Abstract
Objectives
As the understanding of the pathophysiology of chronic rhinosinusitis has increased over the last few years, it has been discovered that biologic therapy targeting type 2 inflammatory molecules can be beneficial for those with difficult-to-treat chronic rhinosinusitis with nasal polyposis. Given the recency of biologics, there is somewhat limited evidence on when to prescribe these medications and who should receive them. A recent consensus paper published by a group of Canadian rhinologists gave recommendations regarding prescribing biologics to patients with chronic rhinosinusitis with nasal polyposis (CRSwNP). This study aimed to outline current practices and prescribing patterns of biologics for chronic rhinosinusitis in Canada and contrast this to existing guidelines.
Materials/Methods
An online survey was distributed to members of the Canadian Rhinology Group of the Canadian Society of Otolaryngology—Head and Neck Surgery. The survey contained 44 questions, with 17 questions covering various topics related to criteria for initializing biologic therapy, investigations ordered prior to initialization of a biologic, and follow-up schedules. The responses were subsequently compared to clinical guidelines.
Results
Twenty-one physicians responded to the survey. One response was discarded for this analysis as the respondent did not prescribe biologics. The biologic agent of choice for the majority of physicians (85%) was Dupilumab (Dupixent). The most used patient-reported outcome measure used was the SNOT-22 (80%), and most physicians (90%) confirm that their patients have undergone adequate sinus surgery with a computed tomography (CT) scan prior to considering biologic therapy. Investigations ordered prior to therapy and follow-up schedules varied. Only 10% of respondents consistently refer their patients to a multidisciplinary clinic prior to prescribing biologics despite recommendations in guidelines, but 70% involve an allergist and/or a respirologist in the care of these patients.
Conclusions
Most respondents follow the recommendations made by the Canadian and international guidelines [European Position Paper on Rhinosinusitis and Nasal Polyps (EPOS), European Forum for Research and Education in Allergy and Airway Diseases (EUFOREA)]. The lack of multidisciplinary clinics across the country could be contributing to the low number of physicians using this resource for their patients despite recommendations. As our knowledge about biologics and CRSwNP improves, and with updated guidelines recently published, this survey should be expanded and repeated in the future to revaluate biologic prescribing practices in Canada.

Introduction
Chronic rhinosinusitis (CRS) is an inflammatory condition that presents with inflammation of the nose and paranasal sinuses, with sinonasal symptoms present for greater than 3 months. 1 It is a heterogeneous disease with a variety of different phenotypes. 2 As understanding of the pathophysiology of CRS has increased over the last few years, biologic therapies targeting the type 2 inflammatory pathway have been proven effective in treating CRS with nasal polyposis (CRSwNP), and these can be used in patients who have severe and/or recalcitrant disease. 3 Generally, biologics work by inhibiting certain parts of the immune system and suppressing inflammatory pathways. There are currently 3 biologics approved for CRSwNP in Canada: dupilumab (Dupixent), omalizumab (Xolair), and mepolizumab (Nucala), targeting IL-4/IL-13, IgE, and IL-5 respectively.4-7
While effective, there is a substantial price to these medications, with costs approaching nearly $50,000.00 CAD per year for some agents.8,9 In comparison, the cost of endoscopic sinus surgery (ESS) in 2013 was approximately $3500.00 CAD which is estimated to be approximately $4550.00 CAD in 2024.10,11 Recent cost-comparison and quality-of-life studies in the United States of America have found that ESS remains the most cost-effective strategy as both ESS and biologic therapy lead to near equal improvements in quality of life, regardless of the number of ESS needed by patients.12,13 Therefore, it can be challenging to determine in which situations the benefits of biologic therapy outweigh the benefits of ESS.
Given the complexities of prescribing these medications, a Canadian consensus paper published in 2021 presented recommendations on the approach to treatment of CRS with biologics. 3 The consensus statements cover suggestions for eligibility for biologic therapy for patients with CRSwNP including evaluation of patients to determine if they are eligible for biologic therapy, and evaluation of the response to treatment among others. 3 Given the recent publication of this consensus paper, it is not known what approach Canadian rhinologists take to prescribe biologics to patients with CRS, and how they follow-up with these patients. This paper aims to better understand how biologics are prescribed to patients with CRS in Canada, and contrast practice patterns to the Canadian guidelines.
Methods
A 44-item survey was constructed to evaluate various practices in otolaryngology, of which 17 questions covered biologic prescribing habits by Canadian rhinologists for patients with CRS. The survey included 8 demographic questions. The survey was distributed electronically to the 71 members of the Canadian Rhinology subgroup (a committee/group within the Canadian Society of Otolaryngology—Head and Neck Surgery). Data was collected over 4 weeks and a reminder email was sent 5 days before the survey closed. Participation in the survey was voluntary and no identifiers were collected. Of note, the authors of this paper did not participate in the study. The survey can be found in Appendix A.
Only complete survey responses were included in the data analysis. Responses were collected via Opinio, an online survey software, and analyzed using the software’s survey reports.
Results
Demographics
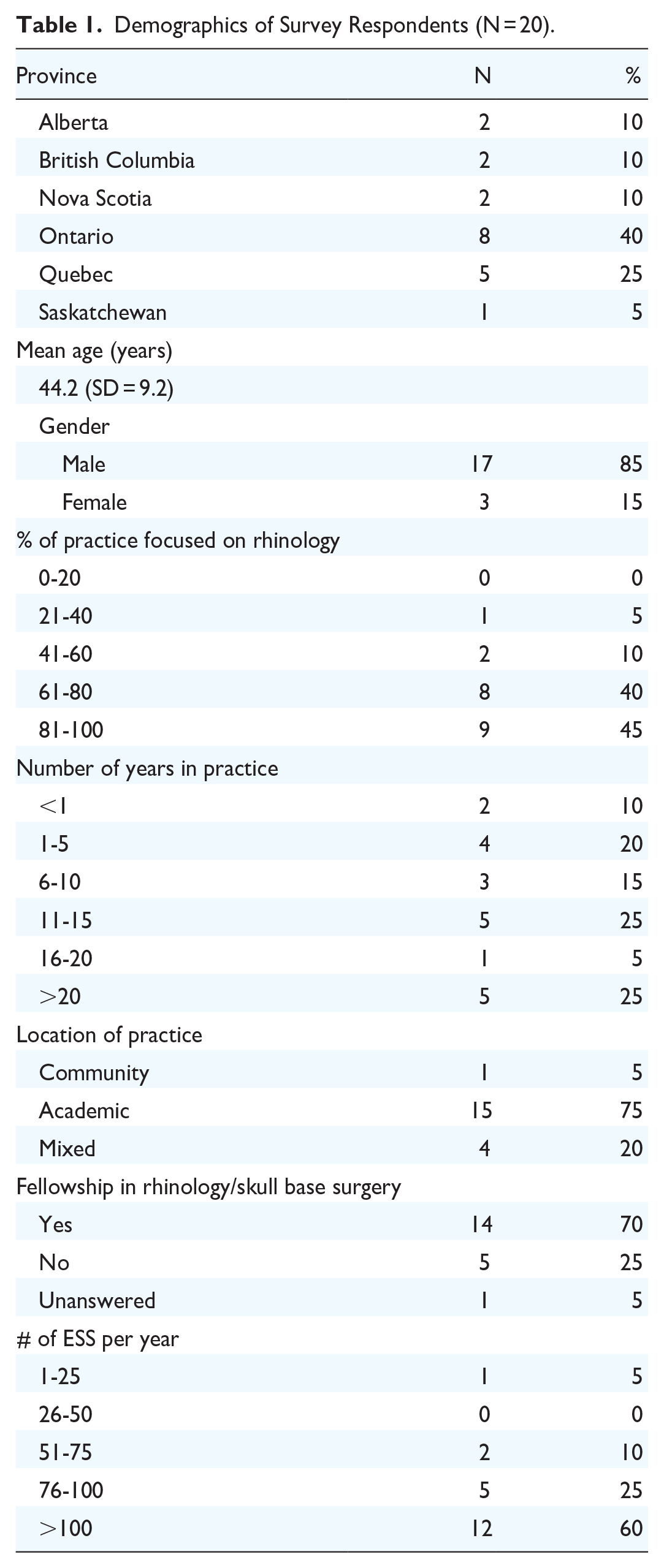
Twenty-one rhinologists responded to our survey from a total of 71 eligible participants (29.6%). One response was not included in the data analysis as the physician did not prescribe biologics in their practice. There were 2 respondents from Alberta (10%), British Columbia (10%), and Nova Scotia (10%) who responded to the survey, 8 members from Ontario (40%), 5 from Quebec (25%), and 1 from Saskatchewan (5%), with the majority working in an academic setting (15 respondents, 75%). One respondent works in a community setting (5%) and 4 work in a mixed practice (20%). Fourteen respondents have a fellowship in rhinology/skull base surgery (70%), 5 report not having this fellowship (25%), and 1 preferred not to answer (5%). The age of respondents ranged from 33 to 65 years, with the average age being 44.21 [Standard deviation (SD) = 9.2]. Most respondents are males (17 respondents, 85%). The complete demographics can be found in Table 1.
Demographics of Survey Respondents (N = 20).
Prescribing of Biologics
Nineteen respondents (95%) report following guidelines when considering biologic therapy for their patients, with 14 respondents using Canadian guidelines, 2 respondents using the European Forum for Research and Education in Allergy and Airway Diseases (EUFOREA) guidelines, and 3 respondents using both guidelines mentioned. The number of patients on biologics in individual practices varied and can be found in Figure 1. Survey respondents were asked to select which patient-reported outcome measure (PROM) they use prior to prescribing biologics if they use one. The most used PROM is the Sinonasal outcome test (SNOT-22), which is used by 80% of respondents. Of those using the SNOT-22, 4 respondents also used the Visual Analog Scale (VAS), representing 20% of respondents. One respondent also uses the Chronic Sinusitis Survey (CSS), and 1 respondent also uses the chronic rhinosinusitis patient-reported outcome (CRS-PRO), along with the SNOT-22. The VAS, CSS, or CRS-PRO were never used as the sole PROM of choice of physicians who participated in this survey. Three respondents (15%) did not use a PROM, and 1 respondent (5%) did not answer. Almost all respondents (N = 19, 95%) consider cost when prescribing biologic therapy, and 90% (N = 18) discuss side effects with their patients.
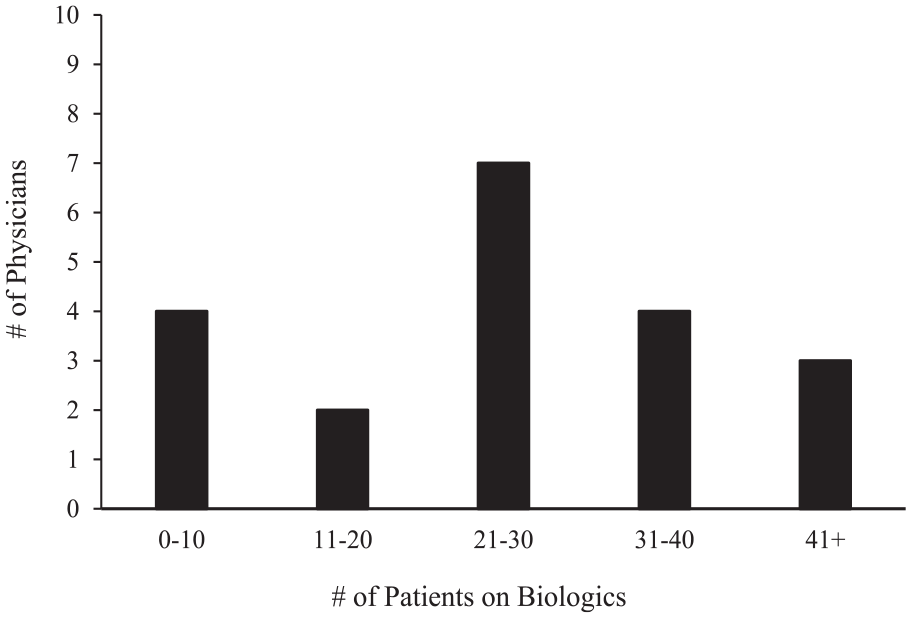
Number of patients on biologic in individual practices.
The survey also asked about investigations ordered prior to initializing a biologic agent. Twelve physicians (60%) report ordering more than 1 investigation. The most common test is an eosinophil count, which is ordered by 14 respondents (70%). Of those, 9 (45%) will also order an IgE count and 3 (15%) will also order a spirometry test. Various combinations of investigations are ordered, ranging from no investigations (3 physicians, 15%) to a full panel with spirometry, eosinophils, IgE, and allergy testing (one physician, 5%). A summary of the investigations ordered can be found in Figure 2.
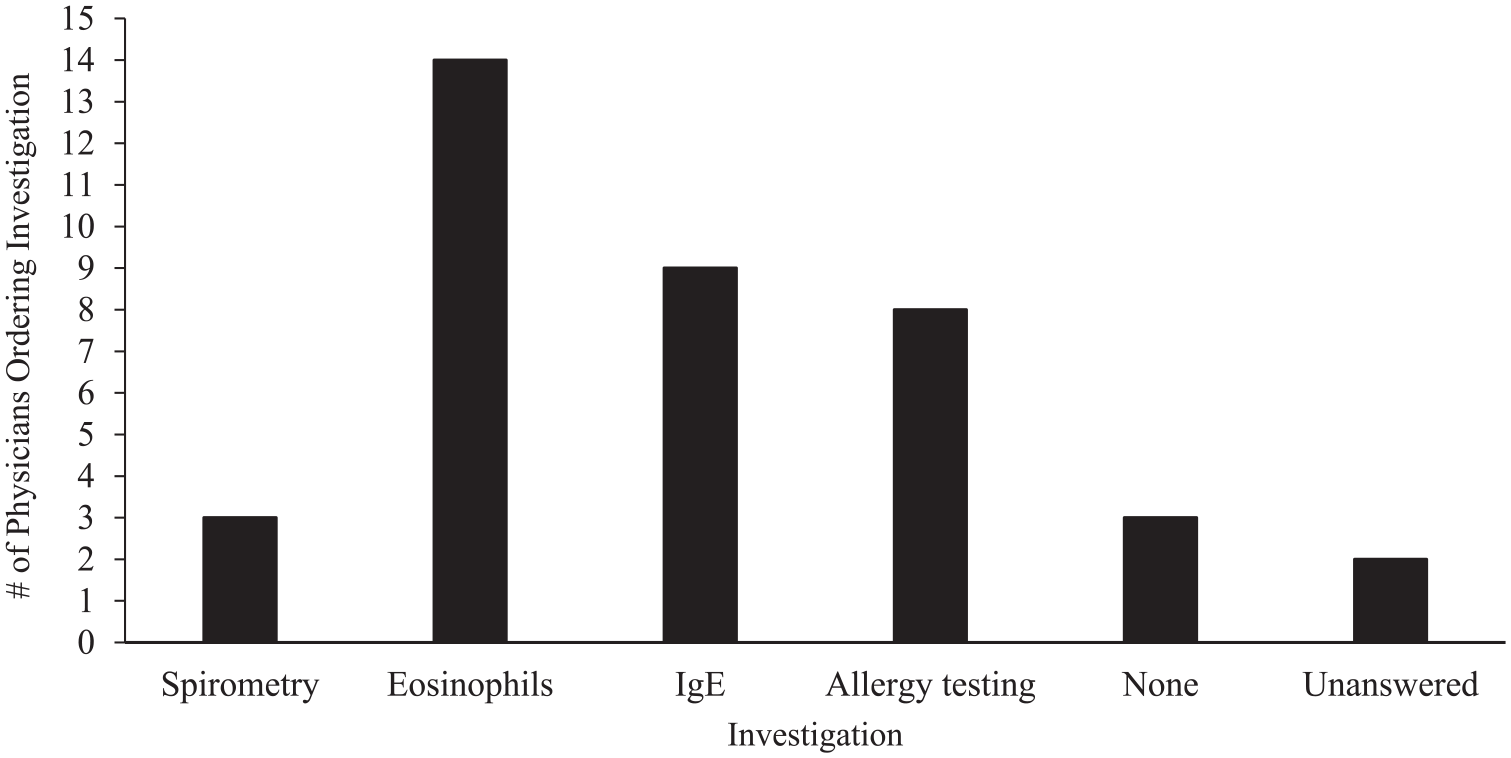
Investigations ordered prior to initializing a biologic agent.
Ninety percent of respondents ensure that their patients have undergone adequate sinus surgery, if fit for surgery, with a CT scan prior to initializing biologic therapy. However, 45% of physicians (N = 9) reported having at least 1 patient on biologic therapy prior to any sinus surgery. Of these 9 responses, 6 physicians (67%) reported that surgery was not needed after initializing biologic therapy, 2 physicians (22%) reported that surgery was eventually needed after a trial of biologic therapy, and 1 physician (11%) did not answer.
Dupilumab is the initial biologic of choice for 85% of physicians (N = 17), while mepolizumab is the initial biologic of choice for the remaining 15% of physicians (N = 3). Omalizumab was not the initial biologic of choice of any respondent. Thirteen respondents (65%) reported offering a second type of biologic if the first treatment failed. Patient follow-up was varied. Follow-up schedules are described in Table 2.
Follow-Up Schedule After Initialization of Biologic Agent.

Multidisciplinary Care
Two respondents (10%) reported that their patients are always evaluated in a multidisciplinary clinic prior to prescribing biologics, 8 respondents (40%) will sometimes send their patients for an evaluation in a multidisciplinary clinic prior to prescribing a biologic agent, and the remaining 50% do not routinely use a formal multidisciplinary clinic. One physician specified that they will ask for a consult with a respirologist if the patient has evidence of poorly controlled asthma. However, 70% of respondents work with an allergist and/or a respirologist when prescribing biologic agents to their patients, just not in a formal multidisciplinary clinic setting.
Discussion
This survey aimed to describe how Canadian rhinologists prescribe biologics for CRS and compare these patterns to the existing guidelines. The majority of the respondents work in an academic setting, have a fellowship in rhinology/skull base surgery, and are male. The respondents represented 6 different provinces in the country, from Nova Scotia to British Columbia. As biologics are a new treatment for CRS, it is important to understand how they are being prescribed and if there are any gaps in the guidelines that need to be addressed. This study compared survey results to various practice guidelines available.
Multiple guidelines outline a variety of things to consider when evaluating and managing patients with CRS, with some specific recommendations for the initialization of biologic therapy. One recommendation made by the EUFOREA guidelines and the Canadian Rhinology Working Group Consensus Statement is using a validated PROM, specifically SNOT-22 or the VAS, to determine the subjective burden of disease on the patient.3,14 Most rhinologists in this study use the SNOT-22, and some use the VAS, CSS, or CRS-PRO, all of which are validated PROMs. Specifically, the SNOT-22, CSS, and VAS are mentioned in the Canadian Rhinology Working Group Consensus statement as being some of the validated PROMs that should be used to prescribe biologics for CRS, all of which are used by the respondents of this survey. 3 Only a minority of respondents do not use a PROM when assessing patients with CRS.
There are currently no known biomarkers that can be used to predict response to treatment with biologics. However, multiple guidelines recommend measuring type 2 inflammatory markers (IgE, eosinophils) prior to initializing biologic therapy, as this can be suggestive of poor disease control.1,14 The most ordered test by the survey respondents is an eosinophil count, followed by an IgE level, as recommended by the guidelines. Furthermore, some guidelines also recommend that patients at high risk of comorbid asthma and/or allergies be seen by a multidisciplinary team consisting of an otolaryngologist, a respirologist, and an allergist, where investigations like spirometry and skin prick tests can be done alongside the type 2 inflammatory markers.3,14 However, only a small number of physicians report consistently referring their patients with CRS to a formal multidisciplinary clinic, and less than half of respondents order an allergy skin prick test for their patients with CRS who are considering biologic therapy, and only 15% order a spirometry test. Of note, the majority do work with a respirologist and/or allergist prior to initializing a biologic agent. Given this data, the low number of patients being seen/referred to these clinics is more likely due to a lack of established infrastructure as opposed to a willingness to utilize a multidisciplinary approach.
The treatment algorithm for CRS recommends that biologic therapy should only be initiated if patients have failed appropriate medical therapy (AMT) and have received minimal relief from sinus surgery if they are eligible for surgery.1,3,14,15 The Canadian consensus statement also mentions the possibility of using a CT scan to determine if patients have undergone adequate sinus surgery. 3 This is because there is a wide variety of surgical procedures that can be completed in the paranasal sinuses. Furthermore, the degree to which the sinuses are opened can greatly influence the delivery of topical medications, as well as disease control rates.16-20 Ensuring patients have received adequate, complete surgery is therefore paramount before deeming a patient as a treatment failure and current research is ongoing about how to best assess this. 21
A vast majority of Canadian rhinologists ensure patients have failed AMT and have undergone adequate sinus surgery with a CT scan prior to initializing biologic therapy, as recommended by the Canadian guidelines. Interestingly, 45% of our respondents have had at least 1 patient with CRS on biologic therapy despite them not having undergone sinus surgery. Surgery was able to be avoided in many, but not all, of these patients. This survey did not evaluate if these patients were ineligible for surgery which could explain why biologic therapy was initiated, or if the biologic was started by a different practitioner for control of a comorbid disease (for example, a respirologist treating severe asthma). The Canadian guideline also recommends considering cost when prescribing biologic therapy.
When a biologic agent is initialized, it is recommended to follow-up with patients within 16 weeks, and then again after 1 year to evaluate the response to treatment.1,14 However, the 16-week mark has not been evaluated by research and is simply recommended to prevent unnecessary continuation of treatment in patients who do not respond to biologics. 14 Follow-up time in survey respondents varied, with the majority following up within 3 to 4 months after initialization of a biologic agent, and some following up earlier (1-2 months) or later (5-6 months). Finally, the Canadian Society of Otolaryngology (CSO) consensus statement agrees that a second biologic therapy may be initiated if the first therapy fails as different biologics target different parts of the immune system, so patients may benefit from a different agent if the first one has failed. 3 This recommendation is followed by most Canadian rhinologists.
This study is the first in Canada to examine the biologic prescribing practices for CRS among Canadian rhinologists. The initial guidelines for biologic use in CRS were published in 2021 but have since been updated in 2023. Future research should aim to administer an updated version of this survey, focusing on how the new guidelines recently published by the CSO are being followed, get a more detailed view of the prescribing process by looking at barriers to implementing and following the guidelines, and how these barriers can be addressed to improve patient care. 22 Additional topics that we hope to investigate further include how the use of biologic medications has influenced views and prescribing practices of otolaryngologists with respect to oral corticosteroids, and if operating room constraints have influenced biologic prescribing patterns.
This study is not without limitations. The small response rate, while within norms in the literature, makes it challenging to generalize biologic prescribing habits to all Canadian rhinologists. The response rate of this survey was approximately 30%, with acceptable response rates for online surveys varying from 25% to 30% in the literature. 23 The low response rate may also be due to the short response time. Given that these practices were evaluated via a survey, response bias may be present and therefore, our analysis may not be truly reflective of the current practices. Furthermore, our survey did not ask about the reasoning behind certain practices. Options for testing or access to different specialists may be limited across regions, which could influence the management strategy of different physicians. Some of these limitations may be addressed in future surveys. Lastly, our survey was focused primarily on rhinologists or those with a strong focus on rhinology in practice, but in the future, we hope to expand it to General Otolaryngologists to evaluate guideline adherence as prescribing of biologics expands.
Conclusions
The majority of Canadian rhinologists who responded to this survey follow some of the recommendations made by the Canadian guidelines and international guidelines [European Position Paper on Rhinosinusitis and Nasal Polyps (EPOS), European Forum for Research and Education in Allergy and Airway Diseases (EUFOREA)] regarding the initiation of biologic therapy for patients with CRS and the management of these patients. The low number of physicians referring patients with CRS who are on biologics to multidisciplinary clinics could be due to the lack of access to these clinics across the country, which creates a barrier to patient care. This study serves as a baseline for future studies on this topic as guidelines evolve. As our knowledge about biologics and CRSwNP improves, and with updated guidelines recently published, this survey should be expanded and repeated in the future to revaluate biologic prescribing practices in Canada.
Footnotes
Appendix A
Acknowledgements
We would like to thank the survey respondents for taking time out of their day to complete the survey.
Author Contributions
AO generated the survey, analyzed the data, and prepared the original draft. All authors contributed to editing the survey and final manuscript. All authors read and approved the final manuscript.
Availability of Data and Materials
Survey questions and survey results described in this article can be obtained upon reasonable request by contacting the corresponding author.
Consent for Publication
Not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: AO received a stipend from the Dalhousie Medicine New Brunswick Summer Studentship fund to complete this research project.
Ethics Approval and Consent to Participate
Approval for this study was granted by the Horizon Health Network Research Ethics Board, ROMEO File # 101666.
