Abstract
Laryngeal squamous cell carcinoma (LSCC) is one of the most common laryngeal tumors, and its incidence is increasing yearly; however, whether lymph node dissection should be performed during surgery remains unclear. We retrospectively analyzed the clinical and pathological data of 246 cases of LSCC and developed a nomogram for the prediction of lymph node metastasis (LNM) of LSCC. The predictive performance and consistency of the model were evaluated using the consistency coefficient (C-index) and calibration curve, respectively. Among 246 cases of LSCC, 52 cases had metastasis with a positivity rate of 21.14%. Multivariate analyses showed that dysphagia, clinical T stage, and pathological differentiation were independent risk factors for LNM in LSCC. The accuracy of the contour map used to predict the risk for LNM was 0.809. Overall, this nomogram model can be used to evaluate LNM in patients with LSCC before surgery to decide whether to conduct neck dissection and improve patient prognosis.
Introduction
Laryngeal cancer (LC) is one of the most common tumors, of which laryngeal squamous cell carcinoma (LSCC) accounts for about 85% to 90% of all cases. 1,2 Previous studies have reported that the crude incidence rate of LC was 1.54/100 000 in 2010 in China, resulting in 0.66% of new cancer cases and ranked 22nd among all cancer types. In addition, the prognosis of LC after surgery during that year was poor, with a 5-year survival rate of only 50% to 60%; about one-third of the cases relapse. 3
Surgical treatment is commonly used in patients with LSCC, and lymph node metastasis (LNM) of LSCC strongly affects patient prognosis 4 ; therefore, surgeons must decide whether to perform cervical lymph node dissection when making surgical plans. However, there is still controversy about whether neck lymph node dissection should be performed. Previous studies have reported that a large proportion of patients with LSCC who are clinically diagnosed with cervical LNM before operation and undergo cervical lymph node dissection during their operation have a negative pathological diagnosis after operation, which resulted in excessive treatment. 5 On the contrary, some clinically negative neck nodes are pathologically positive, leading to delay treatment or even a second operation. 6 Furthermore, indications for neck dissection and salvage laryngectomy for patients with LSCC without regional lymph node involvement before operation remain unclear. 7
At this time, for patients with LSCC who are undergoing open surgery, there is a lack of comprehensive evaluation indicators for LNM in clinical trials to evaluate whether patients require neck lymph node dissection. Therefore, identifying risk factors for LNM of LSCC and establishing an effective prediction model can help clinicians identify high-risk patients, perform neck lymph node dissection, and avoid overtreatment. The nomogram can integrate more clinical and pathological parameters to achieve individualized prediction by providing a computational chart that replaces complex mathematical formulas and is of great significance for individualized and accurate prognosis prediction.
We retrospectively analyzed the clinical and pathological data of 246 patients with LSCC. We determined the independent risk factors for LNM in patients with LSCC using univariate and multivariate analyses and constructed a nomogram model to evaluate LNM in patients with LSCC.
Methods
Patients
This study was approved by the Ethical Committee of our hospital. Because our study was a retrospective study, the committee exempted patients from the need for informed consent, while all data were collected and analyzed anonymously without causing any potential harm to patients. Through the electronic medical record system of our hospital, we retrospectively analyzed the clinical and pathological data of patients with LSCC who underwent open surgery and bilateral neck lymph node dissection by the same senior otolaryngologist team (who had more than 20 years of clinical experience for the treatment of LSCC) from January 2014 to December 2018. The inclusion criteria were as follows: underwent standard clinical examinations (routine auxiliary examinations, laryngoscopy, neck ultrasound, cervicothoracic contrast-enhanced computed tomography, and magnetic resonance imaging); preoperative biopsy under electrolaryngoscope revealed LSCC; clinical T classification assessed preoperatively; and complete follow-up. The exclusion criteria were as follows: preoperative biopsy under electrolaryngoscope revealed a pathological type of nonsquamous cell carcinoma; history of head and neck cancer; and history of preoperative radiotherapy or chemotherapy. Finally, in 4 years, we treated 279 patients with LSCC. Of these, 33 were excluded, 15 with nonsquamous cell carcinoma, 11 with the electronic medical record system did not contain all the information, 2 with a history of head and neck cancer, and 5 with a history of preoperative radiotherapy or chemotherapy. Thus, a total of 246 patients were included in our study.
Surgical Procedure
All patients underwent biopsy under an electrolaryngoscope before surgery to determine the pathological type and degree of differentiation. All patients underwent standard total or partial laryngectomy and bilateral neck dissection. The resected lymph nodes were routinely sent for pathological examination to determine LNM.
Data Collection
Our study collected clinical and pathological data from all patients including sex, age, body mass index (BMI), smoking, drinking, and dysphagia. We evaluated dysphagia using 2 tests 8 : (1) a water-swallowing test, in which patients were asked to drink 5 mL water and then 50 mL water; and, for those who exhibited cough or wet/hoarse voice in this test, (2) the swallowing provocative test, 9 in which we injected 0.4 mL distilled water followed by 2 mL into the suprapharynx through a small nasal catheter. This second test determined dysphagia according to Teramoto and Fukuchi 10,11 if the latency of swallowing after either of the water injections was ≥3 seconds. Patients who were abnormal in both tests were diagnosed with dysphagia. The time from detection to diagnosis, tumor subtypes (supraglottic, glottic, and subglottic type), clinical T classification (divided into I, II, III, and IV stages in accordance with the 7th edition of the TNM classification established by the American Joint Committee for Cancer), and pathological differentiation of tumors (poorly differentiated include poorly and medium-poorly differentiated tumors, medium differentiated include medium and highly medium differentiated tumors, and highly differentiated).
Statistical Analysis
Data analyses were performed using SPSS version 22.0 for Windows software (SPSS Inc, Chicago, Illinois). For continuous variables, Kolmogorov-Smirnov test was used to determine whether the data follow a normal distribution. For normal-distribution variables, an independent sample t test (student t test) was used to calculate and compare the mean values; otherwise, Mann-Whitney U test was used to compare the 2 groups. The χ2 test was used to analyze qualitative variables. Univariate analyses were initially used to identify variables that may affect LNM. All variables with a correlation value of P < .05 were considered candidates for multivariate analyses to identify independent risk factors associated with LNM of LSCC. All independent variables were incorporated into the model using the method of “Enter.” The final independent risk factors were incorporated into the R3.1.2 software (R Foundation for Statistical Computing, Vienna, Austria) to establish a nomogram prediction model. The consistency index (C-index) was used to evaluate the predictive performance of the model and the calibration curve was used to judge the predictive consistency. The range of C-index value was 0.5 to 1.0, with the following cutoffs: 0.5, the model has no prediction ability; 0.5-0.7, low accuracy; 0.70-0.9, high accuracy; > 0.9, very high accuracy. The calibration curve included image comparison of predicting risk and patients transfer risk. The closer the predicted risk to the standard curve, the better the conformity of the model is. In all analyses, P < .05 was considered to indicate statistical significance.
Results
A total of 246 patients with LSCC were enrolled in our study. Among them, 52 (21.14%) had LNM. The demographic data of the patients are shown in Table 1. There were no significant differences between the 2 groups in terms of sex, age, smoking, drinking, dysphagia, time from detection to diagnosis, and tumor subtypes. It is worth mentioning that patients with LNM have lower BMI and higher incidence of dysphagia than those without LNM (P < .001, Table 1). The number of patients with T stages I, II, III, and IV were 0, 4, 14, and 30, respectively, in the LNM group and 70, 84, 28, and 12 in the group without LNM (P < .001, Table 1). Similarly, the numbers of highly, medium, and poorly differentiated tumors were 6, 30, and 16, and 128, 56, and 10, respectively (P < .001, Table 1).
Comparison of Laryngeal Squamous Cell Carcinoma Patients With or Without Lymph Node Metastasis.
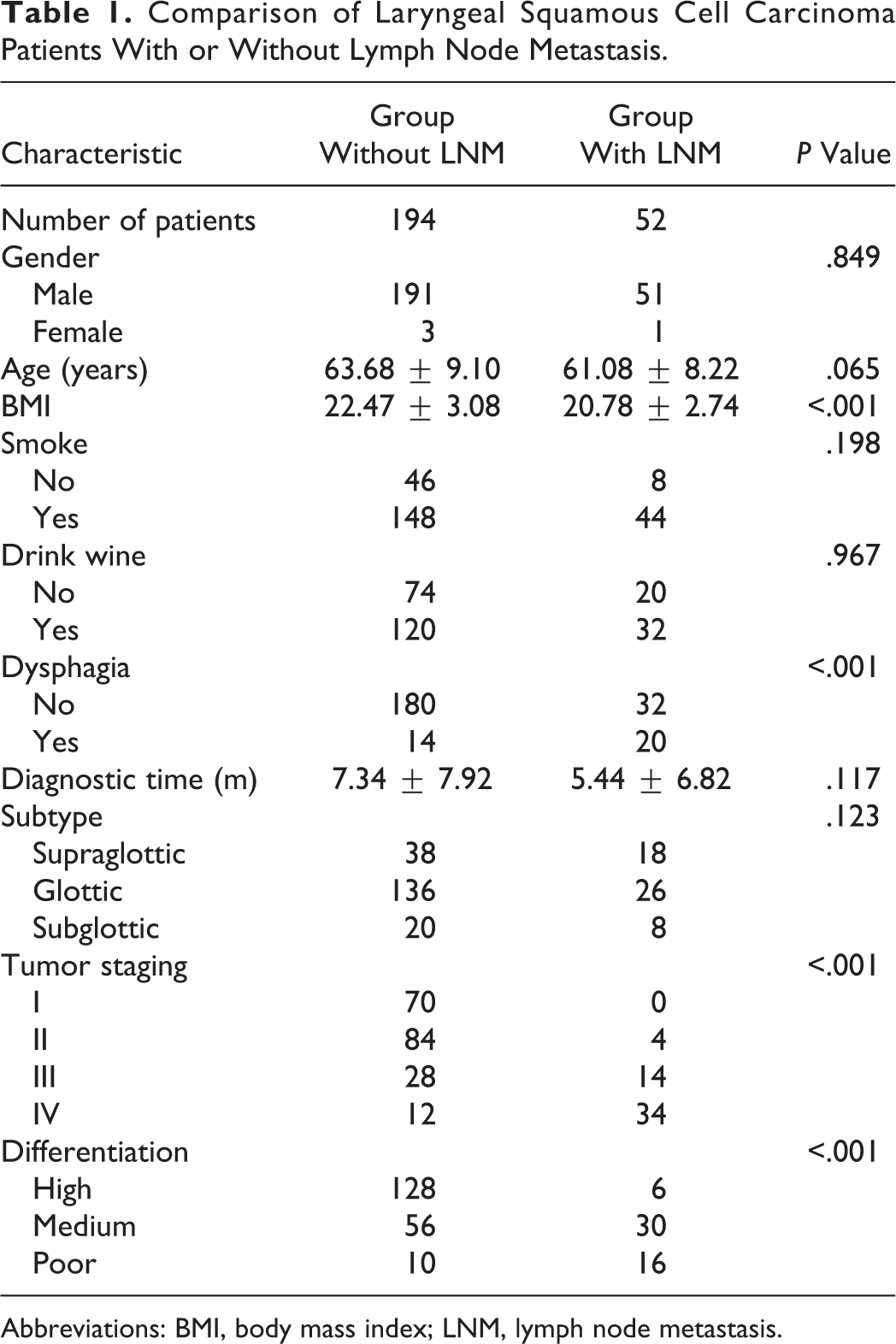
Abbreviations: BMI, body mass index; LNM, lymph node metastasis.
Table 2 shows the results of univariate analyses of the 10 potential risk factors mentioned earlier and LNM of LSCC. Dysphagia, BMI, clinical stage, and differentiation of tumors were included as potential risk factors in multivariate analyses. Table 3 shows the results of multivariate analyses. Ultimately, our preliminary data suggest that dysphagia, clinical T staging, and differentiation of tumors were independent risk factors for LNM of LSCC. According to the variables selected by multivariate analyses (dysphagia, T staging, and differentiation of tumors), a nomogram was established to predict the risk for LNM in LSCC. T stage had the highest score, which indicated that it was important to conduct neck dissection concurrently with surgery as precautionary measure (Figure 1). The total score of prognostic indicators was obtained by summing the corresponding scores, and the corresponding LNM probability was obtained based on the total score. For example, a patient with T stage III complicated with dysphagia symptoms with postoperative pathology was suggestive of medium differentiation. The corresponding T stage III score of the patient was 85, the dysphagia score was 25, and the medium pathological differentiation score was 10, with a total score of 120, corresponding a risk for LNM of 80%.
Univariate Analysis of Lymph Node Metastasis in Laryngeal Squamous Cell Carcinoma.
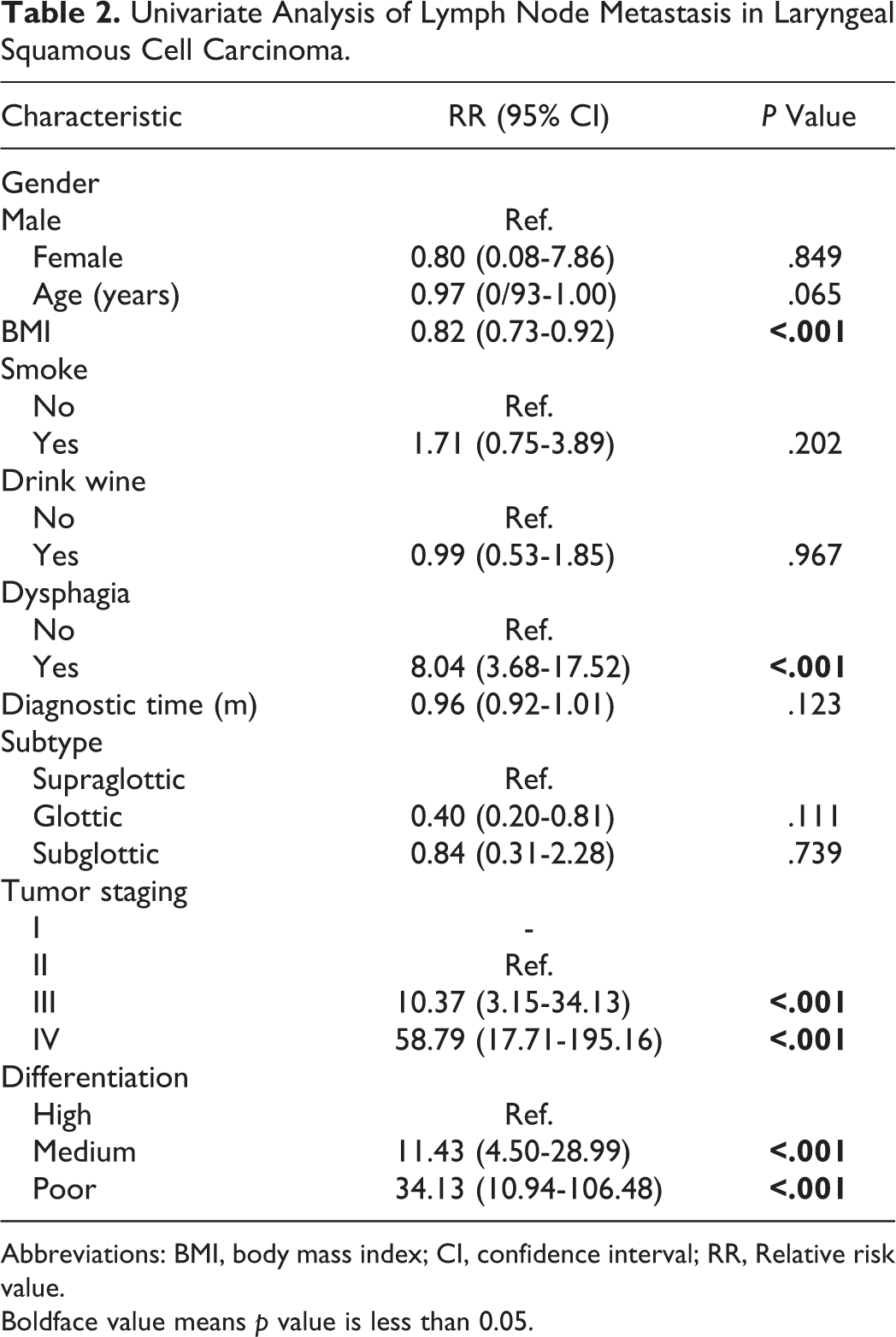
Abbreviations: BMI, body mass index; CI, confidence interval; RR, Relative risk value.
Boldface value means p value is less than 0.05.
Multivariate Analysis of Lymph Node Metastasis in Laryngeal Squamous Cell Carcinoma.
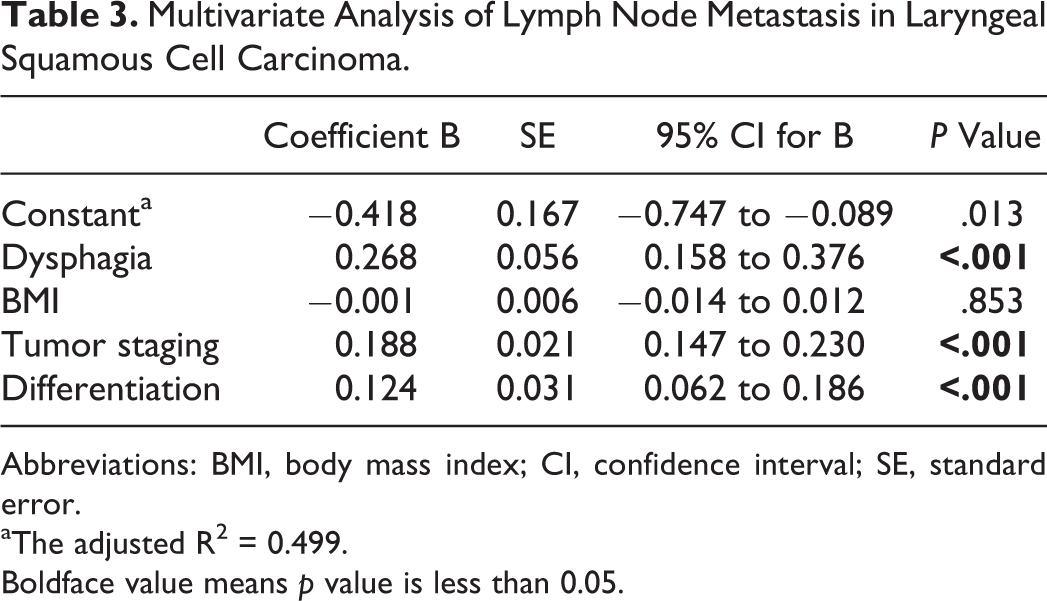
Abbreviations: BMI, body mass index; CI, confidence interval; SE, standard error.
aThe adjusted R2 = 0.499.
Boldface value means p value is less than 0.05.
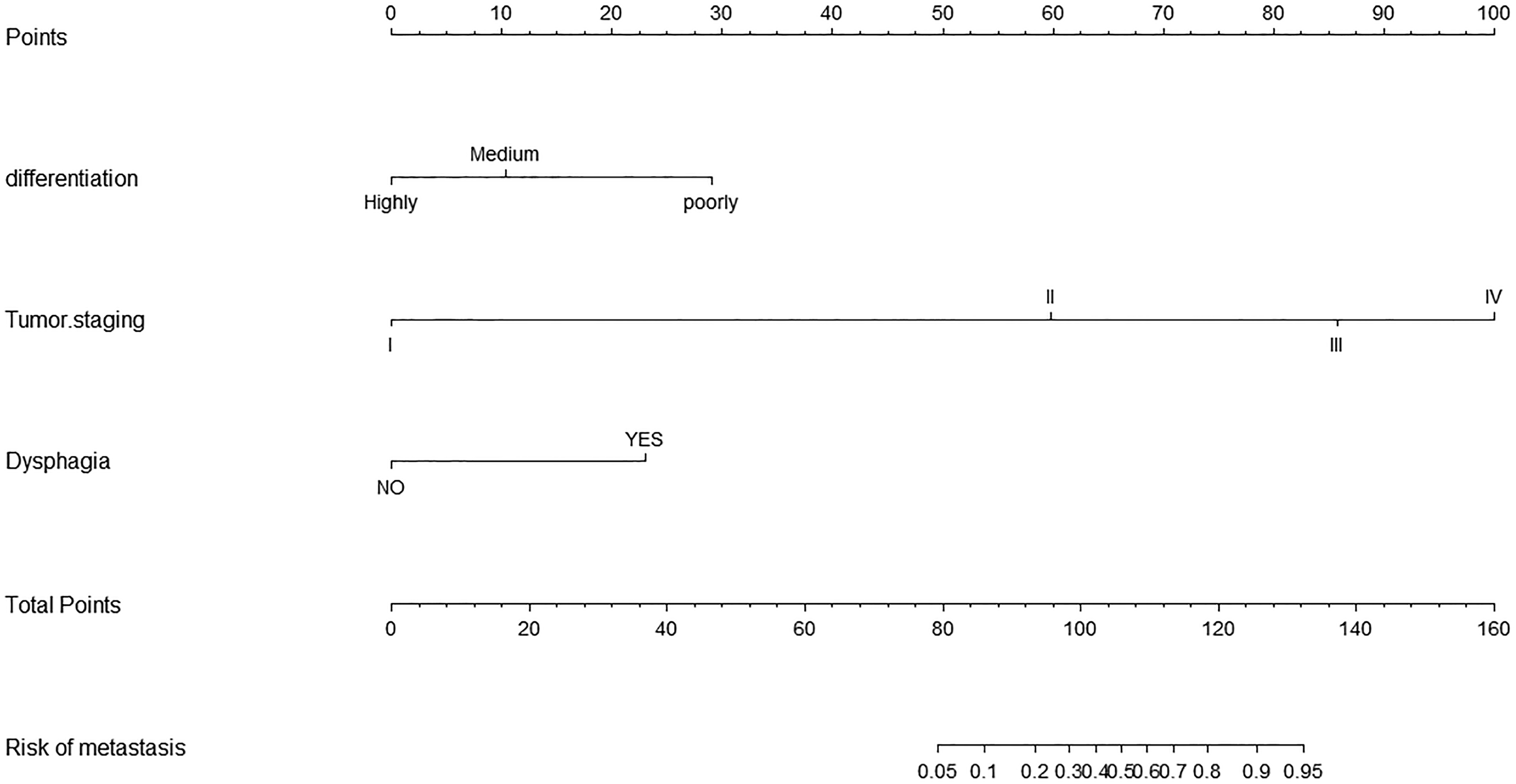
A nomogram to predict lymph node metastasis in patients with laryngeal squamous cell carcinoma.
The C-index value of the model was 0.809 after 400 internal verifications of bootstrap self-sampling, which indicated that the consistency between the predictions and the actual observations obtained from this chart is in agreement with the standard and that the chart has a standard resolution. The calibration curve is shown in Figure 2. The average absolute error of the coincidence between the predicted value and the real value was 0.022. The predicted risk was close to the actual risk and the coincidence was good.
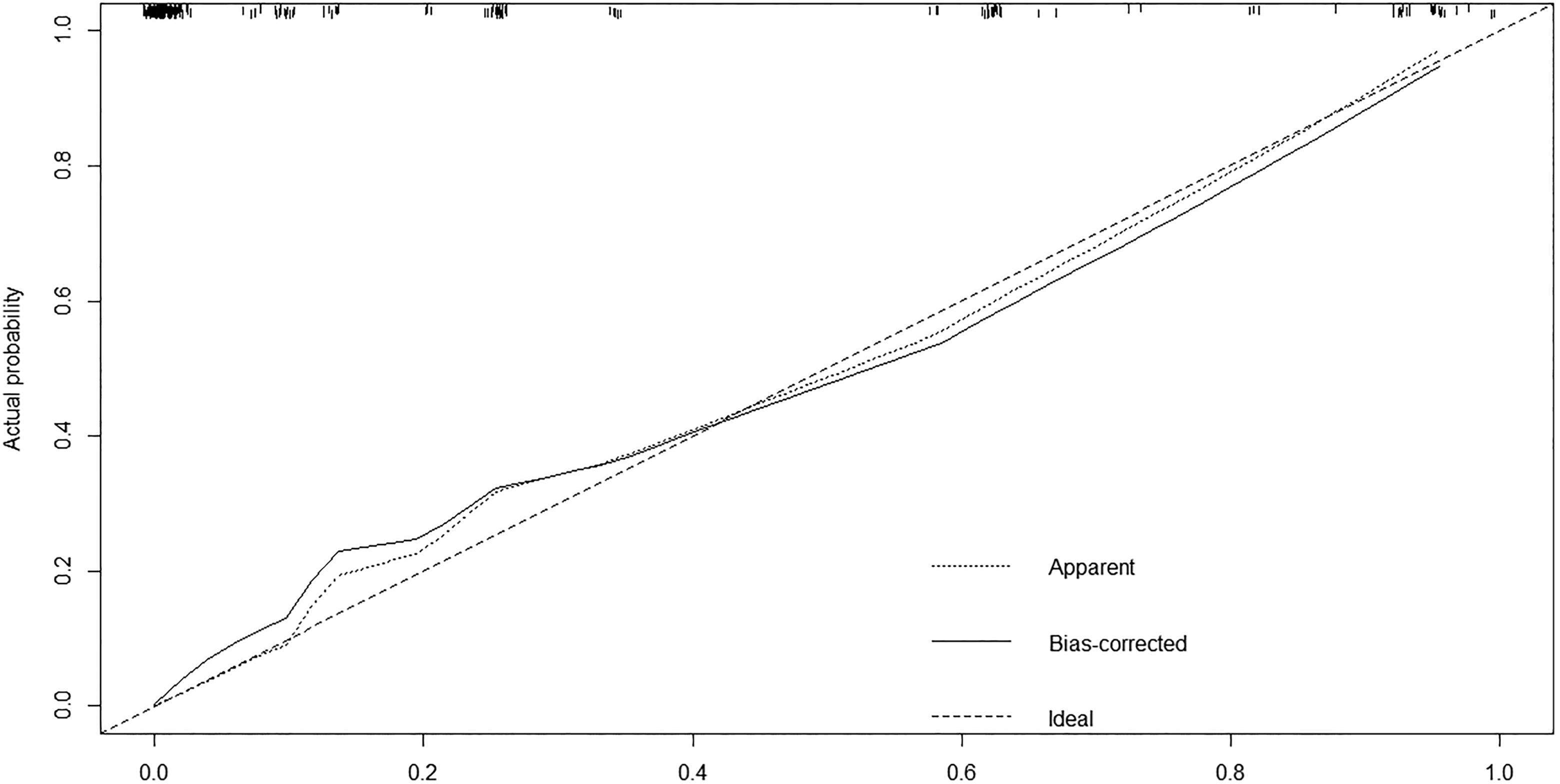
Calibration curve for nomogram prediction of lymph node metastasis in laryngeal carcinoma.
Discussion
Laryngeal cancer is one of the most common head and neck tumors, among which squamous cell carcinoma accounts for 85% to 90%. 12 Moreover, similar to other head and neck cancers, lymph node involvement reduces the survival rate of LSCC by about 50%. 13 Cervical lymph node dissection is considered an appropriate treatment for patients with LNM. However, there is no accurate method to evaluate cervical LNM; thus, there is no consensus as to whether cervical lymph node dissection should be performed in LSCC. 14 Therefore, establishing a predictive model of LNM is helpful to guide whether neck lymph node dissection should be performed during surgery. Using this approach will provide more appropriate treatment for patients with LSCC, avoiding a certain degree of overtreatment and improving patient prognosis.
Our preliminary results showed that cervical LNM of LSCC was associated with T stage, consistent with previous literature. Li et al 15 retrospectively analyzed the clinical and pathological data of 91 cases of glottic LC to explore the relationship between different T stage, differentiation degree, location, and cervical LNM. They showed that cervical LNM in cT2-T4 N0M0 (cN0) patient with supraglottic carcinoma is affected by T classification whereas cervical LNM of T3 and T4 glottic carcinoma is not entirely affected by T stage and differentiation degree. Moreover, Ma et al 16 retrospectively analyzed a total of 121 patients who received surgery to investigate factors that contribute to LNM from clinical cN0 supraglottic laryngeal carcinoma and to predict the risk of occult metastasis before surgery. They believed that the rate of cervical LNM increased with T stage in patients with supraglottic LC and recommended neck dissection for these patients.
In addition, our results suggest that the degree of differentiation of tumors was an independent risk factor for LNM in LSCC. Mutlu et al 17 investigated laryngectomy (partial and total) and neck dissection (functional, radical) in patients undergoing laryngeal surgery identified based on primary tumor localization, size, stage and histopathology, and the relationship with LNM. Their results showed that the rate of cervical LNM is more common in advanced T-stage tumors than in early T-stage tumors (P = .027), and cervical LNM has been determined as 27.3% in good differential (G1) tumors, 53.4% in medium differential (G2) tumors, and 74.1% in bad differential (G3) tumors (P = .005). We considered that the relevance of LNM of LSCC to tumor differentiation may be associated with the expression of specific genes. Gene expression affects differentiation and metastasis. Shen et al 18 reviewed the data of 506 cases of cN0 LC in their institution from March 2011 to March 2018. They found that the incidence of cervical LNM in poorly differentiated patients with carcinoma (17/42, 40.48%) was higher than those in medium differentiated (26/205, 12.68%) and highly differentiated patients (12/246, 4.88%; P < .01). Lu et al 19 studied the expression of osteopontin (OPN) and integrin alpha v (ITGAV, the main receptor of OPN) in laryngeal and hypopharyngeal squamous cell carcinomas, and found that the expression of OPN and ITGAV in primary and metastatic carcinoma was significantly higher than that in normal tissues. In addition, the expression of OPN and ITGAV in the highly differentiated group was significantly lower than that in medium and poorly differentiated group, and the expression of OPN and ITGAV in the LNM group was significantly higher than that in non-LNM group. They believe that the overexpression of OPN and ITGAV may lead to the invasion and metastasis of laryngeal and hypopharyngeal squamous cell carcinomas, and that targeting OPN and ITGAV during laryngeal and hypopharyngeal squamous cell carcinomas treatment has certain application value. Meanwhile, Wang et al 20 studied the gene expression of vascular endothelial growth factor C (VEGF-C) and vascular endothelial growth factor receptor-3 (VEGFR-3) in LSCC and the association with clinic pathologic features. They reported that gene expression of VEGF-C and VEGFR-3 were different among normal laryngeal mucosa tissue, primary laryngeal carcinoma, and cervical lymph node in the same patient. In primary laryngeal carcinoma, the expression of VEGF-C and VEGFR-3 was significantly higher in lymph node–positive group than in lymph node–negative group and was associated with histological grade of differentiation.
Mutlu et al 17 reported cervical metastasis ratio of 55.8% for tumors larger than 4 cm and 27.27% for those smaller than 4 cm (P = .002). Tumor size may be related to LNM. Furthermore, Li et al 21 confirmed that the most significant risk factors for cervical LNM were the size and histological grade of primary tumor. In our study, because it was difficult to assess the actual size of tumors based on preoperative imaging alone, we explored dysphagia as a more accessible clinical parameter. Larger tumors can compress the surrounding tissues of patients with LSCC, causing dysphagia and other clinical manifestations. Previous studies have showed that various clinical and pathological predictors are considered risk factors for LNM in LSCC. However, no study has provided a visual presentation of these risk factors using a nomogram. Compared to the traditional multiple regression model, the advantage of a nomogram is that all the key prediction factors are graphically displayed. According to our results, the establishment of a nomogram can be used to evaluate LNM in patients with LSCC, allowing for individualized treatment. Our study had certain limitations. First, it was based on retrospective data and the sample size was relatively small, which will inevitably be affected by the inherent data. Thus, a prospective study is required to validate our results. Furthermore, at present, this nomogram model has only been validated internally and still needs to be validated externally.
Conclusions
We showed that dysphagia, clinical T-stage classification, and differentiation of tumors were independent risk factors for LNM of LSCC. Based on this information, a nomogram model was constructed. This nomogram model can be used to evaluate LNM in patients with LSCC before surgery to determine whether to conduct neck dissection before surgery and to avoid overtreatment and improve patient prognosis.
Footnotes
Authors’ Note
J.-F.C. contributed to study design; L.-Y.C. contributed to analysis and interpretation of data and drafted the manuscript; W.W. contributed to data collection and analysis; W.-B.W. critically revised the manuscript and searched the literature. All authors read and approved the final manuscript. The data sets analyzed in the study are available from the corresponding author on reasonable request. All procedures performed in studies involving human participants were in accordance with the ethical standards of the Ethics Committee of First Affiliated Hospital of Wenzhou Medical University and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. Since this study was a retrospective study, the Committee exempts patients from the need for informed consent, while all data was collected and analyzed anonymously, without causing any potential harm to patients.
Acknowledgments
The authors thank all the colleagues for their valuable assistance during the implementation of this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
