - Head & Neck Surgery
Footnotes
Acknowledgements
We sincerely thank all the patients who participated in this study. We acknowledge Dr Nadim Saydy’s contribution to the illustrations (Figure 1).
Author’s Contribution
Béatrice Voizard, MD, MSc, FRCSC: Study design, Data collection, Manuscript redaction, Manuscript revision. Gabriel S. Dayan, MD: Data collection, Statistical analysis, Manuscript redaction, Manuscript revision. Olguta-Ecaterina Gologan, MD, FRCPC: Data collection, Manuscript redaction, Manuscript revision. Tareck Ayad, MD, FRCSC: Data collection, Manuscript revision. Eric Bissada, MD, DMD, FRCSC: Data collection, Manuscript revision. Louis Guertin, MD, FRCSC: Data collection, Manuscript revision. Paul Tabet, MD, MSc, FRCSC: Data collection, Manuscript revision. Guillaume B. Cardin, MSc: Data collection, Manuscript redaction, Manuscript revision. Laurent Létourneau-Guillon, MD, MSc, FRCPC: Statistical analysis, Manuscript redaction, Manuscript revision. Apostolos Christopoulos, MD, MSc, FRCSC: Study design, Data collection, Manuscript revision.
Consent to Participate
Written informed consent was obtained from all participants at the time of recruitment. This was approved by the IRB.
Consent for Publication
Although there are no individual identifiable participant details, images, or videos, consent for publication was obtained with each participant, at the time of recruitment.
Data Availability
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the Dr Azar-Angelil Research Chair in Head and Neck Oncology of the Université de Montréal (chairholder Dr Apostolos Christopoulos).
Ethical Considerations
This study was performed in accordance with the Declaration of Helsinki. This study was approved by the institutional review board of the Centre Hospitalier de l’Université de Montréal (CRCHUM IRB#: 18.296; January 31, 2019). Written informed consent was obtained from all participants.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
