Abstract
Introduction:
Vincristine (VCR) is a chemotherapeutic agent used widely in the treatment of hematologic and solid tumors, known to result in neurotoxicity, especially with cumulative administrations. Bilateral vocal fold palsy (VFP) is a rare but life-threatening complication of VCR. We report 2 patients with hepatoblastoma presenting with stridor following VCR treatment and propose a management plan.
Methods:
Electronic medical records of oncology patients treated at a tertiary hospital with VCR-induced VFP were reviewed. Literature review was performed in PubMed using the terms: hoarseness, VFP, stridor, vincristine.
Results:
A total of 23 children with VCR-induced VFP were identified from the literature review and adding on our 2 cases. Seventeen (77.3%) were male and 5 (22.7%) were female. The median presenting age was 36.0 months (5-204 months). Acute lymphoblastic leukemia, 15 of 23 (65.2%), was the most common malignancy. Eighteen patients (78.3%) had bilateral VFP and 5 (21.7%) had unilateral VFP. The mean time to VF function recovery was 167.3 days (median: 200.5 days, range: 7-270 days) in the intervention group versus 72.1 days (median: 31.5 days, range: 3-240 days) in the conservative group. One patient in the intervention group had persistent VFP. Sixteen patients (69.6%) were observed, 4 (17.4%) underwent tracheostomy, 1 (4.35%) was intubated, 1 (4.35%) underwent cordectomy, and 1 (4.35%) required positive pressure support. Vincristine was restarted in 12 patients (54.5%), of which 4 developed recurrence of airway symptoms and had to stop VCR.
Conclusion:
A new-onset hoarseness or stridor in a child on VCR should raise the suspicions of VFP. The assumption of an upper respiratory–induced hoarseness or stridor should be avoided. Decisions regarding readministration of VCR and possible airway interventions should be made via a multidisciplinary team approach.
Introduction
Vincristine (VCR) is a chemotherapeutic agent belonging to the family of vinca alkaloids. Since the early 1960s, it has been used widely in the treatment of hematologic malignancies and solid tumors. 1 However, it is well known for its neurotoxicity, and previous literature reports have demonstrated a cumulative effect. 2 The incidence of neuropathy due to VCR is clinically significant among pediatric patients, with some studies reporting up to an incidence of 86.5%.3,4 Vinca alkaloids act as mitotic inhibitors by binding to the protein component of microtubules. The neurotoxicity of VCR has been postulated to be a result of binding with the tubulin, resulting in disruption of the microtubule assembly, axonal transport, and secretory function.5,6 Vincristine-associated complications can be categorized into peripheral neuropathy, autonomic dysfunction, and cranial neuropathy. Very rarely, central neurotoxicity manifesting as encephalopathy seizures and syndrome of inappropriate antidiuretic hormone secretion can occur. 7 The presence of preexisting liver dysfunction, hereditary motor sensory neuropathy (Charcot-Marie-Tooth syndrome), 8 and concomitant use of drugs such as azoles can potentiate the problem. 9
There are several clinical reports of using pyridoxine, pyridostigmine, and glutamic acid to alleviate the VCR-associated neurotoxicity. Baker and Lipson reported a complete recovery of VCR-induced neuropathy using pyridoxine, levocarnitine, and N-acetylcysteine. 10 Despite the exciting promise of proposed drugs and therapies, there is a lack of randomized controlled studies on treatment options available to treat VCR-induced neuropathy.
This is the first study describing VCR-induced vocal fold paralysis (VFP) in children with hepatoblastoma. Most studies have recommended discontinuing VCR when clinical symptoms are suspicious of VFP and it should be reintroduced at reduced dosage and frequency. 11 The goal of this study is to highlight the controversies in the management of children with VCR-associated VFP and provide the first guidelines for airway intervention and the reintroduction of VCR.
Methods
A comprehensive literature review was performed using PubMed/MEDLINE, SCOPUS, and using keywords “vocal cord paralysis,” “vocal fold paralysis,” “recurrent laryngeal nerve paralysis,” “pediatrics,” “children,” and “vincristine.” Three studies were excluded in the review (Oman, Yasmin, and Argawal). The articles that were excluded did not include documentation of vocal fold status on flexible scope at diagnosis and at symptom resolution. Two patients diagnosed with hepatoblastoma who developed VFP following treatment with VCR were identified and described.
Results
Case Reports
Patient 1
An 18-month-old boy was diagnosed with stage 3 hepatoblastoma with lung metastases following presenting symptoms of weight loss, lethargy, and an abdominal mass. He was started on a chemotherapy protocol including cisplatin, VCR, fluorouracil, and doxorubicin. After 7 doses of VCR at 1.5 mg/m2, he developed tachypnea, increased work of breathing, and was diagnosed to have metapneumovirus bronchiolitis. Otolaryngology was consulted due to persistent tachypnea and increased work of breathing. Flexible laryngoscope performed at bedside showed bilateral vocal fold palsy (VFP). Over the next few days, the patient was supported with racemic epinephrine, nebulized levalbuterol, and intranasal oxygen. The subsequent dose of VCR was withheld in view of the newly diagnosed VFP. A repeat flexible laryngoscope was performed 1 month after initial evaluation. At the time of examination, the child was well with no stridor and retractions. The flexible laryngoscope demonstrated improved vocal fold movements bilaterally. The child was restarted on VCR at 50% reduced for 2 doses. A few days later, the child was reported to have coughing and choking with oral intake. Ear Nose and Throat (ENT) was called to perform a repeat scope, which showed mildly reduced vocal fold movements bilaterally. Vincristine was omitted for subsequent cycles because of new-onset peripheral neuropathy.
Patient 2
A 2-year-old boy was diagnosed with hepatoblastoma with pulmonary metastasis after presenting with increasing abdominal mass. After 5 doses of VCR (1.5 mg/m2), he presented with peripheral neuropathy, stridor, and shortness of breath. Otolaryngology consult was made and a bedside flexible laryngoscope confirmed bilateral VFP. In view of the clinical symptoms and recent flexible laryngoscope findings, the next dose of VCR was withheld. Postoperatively, after his hemi-hepatectomy, he was unable to be successfully extubated. After a multidisciplinary discussion, decision was made to perform a tracheostomy. Vincristine was omitted from all of the subsequent chemotherapy cycles. His last ENT flexible laryngoscope showed improved vocal fold mobility. The patient was decannulated after completion of chemotherapy, with no subsequent VCR administered.
Literature Review
Over the past 28 years, there were 10 studies that met criteria and were included in the review. The demographics, diagnosis, airway intervention, swallowing dysfunction, time to symptom resolution, associated peripheral neuropathy, and VCR reintroduction were summarized (Table 1).1,8,11-18
Summary of Patients With VCR-Induced VFP.
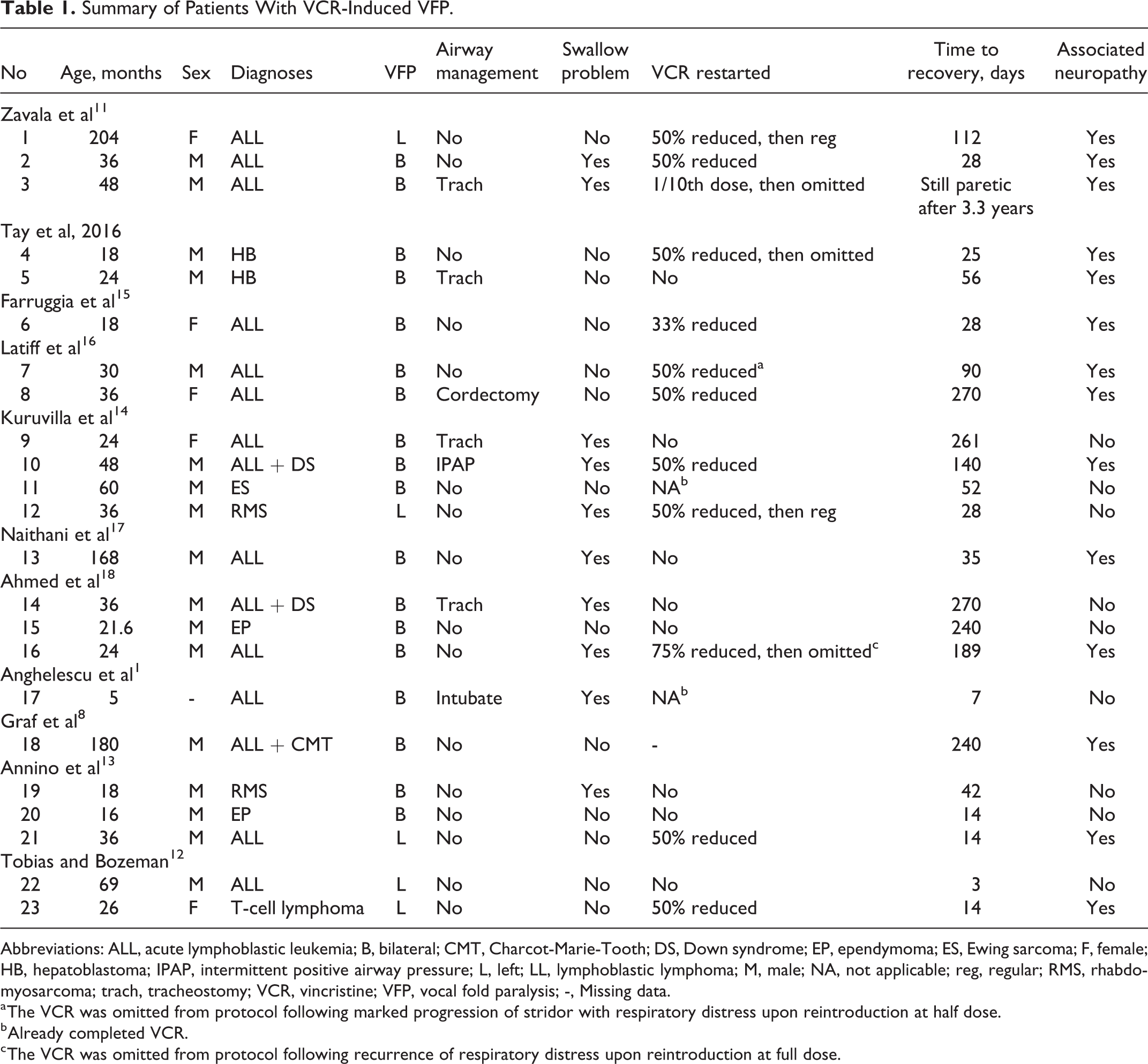
Abbreviations: ALL, acute lymphoblastic leukemia; B, bilateral; CMT, Charcot-Marie-Tooth; DS, Down syndrome; EP, ependymoma; ES, Ewing sarcoma; F, female; HB, hepatoblastoma; IPAP, intermittent positive airway pressure; L, left; LL, lymphoblastic lymphoma; M, male; NA, not applicable; reg, regular; RMS, rhabdomyosarcoma; trach, tracheostomy; VCR, vincristine; VFP, vocal fold paralysis; -, Missing data.
a The VCR was omitted from protocol following marked progression of stridor with respiratory distress upon reintroduction at half dose.
b Already completed VCR.
c The VCR was omitted from protocol following recurrence of respiratory distress upon reintroduction at full dose.
Demographics
Seventeen patients (77.3%) were male and 5 (22.7%) were female. The median age at presentation was 36.0 months (mean: 51.4 months, range: 5-204 months). Acute lymphoblastic leukemia (ALL) was the underlying diagnosis in 15 patients (65.2%). The rest had hepatoblastoma, 2 (8.70%) of 23, ependymoma, 2 (8.70%) of 23, rhabdomyosarcoma, 2 (8.70%) of 23, Ewing sarcoma, 1 (4.35%) of 23, and T-cell lymphoma, 1 (4.35%) of 23. Out of the 15 patients with ALL, 2 of them also had Down syndrome and 1 of them had Charcot-Marie-Tooth disease.
Type of VFP, Swallowing Issues, and Associated Peripheral Neuropathy
Eighteen (78.3%) of 23 patients had bilateral VFP and 5 (21.7%) of 23 had unilateral VFP. All of them had left VFP. The mean time to recovery of the VF function was 98.1 days (median: 47 days, range: 3-270 days). Only 1 patient had persistent cord palsy. There were 10 patients (40.0%) with swallowing dysfunction. Nine out of the 10 patients had bilateral VFP, and 1 out of 10 patients had left VFP. Fourteen (60.9%) out of 23 patients had concomitant peripheral neuropathy.
Airway Intervention
Sixteen patients (69.6%) were managed with close monitoring. Four (17.4%) out of 23 patients required tracheostomy, 1 (4.35%) of 23 patient was intubated, 1 (4.35%) of 23 underwent cordectomy, and 1 (4.35%) of 23 was treated with positive pressure support. All the patients who required surgical intervention, intubation, or positive pressure support had bilateral VFP. The mean time to recovery of VF function was 167.3 days (median: 200.5 days, range: 7-270 days) for the group of patients who received intervention versus 72.1 days (median: 31.5 days, range: 3-240 days) in the group managed conservatively. One patient in the intervention group had persistent VFP.
Vincristine Administration
Vincristine was stopped completely in 8 patients (36.4%). Twelve patients (54.5%) had VCR restarted after a period of holding. Four (33.3%) of these 12 patients had to stop VCR after restarting because of recurrence of airway symptoms. Two patients (9.09%) had already completed treatment when VFP developed.
Discussion
Our first patient had recurrence of symptoms when restarted on VCR at 50% reduced dosage, and VCR was omitted for subsequent cycles because of new-onset peripheral neuropathy. In our second patient, we decided to perform tracheostomy because of his bilateral VFP following VCR administration and he was unable to tolerate extubation following his hepatectomy. The prognosis of both patients had remained unchanged despite decision to stop VCR. We would like to emphasize in this publication that the management strategy should be individualized based on clinical presentation of the patients and keeping in mind the diversity in symptoms and at times the nonproportional severity of airway symptoms in relation to the glottal gap observed in patients with bilateral VFP.
Bilateral VFP is a rare but potentially life-threatening complication of VCR treatment. It may occur as an isolated complication or in one-third of patients with other manifestations of neurotoxicity. 15 Withdrawal of the drug usually leads to resolution of symptoms. However, there have been varied outcomes on reintroduction of the drug. Based on a review by Latiff et al, at half dose, only 1 out of 6 cases had recurrence of symptoms. 16 Vincristine is commonly used in pediatric oncology regimens due to its low myelotoxic effects and proven efficacy. In our review of the literature, patients with ALL (15/23) were the most reported to have VFP. Children with concomitant medical conditions such as Down syndrome and other preexisting neurologic diseases may be more at risk of developing complications. Both of the patients with ALL and Down syndrome in this review required airway intervention. The administration of VCR in this group will require close monitoring and clinicians should maintain a high index of suspicion. 14
It has been shown that the toxicity level of VCR has a cumulative effect. High dosage (>2 mg/m2/wk) and frequent dosing (more than once a week) increase the toxicity. There are currently no proven methods of reducing the toxic effects of VCR. Folinic acid and pyridoxine have been tried previously but failed to have any protective effect.19,20
We suspect that VFP is underdiagnosed and recommend that all children who are receiving VCR and developed airway and swallowing difficulties should be evaluated for VFP. Immunocompromised child on chemotherapy are at a high risk of developing upper and lower tract respiratory infections. If the patient’s respiratory symptoms seem out of proportion to their illness, or they do not recover as anticipated, evaluation for VFP should be strongly considered.
From our review of the literature, it is concluded that 60.9% of patients with VCR-related VFP had concomitant peripheral neuropathy, and there should be a low threshold to consult otolaryngology for evaluation of VFP in pediatric oncology patients with peripheral neuropathy.
Most of the VFP seems to be reversible with full recovery following cessation of VCR. Only 1 patient in the review had persistent VFP. Given the temporary nature of the condition, supportive measures may be preferred over invasive procedures such as cordotomy or tracheostomy. From this review, only 4 (17.4%) out of 23 patients required tracheostomy, 1 (4.35%) of 23 patient was intubated, 1 (4.35%) of 23 underwent cordectomy, and 1 (4.35%) of 23 was treated with positive pressure support. Early recognition and discontinuing the use of VCR can prevent the worsening of the airway problems and further damage to the lower airways.
Here, we provide the first set of guidelines for assessing and managing VCR-induced VFP in children. First, assess VF status with a bedside flexible laryngoscope and document VF mobility prior to restarting VCR. If VCR needs to be restarted, administer at a 50% dose reduction. Following the readministration of VCR, carefully monitor for signs and symptoms of VFP, with a low threshold to perform a repeat flexible laryngoscope. As the majority of children in the review did not require tracheostomy or intubation, we recommend beginning with a conservative approach. (Evidence level 4) For each case of VCR-associated toxicity, the risk to benefit ratio has to be carefully considered before discontinuation of the drug (Figure 1).
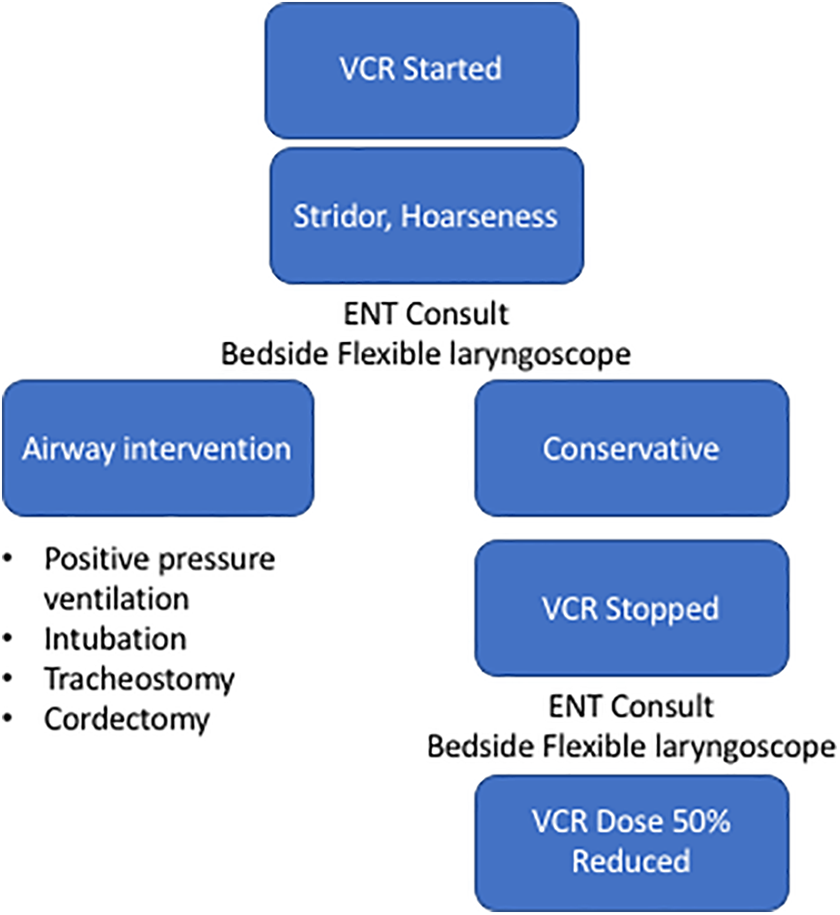
Management guidelines for oncology patients with vincristine (VCR)-induced vocal fold paralysis (VFP).
Conclusion
We recommend an otolaryngologist evaluation of any pediatric oncology patients receiving VCR who presents with stridor, increased work of breathing, or dysphonia that is not consistent with the typical infectious course, prior to continuation of VCR. In addition, pediatric oncology patients who develop VCR-induced peripheral neuropathy should be clinically evaluated for VFP.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
