Abstract
Background:
Oral corticosteroids are often used in the medical management of chronic rhinosinusitis (CRS) with and without polyps. The purpose of our study is to review the literature for studies reporting the dosage of oral corticosteroids as part of the appropriate medical management prior to, immediately before, and after surgical intervention.
Methods:
We reviewed the literature for oral corticosteroid regimens given to patients with CRS from March 2012 to September 2018. Studies that did not disclose the exact doses of the regimen were excluded from our analysis.
Results:
Our search resulted in 7 articles with 4 studies of Level of Evidence (LOE) 1b, 2 studies with LOE III, and 1 study with LOE IV. The daily doses varied from 15 mg to 1 mg/kg, and with total doses ranging from 150 to 352 mg. In addition, several studies gave the same regimen to both subtypes of CRS. There was no mention of side effects in most of the studies.
Conclusion:
There is a wide variation in the steroid doses given to patients with CRS and prospective or randomized controlled trials are needed to provide better improved evidence.
Introduction
Chronic rhinosinusitis (CRS) is a chronic disease that affects as much as 15.5% of the population in the United States. 1 It is associated with greatly impaired quality of life and significant health-care costs. 2 Per the American Academy of Otolaryngology—Head and Neck Surgery, CRS is formally defined as at least 12 weeks of persistent sinonasal symptoms, nasal obstruction, nasal drainage, facial pressure, or compromised sense of smell, in conjunction with endoscopic or computed tomography (CT) evidence of mucosal inflammation. 3 The diagnosis is confirmed with a positive nasal endoscopy or imaging consistent with inflammation or mucosal changes within sinuses. Chronic rhinosinusitis is clinically divided into CRS with nasal polyps (CRSwNP) and CRS without nasal polyps (CRSsNP).
The pathogenesis of CRS remains ambiguous with several hypotheses emerging in an attempt to unveil the causative factors for this condition. Over the past several years, there has been a paradigm shift in viewing CRS as an infectious pathogen-derived condition to an inflammatory condition. Medical therapy is the initial step in the management of CRS; it may include the use of oral antibiotics, intranasal steroids, and oral corticosteroids. 2 The International Consensus Statement on Allergy and Rhinology: Rhinosinusitis gives a grade A recommendation for the use of short-term oral corticosteroids in the management of CRSwNP but does not recommend long-term use. In addition, the guidelines make no recommendation for the use of oral corticosteroids in the management of CRSsNP due to a lack of studies addressing oral corticosteroid management alone. 4 Likewise, the guidelines in the European Position Paper on Rhinosinusitis and Nasal Polyps (EPOS) published in 2012 have given a grade A recommendation, corroborated by level 1a evidence, for the use of oral corticosteroids but without dosing recommendations. 2 For patients who remain refractory to medical management, sinus surgery is an option.
Our focus in this article is to review the literature on the variation in the dosage of oral corticosteroids in the managing patients with CRS following the publication of EPOS in 2012. Given the heterogeneity and continued evolution in CRS phenotype definitions, otolaryngologists still vary in their use of oral corticosteroids as part of their appropriate medical therapy for CRS. Indeed, a survey by the American Rhinologic Society in 2007 found ta variety of regimens being prescribed with a mean peak dose of oral prednisone of 51.7 mg. 5 Given the potential side effects of systemic steroids associated with their chronic use, there is a need for high-level studies to define the safest and most effective treatment regimen. We sought to review studies that involved prescribing oral corticosteroids in patients with CRSwNP or CRSsNP since the introduction of the EPOS guidelines in 2012.
Methods
An electronic search through MEDLINE/PubMed was used to identify papers published between March 2012 and September 2018 containing the keywords “chronic rhinosinusitis” and “steroids.” This initial search yielded 252 articles. The results were screened for studies that involved the administration of oral corticosteroids as part of appropriate medical therapy in adult patients with CRS with or without polyps preoperatively, perioperatively, and postoperatively. This excluded 168 publications that did not involve oral administration of steroid therapy. Published articles that did not disclose dose or involved pediatric patients were excluded. This resulted in 7 publications that met all our criteria. Of the 7 publications that met criteria, 2 studies had included patient populations who had CRSsNP or CRSwNP who had oral corticosteroids administered; the other 5 studies strictly had patients with CRSwNP with oral corticosteroid treatment (Figure 1).
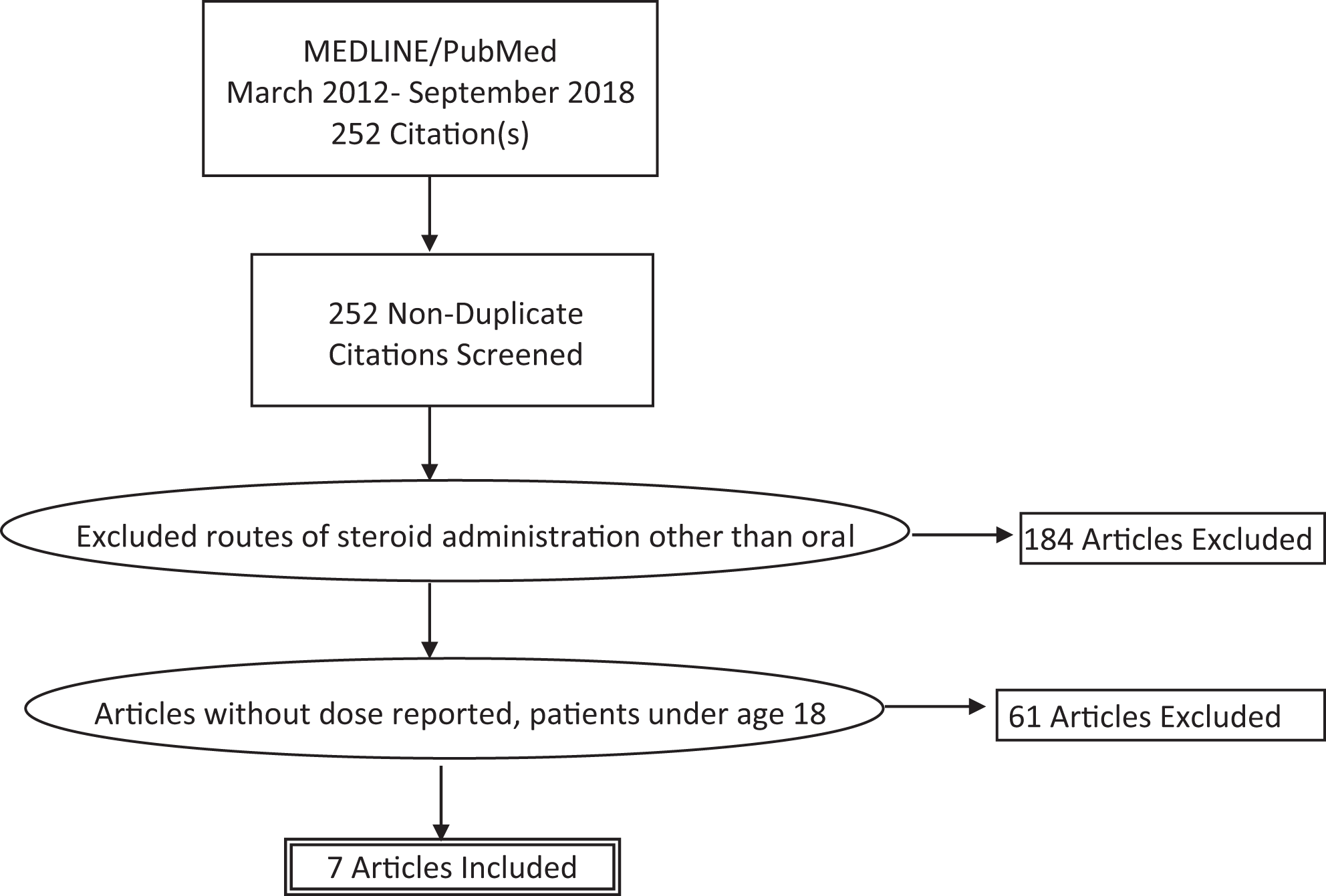
Selection method and inclusion/exclusion criteria for the studies reviewed.
Results
We identified 7 articles that fit our criteria (Table 1). Four of the studies were randomized controlled trials (RCTs) of which 2 were nonblinded, and 3 were retrospective studies. Therefore, 4 studies had a Level of Evidence (LOE) Ib, 2 studies had LOE III, and 1 study with LOE IV. The total number of patients in the studies ranged from 18 to 124, with the largest treatment group containing 86 patients treated with oral corticosteroids. All 7 studies used oral corticosteroids, including prednisone, prednisolone, and methylprednisolone; however, doses and regimens varied greatly. Total doses given to patients ranged from 150 to 352 mg (Figure 2).
Summary of the Studies Included in Our Search Criteria.
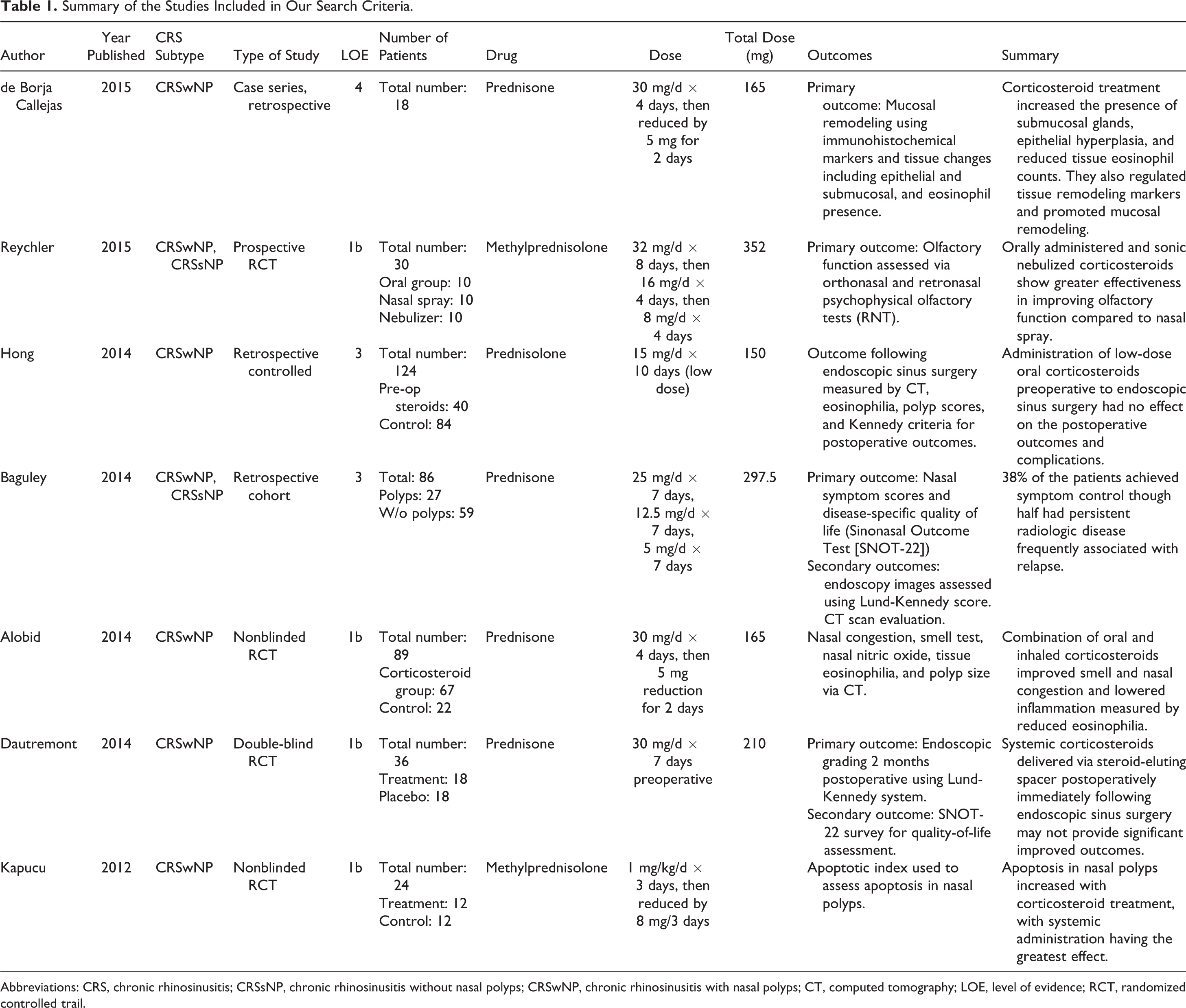
Abbreviations: CRS, chronic rhinosinusitis; CRSsNP, chronic rhinosinusitis without nasal polyps; CRSwNP, chronic rhinosinusitis with nasal polyps; CT, computed tomography; LOE, level of evidence; RCT, randomized controlled trail.
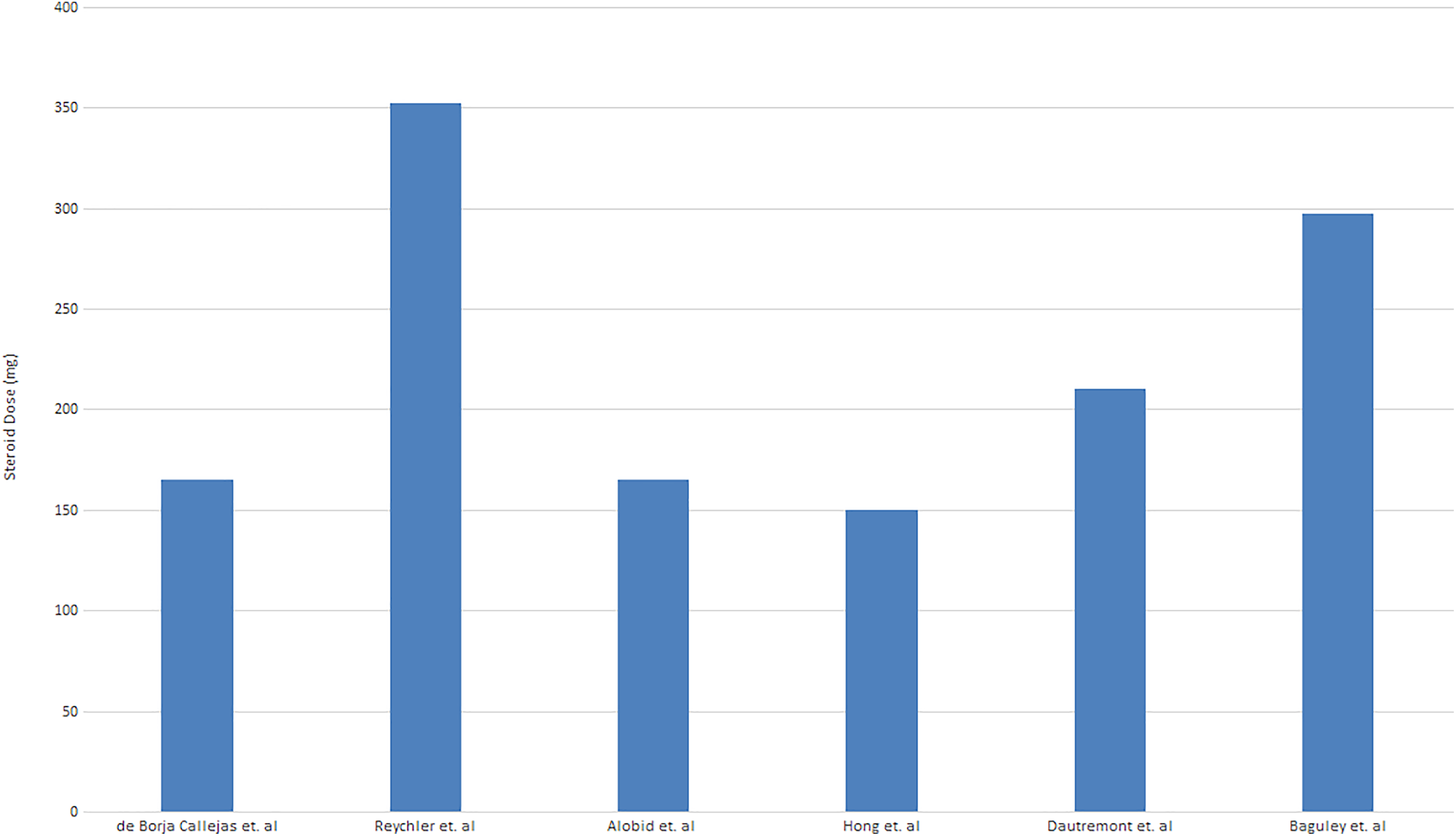
Total dose of oral corticosteroids administered throughout the treatment in studies identified.
The first paper is by Kapucu et al published in March 2012. 6 This nonblinded randomized placebo-controlled trial (RCT) aimed to elucidate the efficacy of treatment of CRSwNP through different steroid administrations. Forty-eight patients were assigned into 4 groups of 12. These groups were then treated with oral corticosteroids, topical steroids, direct administration into the polyps; and a control group. Their primary outcome measure was apoptosis in the nasal polyps to determine efficacy. It must be acknowledged that the end point of this article, apoptotic index is determined by terminal deoxynucleotidyl transferase-mediated dUTP Nick-End Labeling assay. Although it provides a useful quantitative measure of treatment response, it is not a practical option for measuring clinical symptomatic response. The oral dose that was used was methylprednisolone 1 mg/kg/d for 3 days and tapered at a rate of 8 mg/3 days. As a result, patients received different total doses depending on bodyweight, and the authors did not describe a maximum dose given to the participants. The study was included because it provides high-quality evidence on appropriate medical management of CRSwNP and follows EPOS consensus on implementation of systemic steroids. Although the study was not blinded and therefore the results could be susceptible to research bias, it is one of the few prospective randomized controlled studies that explores oral steroid dosing and efficacy as represented by apoptotic index. This prospective study adhered to EPOS guidelines by achieving appropriate medical management while minimizing adverse side effects in patients receiving oral steroids. The authors did not report any side effects to the oral steroid treatment. The study utilized appropriate exclusion criteria, including, but not limited to, contraindications for steroid usage: osteoporosis, uncontrolled diabetes mellitus, and glaucoma. Ultimately, patients who received oral corticosteroids were able to achieve the greatest apoptotic index. All the patients in the study had CRSwNP; therefore, the study does not shed any light on appropriate dosing for CRssNP. In addition, although the safety of intranasal polyp injections has been noted in some studies, the concern for blindness as a side effect makes this route of administration rare in clinical practice and thus limits some of the conclusion that may be drawn. 7
This article also had some potential risks of bias identified via the Cochrane risk of bias (ROB 2) tool for randomized trials (Table 2). In particular, there are some concerns over the randomization process as well as the measurement of the outcomes domains. Although the authors described the study as a randomized study, no information was given on the randomization process and concealment methods, as well as whether the assessors were blinded to the study.
Risk of Bias Assessment of Randomized Controlled Trials. The Revised Cochrane Risk of Bias Tool for Randomized Trials (ROB 2) was Used in the Assessment of the Individual Studies.
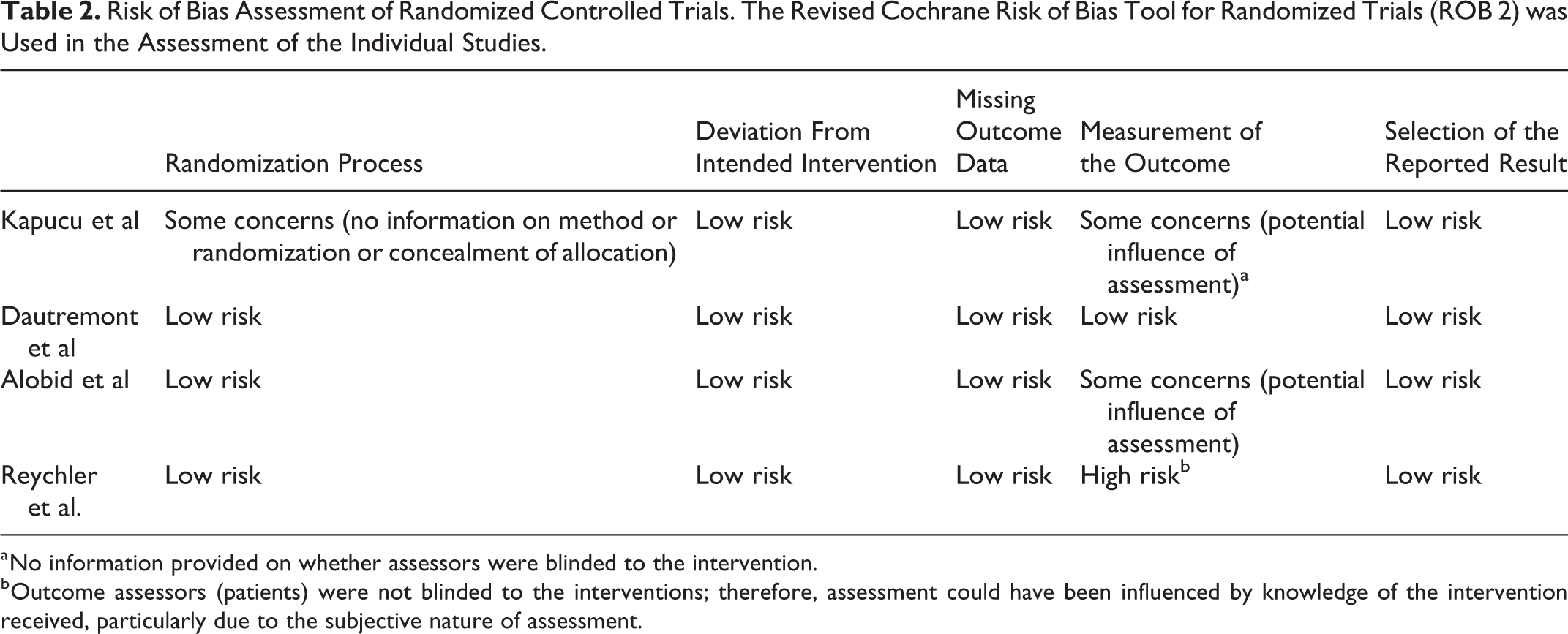
a No information provided on whether assessors were blinded to the intervention.
b Outcome assessors (patients) were not blinded to the interventions; therefore, assessment could have been influenced by knowledge of the intervention received, particularly due to the subjective nature of assessment.
The second paper is a randomized, double-blind, placebo-controlled trial published in January 2014 by Dautremont et al. 8 The study aimed to reveal the role of immediate postoperative oral corticosteroids when utilizing a steroid-eluting spacer on outcomes of endoscopic sinus surgery. The primary outcome reported was endoscopic grading 2 months postoperatively using the Lund-Kennedy system. The secondary outcome was improvement in the Sinonasal Outcome Test (SNOT-22) survey for quality of life assessment. A total of 36 patients with CRSwNP were divided into 2 equal groups: one given oral prednisone 30 mg/d for 7 days and the other receiving placebo. The total dose of steroids given over the treatment period was, therefore, 210 mg.
The third paper is a nonblinded RCT published in January 2014 by Alobid et al discerning the effects of oral and intranasal corticosteroids on patients with CRSwNP. 9 The authors report the use of both subjective and objective measurements as end points of the study. Subjective measurements included a smell test designed by the authors termed the Barcelona Smell Test 24, and the Likert scale for nasal congestion. Objective measurements reported were allergy study, polyp biopsy for eosinophilia, nasal nitric oxide inhalation, polyp size scored based on the Lildholdt score using rigid endoscope, and CT sinus staging via the Lund-Mackay score. The treatment group containing 67 patients received 30 mg/d of oral prednisone for 4 days followed by a 5 mg reduction for 2 days. The total dose of oral steroids was 165 mg. Notably, the treatment group also received intranasal budesonide for 12 weeks. The control group consisted of 22 patients for a total of 89 patients overall. The authors did not mention whether the assessors of the outcomes were blinded to the study and therefore there were some concerns over the measurement of the outcome domain of the ROB assessment. In addition, the subjective methods reported by the authors raise concerns of external validity (Table 2).
The fourth study is a retrospective cohort study by Baguley et al published July 2014 assessing CRS outcomes following appropriate medical management with oral corticosteroids. 10 A total of 86 patients (27 with polyps and 59 without polyps) identified as having CRS symptoms according to the EPOS criteria were given a 3-week course of oral prednisone totaling 297.5 mg at 25 mg/d for the first week, followed by 12.5 mg/d for the second week, and finally 5 mg/d the third week. The primary outcome measure was sinonasal quality-of-life questionnaire using the SNOT-22, and the secondary outcome was endoscopic and CT scan score via Lund-Kennedy and Lund-Mackay grading, respectively. In addition, the authors used EPOS 2012 definitions for a positive endoscopy. There was no mention of side effects from the steroid therapy. The Risk of Bias in Non-randomized Studies of Interventions (ROBINS-I) tool was used to assess the ROB in nonrandomized studies. This study was found to have a moderate ROB in postintervention domain due to risks in measurement of outcomes (Table 3).
Risk of Bias Assessment of Nonrandomized Studies. The Risk of Bias In Non-Randomized Studies of Interventions (ROBINS-I) Tool Was Used to Assess the Risk in Each Study.
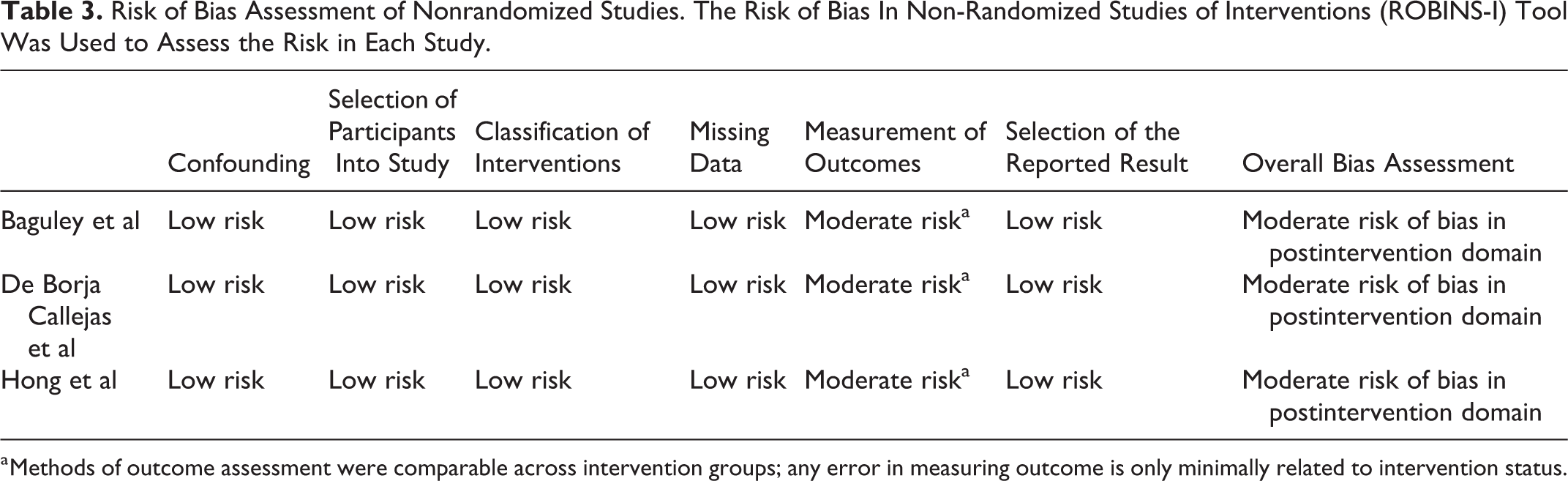
a Methods of outcome assessment were comparable across intervention groups; any error in measuring outcome is only minimally related to intervention status.
The fifth study is a retrospective chart review published in November 2014 by Hong et al that sought to analyze the outcome of endoscopic sinus surgery in patients with CRSwNP after preoperative systemic steroid (PSS) treatment. 11 The authors included patients with CRS as identified by the American Academy of Otolaryngology—Head and Neck surgery criteria. The clinic had 40 patients who underwent PSS treatment and 84 patients without such a regimen, and the patients were classified as CRSwNP or CRSsNP postoperatively. The PSS treatment arm was comprised of 15 mg/d of prednisolone for 10 days totaling a low dose of 150 mg over the treatment period. The control group contained 84 patients for a total of 124 patients. The primary postoperative outcome measures were the presence of atopy as measured by multiple allegro sorbent test or skin prick test, eosinophilia defined as when eosinophils account for >20% of inflammatory cells, polyp scores based on endoscopic findings, and radiologic findings of paranasal sinus CT scored by Lund-Mackay scoring system. The study was found to have the same postintervention risks as the previous study according to the ROBINS-I tool (Table 3).
The sixth paper included in this review is an RCT published in March 2015 by Reychler et al that examined oral, intranasal, or nebulized administration of corticosteroids to patients with CRSwNP or CRSsNP (identified via endoscopy) and the subsequent effect on olfactory function. 12 The authors did not disclose the criteria for CRS used in selecting the patients. The study involved 30 patients in total who were randomized into 3 equal groups depending on modality of corticosteroid being given. The oral steroid group was given 32 mg for 8 days, followed by 16 mg for 4 days, and finally 8 mg for 4 days, resulting in a total dose of 352 mg of methylprednisolone. The authors did not report any side effects for any of their groups. This study was assessed to have a high ROB according to the ROB 2 tool in measurement of the outcome due to the nature of the study. Since the study is examining different administration methods of the steroids, participants were aware of the intervention they were receiving, and none of the groups involved a placebo (Table 2). Thus, outcomes of olfactory function could have been influenced by participants’ impressions of the interventions.
Finally, de Borja Callejas et al published a retrospective case series in May 2015 analyzing the effects of corticosteroids on mucosal remodeling in patients with CRSwNP. 13 In this study, 18 patients with CRSwNP according to EPOS criteria were given 30 mg/d of oral prednisone for 4 days, followed by a reduction of 5 mg for 3 days. The total dose was a low 165 mg due to administration of intranasal budesonide for 12 weeks as well. The overall risk assessment was a moderate risk of postintervention bias due to moderate risk in the measurement of outcomes domain according to the ROBINS-I tool (Table 3).
Discussion
This review aimed to assess the perioperative use of oral corticosteroid in patients with CRS following the publication of the EPOS in 2012. Although there is compelling evidence supporting the use of oral steroids in the management of CRSwNP, a clear consensus on regimen and dosage has yet to be reached. Currently, EPOS guidelines recommend that preoperative management of CRSwNP must consider balancing the short-lived benefits of systemic corticosteroid with the potential long-term side effects. Based on their recommendations, it would appear that there is no definitive appropriate dose. Consequently, several of the studies that were alluded to in EPOS had great variation in oral steroid dosing, ranging from 25 mg daily for 2 weeks to 50 mg daily for 6 weeks. However, oral steroids for treatment of CRSwNP have LOB Ia. There is also LOB Ia for use of oral steroids postoperatively in patients with CRSwNP. In a randomized placebo-controlled study, patients who received oral corticosteroids perioperatively (for 5 preoperative and 9 postoperative days) had much healthier looking cavities at 6 months follow-up compared to placebo. 14 Conversely, due to the paucity of studies that support use of oral steroids in CRSsNP, EPOS designed an LOB IV for its use preoperatively and postoperatively.
Poetker et al in 2013 had evaluated studies prior to the publication of EPOS 3 and corroborated the use of short-term oral corticosteroids in the management of CRSwNP and, in some cases, CRSsNP. The articles we identified had unanimously reported improved outcomes with the use of oral steroids; however, several caveats exist. There is a lack of high-quality studies evaluating the therapeutic effects of progressively increasing doses of oral steroids, as well as implementation of consistent treatment protocols currently in the literature. Of the studies we identified, only 1 was a double-blinded randomized controlled study. Four studies had an LOE Ib, 2 studies had LOE III, and 1 study with LOE IV, yet most the studies were either nonblinded or nonrandomized, leaving very few powerful studies. In addition, there are no studies to our knowledge that have ideal oral steroid regimen as the primary end point. Although there have been studies performed that have compared the efficacy of different modes of administration of steroids, there is a clear lack of evidence supporting an ideal dose for symptom relief. No consensus exists on this front and this is further complicated by the wide variety of dosages used in the studies identified. The study performed by Dubin et al attempted to explore various thresholds of “maximal medical therapy.” 5 There is general agreement that “maximal” therapy is anecdotally referenced, and it has not been studied systematically. 15 Consequently, from the 30-item questionnaire that was administered to 793 nonresident physicians, oral steroids were reported as “usually used (50%-90%)” as the median value, with prednisone dose ranging from 10 to 120 mg.
Another important aspect of steroid therapy is side effects. While most treatment protocols used involve only a short-term administration of systemic steroids, evidence suggests that the relief may be temporary with symptoms eventually relapsing. 13 The main goal of medical therapy in CRS is symptom relief and improved quality of life. Thus, the use of oral steroids may be warranted, potentially for a long term. Given the well-established side effect profile of systemic corticosteroid use such as delayed wound healing, increased risk of osteoporosis, immunosuppression, and hyperglycemia, we believe such research is highly relevant. While the side effects of long-term systemic steroid usage can be extrapolated from studies evaluating treatment of other chronic inflammatory diseases, it still serves as a useful secondary end point, especially in ascertaining the ideal oral corticosteroid regimen in CRSwNP. In the study performed by Reychler et al, there is discussion of the comparable efficacy of steroid nebulization to conventional oral steroids, with decreased risk for side effects due to lower systemic absorption. 15 Dautremont et al conceded that their study may have potentially missed a late effect of oral prednisone, beyond the 2-month period. 11 Interestingly, most studies we identified did not mention any side effects, though once could not assume there was none. Therefore, an ideal therapeutic range for steroid dosing is warranted.
The cornerstone of medical therapy in CRS is strictly symptom management. Much like any other chronic diseases of the airway, such as chronic bronchitis, CRS treatment revolves around symptom relief rather than cure. This can be a difficult task as studies have shown that suboptimal medical management can increase the likelihood for disease relapse. 16 The chronicity of symptoms in CRS is heavily determined by the burden of disease following appropriate medical therapy. Therefore, treatment should be aimed at not only symptomatic relief but should also minimize tangibly evident radiological or endoscopic disease. One of the most debilitating symptoms that is associated with CRS is hindrance, as seen in CRS, and has been linked to loss of smell regardless of whether there is any nasal obstruction to odorant molecules. Oral corticosteroids reduce epithelial inflammation and nasal polyposis, thereby providing symptomatic and radiologic improvement. The major limitations of our study are related to the number and nature of the studies that met our criteria. It was expected that the number of reputable studies would have been greater than what we found, and we believe this further corroborates our argument for the need of high-level studies addressing this topic. Due to the lack of high LOB studies on the use of oral corticosteroids, it is difficult to draw conclusions on the ideal dose for appropriate medical management of CRS. Although the studies that were identified in this review had overall low bias, there is a strong need for high-level RCTs to determine the safest and most effective treatment regimen in the medical management of CRS.
Conclusion
There is currently no consistent steroid regimen in the treatment of CRSsNP and CRSwNP. The oral steroid dosages ranged from 150 to 352 mg—a large disparity that needs to be further explored to minimize adverse side effects, optimize therapy, and avoid implementation of potentially ineffective treatment. In many of the studies we identified, patients with CRS were all treated with the same protocol without distinction between polypoid and nonpolypoid patients, it is pivotal that the distinction be made in future studies in order to optimize medical management given the differing disease profiles.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
