Abstract
Introduction:
The most basic question to be answered in each case in which the choice of using a pressure equalization tube (PET) is being considered is: “what is the underlying pathophysiology of the middle ear disease being addressed?”
Methods:
We will evaluate the hypothesis that the Eustachian Tube (ET) may become “dysfunctional” due to allergic mucosal edema and obstruction. We review the literature that evaluates the role of ET, the proposed affect that allergy may contribute to ET dysfunction (ETD), and the relation of allergic rhinitis to otitis.
Results:
Proof that allergy affects the middle ear was supported by (1) over a dozen investigators using objective immunotherapy demonstrating over the past 70 years that 72% to 100% of the children with otitis media with effusion (OME) are atopic, (2) an association of allergic Th2 immune-mediated histochemical reactivity within the target organ itself, (3) establishment that inflammation within the middle ear is truly allergic in nature, and (4) direct evidence of a dose–response curve and consistency of results, which confirm that OME resolves on allergy immunotherapy.
Conclusion:
Current medical evidence should heighten the awareness of physicians of the physiology that underlies ETD. The evidence supports the link between allergy and OME. The middle ear behaves like the rest of the respiratory tract, and what has been learned about the atopic response in the sinuses and lungs may be applied to the study of the immunologic mechanisms within the middle ear that lead to ETD requiring the use of PET.
Keywords
Introduction
Recurrent middle ear infections have been a scourge of humanity for millennia, as they often bode chronic hearing loss or worse—mastoidits that most often resulted in death. Major advances began with Astley Cooper’s pioneering operation of piercing the human membrane tympani as written up in the Royal Society’s publication, Philosophical Transactions, in 1801. Secretory otitis media (OM) was first described by Politzer in 1869. Observations as to its incidence, etiology, pathology, and therapy were reported with increased frequency through the 1950s and 1960s following the advent of antibiotics in the 1940s and the development of tympanostomy tubes by Shea in 1952. 1
Pressure equalization tubes (PET) or tympanostomy tubes have been a remarkable advancement, and many efforts to improve upon their design, material, and application have provided significant advances in the treatment of Eustachian tube (ET) dysfunction (ETD) and its concomitant middle ear pathology. Yet those efforts seem to have reached an asymptote in effectiveness. The most basic question that must be answered in each case in which the choice of using a PET is considered is: “What is the underlying pathophysiology of the middle ear disease being addressed?” This article will present a most concise review of such considerations.
The basic function of a PET is to create a bypass of the ET so as to aerate the middle ear cavity. But to what can we attribute the underlying ETD? Damm notes that “besides infection-related adenoid hypertrophy, the differential diagnosis should include ciliary dysfunction, chronic rhinosinusitis, craniofacial malformations, gastroesophageal reflux, 2 tumors and cancer treatment in the nasopharynx and in particular allergies.” 3 Small mastoid cavities seen on X-ray suggest the involvement of the whole middle ear cleft in OM. 4 During chronic otitis media with effusion (cOME), mastoid growth is slowed, but mastoid development can be restored if aeration by ventilation tube therapy is instituted early. 5
Otitis media is an inflammatory disease involving the presence of noninfective fluid within the middle ear space persisting for greater than 3 months. It is one of the most prevalent childhood diseases across the globe. 6,7 Chronic suppurative OM incidence rate is of 4.76% or 31 million cases, with 22.6% of cases occurring annually in children under 5 of age. 8 Otitis media-related hearing impairment has a prevalence of 30.82 per 10 000. Each year 21 000 people die due to complications of OM. 6
Role of the ET
Eustachian tube dysfunction, either extrinsic or intrinsic, has been regarded for decades as the underlying pathophysiologic event that leads to chronic middle ear disease. Obstruction of the ET in humans has been clearly demonstrated to result from antigen challenge. 9 The ET has been shown to be involved functionally and morphologically in type I reactions of the nose. 10 In a double-blind protocol, Friedman et al 11 demonstrated that intranasal pollen challenge of atopic individuals produced allergic rhinitis (AR) followed by ET obstruction, which did not occur with the placebo. The allergic reaction inhibited even transient dilations of the ET during swallowing. Several other double-blind protocols with intranasal allergen or histamine challenge produced similar severe functional obstruction of the ET. 12 All this confirms the hypothesis that the ET may become “dysfunctional” due to allergic inflammation causing intrinsic mucosal edema and obstruction. Skoner concluded that “Allergen-induced blockage subverts the normal mechanism of gas exchange between the middle ear and the environment, thus setting the stage for development of middle ear underpressured OME.” 13
Patients with symptoms related to obstructive ETD are often young children who may have only mild or no additional allergy symptoms. Consequently, many physicians do not investigate the possibility of allergic disease as an etiological factor in ETD, with or without cOME. The reported incidence of allergy being related to ETD and/or OME, as determined by allergy testing, ranges from 15% to 93% in children up to 18 years and up to 35% among adults. 12,14 Meta-analysis of clinical evidence suggests a strong correlation of AR and OME among children. 15 Exposure to allergen challenge has consistently resulted in a dose-dependent decrease in ET patency, regardless of whether a seasonal (ragweed) or a perennial (dust mite) allergen challenge was used. 9 Evaluation of the records of 2.4 billion children found allergy to be associated with a 2- to 4.5-fold increased incidence of OME. 16 Modern research in immunology leading to the application of allergy immunotherapy (AIT) to refractory cases of OME as proposed in 1990 by Hurst 17 has been extremely effective in curing chronic OME in both children and adults. 18
Certainly, there are some genetic and environmental predispositions as seen among Australian Aborigine people and Native Inuits of Greenland that account for the unusual high incidence of cOME in these populations. They present unique situations and are not the focus of this article.
Several critical questions require answers: “Why is it that 5-10% of patients with acute otitis media progress to chronic OME despite adequate antimicrobial therapy?” 19 “Why, despite a reduction of 50% to 70% of acute otitis media(AOM) cases after the introduction of Pneumococcal vaccines has there been no significant change in the incidence of OME?” “Why despite having had an adenoidectomy do children continue to get OME?” “Why do 20% of children require a second set of tympanostomy tubes or develop otorrhea?” 20 “Why do some children with no obvious viral or bacterial infection develop OME?” “Why, despite positive cultures, are antibiotics no more effective than placebo in patients with cOME?” 21 “Why do patients with OME have 4 to 5 times the expected incidence of allergies?” 10,21 -23 “Why is OME more typical of older children who have reached an age at which they would have been expected to have outgrown an immature ET morphology?” “To what degree then is allergy a risk factor?” “If allergy itself is not the ‘actual cause’ of an effusion, is it more than a mere ‘contributing factor’ or is allergy a catalyst essential to the formation of persistent effusion?”
The short answer to these questions requires an understanding of the pathophysiology of mucous membrane itself. There are many similarities in the pathophysiology of cOME and allergic sinusitis. The middle ear space is an anatomic extension of the upper airway by way of the ET, and because the middle ear is capable of mounting an inflammatory response similar to other areas of the respiratory tract, it has been proposed that the middle ear is part of the Unified Airways Concept. 24 Both the middle ear and the sinuses are largely bony cavities lined by similar pseudostratified, ciliated, columnar, or cuboidal epithelium, and both have been found to have a similar response to allergic challenge. The middle ear has been shown to have degranulating mast cells and eosinophils, 25 just as in the sinuses.
Shubich showed that patients with OME and allergies who had allergy treatment had half as many infections while their tubes were in place and required half as many replacements of tympanostomy tubes as the group with non-treated allergy. 26 Irander et al found that among 54 Swedish infants, 38% with OME had respiratory tract allergy. 27 Infants with allergy symptoms were 5 times more likely to develop OME than nonatopics. Jero et al found similar results in Finland where allergy posed a risk factor of 4.4 for children failing to clear an acute otitis. 28
Otitis media with effusion is a multifactorial disease, of which allergy is only one risk factor. The theory that allergy is an important factor in the development and maintenance of OME is elegant in its simplicity. It is a theory that seems to tie in the various, seemingly unrelated risk factors that are known to be associated with this disease, namely, a history of previous AOM, rhinitis or asthma, family smoking, family history of atopy, snoring, and/or an associated viral infection. 29 -31 The risk of OME increases 2.8 to 4.2 times if parents smoked 2 or 3 packs of cigarettes. Children were 2.2 times more likely to be atopic to foods if exposed to parental smoke, and 1.8 to 2.2 times more atopic than controls if exposed to cats or dust mites. 31 Allergy adds unique comorbidity and is by far a greater risk factor than other identified factors, conferring a 2- to 4.5-fold increased incidence of OME compared to incidence of OME in nonallergic people. Thus, a child who gets an episode of AOM is up to 3 times more likely to develop OME if that same child is also allergic.
In people with certain inherited genetic factors, their mucosa is programmed to respond to environmental allergens (dust, pollen, molds, and pets) by making antibodies. In nonallergics, this mucosa cannot be sensitized to normally occurring substances, but in the mucosa of allergic individuals the cells are genetically programmed to be hyperreactive and will often respond to allergen exposure by producing various inflammatory mediators. The mucous membrane lining the nose, sinuses, middle ear, ET, and lung all have the potential to act in the same way, and all may become hyperactive in allergic people. The hyperreactive cells of allergic mucosa puts a patient at risk for recurrent sinus infections, as it adds to conditions that can lead to an environment that is suitable for mucostasis, bacterial overgrowth, and chronic inflammation. 32 It has been reported that among 97 patients with OME, 62% had documentation of additional atopic signs and symptoms, including asthma 22%, AR 48%, eczema 4%, and chronic nasal congestion 8%. Importantly, 38% had otitis as their only allergy symptom, perhaps resulting from locally produced immunoglobulin E (IgE). 25
There remains a diversity of opinion within the medical community as to whether or not OME is related to allergy. In part, this information has not been collated. In part, otologists simply are unaware of the humoral mechanisms of Th2 immune hypersensitivity reactions. In part, there is confusion in use of the terms “allergy” which refers to “symptomatology” of asthma, rhinitis, or otitis with “atopy” which is a “sensitivity” as reflected by elevated IgE antibodies to various allergens without the patient necessarily having symptoms present. In part, many patients are tested by the low sensitivity skin prick test (SPT) with its 50% false-negative rate rather than by the more sensitive intradermal testing (IT). The Cochrane meta-analysis by Griffin and Flynn 33 analyzed the data from several randomized controlled trials. These trials assessed the outcome from using antihistamines, decongestants, or antihistamine/decongestant combinations as treatment for OME in children. Outcomes such as tympanometry and physician diagnosis of OME were not improved by using these medications. Thus, a disparity arises in that the usual treatments for AR such as intranasal steroids do not seem to be as effective against chronic ETD as for nasal and sinus complaints, as acknowledged in the current guidelines. 34
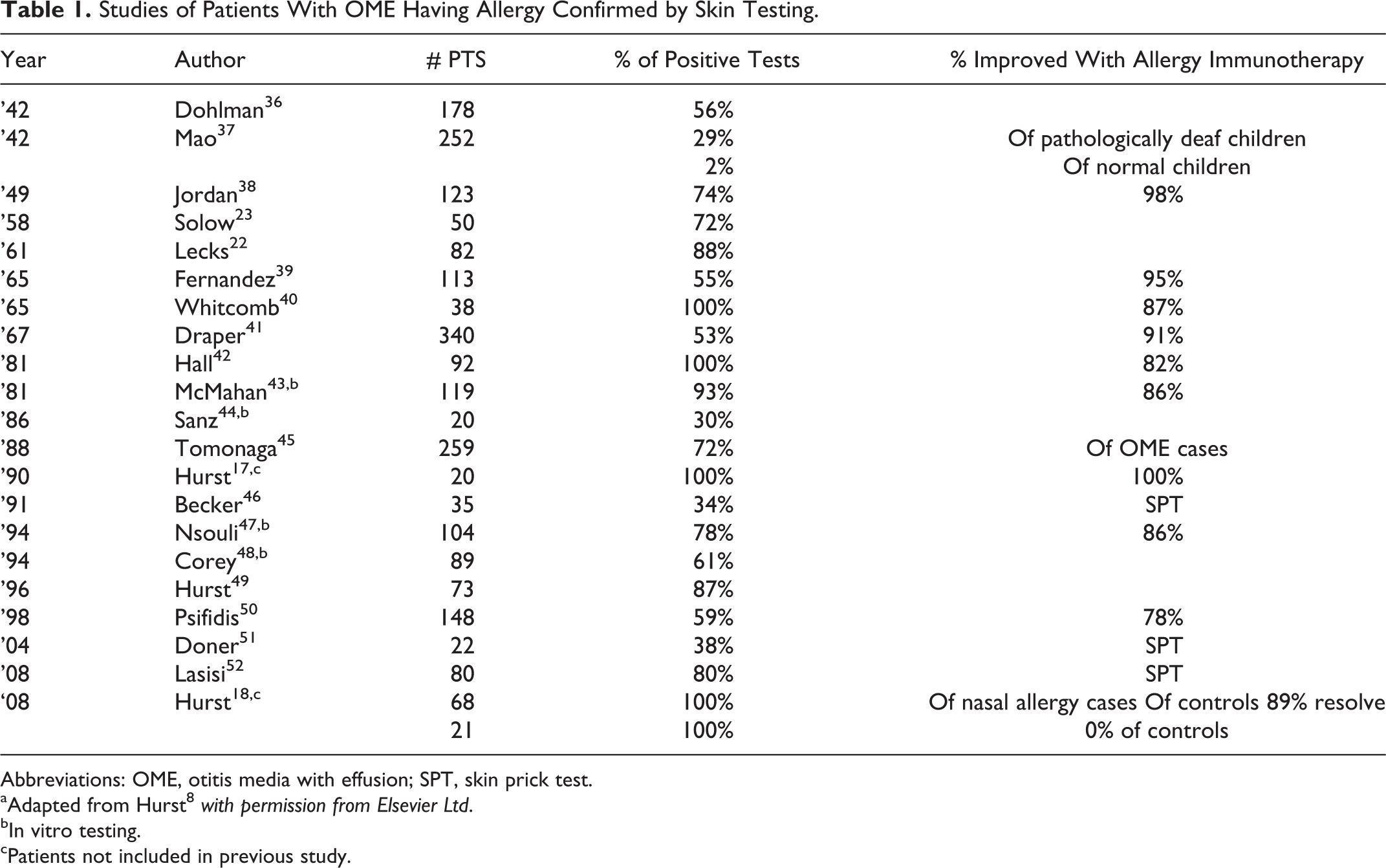
Much of the support for an allergic etiology for cOME has come from 3 observations: (1) the apparently greater prevalence of AR in patients with cOME when compared to the general population 35 ; (2) the reported higher IgE levels, Th2 cytokines, mast cell, and eosinophil-derived immune mediators in middle ear effusion (MEE) from patients with cOME and allergy when compared to those with cOME but no allergy 25 (Table 1); and (c) Hurst’s report that allergic desensitization based on interdermal dilutional testing (IDT) cures OME and prevents future OME episodes. 18
Studies of Patients With OME Having Allergy Confirmed by Skin Testing.
Abbreviations: OME, otitis media with effusion; SPT, skin prick test.
aAdapted from Hurst 8 with permission from Elsevier Ltd.
bIn vitro testing.
cPatients not included in previous study.
The current hypothesis of a common mucosal immune system suggests that activated T lymphocytes are able to migrate from one mucosal site to another. Exposure of one epithelial surface to an infectious or antigenic agent leads to secretion of locally produced antibodies. 53 Other studies support the hypothesis that an abnormally increased absorption of allergen is pathogenic and may trigger distant airway diseases of rhinitis and asthma. 54 This again demonstrates a “unified airway” response.
The Relation of AR to OM
A relationship of AR with OME was first proposed by Proetz 90 years ago. 14 Most of the recent literature confirms the role of AR in the development of OME. 55 Kwon et al evaluated 370 patients with OME and a control group of 100 other children with no history of OME. Children with OME had a significantly higher incidence of AR (33.8%) when compared to those without OME (16%; P < .05). 35 The Danish birth cohort study found OME to be diagnosed in 39% of the cohort and was associated with AR but not with nasal mucosal swelling, non-AR, or asthma. Thus, they concluded that OME was associated with AR caused by allergic inflammation but not by mechanical mucosal swelling in the nose. 56 The observed prevalence of AR in patients with chronic OME has been reported to range from 24% to 89%. 57 Biologic markers of allergy have been found to be similar in the middle ear as in the nose and upper airway. 58
Pelikan reported that nasal allergen challenge in sensitized adults provoked abnormal middle ear pressure only in those with a history of OM. 59 It has recently been shown that mucociliary transit times within the ET are significantly prolonged in atopics versus normal children. 60 A study in Mexico found that 15% of 80 children with positive skin tests to dust, corn, and cockroach had abnormal tympanograms when compared to 50 controls who all had normal type A tympanograms, and all were SPT negative for the same 3 allergens. Among children with rhinitis, allergy presented an increased risk for difficulty in opening their ETs. 61
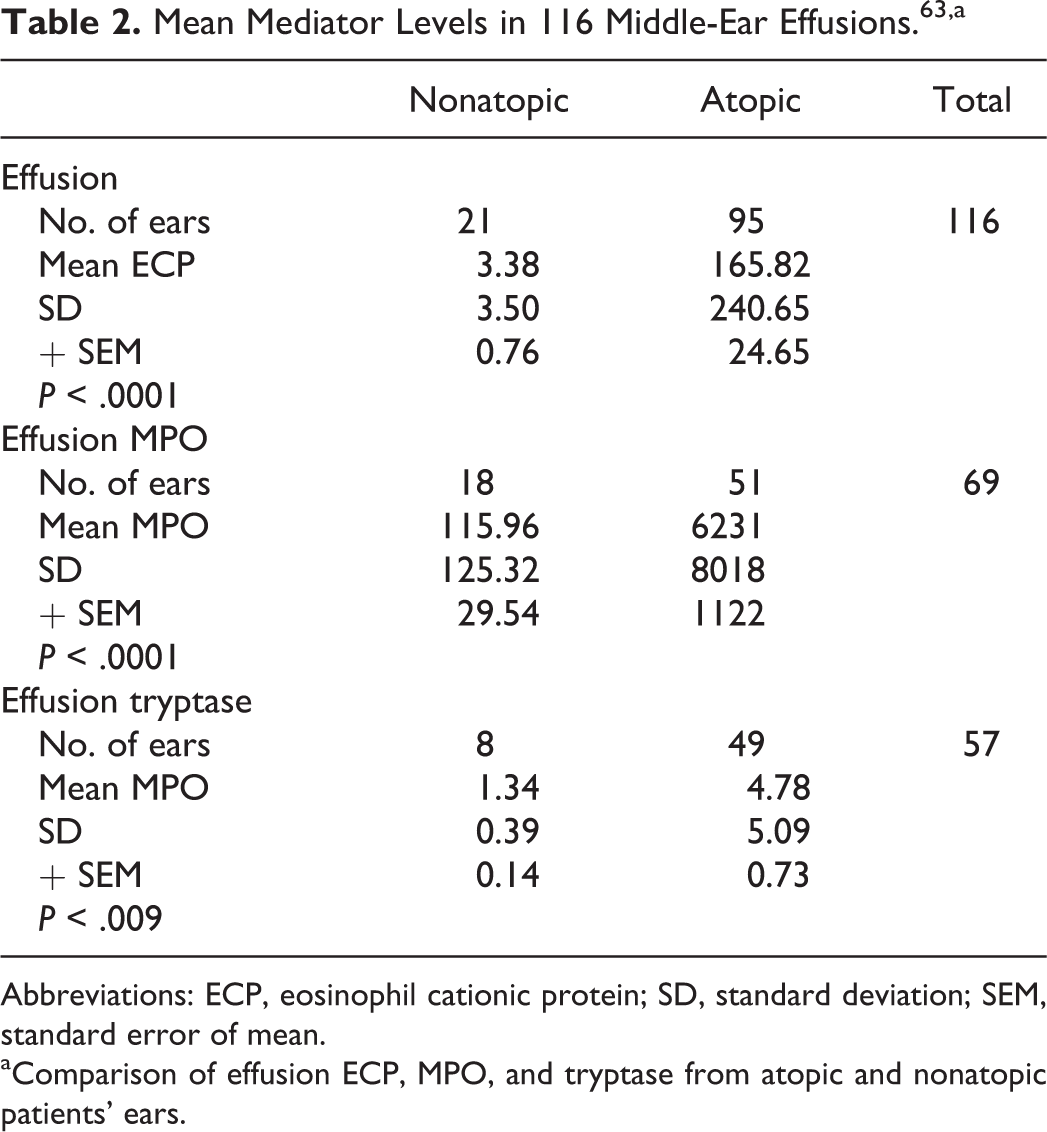
An immune response involving IgG, complement activation, neutrophils, eosinophils, and mast cells has been implicated in the generation of immune-mediated OME by Suzuki et al. 62 Hurst similarly demonstrated neutrophils, eosinophils, and mast cells and their mediators in human MEE in atopics (Table 2). 25,63
Mean Mediator Levels in 116 Middle-Ear Effusions. 63 ,a
Abbreviations: ECP, eosinophil cationic protein; SD, standard deviation; SEM, standard error of mean.
aComparison of effusion ECP, MPO, and tryptase from atopic and nonatopic patients’ ears.
Proof That Allergy Affects the Middle Ear
Proof of the hypothesis that cOME is an allergic disease requires 4 steps. First, establish a relevant, associated, objective diagnosis of atopy in patients with persistent effusion or middle ear drainage. The low sensitivity of conventional prick or in vitro testing hampers diagnosis of this low level IgE-mediated disease. 18,64 Over a dozen investigators using objective IT have demonstrated over the past 70 years that 72% to 100% of the children with OME are atopic (Table 1). 8
Second, establish an association of allergic Th2 immune-mediated histochemical reactivity within the target organ itself. Interleukin 5 (IL-5) is produced predominately by stimulated Th2 and not Th1 cells 65 and is increased during the late-phase reaction of chronic middle ear disease. 66 Interleukin 5 messenger RNA in middle ear mucosa biopsies provides strong evidence that the middle ear is actively participating in a Th2 response. 67 Recent meta-analysis supports the unified airway hypothesis and finds “Epidemiology and current understanding of inflammatory, humoral, and neural processes make links between AR and disorders including asthma, otitis media, nasal polyposis and chronic rhinosinusitis plausible.” 24 Johansson et al postulated in 1972 that more than 90% of IgE in nasal and bronchial washings was produced locally. 68
Atopic children have increased circulating levels of eosinophils, T lymphocytes, IL4+, and IL5+ cells compared to nonatopics. These products have been found to stimulate allergic cell-mediated inflammation within the middle ear. 67 Tian et al reported both specific IgE and eosinophil cationic protein (ECP) to be elevated in MEE. 69
Johnson et al reported the independent production of cytokines associated with a Th2 immunologic response in OME ears. 70 Several immune mediators have been consistently identified in the middle ear fluids of children with cOME. For example, cytokine concentrations of tumor necrosis factor α, IL-1β, IL-4, IL-5, IL-13, IL-6, and IL-8 are elevated in middle ear fluid in children with cOME. 71,72 Smirnova and her colleagues have demonstrated that the cytokines present in middle ears play a key role as molecular regulators of middle ear inflammation. 73 In a very complicated paper, she explains in detail the immunologic mechanisms, wherein cytokines can switch the acute phase of inflammation to the chronic stage and induce molecular and pathologic processes leading to the histologic changes demonstrated in ears with chronic effusion.
There are at least 9 distinct mucins within the human body. Data suggest that the local genes are upregulated in cases of chronic OM. Mucin genes, MUC5B and MUC4, from middle ear fluid collected were upregulated 4.2- and 6-fold, respectively, in middle ears with chronic OM compared to controls. 74 Studies have shown that specific cytokine production can induce mucin production. 75,76 Many of these inflammatory mediators have been identified in middle ear fluid in those with chronic effusion. 71,72,77
The third step needed to prove that the middle ear behaves as a target organ of allergy is to demonstrate that the inflammation within the middle ear is truly allergic in nature. Cellular immunology enables us to differentiate allergic inflammation (Th2 response) from bacterial infection (Th1 response). Demoly et al note that “chronic sinusitis, which occurs in some patients with AR, can be distinguished on the basis of T cells and intraepithelial mast cells.” 78 He found 8 times more T cells in patients with AR than in patients with non-AR. Similarly Wright et al demonstrated that 8 of 8 atopic patients with cOME had increased CD3+ T cells, versus none of the controls. 67 This, coupled with the presence of degranulating mast cells 67 and EG2-positive eosinophils 79 in middle ear mucosa, provides further evidence that the inflammatory response described in the middle ear represents Th2-mediated allergic disease.
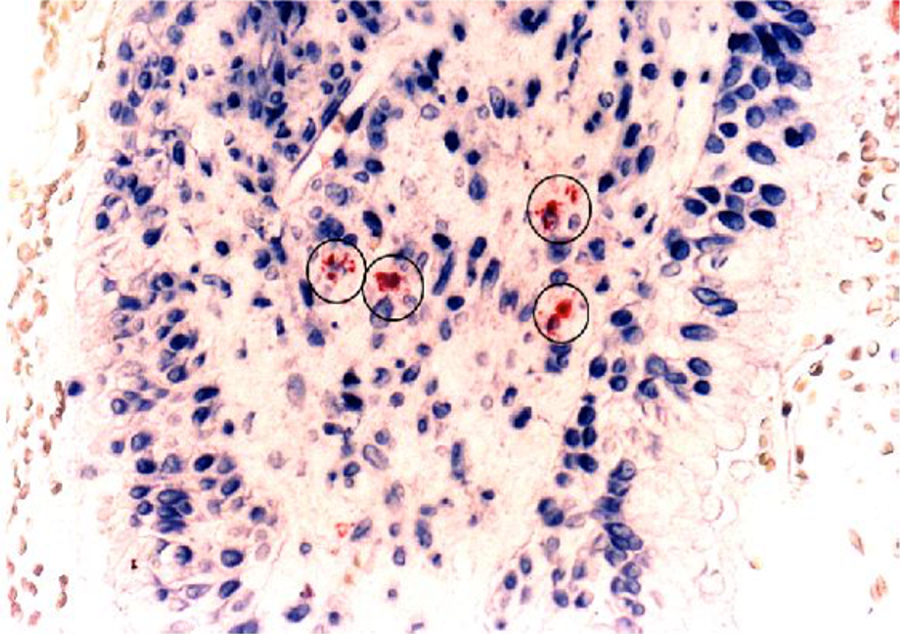
The presence of mast cells in addition to eosinophils is essential, although not pathognomonic, for a disease process to be considered the result of atopy. Anti-tryptase antibody (AA1) staining 49 has shown that mast cells are present in the mucosa and submucosa in patients with atopy and not in controls (Figure 1). Direct demonstration of serum and middle ear immunoglobulins in association with a Th2 response in the middle ear itself has been provided by Hurst, 63,80 Nguyen, 58 Tian, 69 and Lasisi et al. 52 Luong and Roland’s review (54 references) summarized significant data supporting “the concept that chronic OME in atopic patients represents a local Th2 allergy response.” 81 Thus, allergic inflammation in the ear of a patient with OME is similar to that seen in other target organs of allergy and supports the concept that the ear is part of the unified airway.
Efficacy of AIT for OME
The fourth and final step needed to prove the hypothesis of an allergy connection requires direct evidence of a dose–response curve and consistency of results. Recognizing the ineffectiveness of current medical treatments 34 and the limited effectiveness of surgical treatments, including the insertion of PET for OME, 20 we believe that new approaches to both ETD prevention and treatment need to be defined that target specific pathways in the disease pathogenesis. This requires identifying a causal etiology susceptible to intervention, describing the pathways linking that etiology to ETD and introducing targeted therapies characterized by high-expected efficacy and efficiency.
Allergy Therapy
Immunotherapy has been proven over almost a hundred years to be the only way to actually cure rather than just provide symptomatic relief for a variety of allergic conditions including AR and asthma. 82 It is now accepted fact that “Allergen immunotherapy has been thoroughly documented in randomized controlled trials. It is the only immune-modulating and causal treatment available for patients suffering from IgE-mediated diseases such as allergic rhinoconjunctivitis, allergic asthma….” 83 Otitis media with effusion has also been shown to resolve on AIT. 18,24
The association of food allergy in cases of OME have been established. Aydoğan et al using a case–control design found the prevalence of food allergy approached 50% of the children with OME. 84 Nsouli and colleagues 47 found food allergy to be significant in 81 (77.8%) of 104 children. Children with OME were evaluated via SPT, specific IgE tests, and food challenge. Of the 104 patients, 81 (77.8%) had a positive screening for food allergy. Patients were then placed on an elimination diet avoiding trigger foods for 16 weeks. Following elimination of the offending food, 86% of cases resolved. These children were then rechallenged at 4 months resulting in a 94% recurrence of effusion. McMahan et al found 111 (93.2%) of 119 children with OME to be atopic. 43 Following allergy treatment with immunizations and food avoidance, 92% resolved. Nsouli et al found that following elimination of the offending food, 86% resolved. 47 He then rechallenged these children 4 months later, and 94% experienced recurrence of their effusion (Table 1).
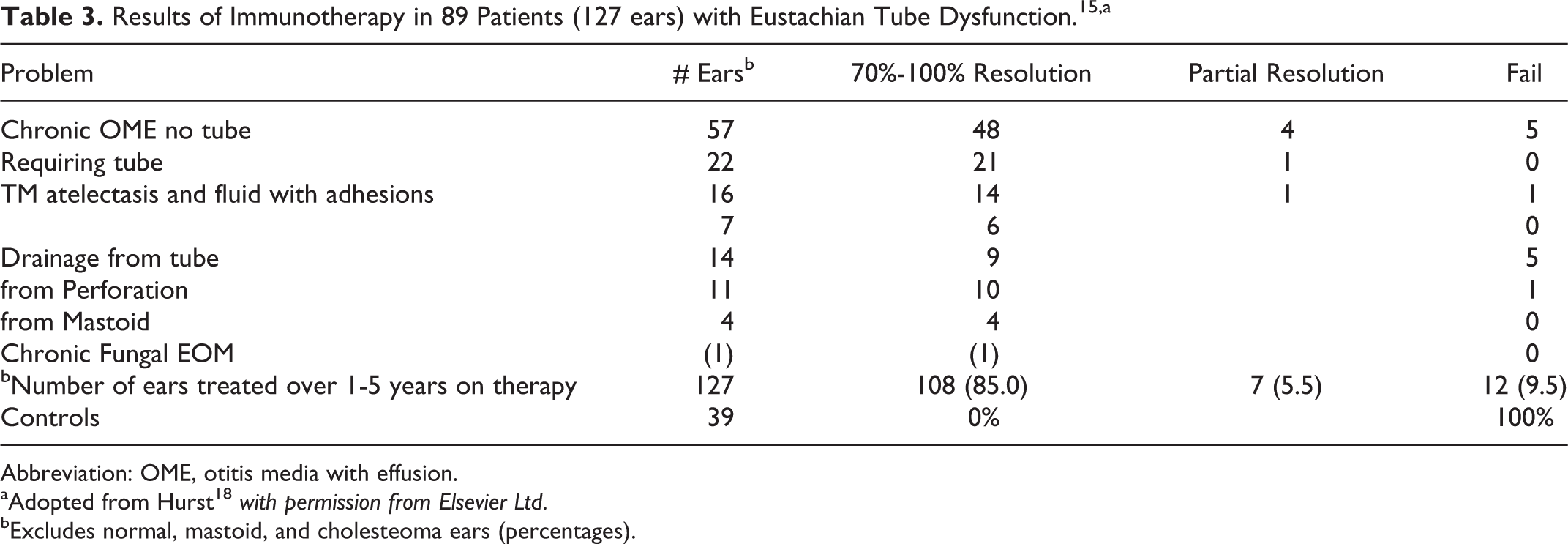
In a study of 89 patients aged 4 to 70 with intractable middle ear disease who presented with chronic effusion or chronic draining perforations or having had 1 to 15 sets of PET—all proved to be atopic by IDT. 18 All were offered AIT based on the results of their IT. Twenty-one individuals self-selected to be a “control cohort.” They chose for a variety of reasons not to proceed with AIT. Specific AIT completely resolved 85% of 127 diseased ears and significantly improved an additional 5.5% (Table 3). All children <15 and most adults in the treatment group resolved within 4 months and remained free of disease while on AIT for 2 to 8 years of follow-up. None of the controls’ 39 ears resolved spontaneously (P < .001).
Results of Immunotherapy in 89 Patients (127 ears) with Eustachian Tube Dysfunction. 15 ,a
Abbreviation: OME, otitis media with effusion.
aAdopted from Hurst 18 with permission from Elsevier Ltd.
bExcludes normal, mastoid, and cholesteoma ears (percentages).
Conclusion
It is not the goal of this article to review in depth all the immunologic mechanisms that lead to ETD requiring the use of PET, but rather to heighten the awareness of physicians of the physiology which underlies ETD and to present the data that supports the consideration that allergy is not only responsible for many cases but that when PETs have been recommended, especially for 2 to 5 times, allergy testing and immunotherapy should be considered. Current medical evidence supports the link between allergy and OME. The middle ear behaves like the rest of the respiratory tract, and what has been learned about the atopic response in the sinuses and lungs may be applied to the study of the middle ear to help in understanding OME.
Histologic, epidemiologic, and clinical studies based on objective allergy testing (Table 1) have thus far (1) established that the majority of patients with OME are atopic, (2) demonstrated that all the mediators necessary for a Th2 allergic response are present in the middle ear (Table 2), 63 (3) provided medical evidence that the middle ear is part of the unified airway conclusive enough for the 2016 guidelines to conclude that the middle-ear epithelium of patients with atopy has all the components required to behave in a manner similar to that of the rest of the upper respiratory system and that “like other parts of respiratory mucosa, the mucosa lining the middle-ear cleft is capable of an allergic response,” 85 and (4) shown that chronic middle ear disease does resolve with allergy immunotherapy based on IT results in over 85% of cases when compared to 0% of a control cohort (P < .001). 18
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
